With his three trachea transplant clinical trials suspended, the British laryngologist and UCL professor Martin Birchall now tries to pull at least the clinical trial RegenVox, which is about the “regenerated” voice-box (larynx) and upper trachea, out of the bog. In order to get an approval to transplant human patients, UCL’s chief trachea transplanter tries to prove in several papers to his funder MRC that the technology is safe in pigs. For this he seems to be reusing data in his recent papers from 2017, to illustrate utterly different experiments, in fact some of this data is at least 7 years old. One wonders if Birchall fibbed the authorities and the scientific community about the novel nature of his results. Was the confused scientist simply salami-publishing and re-interpreting the preclinical data he actually obtained long before he received MRC funding for RegenVox? The evidence of the larynx transplant data reuse across two recent papers and the grant proposal document from 2010 follows below, background on Birchall’s RegenVox and his trachea transplant adventures is here and here.
The UCL laryngologist uses the technology which developed with his former partner, the scandal trachea transplant surgeon Paolo Macchiarini. The method is to strip the cadaveric donor organ of all cells (in a process called decellurisation) and then bring it back to life as recipient’s own organ using the magic of bone marrow cells. In Birchall’s own words, those magic cells recognise the decellurised organ by its shape and know what to do then:
“Having found themselves in certain environments a stem cell will look around and say ‘This has got the correct feel to it, the right stimuli, shape of a muscle, a piece of cartilage, a piece of bone’ and will therefore differentiate in those ways. So, for example, if it’s an environment with lots of other cartilage cells it’ll become a cartilage cell too”.
In UK, Birchall was funded for two phase 1 clinical trials, Inspire for trachea transplants, and RegenVox for the larynx transplant. He also leads a EU-wide consortium which is supposed to be the phase 2 counterpart to Inspire, named TETRA. That one might never happen, the EU stubbornly refuses to comment on its future, while the clinical trial site in Austria, the Medical University Vienna, quietly jumped ship. Inspire was put on hold due to my reporting, while RegenVox was supposed to continue recruiting patients. According to a MRC letter from December 15th 2017, in 2017 one patient agreed to a cadaveric larynx transplant, but the operation did not happen. Now also RegenVox is suspended, while Birchall tries to save it by publishing new papers which claim to be very new, very promising preclinical experiments, but actually show data which already featured in the 2010 RegenVox application.
The 2017 UCL investigation into trachea transplants was quite convinced that the trachea regeneration technology works in principle very well, and that the only problem was the use of plastic material for trachea replacement by shifty foreigners like Macchiarini and the sacked UCL nanotechnology specialist Alexander Seifalian (who is not even a doctor, but physicist). A photo of Birchall proudly holding up a plastic trachea which Seifalian made was removed from the UCL website and even from this MRC article.

Birchall and his UCL colleagues were given a go-ahead by the UCL investigators to transplant tracheas, or at least larynxes, because those were deemed as less dangerous, and hence worth a try (you will likely survive a failed larynx or upper trachea transplant, but you cannot live without a trachea in your chest). On November 8th, UCL Board of Directors decided (see here page 143):
“In light of the report of the special inquiry into regenerative medicine research at UCL (29 September 2017), a review of the status of Professor Martin Birchall’s suspended trials was undertaken. The suspension on the RegenVox trial has been lifted as no concerns were raised about this trial however the Inspire trial remains paused by the Joint Research Office (JRO) as the commercial sponsor has suspended this trial pending their review”.
The website ClinicalTrials.gov lists RegenVox as “recruiting”, 10 patients are expected. But Birchall and UCL probably celebrated too early. The British Parliament began looking into the trachea transplant activities at UCL, after receiving a letter from two Liverpudlian scientists which I reproduced here. The Medical Health products Regulatory Agency (MHRA) announced in December 2017:
“the sponsors of these studies voluntarily halted all trial-related activity while the UCL special inquiry was ongoing. This action was supported by MHRA and the relevant ethics committees, and we have been in close dialogue with the sponsors to actively monitor the situation whilst the benefit:risk profile for these studies has been reviewed.
Should the sponsors of these trials wish to restart their respective studies, a substantial amendment to their clinical trial authorisation will need to be submitted for prior review and approval by MHRA and ethics. While we cannot pre-judge the outcome of such a request, the review will take account of all available information and MHRA will also seek independent expert advice from an expert advisory group of the Commission on Human Medicines. […] these trials currently pose no threat to public health with no patients having been treated to date, that no trial-related activities are ongoing, and that any restart of the trials will need prior MHRA and ethical approval, including input from independent experts. We will only allow these trials to re-start if all of the evidence currently available suggests that the potential benefits to the patients outweigh the risks”.
Even if MHRA should re-approve Birchall’s RegenVox, the funder MRC announced to be having a closer look and still demand an external assessment before RegenVox can resume. How will MRC now react to those image duplications, which suggest Birchall was pulling their leg all these years?
The papers in question are as follows, one of them was already plucked apart on PubPeer, and the critic then had no clue of the data duplications to come:
P. Herrmann, T. Ansari, A. Southgate, A. Varanou Jenkins, L. Partington, C. Carvalho, S. Janes, M. Lowdell, P.D. Sibbons, M.A. Birchall
In vivo implantation of a tissue engineered stem cell seeded hemi-laryngeal replacement maintains airway, phonation, and swallowing in pigs
J Tissue Eng Regen Med. 2017 Oct 19. doi: 10.1002/term.2596
Ansari T, Lange P, Southgate A, Greco K, Carvalho C, Partington L, Bullock A, MacNeil S, Lowdell MW, Sibbons PD, Birchall MA
Stem Cell-Based Tissue-Engineered Laryngeal Replacement.
Stem Cells Transl Med. 2017 Feb;6(2):677-687. doi: 10.5966/sctm.2016-0130.
As well as the black-and-white scans of the original RegenVox proposal I once obtained, which is available here. In particular, note the file “RegenVox3“, stamped “April 2010” where you can find images of allegedly novel data which was freshly produced after 2012, when Birchall applied to MRC for extra funding in order to conduct further preclinical testing of cadaveric larynxes on pigs. Now observe:
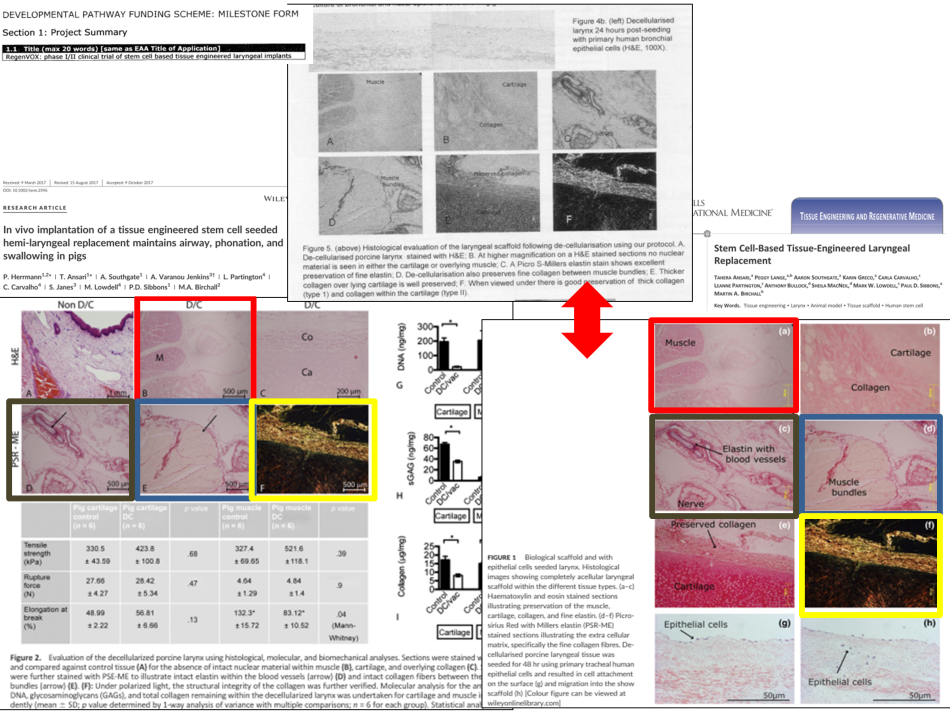
The two papers Herrmann et al 2017 and Ansari et al 2017 show clearly different pig experiments, well, at least they are supposed to. The former is said to have used 8 pigs, where the decellurised graft was seeded with human bone marrow and epithelial cells and those pigs received directly “a full thickness segment of de‐cellularised larynx directly into a defect created in the larynx“. The latter paper used 6 pigs, where each pig
“received a full-thickness segment of decellularized larynx in a two-step procedure directly into a defect created in the larynx. Each decellularized hemilarynx was initially seeded with human mesenchymal stem cells (MSCs) and implanted into a sternomastoid muscle fascial pocket for 1 month (stage 1). Thereafter, the scaffold was relocated into a full-thickness defect created in the cricoid cartilage. Thereafter, the scaffold was relocated into a full-thickness defect created in the cricoid cartilage […] (stage 2)”.
So the method of operation was completely different (1-step vs 2-step implantation), and Birchall even took care to explain in Herrmann et al 2017 the difference to his previous research published earlier same year in Ansari et al 2017:
“Our previous study (Ansari et al., 2016) successfully implanted porcine hemi‐larynx into a porcine model with good structural and physiological outcome but used a two‐step surgical procedure in order to promote vascularisation of the implanted graft prior to in situ relocation. This study has shown that it is possible for complete epithelialisation of the mucosal surface to occur without prior attempts at vascularisation of the scaffold. […] Because both histological and molecular characterisation of the scaffold has been previously reported (Ansari et al., 2016), only histological confirmation is presented. Although each scaffold was seeded with human cells prior to implantation, it was not possible to follow the fate of these cells postimplantation and is a recognized limitation of the study”.
It was however perfectly possible for Birchall to use the images from over 7 years ago, in both papers, representing two utterly unrelated new experiments with two different cohorts of pigs. Here is another example:
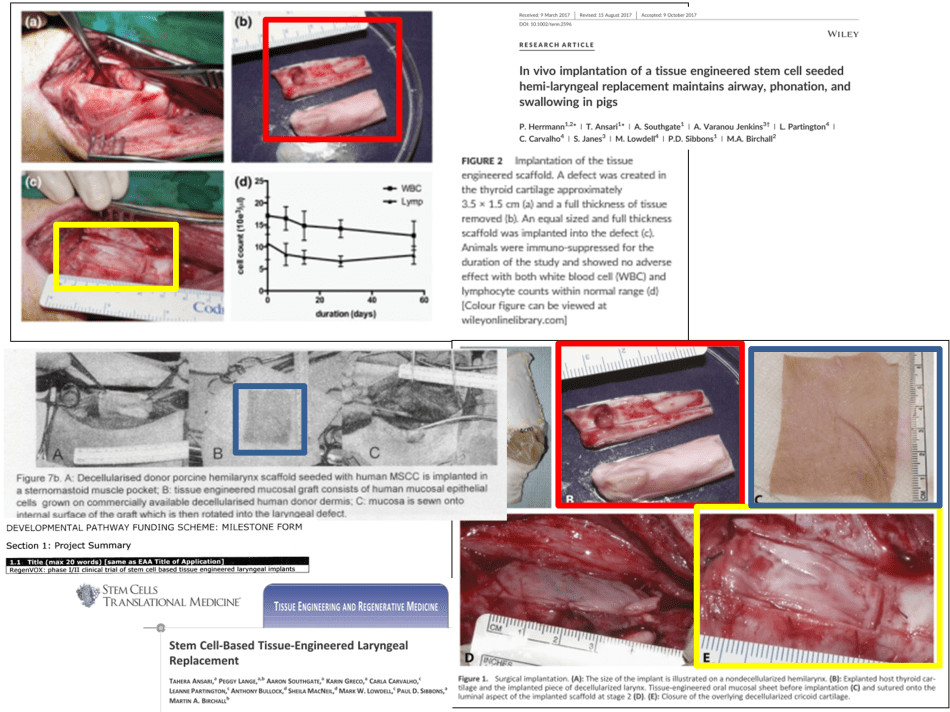
Herrmann et al 2017 speaks of 8 pigs, yet declare “Animal 16 was terminated early on Day 51 postimplantation due to respiratory problems”. Why name a pig Nr 16 if there were only 8 of them? Well, the RegenVox proposal from 2010 (from which some of the data was reused in Herrmann et al 2017) reports “We gave 16 pigs seeded laryngeal implants (Fig. 7b). Animal survival to 2 months was 81%“, which would mean 13 pigs survived out of 16. Sadly, Hermann et al 2017 is less positive (while apparently using same data) and reports only a 75% (6 out of 8) survival rate. What happened to the other 8 pigs of that original herd of 16?
Well, in 2010 MRC funded Birchall for a pig study of different materials in order to develop a ‘tissue-engineered larynx’ where both plastic and cadaveric larynxes were used. In fact, as MRC’s Chief of Strategy Declan Mulkeen explained in a letter from December 15th 2017, “plastic larynxes were placed in eight pigs and cadaveric larynxes in another eight pigs“; but what with the Macchiarini affair around plastic tracheas, “no subsequent development or testing of the plastic implants occurred with MRC funding“. So now we can guess how those other 8 pigs got lost on the way: they received unpublishable graft material, namely plastic. Also, how does it look when 7 out of 8 plastic larynx pigs survived, but only 6 out of 8 pigs with cadaveric larynx? When you know plastic is deadly, how can you sell cadaveric alternative to anyone after more pigs died from it?
Unless, you decide those dead pigs never existed in the first place. In Ansari et al 2017, all six pigs transplanted with cadaveric larynxes were a success until sacrificial analysis at after 6 months. This was probably only made possible because the two pigs from this pre-2010 cadaveric larynx cohort of 8 piggies were disqualified from participating in that paper. A 100% survival rate, as Ansari et al 2017 declares:
“There were no clinical adverse effects due to the implanted scaffold; one pig was killed because of an ear infection relating to a long-term underlying pathology exacerbated by immunosuppression.”
Right after, the 6 pigs came back, together with their two deceased colleagues, to make a come-back in Herrmann et al 2017:
“Six out of the 8 animals survived to the planned termination date; 2 animals were terminated due to mild stenosis and deep tissue abscess formation, respectively”.
Only that here is another case of data reuse, between Herrmann et al 2017 and the 2010 grant proposal:
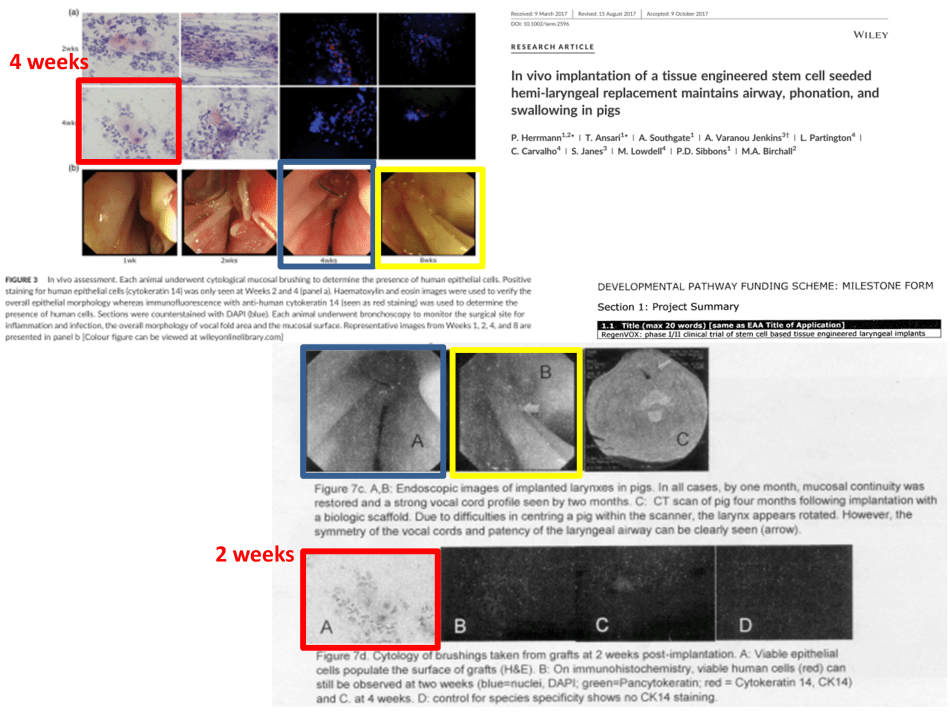
Did Birchall and his UCL partners perform any new experiments on pigs at all? Are they taking the MRC and the scientific community for the proverbial (piggy-back) ride?
And finally, if 2 out of 8 pigs which received a cadaveric larynx graft died after 2 months, where exactly is the benefit for human patients Birchall seeks to treat, who according to RegenVox inclusion criteria must have a life expectancy of at least 5 years?
Note 4.01.2018: this article has been updated with a reference and hyperlink to this letter by MRC, now published by the UK Parliament, which I previously only referred to as an unspecified source.
Update 20.01.2018. I must issue a correction! Image reuse is not Birchall’s new low, but an old one.
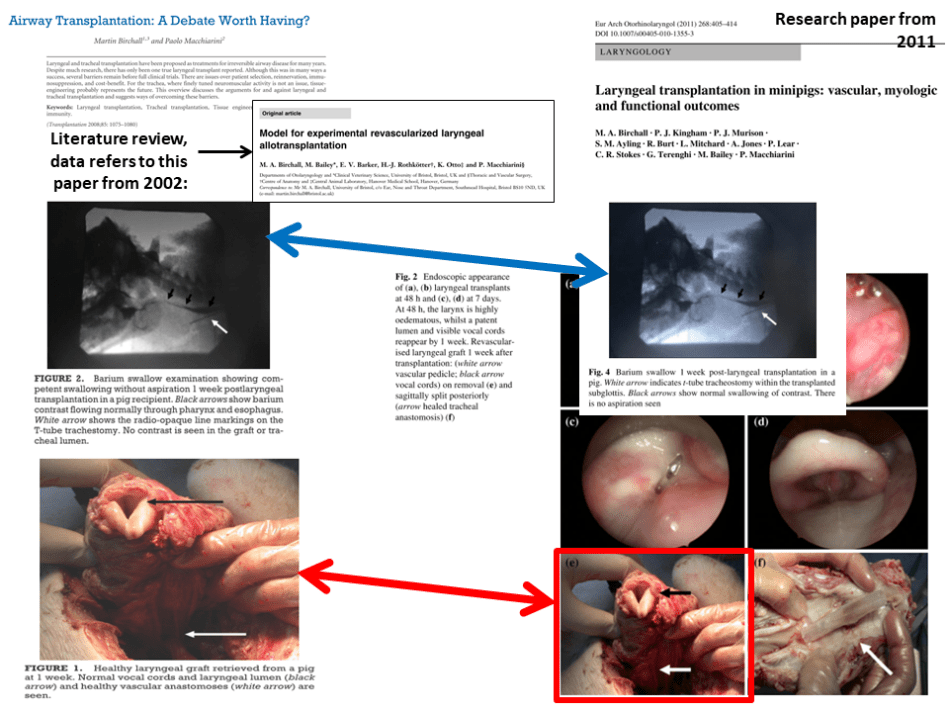
In 2011, Birchall and Macchiarini published this preclinical larynx transplant study on 34 minipigs, results were presented as brand new and freshly obtained:
M. A. Birchall, P. J. Kingham, P. J. Murison, S. M. Ayling, R. Burt, L. Mitchard, A. Jones, P. Lear, C. R. Stokes, G. Terenghi, M. Bailey, and P. Macchiarini
Laryngeal transplantation in minipigs: vascular, myologic and functional outcomes
Eur Arch Otorhinolaryngol. 2011; 268(3): 405–414. doi: 10.1007/s00405-010-1355-3
That paper however reuses images from a literature review “Airway Transplantation: A Debate Worth Having?” by Birchall and Macchiarini, published in Transplantation in 2008. As this is a review, it cites as the source for the data in figures this 2002 publication, where a different number of minipigs was used:
Birchall MA1, Bailey M, Barker EV, Rothkötter HJ, Otto K, Macchiarini P.
Model for experimental revascularized laryngeal allotransplantation.
Br J Surg. 2002 Nov;89(11):1470-5.DOI:10.1046/j.1365-2168.2002.02234.x
The 2011 paper indicates the work was done at the University of Bristol, UK, and declares to have used the technology described in the 2002 paper. That however lists ethics permissions indicating that the experiments were performed in Bristol and at Hannover Medical School (MHH) in Germany, which were Birchall’s and Macchiarini’s affiliations at that time.
Update 28.01.2018. This is a bit strange. Data from a published Ansari & Birchall paper was reused in a patent submitted later. This is the paper:
Lange P, Greco K, Partington L, Carvalho C, Oliani S, Birchall MA, Sibbons PD1, Lowdell MW, Ansari T.
Pilot study of a novel vacuum-assisted method for decellularization of tracheae for clinical tissue engineering applications.
J Tissue Eng Regen Med. 2017 Mar;11(3):800-811. doi: 10.1002/term.1979.
It was submitted on April 16th 2014 and appeared online on February 17th 2015. COI statement: “The authors have declared the there is no conflict of interest”.
The patent on this same technology (here) which uses exactly same images, but refers to different experiments, like different numbers of pigs used and correspondingly with different quantification results, was filed right after the paper was submitted, on June 16th 2014, received a file number on November 19th 2015 and was published on April 6th 2016.
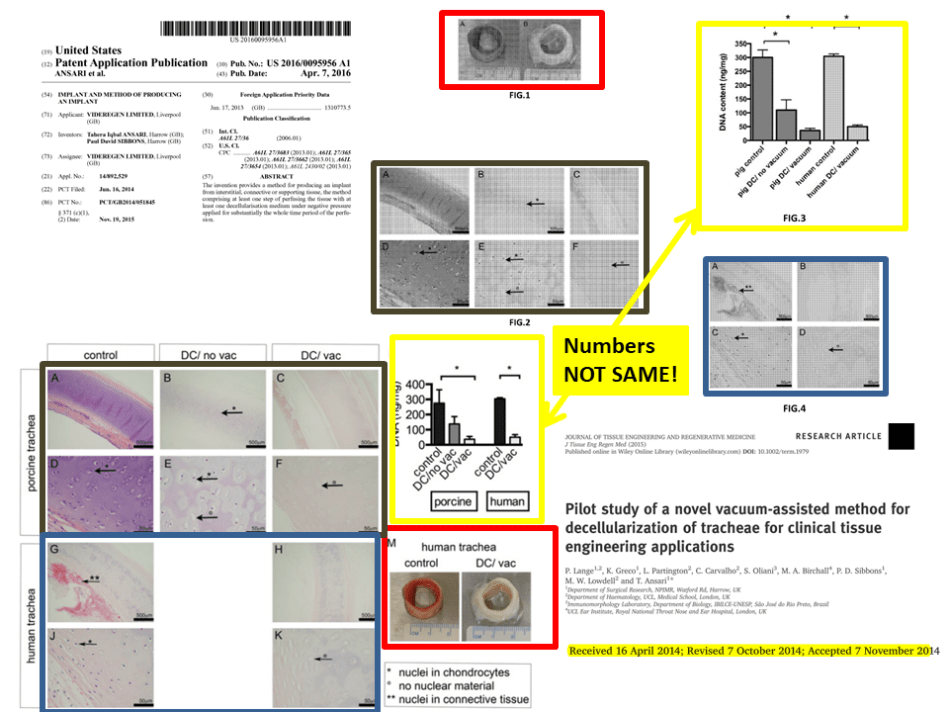
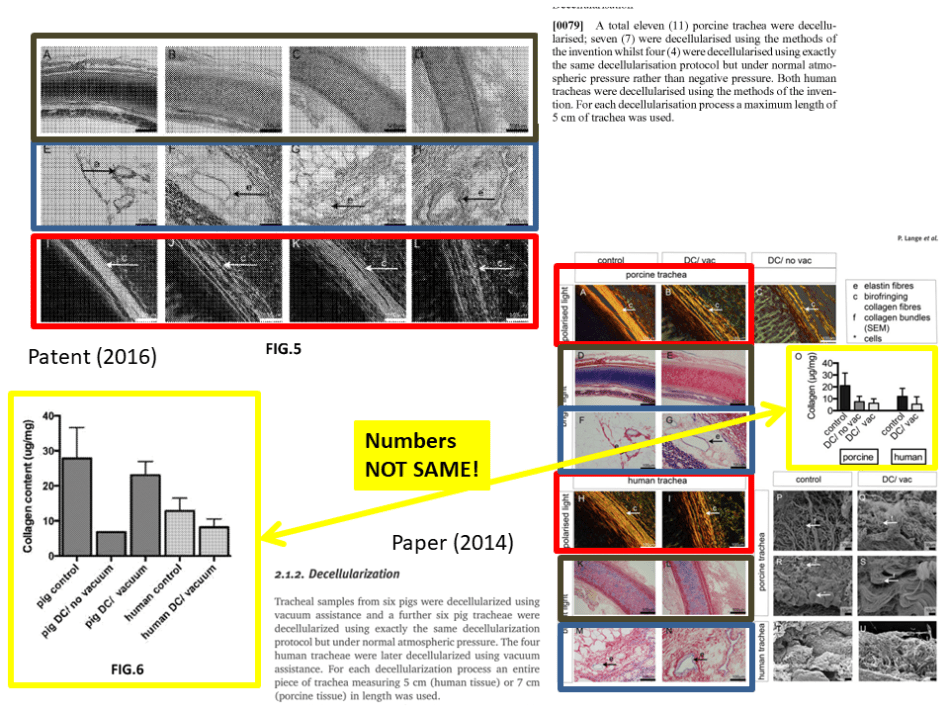
Update 31.03.2018. I received a set of documents concerning RegenVox, from the UK Health Research Authority (HRA-NHS). They include the original RegenVox grant application from April 2014 (starting page 12 here),where the Ansari et al 2017 study featuring 6 pigs is referred to as having had 8 pigs, one of which is said to have died of an unrelated ear infection. It seems, the sty window was left open and two pigs flew away before the paper was published.

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00

So, for example, if it’s an environment with lots of other cartilage cells it’ll become a cartilage cell too”.
Conversely, if a stem cell is placed in a decellularised matrix with no other cartilage cells and indeed nothing but other stem cells, it will presumably remain as a stem cell. The whole point of decellularising a trachea, or a larynx, is to remove all chemical cues that the immune system might recognise as non-self (with unpleasant consequences). That is, there are no cues left to indicate to the naive stem cell what specialty it should follow.
Has Birchall thought this through properly?
or at least larynxes
Please. Larynges.
LikeLike
Pingback: Boletim de Notícias: estudo propõe tecnologia para filtrar ‘fake news’ | Direto da Ciência
Pingback: "He fought the Peer and..." - Ocasapiens - Blog - Repubblica.it
Pingback: Boletim de Notícias: Temer adia novamente prazo para Cadastro Ambiental Rural | Direto da Ciência
Pingback: UCL fibs Parliament about trachea transplants – For Better Science
A reader alerted me to this scandal from 20 years ago, reported in Nature, which shows which kind of medical ethics Birchall’s research team has:
‘Flawed’ infant death papers not retracted
http://www.nature.com/news/2011/110817/full/476263a.html
Some of these co-authors were Tahera Ansari and Paul Sibbons, both working at Northwick Park Institute for Medical Research in Harrow, UK. The Liverpool-based company Videregen, which manages Birchall’s trachea and larynx clinical trials and pays him as scientific advisor, is a spin-off by Northwick Park Institute.
LikeLike
Pingback: Trachea transplanters: Round 2 at UK Parliament – For Better Science
Pingback: UCL’s decellurised tracheas: strong and stable? – For Better Science
Pingback: Birchall’s two dead pigs to prove trachea transplants are safe – For Better Science
Pingback: UCL trachea transplants: Videregen sets lawyers on Liverpool academics Murray and Levy – For Better Science
Pingback: Trachea transplanters without borders – For Better Science
Pingback: Google cleaned of “Defamation Campaign” against Martin Birchall and UCL – For Better Science
Sam Janes is professor and Head of Respiratory Research Department at UCL. He is coauthor on some of papers discussed above. Previously, Janes was Birchall’s and Seifalian’s partner on trachea project:
https://forbetterscience.com/2018/05/31/ucl-trachea-transplant-inquiry-scapegoating-obfuscation-and-a-lost-nose/
Now, a reader alerted me to some image duplications in Janes’ papers on lung cancer research. in these papers:
Teixeira et al Nature Medicine 2019
Giangreco et al J Pathology 2012
LikeLike
Pingback: New government cash for trachea transplanters Videregen – For Better Science
Pingback: Martin Birchall innocent, UCL decides once again – For Better Science