The British laryngologist and UCL professor Martin Birchall is one of the two founding fathers of “bioengineered” trachea, the other one being his former partner, the scandal surgeon Paolo Macchiarini. Birchall presently runs three trachea transplant trials clinical trials, two in the UK and one EU-wide. His patients must however wait to be cured, because the two UK trials, Inspire and RegenVox (with the latter primarily about larynx and upper trachea transplant) were both placed on hold, and the EU funded phase 2 trial TETRA is still far from getting an ethics approval and just now lost one potential trial site after the Medical University Vienna jumped ship. The EU Commission refuses to tell me anything about TETRA, except that it has not applied for ethics approval yet. EU’s Transparence department still evade commenting on how to put this scheduled 48 patient clinical trial in context of the 11 cadaveric trachea transplants by Macchiarini and Birchall, most or maybe even all of which left the patients either dead or mutilated.
In this article, I publish the most recent patient information sheets for the two suspended Inspire and RegenVox trachea transplant clinical trials, which I obtained under Freedom of Information request from the UK Health Research Authority (HRA).
The “breakthrough”
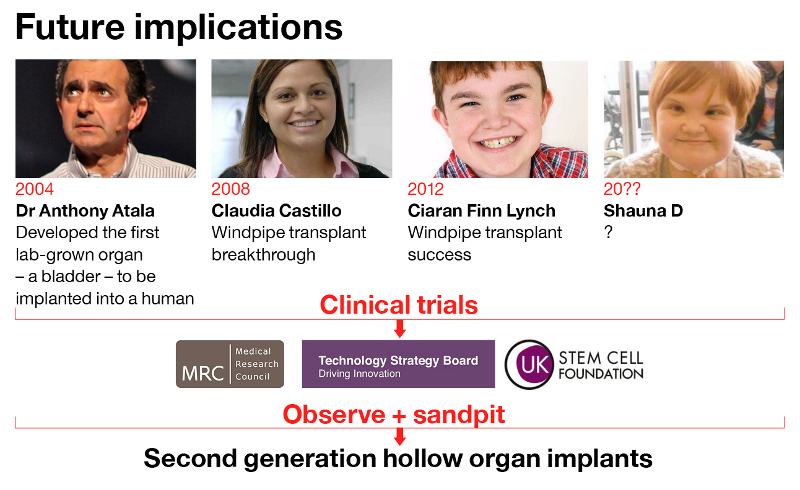
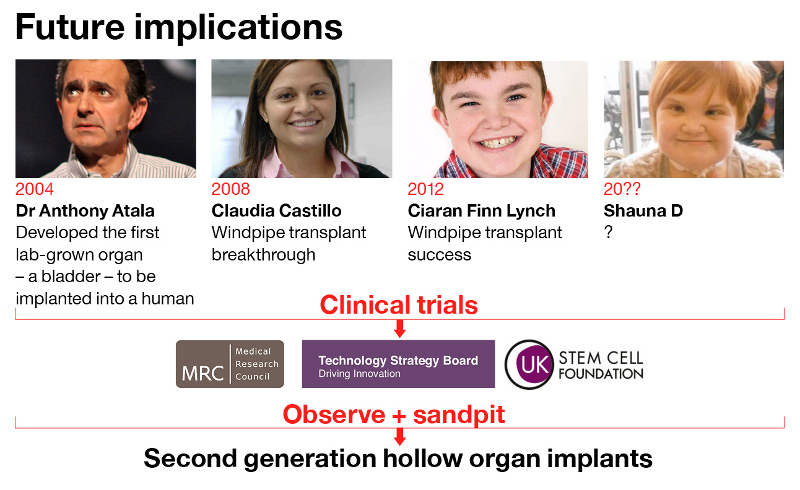
It all started in 2008, when together Macchiarini and Birchall transplanted a young woman, Claudia Castillo, with the first ever circumferential graft made of decellurised cadaveric organ, reseeded with bone marrow cells which were supposed to grow into cartilage and regenerate a piece of dead scaffold into a living, vascularised organ. According to all evidence, Birchall made that graft in his former lab in Bristol, which was a veterinary lab for pig experiments, not certified for any human tissue storage. He obviously did so behind the back of authorities, but because Birchall denies this, all that evidence and common sense are simply disregarded as wrong.
Several other patients followed, which I listed here; many are dead, for others we mostly do not even know if they still alive. Claudia Castillo survived because hers was a bronchus transplant, not a trachea. Last year, her left lung was removed together with the rotting bronchial graft. For Birchall, she continues to be a success story, and so far his word of an Englishman counts more than any evidence.

There is in fact no scientific evidence for bone marrow cells ever regenerating a trachea, in fact a 2015 misconduct investigation at UCL confirmed just that, and reprimanded Birchall for making such claims, while acquitting him from misconduct accusations. Yet a recent, 2017 UCL investigation into trachea transplants recommended for the clinical trials Inspire and RegenVox to continue, based on…, no, not scientific evidence. The 2017 UCL investigative committee freely admitted to me not to have screened or even requested any actual evidence. They relied solely on testimonies, and once again, that of Birchall was the most convincing one, even if it contradicted known evidence or even his own published statements from before (read here). The committee’s conclusion was: because cadaveric tracheas are much safer than plastic ones, they are worth another try on four more patients in the Inspire trial. And because cadaveric voice boxes are even less dangerous, those should be definitely tried out on 10 people in RegenVox.
Don’t mention those
This is what RegenVox offers prospective patients, according to documentation I obtained:
“The implant can be considered ‘living’ because your own cells are grown on it,
and this type of treatment is referred to as ‘tissue engineering’ or ‘regenerative
medicine’.The scaffold along with your own living cells is surgically implanted into you
one month later. Initially it is implanted into a muscle in the forearm (although
it could also be implanted into the neck, chest wall or other site such as the
leg or foot) so that it can develop a good blood supply. Between 2 and 3
months later, a second operation is performed and the implant along with its
new blood supply is moved and used to reconstruct your larynx or upper
trachea”.
That bizarre bit about all these two clinical trials on humans is that they all received their ethics approvals without much scientific evidence that their technology actually works. Inspire got green-lighted based on alleged successes with Birchall’s (and Macchiarini’s) patients Claudia Castillo, as well as Ciaran Lynch, operated in 2010. The UCL trachea transplanter was now forced to mention one more, the deceased Shauna Davison (last section here). A fourth trachea transplant patient of Birchall’s and Macchiarini’s,, Keziah Shorten, remains unmentionable, because of her horrible suffering and death. It was actually that bad, that Birchall’s UCL Hospital (UCLH) refused to answer my related Freedom of Information inquiry as they deemed it vexatious. Now, the central English authority Information Commisioner’s Office ordered UCLH on October 26th to answer me in the course of 35 days or face court. I will sure share that reply.
Who reads papers?
As preclinical data, Birchall served to Inspire reviewers his common paper with Macchiarini, Go et al 2010, where the cadaveric tracheas were tested on pigs. When exactly? Birchall, Macchiarini and his acolyte Philipp Jungebluth (the one who keeps suing me in court) insist those tests were done before the 2008 operation on Claudia Castillo. Yet there is absolutely no actual evidence for this, quite the opposite, everything suggests the experiments were done afterwards. In any case, it seems the Inspire experts who took the cadaveric trachea regeneration technology as proven never bothered to read the actual Go et al 2010 paper. Why should they indeed, it was after all peer reviewed! Here is however another kind of peer review, on PubPeer, excerpt:
“The data lack substance and are too inadequate to support the above claim. In fact, apart from a table indicating the mechanical properties of the tracheas at post mortem, the only data showing the success of the ‘bioengineered’ tracheal graft seeded with both MSC-derived chondrocytes and epithelial cells is Fig. 4 (reproduced here). A major concern is that only a cross-section of the trachea is shown. What should have been shown was a longitudinal section spanning the junction between the recipient’s trachea and the graft. Without this, it is not possible to be sure if the cross-section is from the recipient’s trachea or the graft. However, the logical conclusion to make is that the cross-section of the ‘bioengineered’ trachea (Group IV) shown in Fig. 4 is likely to be the recipient’s trachea, because even if a long segment of the pig’s own trachea were removed and immediately re-implanted, it would not look as good as this, as explained in this review: http://dx.doi.org/10.3978/j.issn.2072-1439.2016.01.85.”
With RegenVox, it is even weirder. There was one patient who received a larynx, thyroid gland and trachea transplant from a donor in UC Davis in US in 2010, with Birchall and Macchiarini involved as surgeons. It was not about bioengineered organs, but live ones, yet somehow this confusion of successful regenerative medicine was created to influence the success of RegenVox and Inspire.
As for the decell-recell larynx- no preclinical data was published before the trial got its ethics approval in 2014. Once the pig data with decellurised voice boxes regenerated with human cells saw the light of the day 2 years alter, it did not look very promising to anyone who actually bothered to read the paper Ansari et al 2016 beyond its title “Stem Cell-Based Tissue-Engineered Laryngeal Replacement” Here again, an excerpt from a peer review on PubPeer:
“The authors indicate that no human cells could be found at the end-point of the experiment (6 months), and there is no evidence for the presence of viable cartilage cells, so why is this called “stem cell-based tissue engineering”? […]
Worryingly, this pig study shows that the implanted scaffold becomes fragmented following implantation. The authors state “In no animal was the intact implanted scaffold visible; however, in each case, white fragments of varying sizes (identified histologically as decellularized cartilage) of the scaffold could be seen.” It is likely that the patients in the RegenVox trial will be receiving implants that are bigger than those in the pig study, the information on clinicaltrials.gov indicating that “the narrow section of voicebox or upper windpipe” will be removed and replaced with the scaffold. Surely the conclusion from this pig study should be that this is not a safe procedure?”
Instead, Birchall simply postulated in 2014, when he obtained the ethics approval for RegenVox:
“We gave 16 pigs seeded laryngeal implants. Animal survival to 2 months was 81%. Decellularised scaffolds showed mild inflammatory responses, but, importantly, clear evidence of remodelled cartilage (quite unlike the `competitor’ aortic allografts used in France and the US). In all, endoluminal repair was excellent with glandular and interstitial regrowth. CT scans showed patent airways. Human cells were identifiable at the implant site for four weeks. Thus, we have shown biocompatibility, safety and efficacy in pigs”
Well, where is this amazing success of preclinical regenerative medicine published? Nowhere, that’s where, to this very day. Neither did anyone ever ask to actually see this promising results when approving RegenVox, even the 2017 UCL investigative committee did not bother. No documents about this pig trials are available, even upon specific request. RegenVox ethics approval to fix 10 patients with cadaveric voice boxes (and if necessary, with upper tracheas) was given to Birchall based on his word of honour.
Both trials suspended
The UK authority Oxford Research Ethics Committee (REC) placed the Inspire trachea transplant trial on hold, on December 2nd 2016, and solely on the bases of my reporting (read here):
“the decision had been to taken to place the study on hold to allow the research team to work with other regulators, researchers and patients to ensure the final study design is as responsible as possible”.
RegenVox was at that time still unaffected and actively recruiting patients. Yet from a communication from the Medical Health products Regulatory Agency (MHRA) I learned:
“Regarding the trial approval, we can clarify that the RegenVox trial has been on temporary halt since March 2017 and that no patients have been treated to date within the trial. The sponsor will need to provide a submission to the MHRA (and ethics committee if appropriate) in the form of a substantial amendment for assessment and authorisation before any restart can be allowed.”
I previously published the original grant proposals for Inspire and RegenVox with this article, please access them in this folder. I also previously made available past Patient Information Sheets for Inspire, dated December 2015 and May 2016, in this article. Those were not really honestly informing patients what risks awaited them. I now procured the most recent Patient Information Sheets, both for Inspire and Regenvox, they are here and here. They are actually not much better.
All tracheas working
This is what Inspire sheet tells prospective patients as background, notice that there is no mention whatsoever of Keziah Shorten:
“Three patients have received a similar trachea in the past few years. However, they did not receive exactly the same type of implant that we are using here. In 2008, a lady underwent partial tracheal replacement and is alive and well today. In 2010, a ten years old boy had his entire trachea replaced and is also alive, growing and well today. In 2011, a 15 years old girl also had her whole trachea replaced, but died a month later of unknown causes, although her trachea was working at the time of her discharge from hospital. All three patients were seriously ill at the time they received their transplants and would have probably died without tracheal replacement”.
Claudia Castillo had post-tuberculosis bronchus constriction and was never in any danger of death, at least not before she received the cadaveric bronchus graft; and Birchall knows it very well. How Ciaran Lynch is really doing : nobody ever bothered to find out independently, not even the 2015 and 2017 UCL investigators, who simply trusted Birchall’s word that the boy was still doing great (actually, he was not, again one should read Birchall’s own papers beyond their titles, here Hamilton et al 2015). Also, that 15 years old girl, Shauna Davison , who was operated in 2012, died only 3 weeks later because her trachea transplanting doctors decided not to put a stent in, and quickly sent her off to Leeds for aftercare, where her tracheal graft collapsed and killed her. This was determined unambiguously by the 2017 UCL investigation.
The description of these patients is less truthful for the RegenVox patient information sheet:
“The technology behind this trial has been tested by our team in three patients who were critically ill. One adult patient and two children received tissue engineered tracheal implants. In each of these patients, a section of trachea was prepared in a similar way and implanted successfully. One of the children died of an unrelated problem soon after having the implant surgery, but for two of these patients this treatment was life-saving. The adult patient is alive with minimal symptoms five years after receiving the implant, and the child is alive and well four years after receiving the implant.
A different group recently performed a tracheal implant using a similar technique in an elderly patient. Though this procedure was initially successful, the patient died from a heart attack several months later”.
You might wonder who that other tracheal implant patient from a different group might be. I guess Birchall means the patient of his bone marrow magic wielding colleagues Michael Olausson and Suchitra Sumitran-Holgersson, both recently found guilty of medical research misconduct by their University of Gothenburg in Sweden (more here).
What’s on offer?
In the old Inspire sheets, stents were discussed as a possible necessity in case of complications, to keep the collapsing airway open. Noteworthy is that the whole argument for the Inspire trachea replacement used to be to liberate the patients from the alternative therapy of stenting and its side-effects. Now, given what really happened to Shauna Davison, stents became a permanent fixture for Inspire:
“The new trachea will be supported by a tube made from medical grade plastic (silastic) called a stent”.
Even the tracheostomy, a hole in the throat to breathe through, is discussed as a likely necessity now. Where is the cure, or in fact where are the benefits for patients aside of the pride to contribute to science, one wonders. The risk and side effects in the patient information sheet for Inspire are certainly greatly understated. A look here shows what the most common outcome for cadaveric trachea transplants in reality was: death. Birchall however insists that his technology is different from Macchiarini’s, as it uses fat tissue (omentum) or muscle flap to try and promote vascularisation, but this is also what Macchiarini claimed to have done in his own unpublished manuscript. These prospects Birchall offers his Inspire patients:
“We cannot give a guarantee the trial will help you, since this is an experimental treatment, but, regardless of your personal outcome, the information we get from this trial will help improve the treatment of people with tracheal malacia or stenosis in the future and increase.”
Indeed, Birchall needs human subjects for his research, because in his last preclinical study Maughan et al 2007 he had to save on rabbits. There were apparently not enough of the furry creatures available to test the bone-marrow regenerated trachea on, so Birchall just compared a non-regenerated decellurised model versus plastic or classic formaldehyde-preserved homograft. They all failed, as the paper concludes:
“Conclusions: There are mirror-image benefits and drawbacks to nonrecellularized, decellularized, and synthetic grafts, such that none emerged as the preferred option. Results from prevascularized and/or cell-seeded grafts (as applied clinically)
may elucidate clearer advantages of one scaffold type over another”.
So humans it shall be then.
Bye, Austrian trial site
The EU seems inclined to let 48 more patients follow Birchall’s scheduled four Inspire patients for the trachea transplants, all my previous requests for information were denied outright (read here and here, also for details of TETRA phase 2 clinical trial). The grafts are to be produced in UK and in Germany, the clinical trial sites were originally supposed to next to UCLH in London also University Hospital of South Manchester in UK, Universita Degli Studi Di Brescia in Italy, Instytut Gruźlicy i Chorób Płuc in Poland and the Medizinische Universität Wien in Austria. And the latter recently abandoned ship. As the Vice-Rector of Medical University Vienna, Michaela Fritz, confirmed to me, while refusing to elaborate:
“The Meduni Wien is not part of the project TETRA anymore for strategical reasons”.
Their Polish colleagues might also eventually become reluctant to deliver patients for Birchall’s big experiment, judging from this communication I received from the project manager Joanna Dziwiszek in Warsaw:
“On behalf of Prof.Orlowski I would like to thank you for your e-mail. Professor is acknowledged with some of your article and is aware that this topic is being now controversial”.
She then added:
“Professor Orlowski informed me that at this moment patients for Tetra are not recruited at the Institute as some of another project’s stage has to be finished”.
Which leaves Birchall with two suspended trials in UK, and who knows what Europe-wide. Do not forget the impending Brexit.
UCL positive
UCL meanwhile issued a white paper defending trachea transplants, naturally in The Lancet, co-authored by Birchall, with a UCL scientist Giulio Cossu as lead author: Cossu et al, 2017. Cossu was himself investigated by UCL and cleared of all suspicions of misconduct in 2013, because this evidence was apparently unconvincing. In this Lancet white paper, it turned out that Macchiarini’s two publications in same journal about Claudia Castillo were absolutely correct and that she enjoys
“Normal mechanical properties and appearance of graft, improved quality of life”.
Who knew your life improves when you lose one lung and spend years in-and-out of emergency room! UCL also recently announced to help their unjustly maligned researchers:
“It is important that the sector nurtures such confidence in the management of integrity issues by responding appropriately to reports, having clear, robust and fair processes for investigating misconduct, following up on any actions required to correct behaviour, and supporting researchers when inaccurate or malicious allegations are made against them”.
Given how both UCL investigations into trachea transplants played out, this London university most likely meant to warn everyone to shut up about Birchall and his multi-million Pound and Euro funded trachea transplant trials. They mean business.
Update 31.03.2018. I received a set of documents concerning RegenVox, from the UK Health Research Authority (HRA-NHS). They include the original RegenVox grant application from April 2014 (starting page 12 here).
We learn that RegenVox was indeed also a trachea transplant trial: Birchall announced to remove the upper trachea together with the larynx of his 10 patients, leaving them with a frehsly cut tracheostomy and his educated guess promising that stem cell magic might revive the decellurised replacement grafts. It also seems, Birchall intended to even recruit children for such transplants and one reads quite a number of his fibs to the funder and the authorities, like that Claudia Castillo was doing great at the time of application (she was most certainly not), and that Shauna Davison’s graft was healthy and well vascularized and she died of unrelated causes (both not true).

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00

Giulio Cossu has this 2013 retraction for image manipulation in a 2006 paper.
http://retractionwatch.com/2013/01/22/clare-francis-scores-a-bullseye-journal-of-cell-biology-paper-retracted-for-image-manipulation/
2006 publication.
J Cell Biol. 2006 Jan 16;172(2):233-44. Epub 2006 Jan 9.
Follistatin induction by nitric oxide through cyclic GMP: a tightly regulated signaling pathway that controls myoblast fusion.
Pisconti A1, Brunelli S, Di Padova M, De Palma C, Deponti D, Baesso S, Sartorelli V, Cossu G, Clementi E.
Author information
1
Stem Cell Research Institute, Hospital San Raffaele, 20132 Milan, Italy.
2013 retraction notice.
http://jcb.rupress.org/content/early/2013/01/21/jcb.2005070832003r
Vol. 172 No. 2, January 9, 2006. Pages 233–244.
After concerns were raised by a reader, the Editors of The Journal of Cell Biology detected the following issues with the data in the above article:
(1) The top panel in Fig. 1 F (NOS-1μ) is identical to the top panel in Fig. 2 B (α-sGC).
(2) The bottom panel in Fig. 1 F (NOS-III) is identical to the bottom panel in Fig. 2 B (GAPDH).
(3) Lanes 2 and 3 of the Follistatin panel for satellite cells in Fig. 4 A are identical to lanes 1 and 2 of the Myostatin panel for satellite cells in the same figure.
(4) A band appears to have been erased from lane 4 of the Follistatin panel for satellite cells in Fig. 4 A.
(5) Lane 4 of the Myostatin panel for satellite cells in Fig. 4 A is identical to lane 1 of the Follistatin panel for E12.5/E15.5 in the same figure.
(6) The E9.5 explants and E12.5/E15.5 panels for GAPDH in Fig. 4 A are identical.
Given the above issues with the experimental data in Fig. 4 A, the quantification data in Fig. S1 A cannot be validated.
No issues were detected with the other figures in the paper nor with the other parts of the figures listed above (Fig. 1, A–E, G, and H; Fig. 2, A and C–E; Fig. 4 B; and Fig. S1 B).
The authors were contacted by the Executive Editor of the journal and provided the following statement:
“Given the time that has elapsed since the original date of publication, we are unable to find the original data used to prepare these figures and are not able to explain these observations. We thus retract the paper and apologize for any confusion it may have caused to the research community. Because of the issues noted above, the conclusions presented in this paper that changes in NOS activity are the consequence of activation and inhibition of enzyme activity and not of changes in protein expression (Fig. 1 F), that regulation of guanylate cyclase occurs through posttranslational events (Fig. 2 B), that there is a role for nitric oxide in expression of follistatin mRNA in cultured cells (Figs. 4 A and S1 A), and that there are roles for nitric oxide and cGMP in expression of follistatin mRNA in vivo (Figs. 4 A and S1 A) cannot be validated. The reported observations on the effects of nitric oxide and cGMP on myoblast fusion in cultured cells, of cGMP on myogenesis in vivo, of cGMP on expression of follistatin mRNA in cultured cells, of cGMP and nitric oxide on expression of follistatin protein in cultured cells, and of cGMP on expression of follistatin protein in vivo are not affected by the flaws detected in the figures.”
As a result of this retraction, no data in this paper should be cited in the scientific literature.
The Authors and Editors have informed the University of Milan of this retraction.
© 2013 Pisconti et al.
LikeLike
If there are two trials on hold and one far from getting ethical approval there must be at least some awareness
LikeLike
Pingback: Il "successo" del Gran Guignol - Ocasapiens - Blog - Repubblica.it
Pingback: Tissue-engineered tracheas: an assessment of the scientific, clinical and ethical implications – For Better Science
Pingback: Image reuse, the new low of UCL trachea transplanter Martin Birchall? – For Better Science
Pingback: UCL fibs Parliament about trachea transplants – For Better Science
Pingback: Trachea transplanters: Round 2 at UK Parliament – For Better Science
Pingback: UCL’s decellurised tracheas: strong and stable? – For Better Science
Pingback: EU trachea transplant clinical trial TETRA “uncertain to take place” – For Better Science
Pingback: Trachea transplanters without borders – For Better Science
Pingback: UCL trachea transplants: Videregen sets lawyers on Liverpool academics Murray and Levy – For Better Science
Pingback: Google cleaned of “Defamation Campaign” against Martin Birchall and UCL – For Better Science
Pingback: New government cash for trachea transplanters Videregen – For Better Science