This is a new instalment of my investigation into UK and EU funded clinical trials on trachea replacement by the throat surgeon Martin Birchall at UCL. The method of using bone marrow cells to regenerate a dead decellurised donor trachea was developed by Birchall together with Paolo Macchiarini, and tested since 2008 on several human patients, with catastrophic results. Macchiarini by now is a pariah sacked by his formerly proud employer, the Karolinska Institute in Sweden, but Birchall was given further millions in British and EU money for his human trachea transplant experiments.
I finally obtained the patient information brochure for such phase 1 clinical trial INSPIRE, which was consistently denied to me by all participants. The information therein, or rather the strategic lack of it and the consequential wilful misleading of prospective patients, is truly scandalous. There is no mention whatsoever of any of the previous trachea transplant experiments Birchall performed and the clinical outcome of which he likely misrepresented in order to obtain this very funding and even his current full professorship at UCL (with the help of Macchiarini, see page 43 of his CV). His described strategy of transplant preparation and implantation seems scientifically nonsensical, while suggestive of medical obfuscation and even deceit. Now that I finally received the INSPIRE patient information sheet, I understand why the consortium partners preferred in to be hidden from public scrutiny.
For the scientific and medical ethics background of this complicated case, please refer to my earlier reporting (in chronological order):
- The stem cell faith healers, or magic inside your bone marrow
- EU threatens to terminate TETRA Consortium funding for phase 2 trachea transplant trial
- Brexiting out of EU research and patient rights
Selective amnesia
My Freedom of Information Act 2000 (FOIA) request to Birchall’s employers and trial sites UCL and UCL Hospital (UCLH) in London, as well as to various funding and regulatory agencies, failed to turn up any documentation, due to confidential nature of this industrially-led clinical trial. David Hill, director of digital services at UCLH, initially chose not to act on my inquiry about the patient information brochure and consent form. He and UCLH simply pretended I never asked for INSPIRE documentation and left it as such. After I brought my complaint to the UK overarching authority, the Information Commissioner’s Office, UCLH was instructed to reply to my FOIA request within a certain deadline. Which Hill now did, with apologies and with blaming a “member of the team”. This is the document he finally shared with me, with a caveat that “the Participation Information Sheet (PIS) is currently under review” and that an updated version might be forthcoming.
Indeed it looks like the document needs some updating. Birchall somehow completely forgot all the earlier patients which were left dead or in permanent emergency care after his and Macchiarini’s trachea replacement surgeries. Back then, all their trachea transplant recipients were allegedly in grave conditions in the immediate threat of death, the interventions were presented by Macchiarini and his colleagues as compassionate case care. Macchiarini and Birchall transplanted their first “regenerated” trachea in 2008, according to all available evidence before any animal experiments took place. The operation on Claudia Castillo was hailed worldwide as a huge success and led to two publications in The Lancet (though the medical reality was different, details of the patient’s true state see here). Two more trachea transplant recipients of Birchall’s died, one lives with a permanent stent. Macchiarini’s trachea transplant technology has been declared “a failure” by the Italian National Transplant Center.

Nevertheless, these operational “successes” were most certainly used by Birchall as argument to procure GBP €2Mio for the INSPIRE phase 1 trial and its follow-up. Even before INSPIRE received its ethics vote, Birchall and his collaborators saw even more cash rolling from the EU, namely the €7 Mio funding for the advance-approved Europe-wide phase 2 clinical trial, TETRA. Meanwhile, INSPIRE is far from even beginning to recruit their four scheduled trial participants. Both INSPIRE and TETRA are officially run by the Liverpool-based company Videregen, who were also assigned a major chunk of this public funding.
Magic bone marrow cells
According to the information brochure I now procured, INSPIRE intends to recruit patients with tracheal stenosis in stable condition who receive standard medical care and whose lives are not in any immediate danger. These patients are supposed to be tricked into believing the technology was never trialled in humans:
“This is a first in human study, being done to investigate the safety and potential benefit of this novel type of tracheal replacement in patients with tracheal stenosis or malacia”.
Interestingly, the prospective INSPIRE patients will read no direct mention of the “mesenchymal stem cells” and their magic, unlike pompously listed at the NHS site. Even Birchall and his team seem to have understood that the stem cell capacities of these bone marrow cells are rather limited. Yet he wants anyway to give it another try (or four, or several hundred). Apparently to placate the patients who are distrustful towards the stem cell hype, the patient information brochure cautiously refers to “cells from your bone marrow” only:
“This trial involves a new experimental technique which uses a combination of a human trachea donated after death that has been stripped of any cells from the original donor (known as the trachea scaffold) and to which your own cells will be added. To make this new treatment, we will take cells from your bone marrow and grow and multiply them in a special laboratory. These cells will then be transferred onto the donor trachea scaffold. During a surgical procedure your own damaged trachea will be removed and the new trachea scaffold (that has been seeded with your own cells previously harvested and grown in the laboratory) will act as the replacement. As this new trachea has your own cells seeded within it, it is thought this new section will become part of your tissue”.
Thought by whom? None of Birchall’s transplanted dead tracheas ever became part of patients’ own tissue, he and Macchiarini failed to produce any trustworthy scientific or clinical evidence. In fact, in some cases the rotting transplants had to be removed, and the first patient Castillo must by now have lost her entire left lung as well, due to complications (details here). The scientific aspect of this tracheal regeneration by bone marrow cells is nil and void. Not only does it lack any experimental evidence, all the existing data, including numerous human experiments by Birchall and Macchiarini show exactly the opposite. There is no regeneration, the dead trachea carcass does not become part of patient’s tissue, the magic mesenchymal stem cells from bone marrow do not help to regenerate mucosal epithelium or blood vessels which are absolutely needed to make a trachea transplant functional.
Trust me, I’m a doctor!
To top things off, also the surgical aspect of the INSPIRE trial is questionable and misleading to the very least. This is what the professor of laryngology and throat surgeon Birchall intends to perform on patients:
“The section of damaged or diseased trachea will be removed using the rigid bronchoscopy technique you would have had at the first screening visit and the new trachea will be grafted in place. During the same operation, the trial doctor will do a surgical technique called a laparoscopy which is a surgical procedure that allows a surgeon to access the inside of the abdomen (tummy) without having to make large incisions in the skin. The trial doctor will perform a laparoscopy to take some omentum (which is fatty tissue around your intestines) and then use this to wrap the tracheal implant with what is known as an omentum wrap; this will act to support the tracheal implant. If however, you do not have enough omentum then the doctor may use as an alternative, a local muscle to wrap and support the new graft. To further support the tracheal implant a removable stent (small tube to prop open the trachea) will also be added”.
It is clear as mud what exactly Birchall announces to do to his four INSPIRE patients, even to a specialist I asked for opinion (note that the target audience of this information sheet are not surgeon colleagues, but medically naïve and desperate patients). Will a part of their trachea be removed and replaced with a “regenerated” cadaveric one, like with the previous patients? Aside of being potentially lethal, such trachea removal is “totally impossible from a technical viewpoint” by the method of rigid bronchoscopy, as explained by Pierre Delaere, professor for respiratory surgery at KU Leuven, Belgium. Delaere has been for years one of the staunchest critics of Birchall and Macchiarini (see his exclusive open letter on my site). To Delaere, it sounds that Birchall instead intends
“to endoscopically remove the stenotic mucosa and preserve the outer part of the native trachea (in an attempt to lower the morbidity and mortality). Then put dead trachea with stem cells inside and then insert the stent to keep the airway lumen open”.
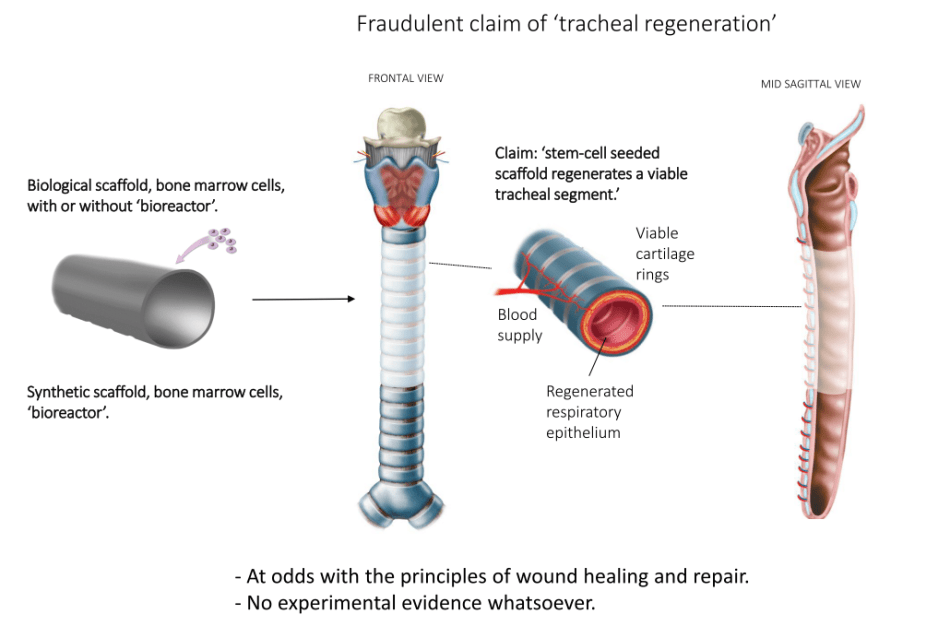
Basically, a dead decellurised donor organ sprinkled with magic cells will be stuffed inside the patient’s own. If you have difficulties understanding why, you are not alone. Obviously Britain and EU are willing to pay millions to see Birchall do this. He himself certainly does not bother to explain this charade to his patients. The carefully disguised fact is: the only medically active ingredient here will be the traditional airway-widening stent, as explained by Delaere:
“A well-known palliative treatment for a tracheal stenosis is the removal of the stenotic mucosal layer by rigid bronchoscopy and introduction of a stent to keep the airway open. The stent will give rise to several complications and therefore it will only be placed for palliation. The INSPIRE proposal uses a similar approach, but they will bring the scaffold in-between the tracheal remnant and the stent. From a theoretical viewpoint this makes no sense. The same result would be obtained with a sheet of paper in between. Both the scaffold and the paper will only make the procedure more complicated without any benefit for the patient. The stent is placed to hamper direct visualization and to make this fraudulent replacement acceptable.
Wrapping the stented trachea with a piece of non-vascularized fat (omentum) or muscle will only bring more damage to the patient. Usually, omentum is used with preservation of its blood supply and this cannot be done through a laparascopic approach”.
However, the use of the fat or muscle tissue omentum for wrapping the carcass would indicate that Birchall does plan to cut out a section of patients’ trachea after all and replace it with a transplant. Again, whether or not the patients will have their windpipe cut out or not, should not be a subject to their guessing, but clearly stated in the information brochure.
In fact, Birchall and his team do not even mention to their prospective patients any possibility that the stent implant would ever be removed for good. The removable stents will only be exchanged from time to time. Once again forgetting all their previous trachea transplant patients, the doctors declare:
“We cannot promise the trial will help you but the information we get from this trial may help improve the treatment of people with tracheal malacia or stenosis in the future. We are hoping that this trial proves that the method of using a donor trachea along with a patient’s own cells could offer patients an alternative, potentially curative treatment which could eliminate the need for repeated surgeries that have limited success. Even if this new technique is not completely curative you could still benefit from an improvement in your quality of life”.
Don’t scare off patients!
Implanting pieces of dead tissue into patients’ unsterile airway environment bears an immense risk of bacterial infection. Another source of danger is immune rejection of the transplant, because it is very difficult, if not impossible, to remove all immune-reactive foreign tissue residues even with most thorough decellurisation. The resulting inflammation can quickly kill or mutilate the trachea recipient. Birchall must know all of this very well. With alternative of either not to implant his beloved decellurised tracheas at all or simply riding it out, he apparently chose to forward the responsibility to the manufacturers of stent and trachea. The way Birchall’s team formulated their risk warnings, a rather predictable medical emergency or even death would be entirely the patients’ own fault, for agreeing to this allegedly “relatively new technique”:
“The risks associated with implanting the trachea scaffold are as follows:
- Potential for contamination of the scaffold or ability for it to develop a harmful immune response in your body. However, the donor trachea is supplied by the NHS Blood & Transplant service who provide robust compliant tissue sourcing and processing capability and it undergoes sterilisation by irradiation to make sure it is free from any germs. The donor trachea goes through a process known as “decellularisation” which means to remove all the cells from the donor trachea so that afterwards only the scaffold remains which has a low risk of developing an immune response in your body.
- The new tracheal scaffold graft will be supported by a stent that will keep the tracheal scaffold open. The stent is CE marked [self-approved by manufacturer, and not “approved as safe by the EU or by another authority”, – LS] and suitable for this use although any stent has a risk of infection and tissue damage.
- […]
- Whilst the tracheal scaffold graft integrates and fuses with the surrounding tissue [trial operators have yet to present any reliable scientific evidence for this claim,- -LS], it will remain vulnerable to infection.
- During the time taken for the tracheal scaffold to integrate with the surrounding tissue, there is the possibility you may suffer from obstruction (blockage) of your airways and as a result develop difficulty in breathing.
- There is a risk of graft failure due to infection and breakdown of its tissue. Tracheal infections will be treated with antibiotics. If the trachea graft breaks down then some of it will have to be removed and a new stent put in place and a tracheostomy (artificial opening into the trachea so that air goes in and out through a tracheostomy tube) may be required. Please discuss this point with your doctor to better understand the potential complications.
There are side effects that you may have that are not listed here and we do not know about”.
The clear and present danger of death is not mentioned anywhere in the information brochure.
Elizabeth Woeckner, head of the Philadelphia-based non-profit organisation Citizens For Responsible Care and Research Incorporated (CIRCARE), lists some past “side effects” which Birchall et al should still be able to recall:
“The consent form says the experimental intervention “is fairly new” yet this seems to sidestep the fact that the approach, if not the specific methods of decellularization and “reseeding” of the donor tracheal graft, has been used (allegedly in compassionate use cases) since 2008, and with uneven if not catastrophic outcomes. As you point out in a previous report, Claudia Castillo is hardly a success.
Mr. Birchall has first-hand evidence of catastrophic outcome in UK patient Keziah Shorten, as he discussed in an interview with Bo Lindquivst in the Swedish Television documentary Experimenten [this patient died after receiving a plastic trachea carcass, see min 36 in the film, -LS]
UK patient Ciaran Finn-Lynch lives with a stent, which is what got him into trouble and required the transplant in the first place, and so is hard to call a success [this “success” patient received a cadaver transplant, but without any “stem cell” treatment, -LS].
In 2012 UK patient Shauna Davison received a transplant and died a few weeks later, apparently of a hemorrhage [this patient died from a “regenerated” cadaver trachea, -LS].
If the investigational intervention in INSPIRE is so different from previous efforts, Birchall et al. need to explain precisely how it’s different and why the catastrophic adverse effects and deaths are unlikely to be at issue in this trial”.

Diagram courtesy of Pierre Delaere, full presentation here
The secretive bureaucrats
The patient information sheet version I received is dated at January 5th 2016. Which means, it was made after the funding for the phase 2 trial TETRA already started flowing from the EU and just two days before INSPIRE received ethics approval from South Central – Oxford A Research Ethics Committee. Mark Ryan-Daly, Research Ethics Service Manager at NHS, confirmed to me that an ethics approval as indeed given as published on January 7th 2016.
Even more peculiarly, the British Medicines and Healthcare products Regulatory Agency (MHRA) wrote to me on June 23rd 2016 that they approved the INSPIRE trial months before that patient information sheet draft was prepared, on October 2nd 2015.
Another INSPIRE partner told me something contrary to the MHRA information, once they deigned to reply to my FOI inquiry more than two months after I placed it. This was what Alison Macdonald, Board Secretary at Royal Free London, wrote me on August 1st:
“Whilst the Royal Free London NHS Foundation Trust is mentioned in the research study information the trust’s role in this study is that the Centre for Cell, Gene and Tissue Therapy, based at the Royal Free Hospital, is contracted by Videregen to manufacture the final product in its state-of-the-art facilities.
In line with EU governance of clinical trials, the Centre for Cell, Gene & Tissue Therapy will provide all of the relevant documents for Good Clinical Practice (GCP) and GMP [Good Manufacturing Practice, -LS] compliance to the Medicine & Healthcare Products Regulatory Agency (MHRA) when the formal clinical trial authorisation application is submitted by the trial sponsor Cell & Gene Therapy Catapult”.
When I asked Macdonald at Royal Free to explain whether she meant that an MHRA approval hasn’t yet even been applied for, she simply answered: “we have nothing to add to our previous responses to you”. Why does Royal Free pretend the MHRA approval is yet to be applied for? Is their GMP lab functional at all? Or maybe it is the Videregen’s trachea-regeneration technology which doesn’t work out as instructed? MHRA, which originally even refused to confirm to me the existence of their approval from October 2015, now reiterated in this regard:
“The MHRA can confirm that the information we provided on 23 June 2016 was correct. The sponsor for the Phase I trial was the Cell Therapy Catapult as part of the INSPIRE consortium. The INSPIRE consortium includes Videregen, Cell Therapy Catapult, University College London (UCL), NHS Blood and Transplant in Speke and the Royal Free London NHS Foundation Trust (this information is public). Further, the plan for the Phase II study is also in the public domain, a link to this has been provided”.
Videregen’s obscure function
Thus, INSPIRE was granted both the manufactural approval from MHRA and the NHS ethical approval for transplantations before the deceitful patient information sheet was even prepared at UCLH as a preliminary version. The two million British pounds which the governmental funder Innovate UK invested into INSPIRE in 2014 might have just as well been flushed down the toilet. At the same time, EU millions from the phase 2 TETRA trial continue rolling. The official industrial sponsor there is Videregen, which is receiving over €1 Million in funding for contributing actually nothing except of “expertise”, since the tracheas are meant to be manufactured by the Royal Free Hospital and NHS Blood and Transplant (NHSBT). This is how Wayne Lawley, Head of External Affairs at the latter institution clarified to me on July 13th their role:
“NHSBT Tissue & Eye Services are partners in the consortium, however our part of the trial is to seek donor consent and retrieve trachea. We then decellularise the trachea following Videregen’s processes and issue this to Videregen for further preparation for the clinical trial. This is the limit of our involvement in the clinical trial”.
A second GMP lab to use Videregen’s trachea regeneration technology is being funded with €900,000 by EU’s TETRA project at the Technical University Munich in Germany. Videregen therefore either has no own GMP laboratory to “grow” windpipes with magic bone marrow cells or is possibly simply afraid of being made legally accountable should future abused patients or even their bereaved families go to court for damages. In the latter case, it would be fully understandable to delegate the entire responsibility to the public-owned GMP laboratories such as the NHS Blood and Transplant and the Munich clinical centre.
Officially, Videregen is also in charge of INSPIRE, or used to be. The above presented information shows that it is the London-based Cell Therapy Catapult which somehow became the INSPIRE sponsor now. For the follow-up TETRA, Catapult might become sponsor as well. I could not find out more, Videregen’s boss Steve Bloor has been ignoring my emails since long. But if his company manufactures or manages nothing, what for does it need over one million Euro from the EU then? Some of it will surely go into Birchall’s private pocket. He is member of the advisory board to Videregen and is paid for “3 x remunerated meetings per year”. Another million Euro was assigned to a biotech service provider, TMC Pharma, whose task is to manage this currently non-existent phase 2 clinical trial. Not much to spend therefore for anyone on anything really, while the EU money continues flowing down the drain.
Making money
As it stands now, INSPIRE, TETRA as well as Birchall’s larynx-replacement trial RegenVox work solely as efficient money-milking setups, with a total of around €13 Million in British and European tax-payers’ funding money invested in them. The only good side to it: no patients will likely be ever treated with “regenerated” windpipe or voicebox transplants (unless Britain really proceeds to abolish the cumbersome patient protection regulation imposed by EU, as cheerfully announced by the new cabinet minister).
Delaere wondered aloud:
“How is it possible that well-respected doctors in the field write this kind of proposals? The only explanation is that they, for their own benefit (research money), continue to mislead both patients and the scientific community”.
The INSPIRE patient information sheet and consent form are available exclusively here.
Update 5.9.2016. The NHS Health Research Authority (HRA), which issued the ethics approval for INSPIRE, provided to me under FOIA documents which expose UCL as dishonest and the entire INSPIRE trial as highly irresponsible to the very least. David Hill of UCL shared with me previously a form dated January 2016 and insisted this to be the most recent available version. This was not the truth.
HRA provided me with a form dated May 2016, and it is different from the other one in one key aspect: no more ambiguity about what exactly Birchall et al plan to do with the patients:
“Your surgery will take place under general anaesthetic. The section of damaged or diseased trachea will be exposed and removed by using an incision within the neck or chest region (will be decided by the surgeon) and the new trachea will be grafted in place”.
Thus, the procedure is largely the same as performed by Birchall together with Macchiarini before: remove patient’s trachea and install a cadaveric one instead. According to Dr. Birchall’s own earlier, unmentioned experience, the likely outcome of this is death or severe complications for the patient.
However, the patients and their family will have difficulties suing the trial sponsor for damages. Catapult declared in the ethics approval form that they lack any insurance.

The main problem and motivation behind whether we are thinking about WB cheater’s, image duplications or trachea transplant experiments, etc. will always be research money: the need to get fast results to get fast funding.
LikeLike
Pingback: Macchiarini investigations outcome: Karolinska University Hospital – For Better Science
Pingback: Macchiarini investigations outcome: the Karolinska Institutet – For Better Science
Pingback: Macchiarini and the bonfire of greed – For Better Science
Pingback: The newly found innocence of Paolo Macchiarini – For Better Science
A pdf always has a creation date signature. I downloaded the 2 pdf, and opened them with Adobe Reader, and read Document “Propriétés”.
The consent form of May 2016 pdf was created and last modified the 05.05.2016 at 15:48:59. The first provided consent form pdf was created the 11.01.2016 and last modified 29.06.2016
• Ask for primary text editor with original date of creation, before each patient operation?
• Ask for oldest generated consent form that was used before patient signature?
A consent form should be kept for long term archiving, and only the original one should be considered as valid.
A consent form should be always for patient protection. In this case, privacy close about sample destruction after one year is not a good thing. Nobody can re-analyze samples. I can agree with this procedure. But a consent form may include something like: in case of suspicion of misconduct, fraud or scientific debate, raw data may be publicly released after high extra anonymization process provided by a licensed service. Open data should help to stop such clinical failures.
LikeLike
Pingback: Claudia’s trachea – For Better Science
Pingback: Collapse of Biotrachea, or how Macchiarini’s greed saved human lives – For Better Science
Pingback: Business interests trump patient safety in EU-funded trachea transplant trial – For Better Science
An amazing piece. I am aghast that this is happening again. Has this reached mainstream media?
LikeLike
Pingback: Birchall’s trachea transplant trial at UCL suspended by health authorities – For Better Science
Pingback: The vexatious life-saving question of cadaveric tracheas – For Better Science
Pingback: Ciaran’s success story – For Better Science
Pingback: Keziah’s deadly trachea transplant: UCL hospital’s secret – For Better Science
Pingback: Unpublished Macchiarini manuscript confirms 5 forgotten trachea transplant patients, Jungebluth’s surgery practice in Italy – For Better Science
Pingback: EU commemorates dead patients of Macchiarini & Birchall with a phase 2 trachea transplant trial TETRA – For Better Science
Pingback: Alexander Seifalian, UCL’s Persian Scapegoat – For Better Science
Pingback: Martin Birchall’s shaky road to mass trachea transplanting – For Better Science
Pingback: Tissue-engineered tracheas: an assessment of the scientific, clinical and ethical implications – For Better Science
Pingback: Image reuse, the new low of UCL trachea transplanter Martin Birchall? – For Better Science