A bit of Frye and Rossignol
“Trapped inside every autist, a normal child with normal cognition is struggling to get out – only needing the right drug or therapy to be released”, – Smut Clyde
By Leonid Schneider, on research integrity, biomedical ethics and academic publishing
“Trapped inside every autist, a normal child with normal cognition is struggling to get out – only needing the right drug or therapy to be released”, – Smut Clyde
“Patients with weak heart function who receive stem cell therapy shortly after a heart attack are at lower risk of developing heart failure and related hospital stays compared with standard care, finds a clinical trial published by The BMJ today.”
“If we can target necrosis, we could unlock entirely new ways to treat conditions ranging from kidney failure to cardiac disease, neurodegeneration, and even aging itself.” – Dr Carina Kern, a genius
Kathleen Seidel analysed two papers by Mark and David Geier, which claimed that mercury in vaccines caused autism
“The magnitude was much greater than we had expected or hoped for,” – Martin Gleave
“David Geier is the ideal fit to the purposes of RFK Jr. For the only reliably loyal underlings are incompetent ones who know they have no future anywhere else. ” – Smut Clyde
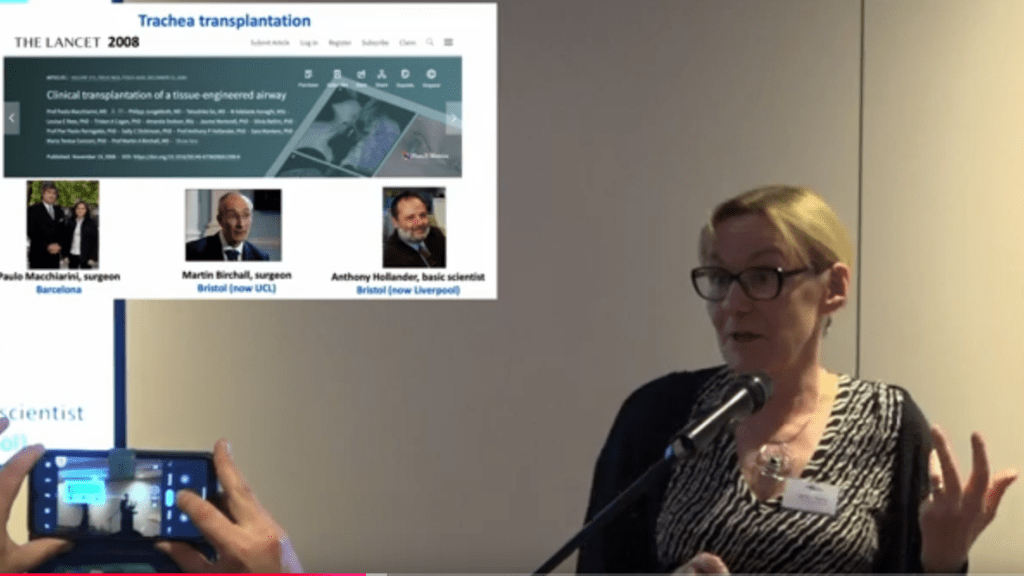
Several years ago, UCL investigated the Macchiarini affair and found their surgeon Martin Birchall was not only innocent, but also entitled to more money and more patients to experiment upon.
Now, Patricia Murray was awarded for exposing this UCL malfeasance, and Sholto David scrutinised UCL papers for data manipulation.
“Imagine what effect might ensue from a young donor body (say, in her 20’s) nourishing with her young blood 24/24, 7/7 the head of an aging body recipient!” – Sergio Canavero
“…request for preventive seizure made on 12.4.2024 by the Public Prosecutor in charge, concerning the article under indictment, still accessible on the website called forbetterscience.com, although it appears to have been removed from the blog.repubblica.it website (referred to in the indictment)…”
“The entire proposition is crazier than a barrel-full of rabid wolverines that have spent a week self-medicating with bath-salts and angel dust. Yet there is this burgeoning literature on mitochondrial transplants!” – Smut Clyde