Swedish investigation spoils Macchiarini cover-up at Lancet
NPOF, again and again: “Philipp Jungebluth and Paolo Macchiarini guilty of research misconduct” Lancet: “Paolo Macchiarini is not guilty of scientific misconduct”
By Leonid Schneider, on research integrity, biomedical ethics and academic publishing
NPOF, again and again: “Philipp Jungebluth and Paolo Macchiarini guilty of research misconduct” Lancet: “Paolo Macchiarini is not guilty of scientific misconduct”
“The board’s conclusion is therefore that the deviations regarding figure 2b and 2c in article 1 constitute serious deviations from good research practice”
Schneider Shorts 23.06.2023 – Italian killer surgeon gets sentenced to 2.5 years in Sweden, Harvard’s honesty expert exposed as fraudster, German university refuses to investigate papermiller, with ACS being naughty again, anti-aging breakthroughs in MDPI, and finally, an accidental sickness in Wuhan in November 2019.
Four years after Ashutosh Tiwari’s scamferences and research fraud were exposed, his impressive-sounding yet fictional “International Association of Advanced Materials”, or IAAM, still opens doors, hearts and wallets.
An academic dynasty of bad cancer research.
“I take issues of research integrity very seriously and shall of course review the concerns posted on PubPeer to establish whether there are any issues that need to be addressed.” Stephen P Jackson.
“The Board assesses that there are no scientifically acceptable explanations for why the notified researchers have fabricated research results in the manner that has occurred in the notified articles. Raw data also does not support the reported results. [..]
In summary, the Board finds therefore that the notified researchers have been guilty of misconduct in research.”
Ashutosh Tiwari’s scamference activities continue. Now the University of Magdeburg in Germany is very excited about a medal from the International Association of Advanced Materials.
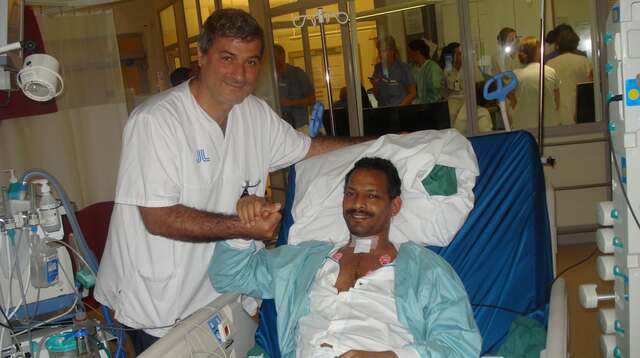
Macchiarini’s victim Paloma Cabeza speaks out again, fearing she doesn’t have much time left. She appeals to the Swedish prosecutor for justice in the deadly trachea transplant scandal.
Swedish prosecutor opened a criminal indictment against Paolo Macchiarini. The scandal surgeon will have to stand court trial for all 3 deadly plastic trachea transplants he performed at Karolinska.