The Science and Technology Committee of the British House of Commons is now dealing with the trachea transplants performed by the scandal surgeon Paolo Macchiarini and his former parter at UCL, Martin Birchall as part of its inquiry into Research Integrity. Two UK scientists from Liverpool initiated this with their written submission from November 21st 2017 which I previously re-published: Patricia Murray, professor in stem cell biology and regenerative medicine, previously a nurse on a Head and Neck unit, and Raphael Lévy, senior lecturer in nanotechnology. Their concerns were not just the past trachea transplants, but also the present clinical trials with bioengineered trachea and larynx which UCL is most keen to start with, following the recommendation of an investigative commission from last year. UCL’s problem is however, that the two phase 1 UK trials are suspended by the UK authorities, and the big phase 2 EU-sponsored trial cannot begin recruiting patients without the results from phase 1.
By now it looks like UCL and their laryngology professor Birchall are in a pickle. Their own reply to the letter by Murray and Levy was not really honest when describing past UCL trachea transplant patients to the Parliament committee. On top of it, it turned out Birchall was reusing data from same old experiments on 16 pigs across different publications which were meant to show separate studies. This, and many more inconsistencies of UCL’s reply to the House of Commons are addressed in the re-published new letter by Murray and Levy from January 30th 2018 below.It is not like Birchall had no proper data to show: as the Animals in Science Regulation Unit at the Home Office of UK government declared, Birchall did use up quite a number of pigs:
“The licence was granted on 23 May 2011 and over the duration of the licence, 76 pigs were used in scientific procedures”.
Whatever studies the missing 60 pigs were used for, their real results remain unpublished, as evident from the reuse of old data. Maybe the transplants didn’t perform too well in those animals, hence the desperate need for human test subjects which UCL keeps demanding. UCL’s line of argument is:
- The transplant trials were approved by Medical Health products Regulatory Agency (MHRA) and are hence safe and reliable. However, Birchall declared to act as “Technology Advisor to MHRA”.
- The proof-of-principle for trachea grafts was sufficiently established by Birchall’s published human experiments with Macchiarini (especially cases of Claudia Castillo [patient A below] and Ciaran Lynch [patient D below]). Problem is those studies are not really entirely honest, while the cases of other patients Keziah Shorten (Patient C below] and Shauna Davison [Patient E below] is where Birchall and UCL simply avoid telling the truth at all costs. And as for preclinical studies with pig larynges: that image reuse is for the ongoing research integrity investigation at UCL to figure out.
- A 2017 investigation found no fault with Birchall and cleared the way for the clinical trials. However, the investigators never screened any evidence, and simply listened to Birchall and his colleagues explain what really happened. As the head of the UCL inquiry, Edinburgh professor of transplantation surgery Stephen Wigmore, admitted in an email:
“The Inquiry took the view that it was beyond our scope to re–explore the individual decisions made by official regulatory authorities such as the MHRA. For this reason we have mentioned the research but not investigated the data in detail”.
One of Wigmore’s team members was his Edinburgh colleague Marc Turner, professor of cellular therapy. Turner is also “Non-Executive Director” of Cell and Gene Therapy Catapult UK, a commercial entity which is key partner on Birchall’s trachea transplant clinical trials and as such has a huge financial COI to get those transplants finally rolling (Catapult’s own submission to the Parliament here). Yet Wigmore saw Turner’s COI actually as a qualification, while praising the organ recellurisation technology as scientific success story in an email:
“Professor Marc Turner who was a member of the Inquiry team with particular expertise in regulation was less concerned about the competency of the MHRA assessment teams and he has very good knowledge and oversight of this process with his involvement with Catapult. We did of course discuss the patient safety issues of both of these trials and were reassured by a number of factors including the regulatory pathway and documentation. With Regenvox the nature of the intervention is such that patient harm is much less likely than with tracheal intervention studies. […] One of themes that ran through the whole Inquiry but which may boo be evident in the report was the huge academic argument over the approach of recellularisilng cadaveric scaffolds. We took the view the the only way to ever resolve some of the arguments was to conduct properly regulated trials with open publication of findings.
No wonder that Murray and Levy remain unconvinced and distrustful. Here is their reply to UCL’s submission, with added illustrations by me.

Birchall, discussing the trachea transplant operation on Shauna Davison. Still from the BBC documentary “Great Ormond Street Series 2, ‘Experimental Surgery”
Supplementary written evidence submitted by Patricia Murray and Raphael Lévy (RES0045)
Further to UCL’s response to our previous written submission (RES0022), we are pleased to provide the following supplementary evidence and further recommendations.
1. In their response, UCL states:
“The Special Inquiry considered the RegenVOX and INSPIRE trials and reported that the appropriate ethics approval and Clinical Trial Authorisation from the MHRA were found to be in place for both trials”.
One of the problems with UCL’s Special Inquiry is that it did not investigate whether correct information was provided to the Research Ethics Committee (REC) and MHRA in order to obtain the necessary approvals. Since our submission, we have discovered that MHRA appear to have been provided with incorrect information regarding the compassionate use cases and the supporting preclinical pig data to secure approval for the RegenVox trial. We have provided MHRA with a full list of the relevant issues but some of the key points are as follows:
(i) MHRA was given misleading information about Patient E’s outcome, the RegenVox application indicating: “A second child [Patient E in UCL inquiry] underwent a successful implant in 2011, but died of non-graft related causes soon afterwards. Her graft was healthy and vascular at time of death…” [1].
In contrast to the above, The UCL Special Inquiry report [2], along with a more recent case study published by the group in 2017 [3], suggest that the death of Patient E was in fact related to her graft because she “..developed ventilatory compromise [was unable to breathe]. During bronchoscopic evaluation, there had been progression of the tracheal graft narrowing.” [3] It can therefore be seen that the team were aware that the graft had collapsed, an event that would have been directly related to Patient E’s death as it would have caused airway obstruction leading to respiratory failure.
(ii) MHRA was not informed of the outcomes of previous recipients of decellularized airways, including the unfavourable outcome of Patient C [1].
(iii) MHRA appears to have been given an overly positive account of the outcomes of the preclinical pig study. Bronchoscopy images in a very recent publication (Herrmann et al, 2017, Fig. 3B) [4] by the UCL team are identical to those presented in Fig 7c (A.B) of the MRC RegenVox proposal [1], indicating that the data are from the same animals.
Yet, the outcomes are reported very differently.
MRC RegenVox proposal[1]: “Decellularised scaffolds showed…clear evidence of remodelled cartilage” and from the legend to 7e: “Complete tissue integration is seen”
Recently published Herrmann study [4]: “The de‐cellularised component of the scaffold was not seen in continuity with the host thyroid cartilage”; “The cartilage component of the de‐cellularised scaffold remained acellular…”; “There was no evidence of regeneration of the de‐cellularised cartilage” [5]
2. UCL states: “The Special Inquiry was satisfied that the review process for compassionate use cases in Great Ormond Street Hospital is appropriate.” and “it found no reason to hold any further separate inquiries into the historic compassionate cases”.
The Special Inquiry should have explored whether the Ethics Committees at Great Ormond Street Hospital (GOSH) and University College Hospital (UCLH) were provided with correct information regarding outcomes in animal models and other patients. For instance, concerning Patient E, whose operation was covered in a BBC documentary [5], it appears that the Ethics Committee were not informed of the unfavourable outcome of Patient C and other recipients of bioengineered tracheas, and were only told about Patient D.

3. UCL mentions that regenerative medicine has the “potential to transform the face of healthcare.”
We wholeheartedly agree with this view, but transplanting decellularized or plastic tracheas into patients is not regenerative medicine. A UCL Impact Case Study from 2014 states:
“We were the first to show that human stem cells could be used to create functional organ replacements in patients. These transplants, first performed to save the life of an adult [Patient A] in 2008, and then repeated to save a child [Patient D] in 2010, have changed the way the world views stem cell therapies.” [6]
There is no evidence to support the claim that stem cells can create replacement tracheas. To present the case studies of Patient A and Patient D in this way is simply misleading.Science hype undermines public confidence and can be demotivating for the majority of researchers who are working conscientiously.
4. UCL states:
“The assumption [of our submission RES0022] appears to be that all the unfortunate characteristics of the Swedish case must apply to all those in the wider airway regenerative medicine field. We do not share this assumption.”
This is not correct. In section 8 of our submission (RES0022) we highlighted examples of specific shortcomings identified in the Karolinska University Hospital (KUH) report that also apply to the transplants performed on the UK patients. We note that UCL has not commented on these specific shortcomings, one example being the administration of untested growth factors to Patient D (see 8.iii of submission RES0022). It should be noted that the KUH investigation considered the use of these untested growth factors to be of sufficient gravity to warrant referral to the Swedish criminal investigation into Macchiarini’s tracheal transplant operations.
5. UCL states:
“…we have some concern that this particular written evidence submission RES0022 is emotive and may not be entirely factually accurate..”
UCL has not indicated any specific examples of factually inaccurate information in our submission. All the evidence in our report (RES0022) is referenced and can be easily verified.
6. UCL states:
“Everything [regarding the compassionate use cases] is in the public domain, including no less than five peer-reviewed papers, covering every aspect of their cases”.
As highlighted in section 8.viii of our submission (RES0022), some of the UCL group’s peer-reviewed papers contain inaccurate reporting of patient outcomes [7,8], while others have misrepresented the role of stem cells [9,10].
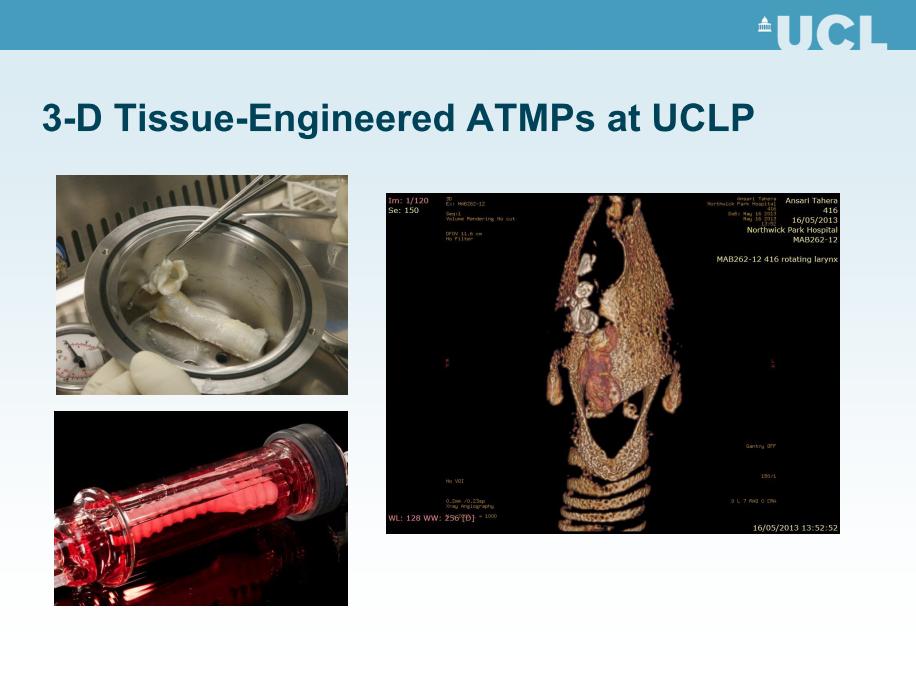
7. UCL indicates that all the compassionate use cases were seriously ill. Patient A was “about to lose a lung”; the Patients C, D, E and F were “about to lose their lives”; “There was a priori evidence that the therapeutic approach adopted in each case might work…”
We agree that these patients were seriously ill, but from what is known about Patients C and E, all evidence suggests that they were not about to lose their lives. According to the Special Inquiry, Patient C had a “slow growing cancer of the trachea“ [2]. Therefore, prior to receiving her decellularized cadaveric trachea, she was in a similar situation to Patient B, who was operated on in Sweden. The KUH investigation concluded that Patient B should not have been implanted with an unproven plastic trachea under ‘compassionate use’ rules because he was not in imminent danger of death. The same reasoning should have been applied to Patient C. As we indicate in our submission (RES0022, section 8.v), Patient E had a good quality of life prior to her transplant, and was not about to lose her life.

UCL also states that Patient A was about to lose her lung. It is noted that this claim is different from what appeared in UCL’s 2014 Impact Case study, which stated that Patient A was about to lose her life (see point 3 above). As revealed by the Special Inquiry, the lack of clarity regarding Patient A’s health status is likely due to the fact that Prof Martin Birchall was not involved in the clinical care of this patient so would not have been in a position to know:
“Professor Birchall asserted during the Inquiry interview that he was not involved in either the surgery or the clinical decision-making regarding the appropriateness or otherwise of the clinical approach, which was determined by Dr Macchiarini and the local team of thoracic surgeons in Barcelona.” [2]
An excerpt from a newspaper interview with Patient A that has been highlighted on Pubpeer [9] suggests Patient A’s health status was not so severe as to warrant treatment with an untested intervention under ‘compassionate use’ guidelines:
“sometimes she visited the Latin dance clubs in Barcelona that she loved, spending the evening doing salsa or merengue.” [9]
Regarding the “a priori evidence”, we note that UCL has not indicated what this was, nor provided any references.
8. UCL states:
“When new technologies are being developed, it is normal for there to be a developmental period during which the clinical and scientific communities learn the best ways to apply the new treatment and learn about the potential safety and efficacy.”
Safety and efficacy should be established in an appropriate animal model before being tested on patients.
9. UCL states:
“It is rare for everything to work perfectly first time, and the introduction of tissue engineered treatments is no exception (see the histories of transplantation and gene therapy for example).”
The use of ‘tissue engineered’ airways on human beings should not be compared with the history of transplantation. The first human heart transplant followed extensive preclinical work using dogs. This preclinical work established the proof of concept for heart transplantation and showed good evidence of safety and efficacy. Likewise, bone marrow transplantation followed extensive experimental work in mice and dogs. With tissue-engineered airways, on the other hand, there are no convincing safety and efficacy data from animal experiments.
10. UCL states:
“However, we note again that three of five patients, including two children, are alive and well today and another was effectively palliated”
Patient A’s graft failed so it is no thanks to her ‘bioengineered’ airway that she is alive and well today. Of the two children who are reported as being alive and well (Patients D and F), very little is known about Patient F who was transplanted in May 2017. Regarding Patient D, a question that needs to be asked is why he was not treated with a chemically fixed cadaveric trachea (‘Herberhold’ graft), which had served him well for seven years, rather than an untested decellularized trachea. The outcomes of patients transplanted with chemically-fixed tracheal grafts appear much better than those transplanted with decellularized ones (see references 11 and 12 and compare outcomes with those listed in reference 13). It is interesting to read the 4-year follow-up on Patient D [14], because Fig.6 in this follow-up paper shows histological analysis of the patient’s Herberhold and decellularized cadaveric grafts. Of note, the epithelial lining of the former appears superior to that of the latter, despite the fact that no attempt was made to recellularize the Herberhold graft using stem cells. This rather suggests that chemically-fixed tracheal scaffolds are likely to be safer and more effective than the decellularized ones.
It is misleading to claim that Patient C was effectively palliated; the only reason she required palliation with a plastic trachea was because a multidisciplinary team comprising surgeons from UCLH and the Careggi Hospital (Florence, Italy) [28] had decided to first treat her with a decellularized cadaveric trachea, the outcomes of which were disastrous [28].
11. UCL states that the Inspire and RegenVox trial are
“based on extensive research, including more than thirty years of published clinical and animal research evidence for INSPIRE, and additional extensive GLP (highest quality) preclinical (animal, pig) research in the case of RegenVOX”.
UCL has not referenced a single paper that shows safety and efficacy in an animal model in support of the Inspire and/or the Regenvox trial. Since submitting our written evidence (RES0022), it has been reported that the UCL group has presented the same preclinical pig data in the MRC RegenVox proposal and two different papers [4,15,16]. Not only are the data duplicated, but they are also meant to demonstrate different decellularization protocols (7 days in the grant proposal compared with 9+ days in the papers) and two very different surgical approaches. This issue is particularly worrying because the data in one of the studies (Ansari et al [15]), were meant to convince the MHRA to give approval for the RegenVox trial:
“It [the Ansari study] was designed after consultation with the U.K. Medicines and Healthcare Products Regulatory Agency to provide data suitable for submission for a Clinical Trials Authorization [the RegenVox trial].” [15],
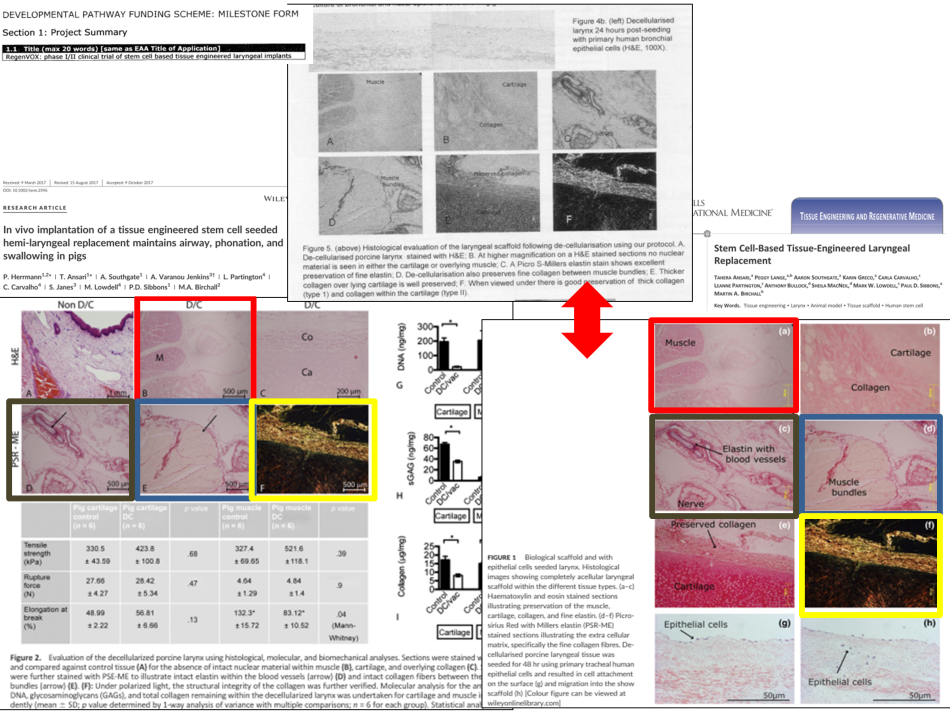
We have reported this concerning issue of apparent research misconduct to UCL, who are dealing with the matter under their guidelines on Research Integrity. A further issue with these publications [4,15] is that the Conflict of Interest statements do not mention that the senior author of these papers is a remunerated member of the Advisory Board of Videregen [17,18], a company that collaborates with UCL. The lead products of Videregen are ‘tissue engineered’ airways [17].
The only research paper cited by the Special Inquiry claiming that decellularized cadaveric tracheas are safe and effective in pigs is by Go et al, 2010 [19]. If one reads this paper, it is difficult to understand how it passed peer review, as there is insufficient evidence to support the authors’ claims. The serious problems with this paper have been highlighted on Pubpeer [19].
12. UCL states:
“The UCL Special Inquiry team was concerned about the accuracy of some of the evidence held by Dr Schneider and also the way that evidence was presented in the blog referenced by the written submission RES0022.”
UCL has not given any specific examples of inaccurate evidence. We found Dr Schneider’s ‘For Better Science’ website to be an invaluable source of official documents relating to bioengineered airways. Of significant value were the copies of the RegenVox proposal, which afforded us the opportunity to scrutinise the data presented to MHRA and MRC from the compassionate use cases (see section 9.i of our submission RES0022) and the preclinical pig studies (see point 10 above). We found problems with how these data were presented and have alerted MHRA and MRC accordingly. Given that the Special Inquiry decided not to scrutinise the RegenVox proposal, these concerning issues relating to the RegenVox supporting data would not have come to light had the proposal not been accessible through the ‘For Better Science’ website. We therefore feel that Dr Schneider provides a very important service to the scientific community and to the general public.

13. UCL states:
“In the UK there was a robust, large and extensively peer-reviewed evidence base for biologic scaffolds.”
UCL has not given a single reference to support this claim. The latest 2017 papers from the UCL group clearly show that the decellularized tracheal scaffolds are neither safe nor effective [3,4,15,20].
14. UCL states:
“UCL can confidently say that there appear to be no trials whatsoever in the UK presently, and arguably anywhere in the world, that have greater levels of approval, oversight, governance and patient protection than INSPIRE and RegenVOX do.”
As indicated in point 1, a key problem regarding the RegenVox trial is that inaccurate supporting data have been provided to the MHRA to obtain approval. The supporting data submitted for the Inspire trial should be scrutinised for inaccuracies.
15. UCL states:
“Denying people the chance of participating in trials with a considerable legitimate evidence and ethical base behind them not only takes away their hope, but also means that the world does not have a chance to see what the real potential is for tissue-engineered solutions to unmet clinical needs”
The key point is that these trials are not evidence-based. UCL has not provided a single reference to show that bioengineered airways are safe and effective in an animal model. Undertaking procedures on patients that have not been shown to be safe and effective in animals amounts to human experimentation, which is unethical.
16. UCL states:
“In accordance with the opinions of MHRA and EMA, the case for additional pig experiments is fallacious.”
The authors themselves indicate that the purpose of the Ansari study [15] was to demonstrate to the MHRA that the laryngeal scaffolds were safe and effective in pigs, in order to be granted approval to run the RegenVox trial. The study showed that the scaffolds were neither safe nor effective, so one would presume that the MHRA and EMA would require the approach to be optimised and re-tested in pigs before being trialled in patients. We therefore suggest that the case for additional pig experiments in not fallacious.
17. UCL states:
“UCL scientists have done as much as they can already to show safety and efficacy in animals.”
The UCL team has not published a single paper that shows convincing safety and efficacy data in an animal model. On the contrary, their 2017 papers [3,4,15,20] show very clearly that this approach is neither safe nor effective (see section 8.i of our submission RES0022). It is important to highlight that despite the unfavourable results, the abstracts and conclusions of some of UCL team’s papers have been written in very positive terms that are not consistent with the actual data [4,15]. Unfortunately, this practice is becoming more common, a recent scoping review revealing that
“abstracts are frequently inconsistent with full reports, and efforts are needed to improve the consistency of abstract reporting in the primary biomedical community” [21].
This is an important issue because reviewers and regulators might only read the abstracts of papers, and not scrutinise the actual data.
18. UCL states:
“The pig is not a model of human airway disease, not least because the structure of the pig trachea is substantially different to the human as it consists of contiguous cartilaginous rings and lacks the trachealis muscle sheet.”
This statement seems to contradict Prof Birchall’s previously published view on the pig model:
“In order to answer scientific questions about the larynx and to develop new operative techniques, large animal preclinical models are invaluable. Dogs, cats and primates have all provided important information. However, pigs are particularly helpful, since they have similar airways to man in terms of size, anatomy and functional characteristics.” [22]
Moreover, if the pig was not a good model, why were the group using it in the first place?

19. UCL states:
“It is unethical to deny a parent the chance of their child having a potentially-life-saving treatment, tissue-engineered or otherwise, when there is substantial pre-existing evidence of efficacy..”
UCL has not given any references for the substantial pre-existing evidence of efficacy in regard to ‘tissue-engineered’ airways. Moreover, other options are available for children with tracheobronchial stenosis [11,12,25,26] (and see point 22 below). We suggest that it is unethical to perform an unproven high-risk intervention on a child when more tried and tested alternatives are available.
20. UCL states:
“All of UCLs publications by Birchall et al in this area of research are fully peer-reviewed, in the public domain and as open to scrutiny and criticism as those of any other authors. All were scrutinised by the Special Inquiry and found to be robust…”
The Special Inquiry overlooked the fact that there were data duplications and misrepresentations in the MRC RegenVox proposal and two of the Birchall group’s 2017 papers [4,15], and that some of the earlier papers had inaccurate descriptions of patient outcomes [7,8].
21. UCL states:
“Patients are not treated as research subjects, but as patients.”
In an interview with two postgraduate students, Martin Birchall refers to the compassionate use cases as “one off compassionate experiments” [23]. This rather suggests that the patients were treated as research subjects. Moreover, the following statement on UCL’s website underneath the photographs of three patients who received decellularized tracheas under compassionate use, strongly suggests that these patients were used to address scientific questions:
“Our progress has been via life-saving breakthroughs in tissue engineering technology in one-off, compassionate patients. At each step, we have refined our methods and asked more focused scientific questions.” [24]
UCL’s 2014 Impact Case Study also suggests that the compassionate use cases were research subjects:
“It [the use of ‘tissue-engineered’ tracheas] further demonstrated the very high scientific importance of the compassionate use of new regenerative medicine technology” [6]
22. In response to Qq197-198: “Whether UCL sees a need to explore further the question of patient safety” UCL mentions that they undertook a Special Inquiry with a panel of five senior scientists and published their terms of reference in a transparent way. Unfortunately, the panel did not include an expert in airway surgery (i.e., a Head and Neck surgeon) and it appeared to rely on testimonies rather than scrutinising preclinical data and patients’ case notes. Did UCL or the Special Inquiry solicit the views of any experienced Head and Neck surgeons in regard to the use of tissue-engineered airways?
A recent review by four UK-based Head and Neck surgeons states the following:
“The intriguing yet unsolved surgical dilemma of tracheal replacement remains a challenge to clinicians. Currently, work from the Leuven group (Delaere et al) have shown promising results with the judicious use of allotransplants. Surgical ingenuity will lead to novel approaches to these problems. However, it is important to note that these techniques should not create more problems than they solve and patients are to be treated as an individual with a duty of care attached to that.” [25]
The authors go on to say:
“Tracheal replacement can be divided into prosthesis, homograft and autogenous tissue reconstruction…[…]…The most convincing evidence has historically been silicone based prostheses, and more recently revascularised tracheal homografts and allotransplants.” [25]
“Work on tracheal regeneration using stem-cell implanted scaffolds [cites 3 references, one of which is the 2008 Lancet paper describing the case of Patient A], which has been the centre of recent controversy, showed questionable data and ultimately poor results.” [25]
Of note, the senior author of this review, Mr Guri Sandhu, was a member of the surgical team who transplanted the plastic trachea into Patient C, so it is disappointing that his views do not feature in the Special Inquiry.
The prospective participants in the RegenVox and Inspire trials will be patients with laryngeal and tracheobronchial stenosis (narrowing), respectively. Presumably, those best-placed to judge the utility of ‘tissue-engineered’ airways for the treatment of these conditions are Head and Neck surgeons themselves. It is therefore worth mentioning a recent text book published in 2015 called ‘Laryngeal and Tracheobronchial Stenosis’ that is edited by Guri Sandhu and Reza Nouraei, with contributions from many other international experts in this field [26]. If the editors and contributors of this text book thought that ‘tissue-engineered’ airways were a viable option for treating laryngeal and tracheobronchial stenosis, one would surely have expected to see a chapter on this topic. However, there is no such chapter; instead, the final chapter on ‘Transplantation and Regeneration of the Trachea’ is written by Pierre Delaere, who suggests that at present, tissue-engineered airways should be regarded as “hypothetical and scientifically unfounded”. [27]
A key finding of the Swedish TV documentary, ‘Experimenten’, was that ‘tissue-engineered’ plastic airways had been implanted into human patients without safety and efficacy first being established in an animal model. Bo Risberg, a former chairman of the Swedish Ethics Council, described the failure to undertake adequate preclinical testing in animals as “the worst crime you can commit” [28]. The same could be said for ‘tissue-engineered’ cadaveric airways. Yet, despite the very poor outcomes in patients treated under compassionate use and the lack of safety and efficacy data from an appropriate animal model, UCL’s Special Inquiry has recommended that the RegenVox and Inspire trials should now proceed, and UCL seem very keen to follow this recommendation, although the MHRA have indicated that both trials are currently suspended (see Appendix).
We conclude with a question that was posed in the first part of the Swedish ‘Experimenten’ documentary by investigative journalist, Bosse Lindquist, in relation to ‘bioengineered’ tracheal transplants:
“How far can you risk a human life in the name of cutting-edge science? And are some of the world’s top medical institutions complicit……?” [28]
Final comment in relation to UCL’s written submission to the Research Integrity Inquiry (RIN0061).
Point 16 of UCL’s submission RIN00661 mentions that
“It is important that the sector nurtures such confidence in the management of integrity issues by responding appropriately to reports, having clear, robust and fair processes for investigating misconduct, following up on any actions required to correct behaviour…”
The first UCL investigation into research misconduct concerning tracheal transplants recommended that Prof Martin Birchall should avoid giving misleading information in his published reports [29], referring specifically to the false claim that stem cells helped regenerate Patient D’s trachea. This investigation took place in 2015, but Prof Birchall was still publishing misleading information in 2016 regarding the outcomes of the trachea transplant patients (Culme-Seymour et al [8]). Also, UCL’s own advertising office has ignored this advice because even today they have this statement on their website: “[Patient D’s] own stem cells were used to build up the donor windpipe and ensure the organ was not rejected” (see reference 35 in our submission RES0022).
Recommendations
In addition to the recommendations made in our previous submission (RES0022), we suggest the following:
- In light of the recent information from the MHRA indicating that the RegenVox and Inspire trials are currently suspended (see Annex), the sponsors and collaborators for these trials (i.e., MRC, UCL, Cell Therapy Catapult and Videregen) should immediately update the “recruitment status” on the ClinicalTrials.gov website to “suspended”. The ClinicalTrials.gov identifiers for RegenVox and Inspire are NCT01977911 and NCT02949414, respectively; these trials are currently listed as “recruiting” and “active, not recruiting”, the latter meaning that “the study is ongoing, and participants are receiving an intervention or being examined”.
- The sponsors and collaborators for the two trials should update their websites and promotional material to make it clear that the trials are suspended. This is particularly important for Videregen, as the information the company is currently providing on the Social Stock Exchange indicates that both trials have MHRA and Ethics approval and are about to start [17].
It is important that these two actions are undertaken immediately so as to reassure the public, and also to ensure that prospective investors in Videregen are given accurate information regarding the status of the two trials before making any investments.
- Our main objective has been to shed light on the specific problems surrounding ‘tissue engineered’ airways in an attempt to prevent more patients from being subjected to these unproven high-risk interventions. However, our final recommendation addresses the broader issue of research integrity. We strongly recommend that an assessment of the measures Universities take to ensure the highest standards of research integrity are implemented and maintained should be a key part of the next Research Excellence Framework. We feel that improvements will only happen if Universities are rewarded for upholding high standards, and financially penalised if they fail to do so. We suggest the following:
(i) Universities should ensure that when researchers submit a paper, all of the raw data underlying the data presented in the paper should be published as supplementary material or in a public repository. This would help to prevent data that is not truly representative from being cherry-picked for inclusion in published articles, would help to prevent data re-use, and would ensure that sufficient numbers of replicate experiments have been undertaken.
(ii) Universities should ensure that complaints of research misconduct (either from internal or external sources) are adequately dealt with. The current trend is for complaints to be ‘brushed under the carpet’. As we have seen from the first UCL investigation into ‘tissue-engineered’ tracheas, even when short-comings are identified by investigative panels and corrective measures are recommended, these are rarely followed up on. Universities need to be far more rigorous in investigating claims of research misconduct, and where evidence of wrongdoing is found, sanctions should be applied. Importantly, in any paper where data has been plagiarised, duplicated, massaged, fabricated or misrepresented; where patient outcomes have been incorrectly reported; or where appropriate ethics and regulatory approvals are lacking, Universities should have a duty to ensure that the scientific record is corrected (retraction or correction) in a transparent and timely manner. The REF should carefully monitor how Universities handle complaints of research misconduct, and check that appropriate actions have been taken.
January 2018
References
- Application forms for the RegenVox trial are available from the following article on the For Better Science website: ‘Business interests trump patient safety, says EU about trachea transplant trial’ https://forbetterscience.com/2016/11/18/business-interests-trump-patient-safety-in-eu-funded-trachea-transplant-trial/
- University College London website ‘Special Inquiry into Regenerative Medicine at UCL’ available on http://www.ucl.ac.uk/news/news-articles/0917/290917-regenerative-medicine. Link to pdf: http://www.ucl.ac.uk/news/news-articles/0917/Special_Inquiry_Final_Report_605109702_7_.pdf
- Elliott MJ et al (2017). Tracheal Replacement Therapy with a Stem Cell-Seeded Graft: Lessons from Compassionate Use Application of a GMP-Compliant Tissue-Engineered Medicine. Stem Cells Translational Medicine 6:1458–64
- Herrmann et al (2017). In vivo implantation of a tissue engineered stem cell seeded hemi‐laryngeal replacement maintains airway, phonation, and swallowing in pigs. Journal of Tissue Engineering and Regenerative Medicine DOI: 10.1002/term.2596
- From the website ‘Subsaga.com’, BBC Great Ormond Street Series 2, ‘Experimental Surgery’: http://subsaga.com/bbc/health/great-ormond-street/series-2/6-experimental-surgery.srt
- Submission of Impact Case Study by UCL to the Research Excellence Framework. ‘The world’s first stem cell based transplants: changing the future of organ replacement’: http://impact.ref.ac.uk/CaseStudies/CaseStudy.aspx?Id=36409
- Weiss DJ et al (2014). Tracheal bioengineering: the next steps. Proceeds of an International Society of Cell Therapy Pulmonary Cellular Therapy Signature Series Workshop, Paris, France, April 22, 2014. Cytotherapy 16: 1601-13
- Culme-Seymour EJ et al (2016). Cost of Stem Cell-Based Tissue-Engineered Airway Transplants in the United Kingdom: Case Series. Tissue Engineering: Part A Volume 22, Numbers 3 and 4; with comments on Pubpeer: https://pubpeer.com/publications/2EFBEF711E8EDE7C90951E94854578
- Macchiarini P et al (2008). Clinical transplantation of a tissue-engineered airway. The Lancet 372:2023-30; with comments on Pubpeer:https://pubpeer.com/publications/2968ADAFF1170575BBB3D0B74FD6CD
- Elliott MJ et al (2012). Stem-cell-based, tissue engineered tracheal replacement in a child: a 2-year follow-up study. The Lancet (2012) doi: 10.1016/s0140-6736(12)60737-5; with comments on Pubpeer:https://pubpeer.com/publications/F7B9D03E2EFC85F8F84D7F02B3C6B4
- Jacobs JP et al (1999). Tracheal Allograft Reconstruction: The Total North American and Worldwide Pediatric Experiences. Ann Thorac Surg 68:1043–52
- Propst EJ et al (2011). Pediatric Tracheal Reconstruction Using Cadaveric Homograft. Arch Otolaryngol Head Neck Surg 137:583-90
- The For Better Science website, ‘Macchiarini’s trachea transplant patients: the full list’: https://forbetterscience.com/2017/06/16/macchiarinis-trachea-transplant-patients-the-full-list/
- Hamilton NJ et al (2015). Tissue‐Engineered Tracheal Replacement in a Child: A 4‐Year Follow‐Up Study. Am J Transplant 15: 2750–2757
- Ansari et al (2017). Stem Cell-Based Tissue-Engineered Laryngeal Replacement. Stem Cells Translational Medicine 6:677-87; with comments on Pubpeer: https://pubpeer.com/publications/9E123595C9AEBBD512BBF1475796DA
- The For Better Science website, ‘Image reuse, the new low of trachea transplanter Martin Birchall?’:https://forbetterscience.com/2017/12/27/image-reuse-the-new-low-of-ucl-trachea-transplanter-martin-birchall/#more-22793
- Videregen’s Impact report on the Social Stock Exchange: http://socialstockexchange.com/wp-content/uploads/2017/06/Videregen-IR-May-2017-v5.pdf; overview of Videregen on Companies House: https://beta.companieshouse.gov.uk/company/07573593
- MRC Declarations of Interest for Prof Martin Birchall: https://www.mrc.ac.uk/documents/pdf/declaration-of-interests/martin-birchall-doi/
- Go T et al (2010). Both epithelial cells and mesenchymal stem cell-derived chondrocytes contribute to the survival of tissue-engineered airway transplants in pigs. J Thorac Cardiovasc Surg. 139(2):437-43; with comments on Pubpeer: https://pubpeer.com/publications/9F32CB65DF8D9983EAEF9D7BE2EEE0
- Maughan EF et al (2017). A Comparison of Tracheal Scaffold Strategies for Pediatric Transplantation in a Rabbit Model. Laryngoscope DOI: 10.1002/lary.26611
- Li G et al (2017). A scoping review of comparisons between abstracts and full reports in primary biomedical research BMC Medical Research Methodology 17:181
- Murison PJ et al (2009). Development of perioperative care for pigs undergoing laryngeal transplantation: a case series. Laboratory Animals 43: 338–343. DOI: 10.1258/la.2009.008101
- “From Bench to Bedside: Interview with Professor Martin Birchall” published on May 29th 2015, quote taken from ~08.34 https://www.youtube.com/watch?v=9cSNfCXP9Fw
- https://www.ucl.ac.uk/immunity-transplantation/diseases-and-treatments/transplantation/organ_regeneration
- Virk et al (2017). Prosthetic reconstruction of the trachea: A historical perspective. World J Clin Cases. 5: 128-33
- Laryngeal and Tracheobronchial Stenosis (2015). Editors Guri Sandhu and Reza Nouraei. ISBN: 978-1-59756-550-9 https://www.pluralpublishing.com/publication_lts.htm
- Delaere P and Van Raemdonck D (2014). Tracheal replacement. J Thorac Dis 8:S186-S196
- BBC News story summarising the Swedish documentary: ‘Paolo Macchiarini: A surgeon’s downfall’ by William Kremer, BBC World Service: http://www.bbc.co.uk/news/magazine-37311038
- Storyville Part 1 ‘Fatal Experiments’: https://subsaga.com/bbc/films/factual/storyville/fatal-experiments-the-downfall-of-a-supersurgeon/part-1.html
- The 2015 UCL investigation report is available on the For Better Science website, ‘Macchiarini and the tracheal regeneration scandal, by Pierre Delaere’: https://forbetterscience.com/2016/04/02/macchiarini-and-the-tracheal-regeneration-scandal-by-pierre-delaere/
- The For Better Science website, ‘UCL fibs Parliament about trachea transplants’ https://forbetterscience.com/2018/01/08/ucl-fibs-parliament-about-trachea-transplants/
The evidence has been prepared by:
Patricia Murray
Professor in stem cell biology and regenerative medicine, previously a nurse on a Head and Neck unit, Institute of Translational Medicine, University of Liverpool
Raphael Lévy
Senior Lecturer in nanotechnology and imaging, Institute of Integrative Biology, University of Liverpool
It should be noted that the evidence has been prepared in a personal capacity by the authors and has not been submitted on behalf of the University of Liverpool.
We acknowledge science journalist, Dr Leonid Schneider (‘For Better Science’ website) and ‘Citizens for Responsible Care & Research’ for providing important advice and information.
Appendix: email correspondence from MHRA regarding status of RegenVox and Inspire trials
From: Clinical Trial Helpline
Sent: 19 December 2017 22:56
Dear Professor Murray,
Thank you for your email of 7 December and for the additional information you have provided.
I can clarify that the sponsors of these studies voluntarily halted all trial-related activity while the UCL special inquiry was ongoing. This action was supported by MHRA and the relevant ethics committees, and we have been in close dialogue with the sponsors to actively monitor the situation whilst the benefit:risk profile for these studies has been reviewed.
Should the sponsors of these trials wish to restart their respective studies, a substantial amendment to their clinical trial authorisation will need to be submitted for prior review and approval by MHRA and ethics. While we cannot pre-judge the outcome of such a request, the review will take account of all available information and MHRA will also seek independent expert advice from an expert advisory group of the Commission on Human Medicines.
I hope that the information above provides you with assurance that these trials currently pose no threat to public health with no patients having been treated to date, that no trial-related activities are ongoing, and that any restart of the trials will need prior MHRA and ethical approval, including input from independent experts. We will only allow these trials to re-start if all of the evidence currently available suggests that the potential benefits to the patients outweigh the risks.
Kind regards,
Clinical Trial Helpline

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00

One of themes that ran through the whole Inquiry but which may [not] be evident in the report was the huge academic argument over the approach of recellularisilng cadaveric scaffolds. We took the view the the only way to ever resolve some of the arguments was to conduct properly regulated trials
“There is intense debate on whether it is safe and ethical to subject human patients to these experimental and probably unworkable procedures. The best way of deciding is to try these procedures on human patients and see if they work.”
LikeLike
This is good for a debate about animal welfare vs human rights in clinical trials and alternatives to animal testing
LikeLike
Pingback: Boletim de Notícias: Metabolismo acelerado faz urso ser mais vulnerável ao clima | Direto da Ciência
Pingback: Oca novella 2000 - Ocasapiens - Blog - Repubblica.it
Pingback: EU trachea transplant clinical trial TETRA “uncertain to take place” – For Better Science
Pingback: Birchall’s two dead pigs to prove trachea transplants are safe – For Better Science
Pingback: Trachea transplanters without borders – For Better Science
Pingback: UCL trachea transplant inquiry: scapegoating, obfuscation and a lost nose – For Better Science
Pingback: Macchiarini victim Zhadyra Iglikova is dead – For Better Science
Pingback: UCL trachea transplants: Videregen sets lawyers on Liverpool academics Murray and Levy – For Better Science
Citation “Regarding Patient D, a question that needs to be asked is why he was not treated with a chemically fixed cadaveric trachea (‘Herberhold’ graft), which had served him well for seven years, rather than an untested decellularized trachea. The outcomes of patients transplanted with chemically-fixed tracheal grafts appear much better than those transplanted with decellularized ones”
There is no big difference in between ‘Herberhold’ graft and Macchiarini’s trachea. Both are decellularized allogeneic trachea. Herberhold applied older chemical approach for decellularization, Macchiarini used a newer one. As it is mentioned in Herberhold article (Tracheal allograft reconstruction: the total North American and worldwide pediatric experiences / J.P. Jacobs [et al.] // Ann Thorac Surgery. – 1999. – Vol. 68. – P. 1043-1052) “Histological studies confirm that all cells in the graft die and all major histocompatibility complex markers are lost”.
LikeLike
It is true that both types of graft are sourced from cadaveric donors and that both are reported to be non-immunogenic.
There is, however, an important difference between ‘Herberhold’ and decellularized cadaveric tracheas. The former is chemically fixed, while the latter is decellularized using enzymes and detergent. As explained by a member of UCL’s trachea transplant team, this process of decellularization damages the tracheal scaffold so that it is inclined to collapse following implantation:
“the decellularising is suitable only for small sections of trachea, rather than an entire organ, and risks damaging their underlying structure, leading to their collapse once they are implanted in the patient.”
Please see: http://impact.ref.ac.uk/CaseStudies/CaseStudy.aspx?Id=30007
This likely explains why the mortality rates in patients implanted with decellularized tracheas were much higher than those implanted with Herberhold grafts.
LikeLike
Pingback: Macchiarini’s trachea transplant patients: the full list – For Better Science
Pingback: Paolo De Coppi and the UCL organ factory – For Better Science
Pingback: New government cash for trachea transplanters Videregen – For Better Science
Pingback: Martin Birchall innocent, UCL decides once again – For Better Science
Pingback: Peter Wilmshurst vs Macchiarini cult at The Lancet – For Better Science
Pingback: Questionable activities of UK company Celixir, by Patricia Murray – For Better Science
Pingback: Celixir-critic Murray: “MHRA is prioritising business interests over patient safety” – For Better Science
Pingback: Macchiarini partner Anthony Hollander chairs mass-sacking committee in Liverpool – For Better Science
Pingback: After oesophagus, a thymus by trachea transplanters – For Better Science