The disgraced surgeon Paolo Macchiarini has now been officially sacked from Karolinska Institutet (KI) in Sweden, while also the entire Ethics Council was dismissed by the new KI Vice Chancellor in the process of creating a new supervisory body, with more extended authority.
European Union terminated in 2014 its FP7 funding to Macchiarini and his partners of the Biotrachea Consortium (yet as I show below, the EU’s follow-up programme Horizon2020 has awarded just now almost €7 Mio for a stage II clinical trial with regenerated trachea by the very same Macchiarini partners at UCL) . The original wording of the EU spokesperson in regard to BIotrachea termination referred to “difficulties related to important deliverables” and declared that “at no time did any EU funding for this project involve activities, tests or investigations with human subjects”. Yet the List of Milestones of Biotrachea contained points like:
- MS10 Report on the in vivo immune responses to tissue-engineered airway grafts in men.
- WT14: Outcome of first trial in man tracheal clinical trial
- WT15: Obtaining of clinical trials authorization for tracheal trial
- WT 16: Obtaining of clinical trials authorization for synthetic tracheal trial
After I confronted EU with this and other documents from Biotrachea suggesting an upcoming clinical trial on human patients, the EU spokesperson issued an update to their earlier statement (the entire EU message at the end):
“A chronological sequence of milestones (MS) and work packages (WP) were proposed under the Description of Action. These defined and described all of the technical activities that were to be carried out. The Commission terminated the project before the milestones and work packages referred to in your message were achieved or delivered”.
With a help of my website reader’s, I received also Financial Declarations submitted by KI to the EU (2012/2013 and 2013/2014), the travel expenses therein reveal some interesting information. One post from the 2nd period is particularly intriguing, described as “Philipp Jungebluth, Hannover 130912, meeting with Dr. Schreiber (lawyer) and Dr. Möller (med. doc and veterinarian) regarding legal issues with tissue engineering (human, animal studies)”. To find out what this was about, I contacted the Medical University Hannover (MHH) where, as I previously reported, Macchiarini is still listed as adjunct professor, and where his acolyte Jungebluth studied medicine. Stefan Zorn, head of MHH press office, explained to me that according to his investigation, “this has nothing to do with MHH but with a Hannover-based company MDSS”. The company provides “high quality service as a competent partner for regulatory affairs”, while its head is called Ludger Möller. Neither Möller nor MDSS replied to my inquiry if he was the same Dr. Möller whom Jungebluth has visited in 2013/2014 to discuss legal issues of human experiments.
Zorn also explained to me on the phone that their adjunct professor Macchiarini never was employed by or received a salary from MHH (and that as such, Macchiarini also cannot be sacked). The surgeon used to earn his income in Hannover by operating patients on the private basis. In fact, Macchiarini stands accused by Italian authorities of having attempted extortion from his Italian patients in Florence, where he requested €150,000 for them to be operated in Hannover. The related trial for fraud and attempted fraud was previously dismissed by the Italian courts, but has now been re-opened.
Another peculiar travelling entry from 2013/2014 was “Philipp Jungebluth, Gothenburg Schweden to pick up trachea”. Was there possibly any collaboration with the University of Gothenburg and its team of rather controversial trachea regenerators Sumitran-Holgersson and Olausson?
Graziella Pellegrini, professor for regenerative medicine at the University of Modena in Italy, was one of the key members of the Biotrachea Consortium. Pellegrini has been very helpful with my inquiry and described her lab’s contribution, namely that of the epithelial stem cells:
“We started primary tracheal epithelial cell cultures from biopsy, approximately 1,5 years into the Biotrachea project. After the approval for biopsy retrieval from Careggi Hospital, the project was transferred to Karolinska, a new approval was requested […] and finally I got it and I received biopsies: at that time I could start primary cultures but the project was stopped approximately 6 months later”.
The biopsy is documented in the Periodic Report Summary 2 submitted to EU by the Biotrachea Consortium. Since the EU has originally specifically agreed to fund Macchiarini’s synthetic trachea experimentation in humans, Pellegrini shared these insights:
“The project contained a plan for human treatments, at a stage of the project when all requirements would have been in place: material, epithelium, cartilage, full characterization, ethical approval, authorizations etc… but we did not reach that point. The project was good, despite the termination, the main technical problem was related to the biomaterial, which was not good at all, neither for trachea nor for epithelial cells”.
The weird thing is, Macchiarini always seeded bone marrow mesenchymal cells onto the biological and artificial trachea carcasses which he then transplanted into his patients. It is not clear what he offered EU to use for his Biotrachea funding, but it surely was not (or not only) bone marrow cells.
According to Pellegrini:
“ the exclusive use of bone marrow derived cells, for trachea transplants was not written in the project. In the proposal, there were included cells able to give cartilage, recruitment factors and epithelial cells from human airway”.
Retrospectively, Pellegrini has this to say about Macchiarini’s claim of trachea regeneration from bone marrow cells:
“The unproven but attractive (both for scientists and entrepreneurs) concept that mesenchymal cells can do everything, probably captivated Paolo Macchiarini also; and he proceeded in that direction. The non- scientific idea that a mesenchymal stem cell can become any cell type, without the need for certain extreme experimental conditions, produced much damage in many different fields of medicine: to patients (some are dead), science, society and its research money and serious scientists.
The artificial trachea material described in the Biotrachea proposal was not working with cells, and Macchiarini suggested a different material already known (so the novelty aspect for this nanomaterial funding call dropped). […] The widespread concept that mesenchymal cells can do everything probably drove Macchiarini to think that his transplants could work anyway”.
Pierre Delaere, professor for respiratory surgery at KU Leuven, Belgium, is one of the earliest and fiercest critics of Macchiarini’s . Yet he sees the potential of clinical stem cell research, even for trachea regeneration:
“In the last decade, miraculous tissue and organ regenerations have been described in humans based on the use of “stem cells”, which are purported to have supernatural therapeutic potential. Medical breakthroughs are and will always occur in small, consecutive steps. In regenerative medicine engineering of corneal implants may be this first step. Corneal epithelial implants can be nourished by diffusion and don’t need blood vessels. Engineering of skin grafts may be a next step. Skin grafts are much more complicated because not only the epithelial layer but also a part of the dermis, with its blood vessels, needs regeneration. Let’s start with these first steps. With our current knowledge, dreaming of whole organ regeneration should be seen as fantasy”.
Delaere has been trying to prevent clinical trachea regeneration trials on human patients whenever he saw those as unscientific and, in his own words, “fraudulent”. Delaere accused of misconduct Macchiarini as well as his close collaborators at UCL Martin Birchall, professor of Laryngology, and Alexander Seifalian, professor of Nanotechnology and Regenerative Medicine. Birchall performed together with Macchiarini tracheal transplant procedures on several patients in between 2008 and 2012, they published their reports subsequently in The Lancet.
Seifalian (his self-portrait at The Guardian here) specialises in the production of artificial tracheas from polyhedral oligomeric silsesquioxane (POSS), which Pellegrini and apparently also EU deemed just as unsuitable as the one produced by Harvard Apparatus together with its inventor Macchiarini. A TED Med talk video shows Seifalian creating an artificial trachea after Macchiarini’s request, apparently without ever having ever seen a trachea previously. Seifalian’s nanotechnology-made organ was then seeded with bone marrow cells and implanted by Macchiarini into a patient at KI in Sweden in 2011.
Some of the evidence Delaere submitted to UCL are available here, here and here.

The UCL internal investigative panel (which consisted of four Birchall’s faculty colleagues) dismissed all accusations and decreed that in case of Birchall and Seifalian “there was no prima facie evidence that any research misconduct had taken place”. Birchall was solely criticised for “a misleading element” in his Lancet paper (Elliot et al,2012), where Seifalian and Augustinus Bader were co-authors. UCL committee decreed:
“Professor Birchall should be urged to give greater consideration to the need for clearer and more representative presentation of information and evidence in his published reports in order to support his assertions”.
As Delaere pointed it out to UCL, while Birchall and his co-authors proclaimed in that Lancet publication “the success of the stem-cell-based tissue engineered tracheal implantation and regeneration procedure that had been employed, they had omitted to clearly identify and acknowledge the stent and surrounding vascularised omentum tissue wrap and the essential role and contribution of these elements”. Simply put, Birchall’s stem cell treatment was at the very best utterly irrelevant to the therapy, which otherwise relied on “traditional” procedures.
It seems that Birchall’s and Seifalian’s UCL colleagues were nevertheless quite convinced by his bone marrow-derived mesenchymal cell approach in tracheal regeneration and recommended:
“That the allegation of research misconduct made by the Complainant [Delaere, -LS] as it pertains to Professor Alexander Seifalian be regarded as having no substance and mistaken, and that no further action therefore be taken”.
I provide the June 2015 dated draft of the entire 9-page UCL report here.
Both Birchall and Seifalian were possibly Macchiarini’s UCL partners at Biotrachea. The loss of this funding cannot be that grave for Birchall though. Further well-funded clinical trials with stem-cell regenerated trachea and larynx are about to commence, under his leadership.
One is the MRC funded project RegenVOX, set to run from June 2013 till April 2018 and funded with almost GBP 3 Mio. There, Birchall is currently recruiting ten “clinical trial volunteers” for “autologous stem cell based, tissue engineered partial (non-moving, but anatomically matched) laryngeal replacement grafts”. According to Delaere, the phase I/II clinical trial RegenVOX uses “stem cell based tissue engineered laryngeal implants based on the same decellularization-recellularization approach” as the tracheal transplants.
And if anyone was thinking that EU has stopped funding human trials on trachea transplants, this is not entirely correct. As announced in December 2015, the EU research funding programme Horizon 2020 awarded €6.8 Million to the TETRA Consortium for the phase II clinical trachea replacement trial. The Consortium is led by Videregen, a company that was established based on the UCL expertise in tracheal transplants. The announcement goes:
“The Phase II trial, which will recruit patients from five clinical sites across Europe, is to assess the safety and efficacy of the replacement trachea product.[…]The project also involves scale-out of stem cell manufacture from the UCL/Royal Free Hospital London Centre for Cell, Gene and Tissue Therapeutics to establish a second GMP [Good Manufacturing Practice, -LS] manufacturing site in Munich, creating a pan-European supply chain. […]
The principal investigator is Professor Martin Birchall (UCL, UCL Hospital NHS Foundation Trust) with additional trial sites at the University of Manchester/University Hospital of South Manchester NHS Foundation Trust (UK), Università degli studi di Brescia (Italy), Medical University of Vienna (Austria) and Instytut Gruzlicy I Chorob Pluc (Poland). Other parties include the Cell Therapy Catapult (pan-European regulatory processes and lead clinical research activities), TMC Pharma (regulatory affairs and lead clinical research activities), NHS Blood and Transplant (tissue retrieval and trachea scaffold manufacture), Klinikum Rechts der Isar der Technischen Universität München (stem cell processing and final product release) and Euram (project management)”.
Update 30.03.2016: according to this press release, the second trachea-production facility in Munich will be the TUMCells centre, led by Martin Hildebrandt:”first patients should be able to participate in the trial in 2017″, with the goal to obtain European market permit in 2019 and to offer the commercial product in 2020.
As Delaere explained, Birchall will be “using the decellularized tracheal scaffold treated with bone marrow for tracheal and laryngeal defects”. This seems in fact the very stem cell technology Macchiarini has been using. Videregen describes their method as:
“Autologous mesenchymal stromal cells and airway epithelial cells are used to reseed the decellularised tracheal scaffold, which is then used to replace the diseased portion of the patients trachea”.
The only problem which may be standing in the way of the success of this EU-funded clinical phase II trial is possibly the actual biology of stem cells and the trachea. As Delaere wrote in his recent publication with his KU Leuven colleague Dirk Van Raemdonck:
“The engineered trachea is an example of blatant scientific deception. […]There is no scientific foundation whatsoever to assume why stem cells would support airway tissue regeneration in this setting. In addition, even if a trachea-like organ would be generated, it would irrefutably fail after implantation if adequate blood supply had not been restored. As expected, the implantation of de-cellularized and synthetic scaffolds resulted in extremely high morbidity and mortality rates (14). At this point in time, this form of airway regeneration should be regarded as hypothetical and scientifically unfounded (15,16)”.
Entire EU statement to me on Biotrachea termination:
“We would like to reaffirm that the Commission services scrutinised every aspect of the work on BIOTRACHEA until termination, and there has been no suggestion that EU funding was used for any purpose that has been harmful towards citizens. A chronological sequence of milestones (MS) and work packages (WP) were proposed under the Description of Action. These defined and described all of the technical activities that were to be carried out. The Commission terminated the project before the milestones and work packages referred to in your message were achieved or delivered.
The policy of the EU on research involving human beings is that all applicants’ research projects and proposals must comply with applicable international, EU and national law and ethical principles that govern the area of research that is the subject of their work, including on human beings. The Grant agreement for Biotrachia stipulated a separate ethical assessment through the establishment of an ethics committee leading to binding obligations that could later on be checked during ethics checks, reviews and audits. However, the project failed to establish the required ethics committee by the time stipulated in the milestones, which was one of the reasons for the early termination of the project.
During Period 1 (from month 1 to month 12), the consortium focussed on developing a number of platform technologies, and a human biopsy sample was obtained. This procedure did not involve the testing in humans of any new technology that the project aimed to create. This can therefore be seen as a normal part of the initial stage of a preclinical development process.
Regarding funding, we can only reiterate what we said in our reply to your original question:
“The procedure in the case of project termination is that the Commission requires reimbursement of all or part of the financial contribution made by its services, taking into account the nature and results of the work carried out and its usefulness in the context of the specific programme concerned. The legal basis for requiring reimbursement of all or part of the financial contribution made by its services in the event of termination is underlined in the FP7 Grant Agreement for the project (Article II.39 : “Financial contribution after termination and other termination consequences”, Annex II General Conditions). As part of the procedure, the consortium must prove that any non-recoverable funds constitute “eligible costs incurred and accepted up to the effective date of such termination and of any legitimate commitments taken prior to that date, which cannot be cancelled”.
The Commission is proceeding with this procedure of requiring reimbursement of all or part of the financial contribution made by its services. “

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00

sigh
number 11, UCL panel report, pp. 4-5, how many trachea transplants and response by Birchall
Birchall and (Seifalian) did not disclose or the committee did not record that the female trachea transplant at UCL in 2012 got Seifalian’s plastic trachea, it failed and she died. NO preclinical work supported Seifalian’s POSS/PCU trachea, the same one that Mr Beyene got at KI.
Birchall is on record in pt 1 of Experimenten
The Experiments Pt. 1 starts around 36 minutes, interview with Martin Birchall (in English): http://www.svt.se/dokument-inifran/se-program/dokument-inifran-experimenten-stjarnkirurgen
Birchall told the committee she died from esophageal complications and pneumonia. He told SVT the trachea loosened at both ends and failed. He told Science (G. Vogel Trachea transplants test the limits 2013, subscription: http://science.sciencemag.org/content/340/6130/266.full.pdf+html ) that she died of cancer. Birchall failed to disclose that she became his patient when the decellularized stem cell trachea transplant performed by Macchiarini at Careggi “broke down”. Birchall wrote in his Research Impact statement for 2014 that the transplant was palliative: http://www.circare.org/info/pm/ucl-impact-2014.pdf
If she had recurrent tracheal cancer and it was incurable, why the hell would he put her through a high-risk grueling experimental surgery? What about approval from UCL research ethics committee and MHRA?
Mom’s fund raising page, Read my Story, suggests that neither Keziah nor her family seemed to think the plastic trachea (re) transplant was “palliative.”
https://www.justgiving.com/tracey-hayter/
Macchiarini surgery: http://www.dailymail.co.uk/health/article-1299877/British-cancer-girl-saved-windpipe-stem-cells.html
How much of a failure was Seifalian’s plastic trachea? A US woman avoided becoming a victim of Macchiarini when “a British surgeon” contacted her husband and told him about a patient of his who had problems. Perhaps it was Mr Sandhu, also interviewed with Birchall in Experimenten:
Avråddes från att opereras av Macchiarini svt Nyheter 2016-02-21 [translated from Swedish to English courtesy of Google Translate; story about U.S. citizen Rachael Phillips and how she was dissuaded from allowing Dr Macchiarini to perform a synthetic trachea transplant by a warning from a UK surgeon ] http://tinyurl.com/jz3zn8k
Fletcher Allen Has a Candidate for Groundbreaking Windpipe Transplant Seven Days Ken Picard 2011-08-03 http://www.sevendaysvt.com/vermont/fletcher-allen-has-a-candidate-for-groundbreaking-windpipe-transplant/Content?oid=2144010
One child had a decellurized-stem cell trachea transplant. The child, 15 yo Shauna Davison was featured in an episode of BBC2’s Great Ormond Street Hospital. The video is no longer online but the the subtitles give a reasonable amount of information
BBC2 Great Ormond Street Experimental Surgery Subtitles 2012-06-19: http://subsaga.com/bbc/health/great-ormond-street/series-2/6-experimental-surgery.srt
Great Ormond Street: how far should surgeons push the boundaries? NPR RadioTimes 2012-06-19: http://www.radiotimes.com/news/2012-06-19/great-ormond-street-how-far-should-surgeons-push-the-boundaries
Great Ormond Street Hospital Trachea Service Procedures and treatments (why does GOSH list trachea transplant as if it were a clinical procedure?)
http://www.gosh.nhs.uk/health-professionals/clinical-specialties/tracheal-information-health-professionals/procedures-and-treatments
Family’s tribute after shock heart attack claims teen Lindsey Sampson Gazette Live 2012-03-13: http://www.gazettelive.co.uk/news/local-news/familys-tribute-after-shock-heart-3675304
Birchall told the committee she died of an acute event unrelated to the trachea transplant, but he couldn’t be certain without an autopsy which the family declined. The young girl died about 1 month after the transplant according to the Gazette Live story above. Birchall told Science (cited above) that she died from a sudden massive bleeding in the chest. One of the experts commented that this could well be related to the transplant.
You noted funding for trachea transplants has been awarded. However funding for a phase 2 trial was announced less than 30 days after funding for the phase 1 trial was announced. It’s even more perverse because the phase 2 trial was funded/announced before there was confirmatory data from the phase 1 and even before the phase 1 trial opened, which is supposed to happen in the first half of 2016. How the hell does this work??? What am I missing here?
Horizon 2020 awards €6.8m to TETRA consortium for pivotal replacement trachea trial EU study to build on Phase I trial of Videregen product & generate approval application Videregen Ltd Press Release 2015-12-10 http://www.videregen.com/horizon-2020-awards-e6-8m-to-tetra-consortium-for-pivotal-replacement-trachea-trial/
Clinical trial for replacement trachea technology to start next year INSPIRE consortium receives UK MHRA approval for study Videregen Ltd Press Release 2015-111-03 http://www.videregen.com/clinical-trial-for-replacement-trachea-technology-to-start-next-year/
LikeLike
Research Impact report 2014 for Saifelian’s group at UCL – keep in mind Andemarian Beyene, who got the POSS/PCU trachea, died on 24 Jan. 2014 following 8 months in hospital.
“Introduction of new therapy: Although it is possible to surgically resect tracheal tumours, most are already of an inoperable size by the time of diagnosis. As such, the synthetic trachea addressed a pressing clinical requirement for alternative therapeutic options. In 2011, a patient with advanced tracheal cancer who had exhausted all existing treatment options was referred to cardiothoracic surgeon Professor Macchiarini at the Karolinska Institutet, Stockholm. In turn, Macchiarini approached Seifalian for help. Seifalian used his POSS-PCU polymer [output 1] and knowledge of developing and seeding 3D scaffolds [output 4] to manufacture the synthetic trachea implant, which consisted of a 12cm trachea along with two bronchi — the largest such implant ever attempted and the world’s first wholly tissue-engineered synthetic organ transplant [a,b]. By using the patient’s CT scans, Seifalian was able to model the implant on the exact dimensions of the patient’s own trachea, making implantation significantly easier. Mechanically, the synthetic trachea had similar properties to a native organ, including ring-like cartilage structures, with porous materials between them allowing 15% stretchability. UCL’s researchers developed a bioreactor for the organ’s development, which dripped the patient’s own stem cells onto the porous scaffold under physiological conditions in an incubator. Because it used his own stem cells, there was no need for the patient to take the immunosuppressive drugs required after donor transplants. The trachea and bronchi were implanted in June 2011; more than two years on, the patient has a functioning organ and is doing very well. Describing the implantation and its effects, Professor of Laryngology at the Royal National Throat, Nose and Ear Hospital said: “This is the first time that a trachea made from a synthetic scaffold (here repopulated using stem cells), has preserved life and quality of life for longer than a few months…follow up is now 2.5 years” [c].
This was the first instance in which doctors had ever been able to manufacture human organs using synthetic scaffolds, and then incorporate autologous stem cells. As such, the work not only saved the life of the patient in whom the synthetic trachea was implanted, but revolutionised the development of organs more broadly. The success of previous lab-generated transplants, which lined a decellularised donor trachea with the patient’s stem cells, had been limited by a number of problems that Seifalian’s trachea was able to overcome. These included the very real difficulties of obtaining the necessary donor organs; even assuming that these could be found, moreover, the decellularising is suitable only for small sections of trachea, rather than an entire organ, and risks damaging their underlying structure, leading to their collapse once they are implanted in the patient.”
“[c] ] The superiority of the tracheal implant over other types of tracheal transplant is evidenced by the statement from a Professor of Laryngology at the Royal National Ear, Nose and Throat Hospital. Available on request.”
http://impact.ref.ac.uk/CaseStudies/CaseStudy.aspx?Id=30007
I’m guessing there’s some reason why Saifelian and that Professor of Laryngology – Birchall – are misleading everyone by claiming a dead patient is doing well, and enjoys good quality of life no less. What reason would that be?
LikeLike
Just noticed that Birchall trashed the approach he’s currently testing on humans in the RegenVox trial and will test in the soon to open INSPIRE (should be EXPIRE) trachea transplant trial.
“The success of previous lab-generated transplants, which lined a decellularised donor trachea with the patient’s stem cells, had been limited by a number of problems that Seifalian’s trachea was able to overcome. These included the very real difficulties of obtaining the necessary donor organs; even assuming that these could be found, moreover, the decellularising is suitable only for small sections of trachea, rather than an entire organ, and risks damaging their underlying structure, leading to their collapse once they are implanted in the patient.”
LikeLike
Correcting non-working URL above: this is the cached version http://tinyurl.com/gu2g3wa
I can’t link to the URL for the pdf download.
Also correcting the spelling of Dr Seifalian’s name, for which I apologize.
LikeLike
Pingback: Cells Weekly – March 27, 2016
Pingback: Macchiarini and the tracheal regeneration scandal, by Pierre Delaere – For Better Science
Harvard Bioscience as shown in the Experimenten documentary, later spunoff to Harvard Apparatus / Harvard Apparatus Regenerative Technology (HART) Nov 2013, (side note, named Harvard b/c they had a trademark licensing deal with the university http://www.harvardbioscience.com/about-us/history/ ), has now changed their name again to “Biostage” as of April 1 2016
https://web.archive.org/web/20151027074700/http://www.harvardapparatusregen.com/
http://ir.biostage.com/2016-03-31-Harvard-Apparatus-Regenerative-Technology-Changes-Name-to-Biostage-Inc-NASDAQ-BSTG
“. . . Based on its preclinical data, Biostage identified life-threatening conditions of the esophagus as the initial clinical application to advance its Cellspan esophageal implant. Biostage plans to file an Investigational New Drug (IND) application with the U.S. Food and Drug Administration (USFDA) in late 2016, seeking to initiate clinical trials in humans.
Biostage is currently expanding its preclinical testing of its Cellspan esophageal implants in collaboration with Mayo Clinic to support its planned IND application. The company anticipates providing an update on its preclinical research collaboration mid-second quarter 2016. ”
David Green, the man in the documentary assuring Julia Tuulik that the product is safe, is apparently still on the board
http://www.biostage.com/board-directors
LikeLike
Just to be clear about D. Green
Scene in “Experimentent” from Inifran / svt.se where David Green meets Julia Tuulik to show her the implant and bioreactor (about 16 minutes into part 2):
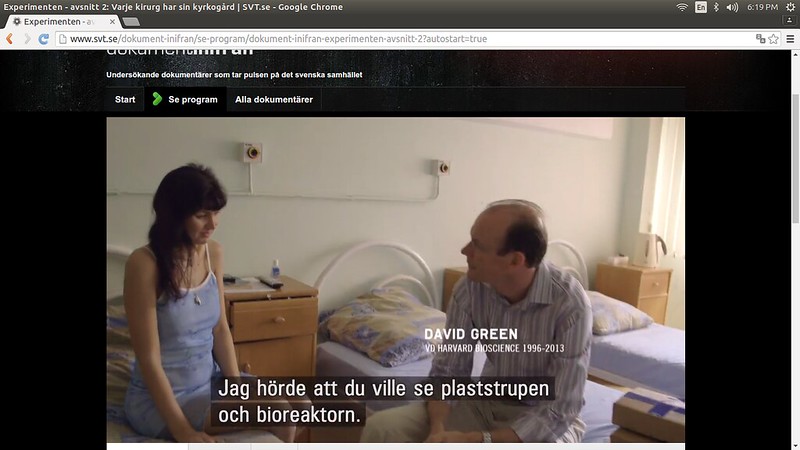
http://www.svt.se/dokument-inifran/se-program/dokument-inifran-experimenten-avsnitt-2
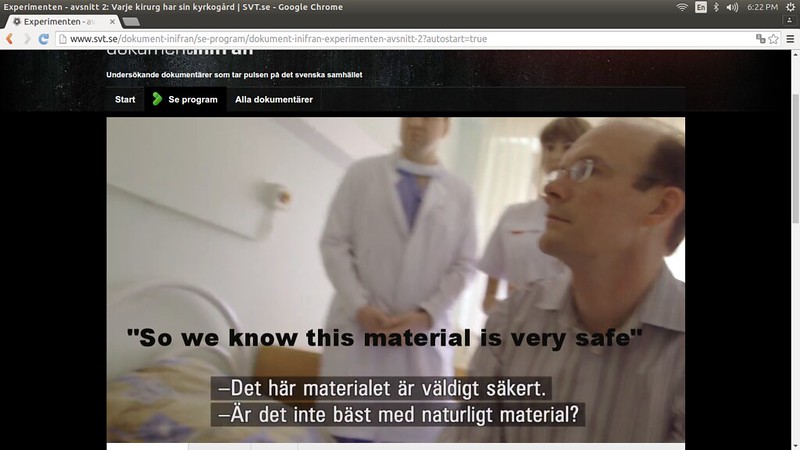
Scene in same where he responds to her comment on the implants appearance by explaining that it is medical grade plastic, saying “So we know this material is very safe”, in English. A few seconds later he says “This you cannot get, naturally, so we have to make it”. Then he shows her pics on his phone of Beyem. “He received his trachea one year ago”.
Biostage biography for Green.
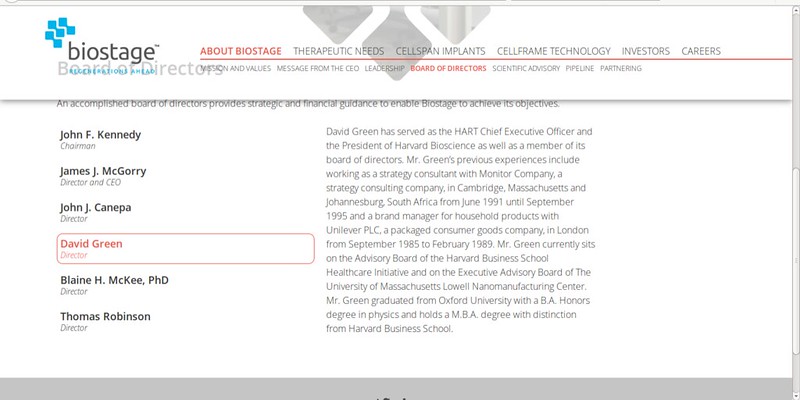
http://www.biostage.com/board-directors
“Mr. Green currently sits on the Advisory Board of the Harvard Business School Healthcare Initiative and on the Executive Advisory Board of The University of Massachusetts Lowell Nanomanufacturing Center”
Linkedin for Green
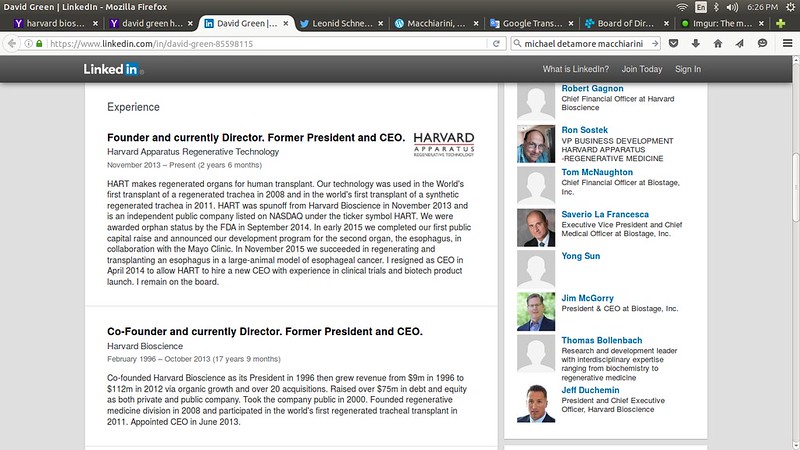
https://www.linkedin.com/in/david-green-85598115
” Our technology was used in the World’s first transplant of a regenerated trachea in 2008 and in the world’s first transplant of a synthetic regenerated trachea in 2011. HART was spunoff from Harvard Bioscience in November 2013 and is an independent public company listed on NASDAQ under the ticker symbol HART. We were awarded orphan status by the FDA in September 2014. In early 2015 we completed our first public capital raise and announced our development program for the second organ, the esophagus, in collaboration with the Mayo Clinic. “
LikeLike
Also, there is an interesting note on Biostage webpage about tracheal implants
“Cellspan Tracheal Implants may allow for the safe reconstruction of any portion of the trachea, potentially restoring its functional integrity. Proof-of-concept was confirmed in man in an earlier generation tracheal product showing that a electrospun scaffold seeded with the patient’s own cells can form a patent airway. ”
I am curious about what “earlier generation tracheal product” they are referring to and what “Proof-of-concept” means.
LikeLike
HART (US company that made bioreactor and some of the artificial trachea scaffolds) put the following statement into their 10-k financial report from March 2015 (side note, 10-k is an annual report required of publicly traded companies by the Securities and Exchange Commission, a result of the reforms of President Roosevelt in the 1930s to prevent corporations from defrauding investors)
“To date, the HART-Trachea has been implanted in five adult human patients. Average survival among the three of these patients who have died to date has been 22 months. This is a significant improvement over the prognosis at the time of implant which was typically just a few months. Of the three patients who have died, none of them have died because of a failure of our scaffold.”
. . .
In June 2012, our InBreath bioreactors were used for the world’s first two successful laryngo-trachea transplants . . . Both of the Russian patients required laryngo-trachea transplants, whereby the scaffold mimics the shape of the windpipe from the larynx to the point where the trachea branches into the two bronchi which lead to the lungs. Both patients had difficulties breathing and talking and had suffered repeated infections prior to the surgeries. . . .
In December 2013 and in June 2014 additional patients received the HART-Trachea in surgeries conducted in Krasnodar, Russia. One of those patients was a re-transplant, where a scaffold that was first transplanted into the patient in June 2012, as described above, was removed and replaced by a HART-Trachea. The other patient had a trachea transplant for the first time.
All these patients have been treated under compassionate-use protocols meaning their prognosis was very poor. Typically, their bodies are very weak as a result of disease, trauma and extensive treatments that often include radiation, chemotherapy and prior surgeries. We believe that patients that undergo such extensive treatments are inherently susceptible to serious medical complications following the transplants. These transplant surgeries are typically the last-resort measure to save the patient’s life. We expect that some transplant patients are likely to suffer serious complications or death following the transplants due to issues that are not directly related to the use of our products. ”
http://yahoo.brand.edgar-online.com/displayfilinginfo.aspx?FilingID=10590352-14548-200209&type=sect&TabIndex=2&dcn=0001144204-15-018746&nav=1&src=Yahoo
March 2016 their new 10-k report has changed a bit. They do not list the operations and do not mention the specifics like the name of Dr Porhanov in Krasnodar. :
“Leading up to the time of the Separation, we engaged in activities with a surgeon, Dr. Paolo Macchiarini, then employed by the Karolinska Institutet in Sweden, one of the world’s most respected medical institutions, and who was at that time considered to be a world-renowned regenerative medicine pioneer. We provided organ bioreactors and organ scaffolds to Dr. Macchiarini’s laboratories to conduct cell biology research and in return Dr. Macchiarini was to provide us with scientific data to advance the development of our bioreactors and tracheal scaffold. We also provided bioreactors and tracheal scaffolds in support of several compassionate care human surgeries performed by Dr. Macchiarini. We collaborated with Dr. Macchiarini in an effort to advance our product research and development with the assistance of a highly-acclaimed researcher and a well-respected institution. Regarding compassionate use human surgeries, we relied on the due process that involved a team of physicians and the Institutional Review Boards of the institutions where the surgeries were performed. We developed no part of any clinical protocol in any manner. Further, all surgeries involving any of our products or product candidates were conducted under the compassionate use system governed by the rules and regulations of each institution. Dr. Macchiarini was not employed by or affiliated with our company, and we did not pay him any wages or consulting fees. In June 2014, shortly after our Chief Medical Officer joined our company we ceased support of any human surgeries with Dr. Macchiarini. In addition, in November 2014, we formally announced that we would no longer be supporting or providing products to the human surgeries being performed in Russia, based in part on our belief that, due to the design of the Russian hospital’s study and the nature and extent of the follow-up medical data made available to us, additional surgeries in Russia would provide less meaningful product development data than the work being done in our U.S. research and preclinical programs at that time. . . . .
Since the time we withdrew from involvement with Dr. Macchiarini, his work has become the subject of at least two investigations by the Karolinska Institutet. Many of the claims Dr. Macchiarini made publicly and published in peer-reviewed articles in reputable medical journals about the post-surgery quality of life, or even the necessity, of certain of his compassionate care surgeries where he used either a HART bioreactor or a HART-Trachea scaffold, or both, have been called into question. We discontinued development of our HART-Trachea product in 2014, and that first-generation product was significantly different from our new Cellframe technology and Cellspan products currently in development. We have focused our development efforts on our Cellframe technology and Cellspan products, which we have and will continue to develop internally, and with our collaborators, via a rigorous scientific development process. As a result, we believe that prior statements by Dr. Macchiarini or others regarding the patients whose surgeries utilized our HART bioreactor or HART-Trachea scaffold, or such products, are not pertinent to our Cellframe technology or Cellspan products, or their respective future development. ”
http://yahoo.brand.edgar-online.com/displayfilinginfo.aspx?FilingID=11291091-882-317878&type=sect&TabIndex=2&dcn=0001144204-16-091643&nav=1&src=Yahoo
I am just a lay-person, but a lot of this doesn’t seem to agree with what Leonid has written on this blog.
Also they kind of don’t mention that the CEO of the company personally was involved in persuading at least one of the patients to undergo operation, which is on video on the svt,se Experimenten documentary
LikeLike
HART press release 2015-11-12, regarding animal studies NOS:
“We are very encouraged by the results of these studies and by the significant advancements we have made across all three indications. In particular, results for the Gen2 implant in the esophagus far exceeded our expectations, particularly given the study’s brief duration. Over a two-week period, all layers of the esophagus, most notably the epithelial and muscle layers, were fully regenerated, and components of esophageal nerves were present.”
http://www.sec.gov/Archives/edgar/data/1563665/000114420415064789/v424594_ex99-2.htm
Wowee an esophagus in two weeks! Since Suchitra Sumitran-Holgersson and her team can regenerate a vein in 7 days, the HART/Biostage claim must be true.
But wait, it gets better. HART annual report for the period ending 2015-12-31:
“We believe that our new technology is unique, in that its mode of action appears to be different from other tissue engineering organ scaffold products developed previously, of which we are aware. Prior to our development of the Cellframe technology, our approach attempted to implant an organ scaffold that would be incorporated into the patient’s body by the surrounding native tissue growing into the scaffold. To our knowledge, all previous research and development efforts by other investigators were based on that same concept. Our Cellframe technology appears to work very differently. We believe that the unique combination of our highly-engineered organ scaffold with a population of the patient’s own mesenchymal stem cells enables an organ to develop new native tissue around our scaffold, but not into it, so the scaffold acts as a sort of frame or staging for the new tissue. As a result, our scaffold is not incorporated into the body. Instead, it is retrieved from the body via an endoscopic or bronchoscopic procedure, not surgically, after sufficient tissue remodeling and regeneration has occurred to restore the organ’s integrity and function.”
http://www.sec.gov/Archives/edgar/data/1563665/000114420416091643/v435009_10k.htm#a_002
Endoscopic retrieval: good luck with that. (if I stand on my head will my Cellframe esophagus fall out?)
HART goes on to explain that the source of cells for the new, aka not the tainted lethal former scaffold, is patients’ adipose tissue. What are these people smoking?
Now we can all have nightmares about growing tracheas in those flabby areas that resist exercise
LikeLike
Aside from the obvious, the other origin of “Cellframe technology”, from the HART annual report for the period ending 2013-12-31
“We have also received correspondence from legal counsel to Nanofiber Solutions, Inc., or NFS, claiming that in developing our scaffold product and related intellectual property, we may have committed misappropriation, unauthorized use and disclosure of confidential information, and possible infringement of intellectual property rights of NFS. We have received correspondence from legal counsel to UCL Business PLC, or UCLB, challenging the validity of the assignment of certain patent applications that have been assigned to us by Dr. Macchiarini. We have also received correspondence from an academic researcher implying that one of our products may violate an issued patent.”
http://www.sec.gov/Archives/edgar/data/1563665/000114420414018938/v371822_10k.htm#tBUS
Nothing like the threat of patent litigation to “discover” new technology.
As the annual report explains, between 2011-12 patients we now know as Chris Lyles, Yesim Cetir, Julia Tuulik, and Alex Zozulya got plastic tracheas made for HART by Nanofiber Solutions. Chris died, Yesim’s plastic trachea failed and Julia and Alex had obstructions and collapse at 6-8 weeks: http://icvts.oxfordjournals.org/content/17/suppl_1/S55.4.abstract
Thereafter HART made its own plastic nanospun tracheas (supposing if only they muttered the correct spells around the cauldron their version of the same thing wouldn’t kill patients). The first use was Hannah Warren in April 2013, and she died three months later. In July 2013 Yesim got a second transplant; it was a failure as well. Then there was a re-transplant in Krasnodar, either Julia or Alex.
In 2013-14 Hannan and Dmitry O. got HART’s plastic trachea and there was another re-transplant in Krasnodar. Hannan is dead (I believe), as are Julia and Alex. Dmitry got a much smaller implant and it was removed.
I count 8 victims, er patients, not five, as in the 2015 10-k filing Donbright quoted above, and 30 month old Hannah is not an adult (“five adult patients”).
Gosh why didn’t the annual report mention Hannah? Gee …
Federal law requires that information filed with SEC be accurate.
What did HART/Biostage tell FDA about their dog-breath airway transplant system, that got orphan drug designation in Sept. 2014?
http://tinyurl.com/jfkz6wf
LikeLike
Pingback: The stem cell faith healers, or magic inside your bone marrow – For Better Science
Pingback: The stem cell faith healers, or magic inside your bone marrow – For Better Science
Pingback: Macchiarini’s patients, the real situation – For Better Science
Pingback: “Notification of Suspected Research Misconduct”, by 4 Macchiarini-whistleblowers – For Better Science
My understanding was that Professor Seifalian was involved with one patient and that patient lived 2 years 8 months. the other was transplanted by other two and POSS-PCU was not used for the other two cases, only for the first patient.
LikeLike
Paolo Macchiarini performed a cadaveric decell-recell trachea transplant on K.S. in July 2010 at Careggi in Florence. This failed and in Sept. 2011 Mr Birchall and colleagues implanted a POSS/PCU trachea in K.S. at UCL. Interviews (in English) at 36.50 in Experimenten pt. 1: http://www.svt.se/dokument-inifran/se-program/dokument-inifran-experimenten-stjarnkirurgen
This paper by Birchall et al. provides information about 3 trachea transplants, one of which is K.S.: https://www.ncbi.nlm.nih.gov/pubmed/26559535
Oddly the authors assert all 3 transplants were performed under Specials licenses approved by the UK MHRA. Specials are limited manufacturing licenses for non-routine use of products not approved for marketing that are dispensed on the authority of a licensed physician. Specials are very similar to UK Hospital Exemptions but one key difference is that unlike products made under a Hospital Exemption license, Specials products cannot be exported. This raises obvious questions about how the POSS/PCU scaffold used in the first transplant at Karolinska was obtained.
LikeLike
Pingback: EU threatens to terminate TETRA Consortium funding for phase 2 trachea transplant trial – For Better Science
Pingback: Brexiting out of EU research and patient rights – For Better Science
Pingback: Regenerating in Hannover, Part 2: Axel Haverich’s “growing” heart valves – For Better Science
Pingback: How UCL throat surgeon Martin Birchall misleads patients and tricks public funders – For Better Science
Any updates on whether Alexander Seifalian is still associated with UCL? He no longer appears on the UCL IRIS profile (Institutional Research Information Service)…
LikeLike
Pingback: Macchiarini investigations outcome: the Karolinska Institutet – For Better Science
Pingback: Macchiarini and the bonfire of greed – For Better Science