Trachea is a very difficult organ to transplant, because its blood supply happens through many tiny blood vessels and it is practically impossible to connect them all (same is true also for the oesophagus). An organ transplanted without blood supply will die, and this is where the regenerative medicine of Paolo Macchiarini and Martin Birchall comes in: a treatment with stem cells and growth factors will somehow magically ensure quick vascularization and bring the dead tissue to life. There is no independent evidence for this, and most patients died as the consequence or became dangerously ill when their new tracheas, made from plastic or from dead, decellurised cadaveric organs predictably failed. One boy has survived: Ciaran Lynch. His current clinical state is not too rosy, even by Birchall’s own admittance, but nevertheless the sole fact that Ciaran (unlike almost all other trachea transplant patients) is alive, sufficed for Birchall to have one or two more patients operated (both dead) and to push through a clinical trial in UK with a much larger follow-up trial EU-wide. The difference though: that boy never had much choice anyway, since he had no real trachea in the first place. He received very early on a pickled trachea homograft from dead human donor tissue which functioned very well for many years, but at some point urgently needed replacement in order to save his life. Now Birchall however wishes to remove the live airways of stable patients outside any life danger, who suffer from tracheal stenosis, and replace them with his regenerated dead ones (see this report). Noone seems to wonder why these tracheas are to be prepared in a radically different way from that of the sole success, Ciaran Lynch: Birchall’s bioreactor vs so-called “bionic” method of Macchiarini.
The easy route to success
Imagine you developed a new technology of tracheal transplant. As an ethical scientist you would first test it on animals, before ever daring to offer it to human patients. Animal tests involve quite a lot of paperwork and ethics approvals, especially tests on large animals like pigs, commonly used in airway surgery experiments. You have to present those results in a peer-reviewed journal and in conferences, and show all data: the statistics of how many pigs were transplanted, how many survived for how long, how many grafts collapsed and needed stenting (widening of the airway with metal or plastic nets), and which were the side effects of the transplants. You also have to do proper controls on your animal subjects to compare your technology with the already established standards. In the case of regenerated dead trachea, the proper control would have to be a homograft, which is basically the same, but without the stem cells. You would have to prove that your stem cell magic works, that your transplant is not lethal or toxic and that it performs significantly better than a homograft. Only when all this is evaluated, published and presented at conferences for peer debate, would you apply to test the method in patients in a life-threatening situation.
Of course, you can also skip all the above. The legal regulations on compassionate use allow doctors to test whatever they seem fit on the patients, provided the patients agree. There seems to be no need for previous animal testing, or in fact any safety tests at all, no strict guidelines on what information on risks and previous experiences must be shared with the patient giving informed consent, and, when the hospital agrees, even no requirement of an independent ethics vote, as the case with tracheal transplants by former Macchiarini partners Heike and Thorsten Walles in Germany demonstrated (see my report here). The doctors who treat the patient decide to apply their methods singlehandedly, as long as no authority interferes and the patients sign whatever the doctors deemed to work as their “informed consent”.
One single surviving patient can be published in an elite medical journal as a case study evidencing groundbreaking success. Noone will come asking what happened to other patients treated with same method, or why this new method is better than established clinical alternatives. Macchiarini operated apparently 10 patients with cadaveric tracheas in Spain, Italy and in Russia as compassionate use cases (see my reports here, here and here), almost all of them are dead, those 1-3 still alive are permanently damaged and mutilated as consequence.

Bioreactor vs Bionics
One such survivor is Claudia Castillo. She was the first trachea transplant recipient of Macchiarini and Birchall, operated in June 2008 at the Hospital Clinic Barcelona in Spain, where Macchiarini led the thorax surgery department. Her graft was made from a decellurised donor organ from Italy, which was regenerated in Birchall’s veterinary lab at the University of Bristol, UK, where he did airway surgery research on pigs (see my report here). According to The Guardian, Birchall “was given permission by the Human Tissue Authority to prepare the cells in a veterinary laboratory, that was not licensed for clinical applications”. However, to me the same authority (HTA) explicitly declared that they had absolutely no clue that a trachea was being prepared for human transplantation by the Birchall team in that unsafe animal research lab. In fact, they even wrote in June 2008 to forbid Birchall to store his patient’s cells in such conditions, and demanded for the cells to be taken away from the veterinary lab. The next day, the organ seeded with cells was urgently flown to Barcelona where Macchiarini implanted in into Claudia Castillo. The operation was hailed worldwide as success for many years, until even Birchall had to admit in a September 2016 conference that that transplant was actually not a success, as a member of audience told me.
But back then, the operation of Claudia Castillo served as a template for a whole series of compassionate use operations for Macchiarini, Birchall and their paediatric surgeon colleague from the Great Ormond Street Hospital in London, Martin Elliott. Ciaran Lynch is officially the 2nd patient who received such a trachea transplant, in March 2010. Yet for some reason, the bioreactor approach was dropped in favour of a novel “bionic” method which Macchiarini based on a patented invention of his former Hannover Medical School (MHH) colleague, Augustinus Bader (read more here). Here, stem cells (or whatever the doctors chose to define as such) are seeded directly onto the graft during the implantation and supplemented by growth factors like erythropoietin (EPO). Bader and Macchiarini published their common bionic method for airway regeneration right after Ciaran’s operation (Bader & Macchiarini, 2010). Macchiarini seemed to have liked it so much that he operated most his trachea patients using a version of the Bader technology (in fact, a 2016 Karolinska University Hospital investigation criticised such use of EPO as dangerous and irresponsible, see report and excerpt here). Birchall however grew fond of the bioreactor method, which is also supposed to serve as basis for his scheduled clinical trials INSPIRE and TETRA (former currently suspended, latter in preparation, see my reports here and here).
Yet why was this bioreactor-free technology used on Ciaran out of the blue if the bioreactor was declared such a success with his predecessor, Claudia Castillo? Macchiarini describes the reasons in this lecture at the Royal Society for Medicine (RSM) from July 2010, where he and Elliott present their operation on Ciaran. Some are a kind of an obscure scientific arguments (i.e., decellurised tracheas are supposed to contain some preserved growth factors which magically promote angiogenesis), but Macchiarini also gives the main rationale to avoid the bioreactor: regulatory headaches. Which is perfectly understandable given the hassle he and Birchall had with HTA and the veterinary lab in Bristol, for which they have been so far very lucky to avoid criminal charges. A bioreactor and all cell culture media and ingredients need to be approved as safe and kept in certified GMP labs, and certainly not where pig cadavers are processed. With the bionic method invented by Bader, you need no approvals and no permits, since it is the patient’s own cells which you extract and right after put back in a different part of same patient’s body.
A secret Barcelona patient
Surprisingly, Ciaran was in fact not the 2nd, but the 3rd trachea transplant recipient of Macchiarini’s, and a second one to get a bioreactor-free, “bionics”-prepared transplant. Yet the fate and even the existence of that second trachea transplant patient, a 51-year old woman, is all but known. Macchiarini talks about her at the above mentioned lecture at the RSM (around time stamp 35:50), she is also briefly mentioned as the apparently permanently damaged patient 2 in this Corriere Fiorentino article. She might be the Spanish woman from the German city of Dortmund mentioned in this BBC article, or someone else. We know she was operated in Barcelona in 2009, yet not at the Hospital Clinic, where permission was denied by its institutional review board, as the hospital’s head Antoni Castells now informed me:
“I did a double double-check. First, I asked our documentation department to review all surgical procedures performed in years 2008 and 2009: only one airway transplant was done. Second, I asked our current chief of the Thoracic Surgery Department if he was aware of a second transplant performed in the Hospital Clínic, and he confirmed me that only one airway transplant was performed in our Hospital. On the other hand, he also told me Macchiarini was working at the Institut Dexeus (a private center in Barcelona, now part of Quiron group) at that time, so it is likely the transplant was done there”.
Indeed, Macchiarini once provided his affiliation with the hospital USP Instituto Universitario Dexeus in a TV programme about artificial organs screened on PBS Nova in January 2011. The Italian star surgeon has a practice of hiding patients: according to the Corriere Fiorentino report, he operated between 2010 and 2011 seven more trachea transplant patients at his next place of work, the Carreggi university hospital in Florence, Italy. 6 out of these 7 patients are dead, one suffered permanent brain damage. Yet only four of those seven are known to the Italian authority National Transplant Centre (details here).
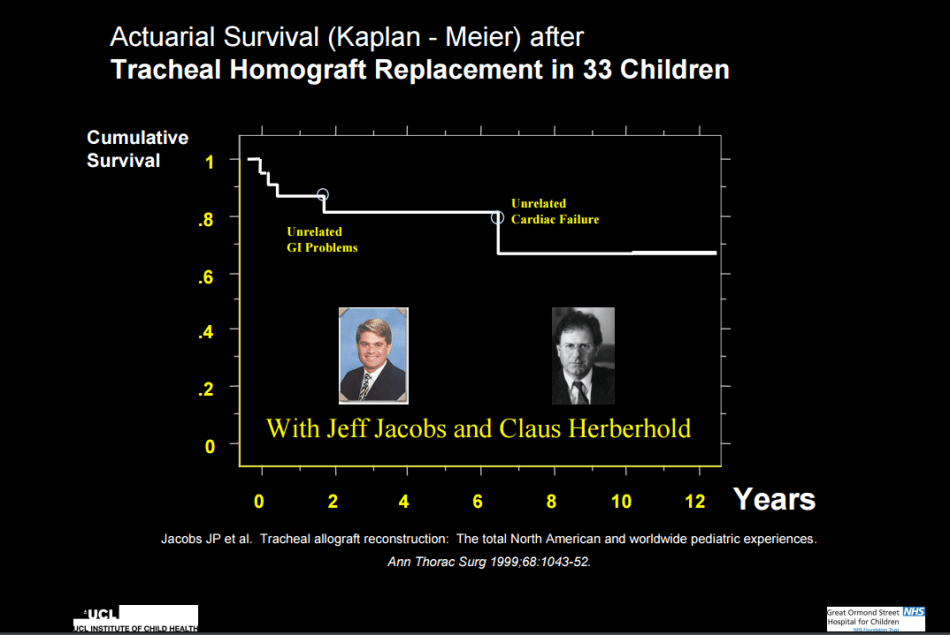
Homograft vs stem cell magic
And of course in March 2010 Macchiarini flew to London to operate Ciaran; also the decellurised donor trachea arrived from Italy, ready to be regenerated in vivo. The team Elliott, Birchall and Macchiarini hit the world news with the story how they saved the life of a boy. Yet for some mysterious reason, when the alleged success story of Ciaran was published in The Lancet (Elliott et al 2013), Macchiarini was not one of the authors (but his bionic-inventor colleague Bader was).
There is no doubt that the transplant saved Ciaran’s life. It is yet another question if that regenerated trachea was any better than the available established technology, or if it instead put the boy under undue extra risk, e.g. though the use of EPO. Ciaran’s previous airway tube was a homograft, which Elliott prepared from a pickled (fixative-preserved) trachea of a dead donor, back when the boy was very little and almost died. The homograft methodology was established in the 1980ies, and provided relatively satisfactory results in cases where no alternative was available to save lives. The grafts often collapsed and needed stenting, and in fact the metal stent inside Ciaran’s homograft eventually eroded into his trachea and almost had him bleed to death. The boy needed a new airway.
In the 2010 RSM lecture, Elliott argued why he could not use a homograft anymore: because the Human Tissue Act banned pickled tracheas, hence the “modern version” of Macchiarini. Weirdly, in yet another talk, on TED in 2015, Elliott says the human-donor-derived homograft was not an available option due to the bovine spongiform encephalopathy (BSE) scare. Why these two things are related, only Elliott might know. Fact remains, Elliot’s homograft worked very well in Ciaran, until it was eroded by the metal stent. In the RSM lecture, Elliott narrates how he established the technology together with his German colleague Claus Herberhold, and that the homografts even got eventually covered with ciliated epithelium. Which is exactly what he and Birchall later on declared to be the most significant and exclusive feature of Ciaran’s transplant, due to the fact that it was regenerated with stem cells. Also, as Elliott admits in his TED talk, the hope that the stem-cell-made graft would grow led nowhere. Despite the regenerative medicine magic, there was no growth at all. Luckily, an adult donor organ was used to operate the 10-year old Ciaran.
Elliot also explains that the metal stent was the main source of danger for Ciaran, but it is difficult to follow this argument of his to justify the Macchiarini/Birchall intervention. Thing is, Ciaran never lived without a stent ever since. In fact, a stent was already inserted into his new “regenerated” airway when it was transplanted, as Elliott tells in his TED talk. Afterwards, there was more stenting as complications and emergencies occurred. In the RSM lecture held only around 4 months after Ciaran’s transplant operation, Elliott admits that the boy’s bronchi collapsed and needed to have permanent stents inserted into them, that there was a massive amount of DNA from necrotic cells clogging his airways, that the boy had recurrent respiratory problems and “all possible infections” and finally, that his graft became “soft”. Which means the graft collapsed and had to be held open by a stent. Elliott also narrates that instead of the old dangerous metal stents they were able to use novel plastic ones, which are specifically custom-made to fit. Was it then really the stem cell magic of Macchiarini and Birchall which saved Ciaran’s life, or the boring modern stent technology which helped to keep his airways open? What was exactly the point of all that regenerative medicine circus, given the evidence?

Only one survivor
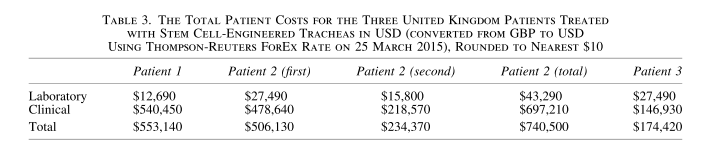
The answer is simple. Ciaran’s survival, which was obviously possible due to the modern custom-made stent technology, which kept his new homograft-like organ open, worked as a ticket to astounding amounts of clinical research funding and industrial engagements for Birchall. There is some circumstantial evidence that Birchall and his UCL colleagues were involved in 2010 in the approving of the cadaveric trachea graft which Macchiarini implanted into the 19-year old British patient Keziah Shorten in Italy. The graft collapsed upon Keziah’s return to UK, it was removed and replaced by Birchall with a plastic trachea at UCL in 2011, which predictably failed to save her live (see my article here). In fact, Birchall and Elliott published a certain paper Culme-Seymour et al 2015 describing the costs of the transplants of Ciaran, Keziah and a juvenile third patient (more about her below). There is this quote about Keziah, aka patient 2:
“For patient 2, the costs are calculated from the graft and 15 months of follow-up care only. The clinical costs relating to the surgical implantation of the first trachea for patient 2 were not costed as hospital admission costs from Italy were unobtainable, hence the total clinical costing for patient 2 is likely to be an underestimate”.
The tables 1 and 3 in this paper describe exactly how much Keziah’s the preparation of first transplant organ cost for its decellurisation and recellurisation. One wonders: how do Birchall and Elliott actually know this, if the trachea was officially made in Italy, especially since Macchiarini was not into any cell-seeding in bioreactors at that time? Is it conceivable that Keziah’s trachea was made in London, like those of the other two patients described in that paper? Made in Birchall’s lab, and then only transported for transplantation to Careggi in Florence? Does this explain what Birchall’s UCL colleague and Keziah’s caretaking physician Paul O’Flynn was doing at Careggi as evidenced by this photograph? UCL Hospital announced to continue withholding the information about that visit and about a related multidisciplinary conference at UCL featuring Macchiarini (which I have reasons to suspect took place), because they deem my request vexatious.

Birchall and Elliott proceeded to parade Ciaran’s alleged miracle cure around to operate at GOSH in February 2012 another patient, the 15-year old Shauna Davison, who was born with one lung and was suffering from various incurable airway and cardiac defects. She also had a tracheostomy. Shauna is patient 3 in Culme-Seymour et al 2015, where the authors conclude about her (and Keziah):
“Patients 2 and 3 had significant comorbidities at the outset, and while both unfortunately died, the causes appeared to be unrelated to the grafts. Before death, both patients had a high quality of life, although for short periods of time, and both would almost certainly have died much earlier if untreated”
and specifically about Shauna:
“”She had immediate resolution of airway problems and was discharged well at two weeks. However, at one month after surgery, she suffered an acute deterioration at her base hospital, and died. A post mortem was not performed. “
It rather indisputable that Keziah died by her plastic trachea, and the failed cadaveric one which left her without an airway and no chance of survival. And as for the cause of death of Shauna, maybe Elliott and Birchall should have read her obituary:
“But in a tragic twist of fate, Shauna’s breathing tube became blocked, depriving her of oxygen and causing a heart attack on March 2. Shauna stayed on life support for four days”.
Revealing BBC documentary
When Shauna was operated, she was not dying. She certainly had more than a couple of weeks to live. This we know from a “Great Ormond Street” series shown on BBC2. One episode features Elliott and Shauna’s operation. The video is not available to public anymore, but I received a copy (which I hesitate to offer here due to copyright issues). There is however a description on RadioTimes, and the show’s transcript, available from CIRCARE). After watching the programme, I understand why it had to be removed. Elliott’s decision to operate Shauna with a cadaveric trachea transplant seems rather questionable, and even more so his evidence to the ethics committee, shown in the film.
Shauna was examined by Colin Wallis, Consultant in Respiratory Paediatrics at GOSH. He spoke to Shauna’s mother and explained in the BBC2 film:
“What’s quite interesting is that mother does perceive the current quality of life as being good. In other words, this is not a situation where we’ve got nothing to lose by going ahead – they’ve got quite a bit to lose and this is going to make for a difficult decision. She’s got a reasonable quality of life, she’s got her good friend, she clearly has a sense of humour and enjoying herself – it makes it harder”.
Wallis assessed that the main danger to Shauna’s life was the stent in her airway, which was eroding her trachea and building scar tissue, which left only a very narrow space for the air to pass though. In this regard it may seem somewhat counterintuitive why Elliott and Birchall decided to remove Shauna’s stented trachea and insert a cadaveric one. They explain it with the previous success with Ciaran. When Elliott spoke to the ethics committee discussing the need for Shauna’s operation, he only forgot to mention that Ciaran never lived without a stent even after his miracle cure. The Lancet paper was not yet published, so there was only Elliott’s word. Elliott also totally forgot to mention the other 7 patients that he was well aware of, according to the RadioTimes article:
“It’s a procedure that has been performed only eight times previously in the world, with mixed results. And so, after extensive deliberation with his team, and with her family, Elliott took the decision that it was worth trying”.
The ethics committee asked Elliott about the rationale to operate Shauna:
“But she’s going to die anyway, even if you do the procedure – what would be the nature of her death if you’ve done the procedure?”
Elliott replied:
“Well, I think that’s saying more than I’m prepared to say. I don’t think she will die if we do the procedure. We’re trying to do the procedure so that she doesn’t. Or at least we prolong her life and her quality of life for as long as possible”.
So Elliott announced to save Shauna from certain death, using a cadaveric trachea made by Birchall. She died just a couple of weeks after the operation. This is also why nobody speaks of Shauna or other dead patients anymore, only of Ciaran.

Onwards
How is Ciaran doing now? We don’t have any objective unbiased information, but this is what Birchall and Elliot concluded in their 4-year follow-up paper Hamilton et al 2015:
“The use of a tissue-engineered tracheal transplant is currently a potential treatment of last resort. In the reported case, all conventional therapies had failed and the technique was used on compassionate grounds in an urgent setting”.
Martin Elliott recently became a champion of clinical research integrity, while promoting Ciaran’s trachea transplant technology as success. The preparations for Birchall’s two clinical trials for patients with tracheal stenosis or malacia (softening of tracheal cartilage) are already under way. Last time I checked, no one was intending to tell those prospective patients about the existence of their predecessors, never mind the collapse of their tracheal grafts and their deaths.
Update 10.05.2017. A reply to my Freedom of Information inquiry from the Royal Free Hospital suggests that Ciaran’s trachea was prepared not in Italy by Macchiarini, but in this London hospital’s lab, led by Mark Lowdell (who also later made Shauna’s trachea). Here the reply from Royal Free regarding the 3 transplants described in Culme-Seymour paper:
“The “Centre for Cell, Gene & Tissue Therapeutics” (CCGTT) was previously “The Paul O’Gorman Laboratory for Cell Therapy” (LCT) and it was as the LCT that it produced/prepared the grafts to which the FOI relates. The LCT and now the CCGTT are wholly owned and operated by the Royal Free London NHS Foundation Trust. Professor Birchall to whom the FOI refers is not employed by Royal Free London NHS FT. Neither the LCT nor the CCGTT has ever been directed by Professor Martin Birchall.
As described in the manuscript to which this POI refers, the first graft [Ciaran Lynch,- LS] was a non-substantially modified tissue and was prepared and released by LCT as a product regulated by the Human Tissue (Quality and Safety for Human Application) Regulations 2007. The product was supplied to Great Ormond Street Hospital as described in the manuscript. It was released on 15th March 2010.
The second graft [Keziah Shorten’s second graft, the UCL-made plastic trachea, -LS] was manufactured to GMP compliance as an ATMP and supplied to UCLH as described in the manuscript. The product was released on 29th September 2011.
The third graft [Shauna Davison, -LS] was also manufactured to GMP compliance as an ATMP and supplied to GOS as described in the manuscript. The product was released on 15th February 2012.
Professor Martin Birchall was not involved in the manufacture or supply of any of these products and none of them were supplied to him as the prescribing clinician or responsible surgeon”.

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00

Would Elliot, Birchall & Manchiarra use their techniques on their own children?
LikeLike
In UK if someone has concerns regarding adult and children safeguarding can contact http://www.cqc.org.uk/content/safeguarding-people
LikeLiked by 1 person
This is a truly horrible story. In essensen this has to be the biggest research skandal in modern times.
LikeLike
I read in latest Nature that China will punish researchers that commit misconduct in clinical research with several years in prison, and even death penalty in cases where patients die. Although I am not supporting death penalty, I think increasing the responsibility for the researchers and define this malignant practise a criminal act is the way to go.
LikeLike
This might be of interest:
Keith Baggerly and C.K. Gunsalus, “Penalty Too Light”, “The Cancer Letter”.
LikeLike
Pingback: Boletim de Notícias, 22/abr: Marcha pela Ciência no Brasil tem adesões em 25 cidades | Direto da Ciência
Pingback: Keziah’s deadly trachea transplant: UCL hospital’s secret – For Better Science
Pingback: Macchiarini’s trachea transplant patients: the full list – For Better Science
Pingback: Unpublished Macchiarini manuscript confirms 5 forgotten trachea transplant patients, Jungebluth’s surgery practice in Italy – For Better Science
Pingback: Alexander Seifalian, UCL’s Persian Scapegoat – For Better Science
Pingback: Martin Birchall’s shaky road to mass trachea transplanting – For Better Science
Pingback: UCL fibs Parliament about trachea transplants – For Better Science
Pingback: Trachea transplanters: Round 2 at UK Parliament – For Better Science
The description of Ciaran’s post-op difficulties with his magic trachea in the Lancet is inconsistent with an earlier publication.
Lancet 2012: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4487824/
“6 weeks after surgery the stent had dissolved and there was mild collapse of the proximal graft. A shorter (10×45 mm) PDO stent was implanted under fluoroscopy. The patient underwent bronchoscopy or balloon dilatation under fluoroscopy, or both, regularly for 6 months (figure 2B). The major reason for further bronchoscopy or balloon dilation was mucus retention and crusting within the native bronchi in which there were still embedded metal stents. At 5 months, after dissolution of the latest stent, we remained concerned about the rigidity of the proximal graft, and so overlapping, self-expanding Nitinol stents (S.M.A.R.T. Control, Cordis, Waterloo, Belgium) were implanted into the trachea. At 6 months after the initial surgery the graft seemed stable, the patient’s airway was patent, and he returned to school.”
In 2011 Vondrys & Elliott et al. published a report on their first experiences using biodegradable (PDO) stents in children. Ciaran is patient 4. (pp. 1873-74)
https://www.ncbi.nlm.nih.gov/pubmed/22051281
“Patient 4
This patient, with an original neonatal diagnosis of long segment tracheal stenosis treated by pericardial patch tracheoplasty, followed a year later by a tracheal homograft transplant and subsequent stenting presented after a further 10 years with hemoptysis, caused by erosion of a balloon-expandable metal stent into his aorta. This was treated by stem cell–supported transplantation of the trachea and repair of the aorta. A polydioxanone stent was used during this operation to support the donor graft. The stent progressively lost its color and strength, and dissolved completely at 17 weeks. There was severe progressive stenosis of the neotrachea, which did not respond to repeated balloon dilation. After tra-cheal predilation to 12 mm, a 12 mm x 65 mm polydioxanone stent was deployed in the neotrachea, but only expanded to about 7 mm, and was therefore too long.
Bronchoscopy showed that the upper end of the stent was in contact with the vocal cords, and it was removed. A 10 mm x 60 mm polydioxanone stent was deployed 1 week later, followed by a fourth polydioxanone stent after a further 6 weeks. Despite this, there was progressive stenosis, and 3 months later, a self-expanding nitinol stent was deployed in the distal trachea, with a dramatic improvement in symptoms.”
There is no indication in the Lancet paper of “severe progressive stenosis of the neotrachea”, nor subsequent “progressive stenosis”. “Mild collapse of the proximal graft” is quite different from severe progressive stenosis.
In light of the earlier paper, this statement in Lancet 2012 appears to be inaccurate:
“The major reason for further bronchoscopy or balloon dilation was mucus retention and crusting within the native bronchi in which there were still embedded metal stents.”
Vondrys & Elliott et al. do not claim these patients were treated under compassionate use but rather (p. 1870) the authors state “This study was approved by our Institutional Research Ethics Committee. Informed consent was obtained from the parents of each child.”
There is no entry for REC approval of this study in the UK Health Research Authority’s research summaries database. Why?
https://www.hra.nhs.uk/planning-and-improving-research/application-summaries/research-summaries/
LikeLike
Pingback: UCL’s decellurised tracheas: strong and stable? – For Better Science
Pingback: Los secretos de Macchiarini en Barcelona – For Better Science
Pingback: Birchall’s two dead pigs to prove trachea transplants are safe – For Better Science
Pingback: Trachea transplanters without borders – For Better Science
Pingback: UCL trachea transplant inquiry: scapegoating, obfuscation and a lost nose – For Better Science
Pingback: How Macchiarini was recruited to Karolinska – For Better Science
Pingback: Megagrant, the Russian docu-novel of Paolo Macchiarini – For Better Science
Pingback: Paolo De Coppi and the UCL organ factory – For Better Science