The Kingdom of Rui Reis
“I think this story shows the debacle and corruption of entire research fields that become just a very expensive show of bad science-fiction. ” – Carabus Maleki
By Leonid Schneider, on research integrity, biomedical ethics and academic publishing
“I think this story shows the debacle and corruption of entire research fields that become just a very expensive show of bad science-fiction. ” – Carabus Maleki
Schneider Shorts 27.03.2026 – degenerative medicine special with London crooks and Californian lunatics, with retractions for mouse torture, concerns for one man’s cancer business, and a Texan Nobelist and his bullying wife.
“Here is the story about one of those institutions where nearly all senior staff members including professors, associate professors and at least 3 former Deans are exposed on PubPeer with extensive evidence of fraud in multiple papers.” – Dayo Maor
“Patients with weak heart function who receive stem cell therapy shortly after a heart attack are at lower risk of developing heart failure and related hospital stays compared with standard care, finds a clinical trial published by The BMJ today.”
How Robin Ali and other London ophthalmologists make blind mice and blind children see.
“If we can target necrosis, we could unlock entirely new ways to treat conditions ranging from kidney failure to cardiac disease, neurodegeneration, and even aging itself.” – Dr Carina Kern, a genius
Schneider Shorts 28.03.2025 – a sexual harassers special, a multimillionaire sponsors a sleuth-on-a-leash, COPE officer loses her only paper, how to troll Elsevier, with cheaters in Florida and breakthroughs in degenerative medicine in Japan and USA.
“Expressions of Concern may be used as an interim notice to flag a potential issue that may be ultimately resolved with another amendment outcome (e.g. retraction or correction) or they may remain as the final outcome in cases where conclusive evidence cannot be obtained. ” – COPE
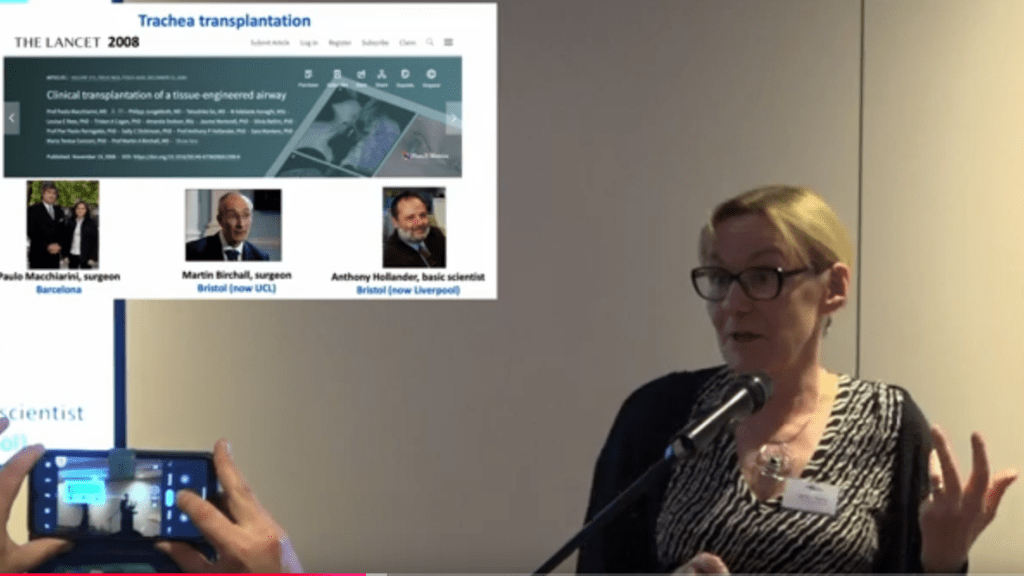
Several years ago, UCL investigated the Macchiarini affair and found their surgeon Martin Birchall was not only innocent, but also entitled to more money and more patients to experiment upon.
Now, Patricia Murray was awarded for exposing this UCL malfeasance, and Sholto David scrutinised UCL papers for data manipulation.
Schneider Shorts 15.11.2024 – trouble in elite Canadian lab, leadership chaos at IEEE, cancer charity money put to good use in Italy, one Elsevier journal untouchable, another cleaned from papermillers, with a Polish professor betrayed, NIH fraudster’s first retractions, and finally, Patricia Murray vs stem cell clinics!