Clare Francis said:
“Ted Hupp and Kathryn Ball may very well feel like kissing David Argyle on both cheeks.”
That’s because when the pseudonymous image integrity sleuth heard of David Argyle’s bullying and the cover-up by the University of Edinburgh in Scotland, UK, Clare Francis started to screen Argyle’s papers for image irregularities. And soon stumbled over Argyle’s postdoc Lisa Pang, who did her PhD at the Cancer Research UK (CRUK) Edinburg Centre and MRC Institute of Genetics & Molecular Medicine under Ted Hupp, the US-born Edinburgh professor, who proved to be Argyle’s collaborator.
Right away, Clare Francis found a treasure trove of questionable research papers from Hupp’s lab. From Hupp’s papers, the sleuth stumbled upon another Edinburgh professor, Kathryn Ball, and her lab’s papers proved a treasure trove as well!
Hupp and Ball are in fact a husband-and-wife team, they both started together as PhD students at the Michigan State University in USA, moved together as postdocs to Scotland, both to the University of Dundee lab of Sir David Lane (who is now at Karolinska Institutet in Stockholm, Sweden, and not replying to emails). Hupp and Ball then both became professors at the same Institute of Genetics and Cancer by CRUK and MRC in Edinburgh. Both work on cell cycle regulation, he on p53, she on the p21 protein. Both sit on the scientific advisory board of the Scottish biotech startup ILC Therapeutics which seeks to cure with interferon drugs various diseases including COVID-19. Both published, together and separately, Photoshopped stuff they should be deeply ashamed for, and both remained silent when I contacted them.
I want to celebrate this power couple.
Here is how Clare Francis was led to playing the Hupp and Ball game. From Argyle to Pang to Hupp to Ball.
Lisa Y. Pang, Mary Scott , Richard L. Hayward , Hisham Mohammed , C. Bruce A. Whitelaw, Graeme C.M. Smith , Ted R. Hupp p21(WAF1) is component of a positive feedback loop that maintains the p53 transcriptional program Cell Cycle (2011) doi: 10.4161/cc.10.6.15012
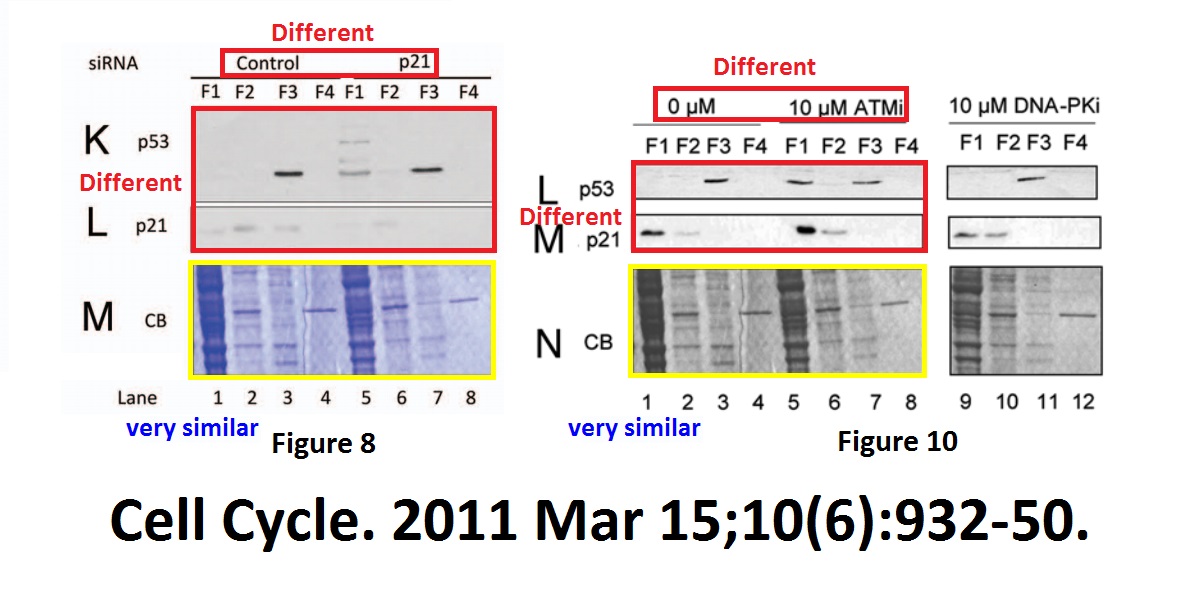
A Ponseau S blot image, “accidentally” reused, once in colour, once black-and-white, for two unrelated experiments. Why couldn’t the correct loading control be used? A mystery.
Here a paper by Hupp and Ball, rather old, raw data unavailable, but even Hupp agrees the gel is falsified:
Ashley Craig , Mary Scott , Lindsay Burch , Graeme Smith , Kathryn Ball, Ted Hupp Allosteric effects mediate CHK2 phosphorylation of the p53 transactivation domain EMBO reports (2003) doi: 10.1038/sj.embor.embor901
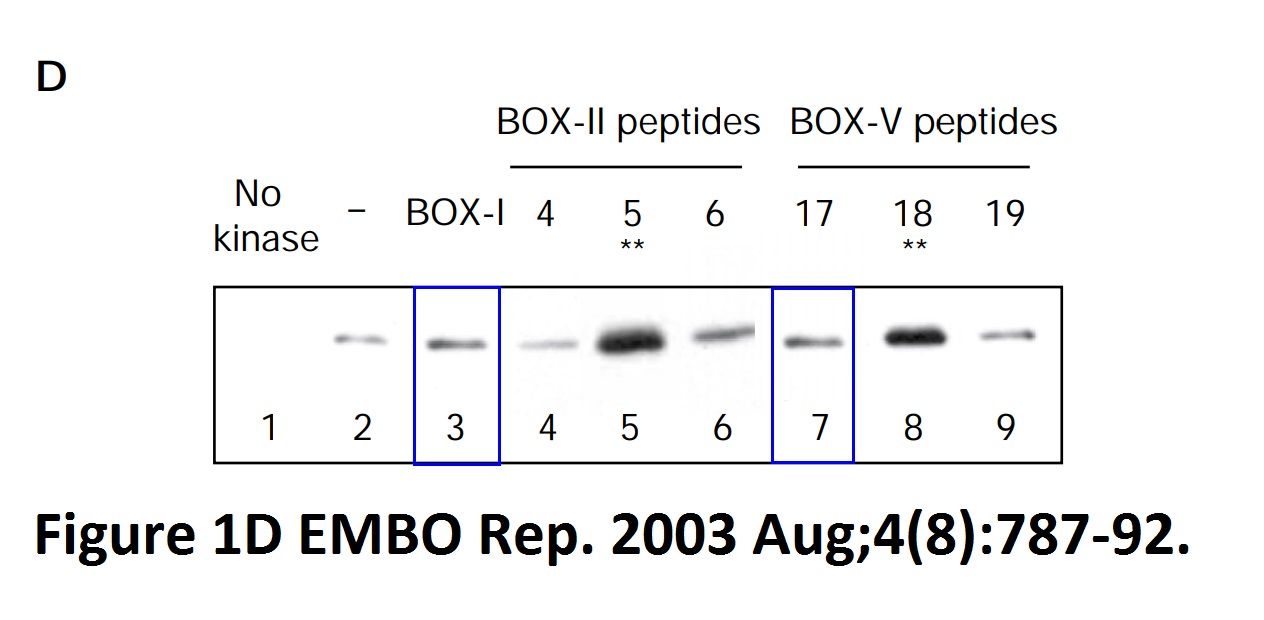
Hupp declared on PubPeer in June 2021:
“The authors think that the apparent similarity resulted from image crop/paste from different gels to condense key data from Fig 1d of the same paper, but the original data are no longer available for analysis. This particular image does not affect the overall data interpretation or the conclusions from the study; regarding stimulation of the chk2 kinase using synthetic peptides (named 5 and 18) that are the best binders to chk2 (fig 1d) so that there is a correlation between binding of peptides to chk2 and chk2 stimulation; the same chk2 stimulatory peptides were also reproduced in other data throughout the paper in question and these same peptides were also used as kinase stimulators reproduced in a follow up study (10.1128/MCB.01595-06) and towards another regulatory protein (10.1016/j.molcel.2006.05.029 ).“
Right-ho. It’s OK to fabricate results if you can reproduce them in your later papers. The first referenced study from Hupp’s lab has a blot duplication and a digitally assembled Chk1 gel, maybe the authors disapproved of the original results.
Ashley L. Craig , Jennifer A. Chrystal , Jennifer A. Fraser , Nathalie Sphyris , Yao Lin , Ben J. Harrison , Mary T. Scott , Irena Dornreiter , Ted R. Hupp The MDM2 ubiquitination signal in the DNA-binding domain of p53 forms a docking site for calcium calmodulin kinase superfamily members Molecular and Cellular Biology (2007) doi: 10.1128/mcb.01595-06
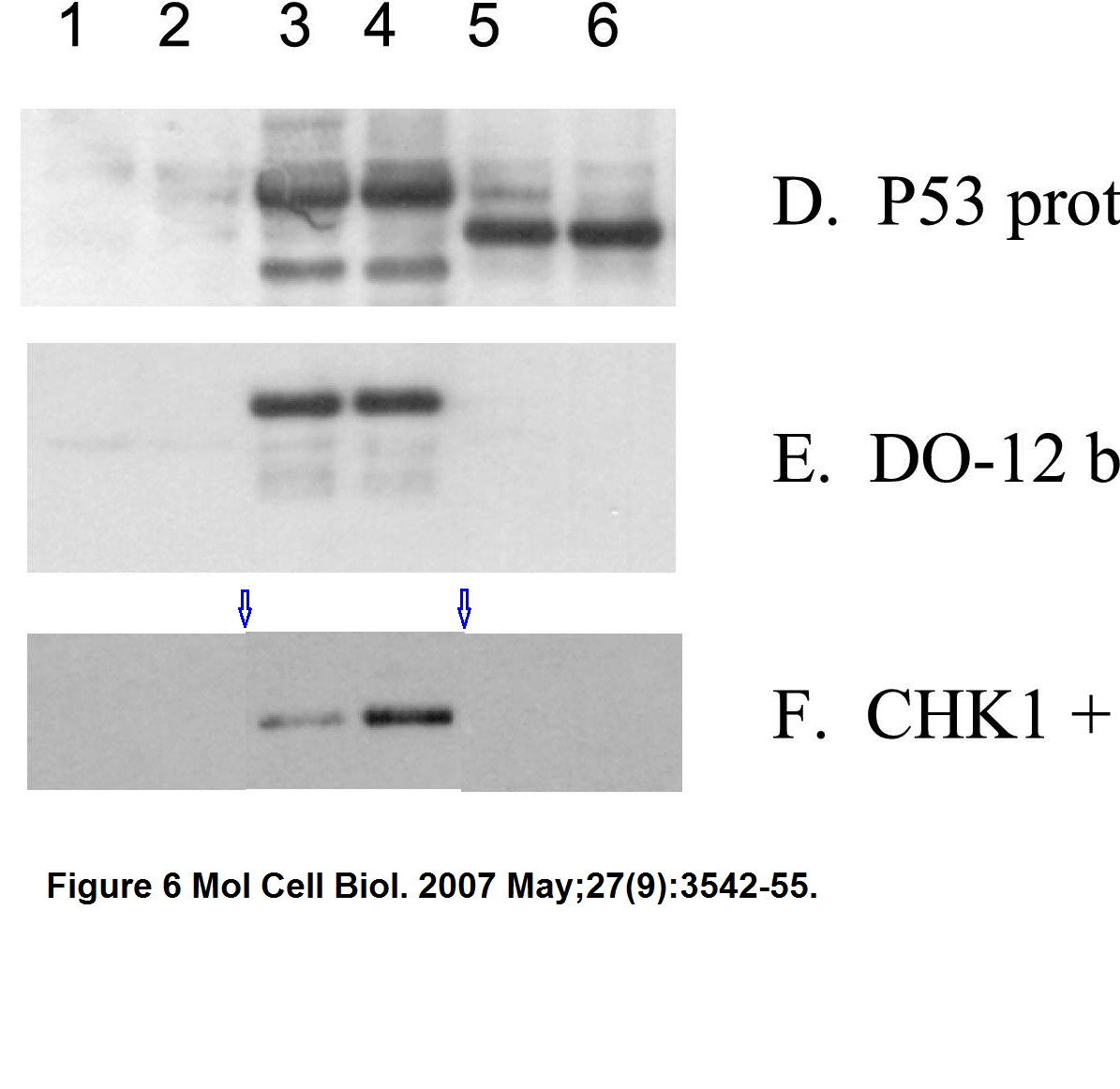
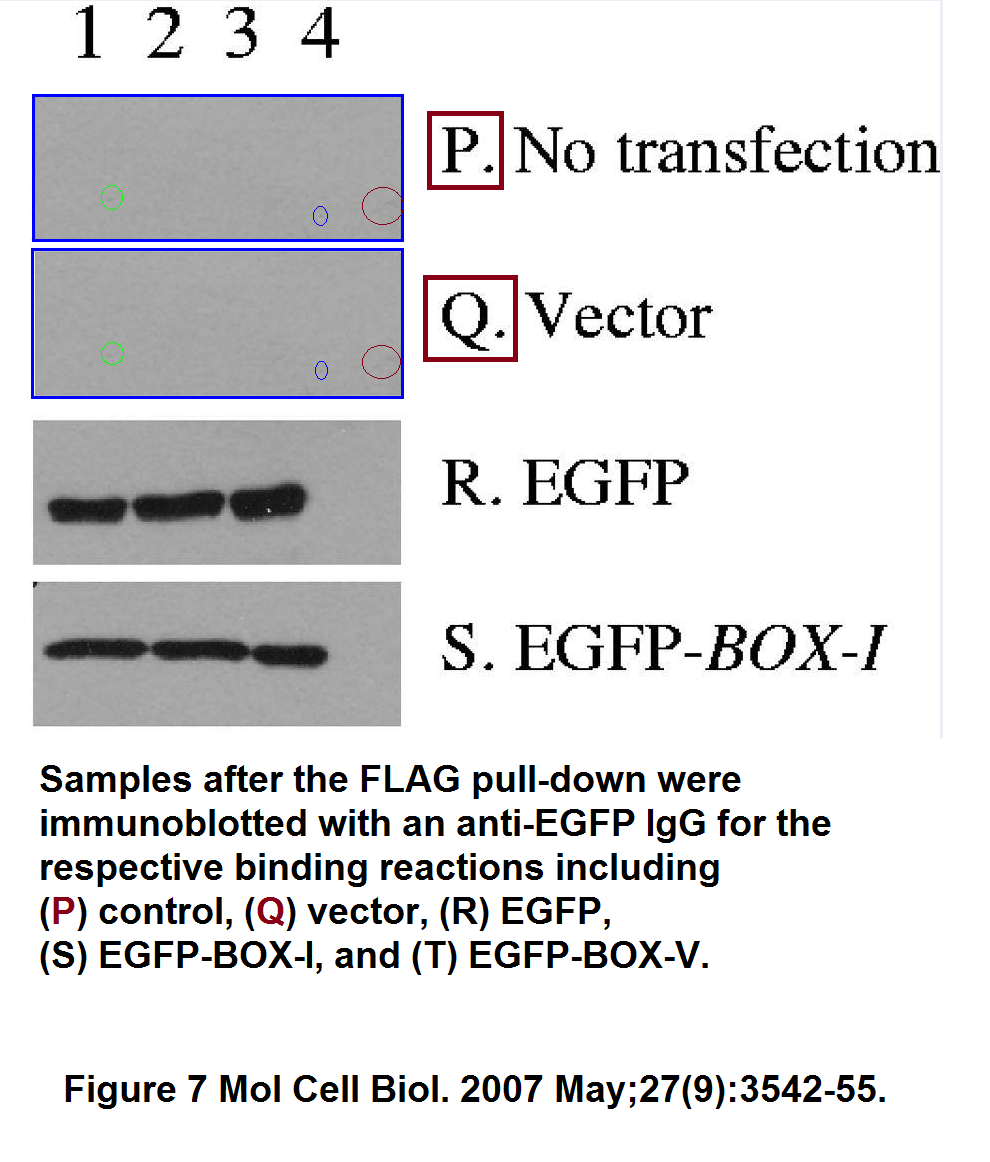
The second referenced paper by Hupp and Ball (Wallace et al 2006) hasn’t been yet commented upon on PubPeer, which under the circumstances of this story is no proof the paper is kosher.
What to make of the next paper though? Does any of this, uhm, reproducibility, affect the conclusions?
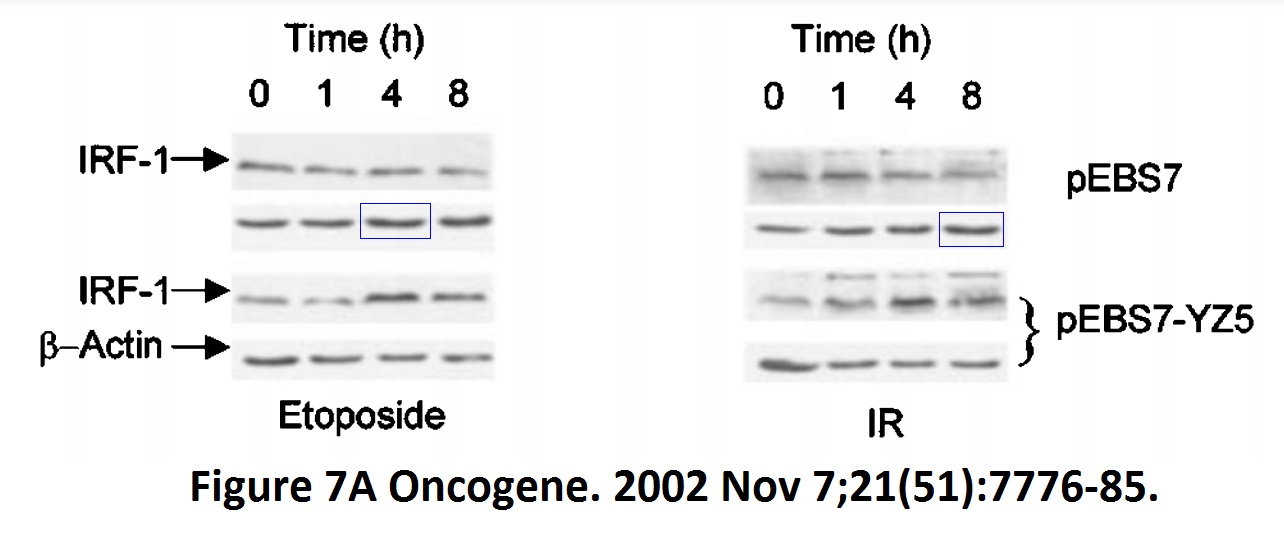
David Dornan , Mirjam Eckert , Maura Wallace, Harumi Shimizu, Eleanor Ramsay , Ted R. Hupp, Kathryn L. Ball Interferon regulatory factor 1 binding to p300 stimulates DNA-dependent acetylation of p53 Molecular and Cellular Biology (2004) doi: 10.1128/mcb.24.22.10083-10098.2004
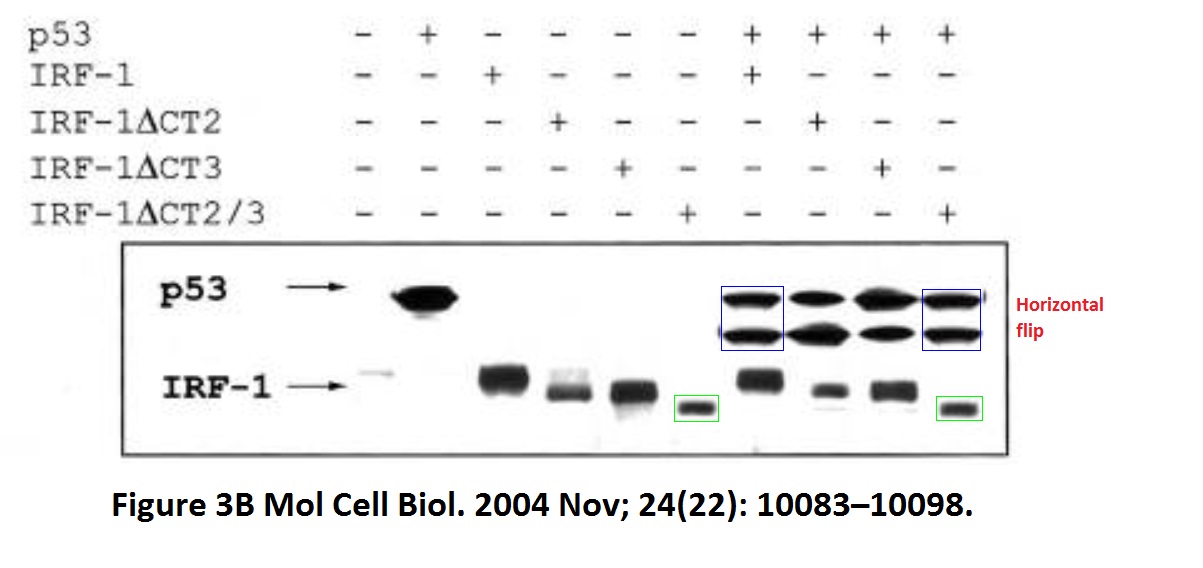

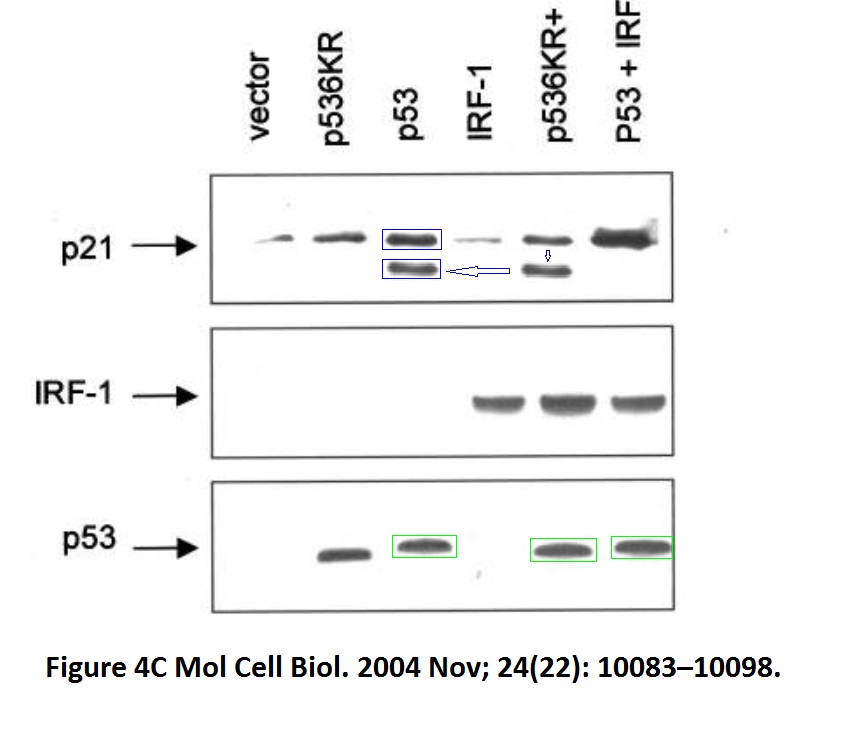
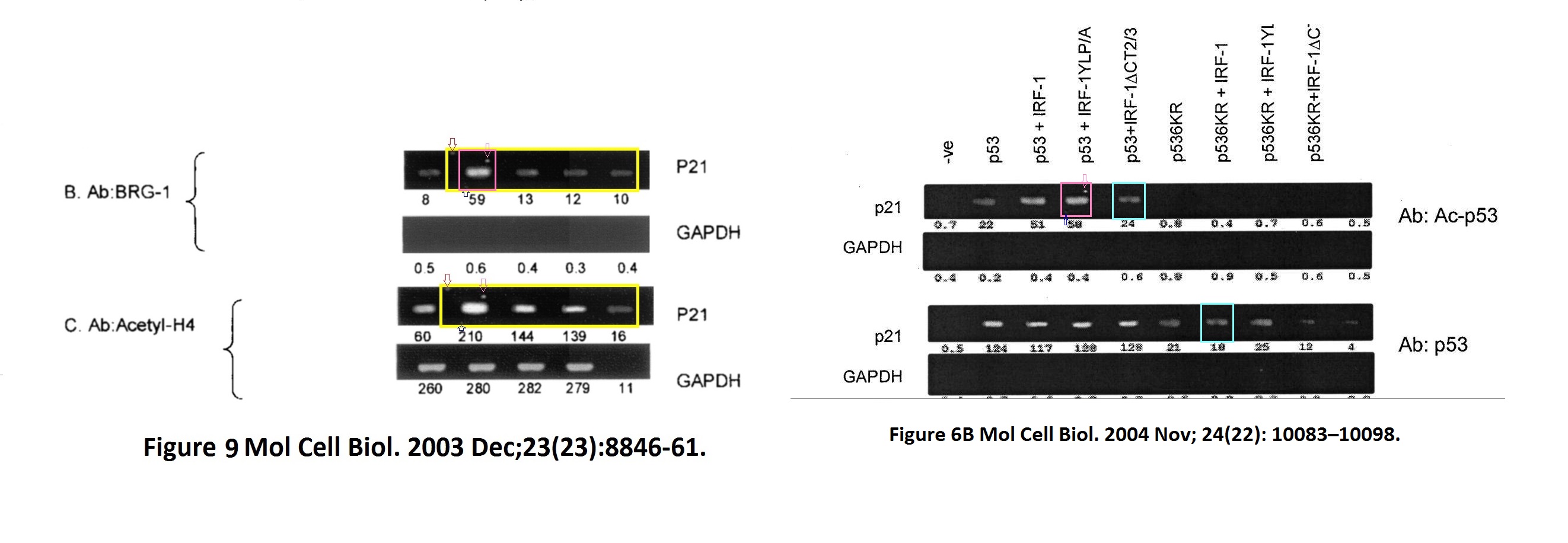

The first author David Dornan is Hupp’s former PhD student and now Chief Scientific Officer of Bolt Therapeutics, where he “is working to harness the exciting technologies at Bolt to discover and develop therapeutics that may exploit myeloid cell biology to generate a robust anti-tumor immune response.” He also didn’t reply to my email, and neither did Bolt.
Here is another example of what Dornan learned in the Hupp lab:
David Dornan , Harumi Shimizu , Lindsay Burch , Amanda J. Smith , Ted R. Hupp The proline repeat domain of p53 binds directly to the transcriptional coactivator p300 and allosterically controls DNA-dependent acetylation of p53 Molecular and Cellular Biology (2003) doi: 10.1128/mcb.23.23.8846-8861.2003
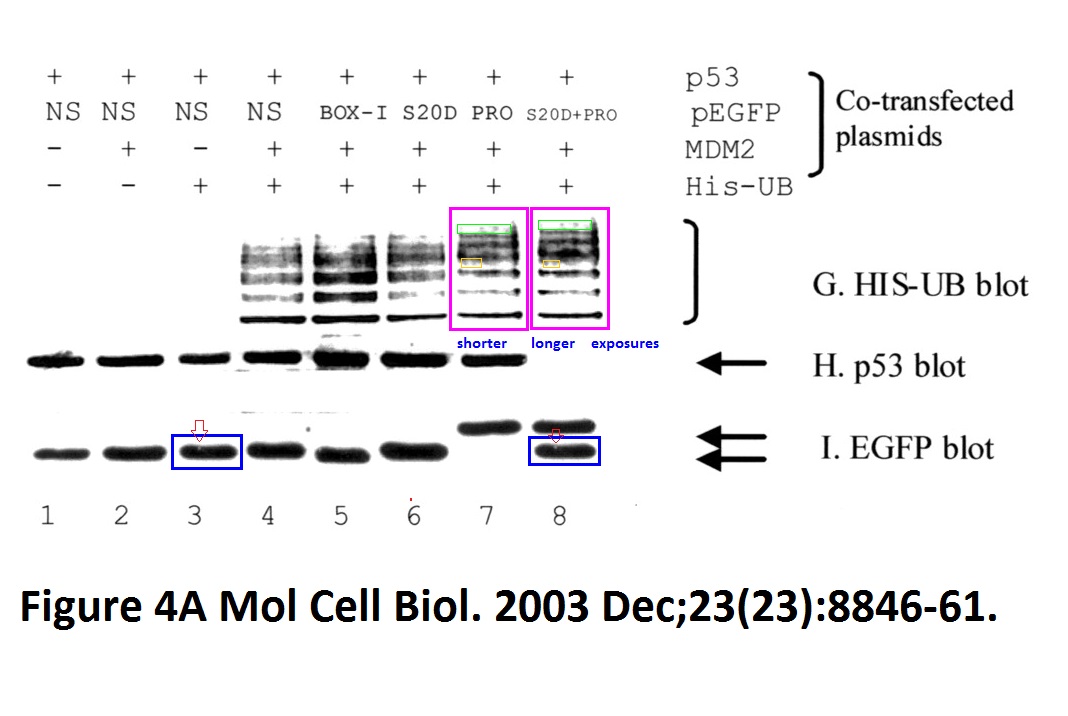
Hupp replied to this finding in June 2021 on PubPeer that yes, the gel is digitally falsified, but it was just a control:
“The authors feel that the apparent similarity likely resulted from mixing images cropped from different gels to assemble the final figure, but the original data could not be retrieved for analysis. This particular image was a negative control and does not affect the overall data interpretation or the conclusions from the study regarding interaction of p300 to proline-derived peptides, including deletion of the proline motifs of p53 that attenuate p300 functions (fig 5/6/7) and proline repeat domain protein promoter recruitment by chromatin ip (fig 8/9). The ability of proline repeat peptides to bind to p300 was also established by other studies (doi; 10.1074/jbc.M210696200; DOI; 10.1074/jbc.275.3.2115; DOI; 10.1016/j.jmb.2005.12.026; 10.1038/onc.2009.71).“
Basically, controls are also there to prove that you didn’t fudge up your experiment to get the result you wanted. Hupp and his mentee Dornan failed the test.
In any case, more was found in the Dornan et al 2003 study:
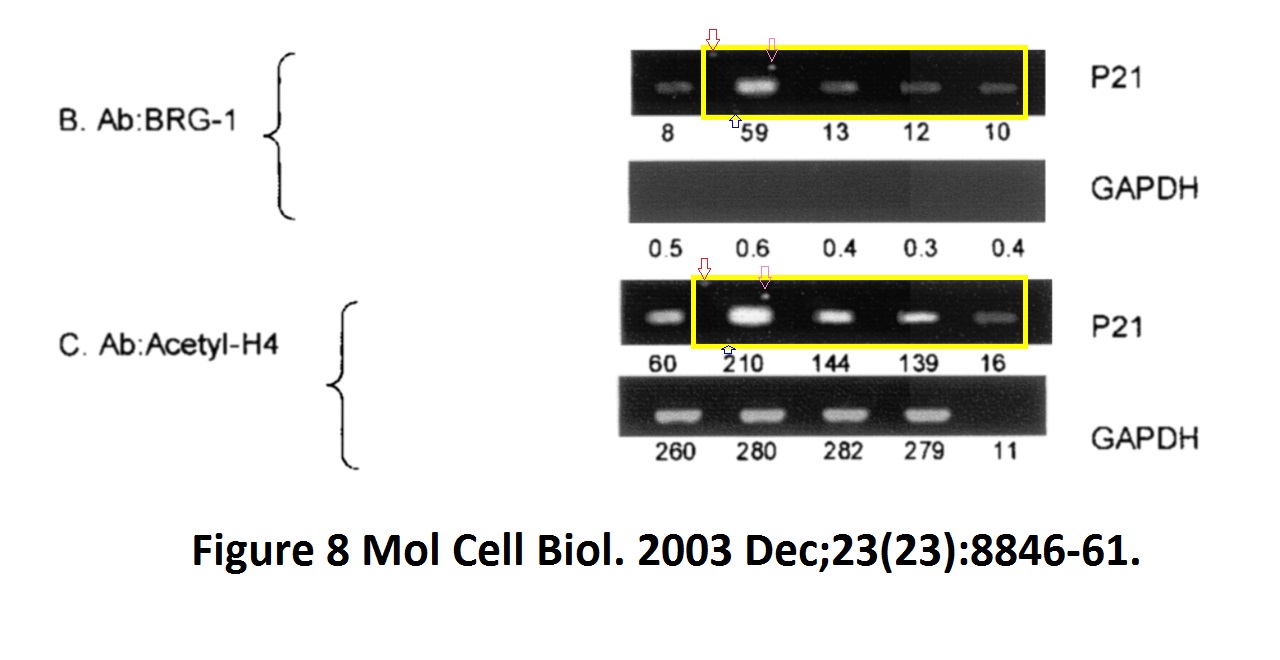
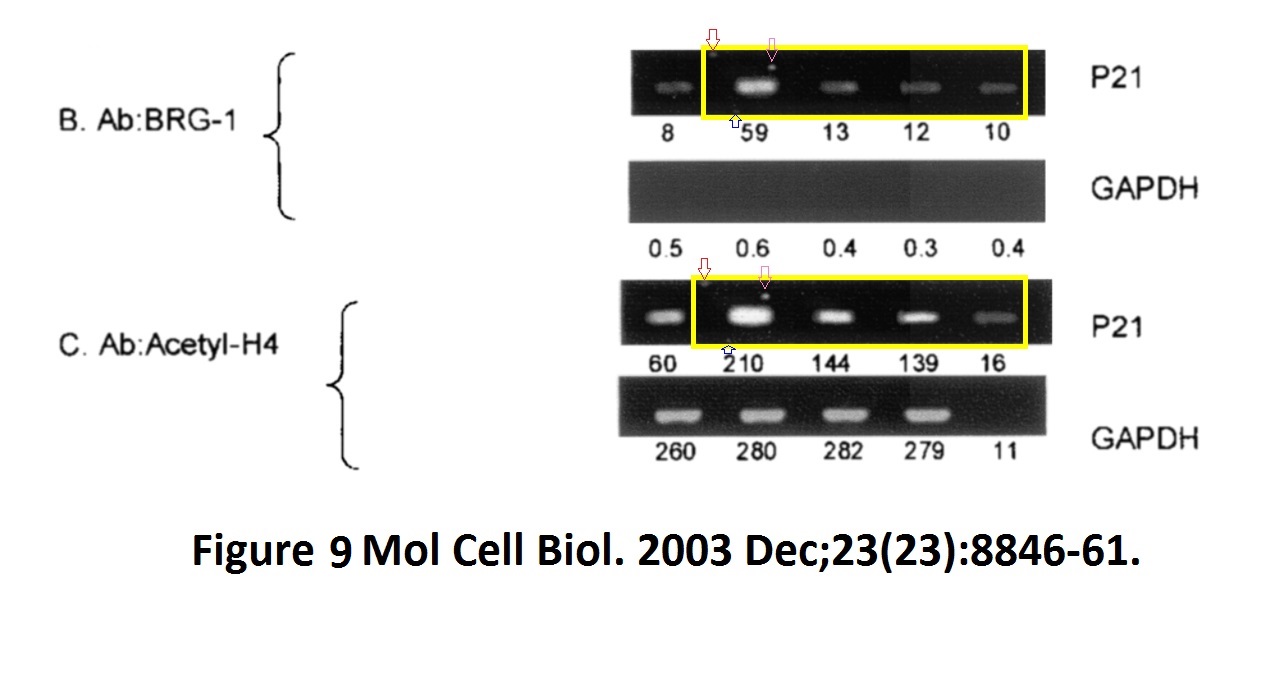


There, Hupp didn’t reply anymore. Also, that Dornan et al 2003 paper shares western blots bands with another Dornan et al 2003 paper:
David Dornan , Harumi Shimizu , Neil D. Perkins , Ted R. Hupp DNA-dependent acetylation of p53 by the transcription coactivator p300 Journal of Biological Chemistry (2003) doi: 10.1074/jbc.m211460200


Neil Perkins, now that name rings a bell. Alarm bells, actually. In 2014, Perkins, then with the Wellcome Centre of University of Dundee, retracted 4 papers, and explained in this regard to Retraction Watch that a) his postdoc Benjamin Barré did it and b) he, Perkins, exposed this fraud all by himself (OK, together with Barre’s PhD mentor). However, Perkins had already at that time several other papers flagged on PubPeer, and those were not co-authored by Barré.
Perkins’ academic career trajectory is interesting. Having spent 12 years as Dundee professor, he suddenly had enough and went to University of Bristol in 2008, but stopped being professor there after less than 2 years. Since 2010, Perkins is with University of Newcastle, where he is now deputy dean of biosciences. Meaning, all misconduct allegations in the faculty go through Perkins’ desk.
Between Hupp and Ball, the husband is the bigger academic heavyweight. In 2017, Hupp (together with a French colleague) received €9.5 Million from the Foundation for Polish Science to set up the International Centre for Cancer Vaccine Science at the University of Gdansk. Hupp runs the Centre’s Twitter account, here is his dear collaborator, the bully Argyle, visiting (right):
Any moment now will Dr Hupp’s Photoshop technology produce a cancer vaccine. Now let’s have a look at what Dr Ball’s lab enriched the science with.
Mary T Scott , Angela Ingram , Kathryn L Ball PDK1-dependent activation of atypical PKC leads to degradation of the p21 tumour modifier protein The EMBO Journal (2002) doi: 10.1093/emboj/cdf684
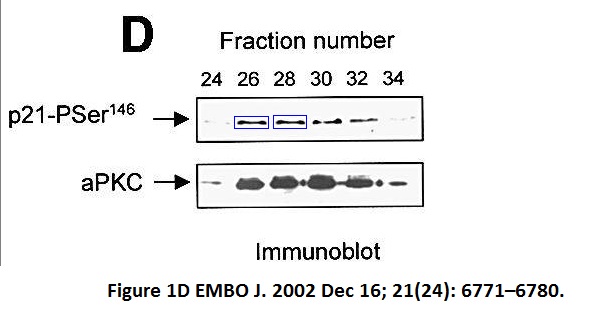
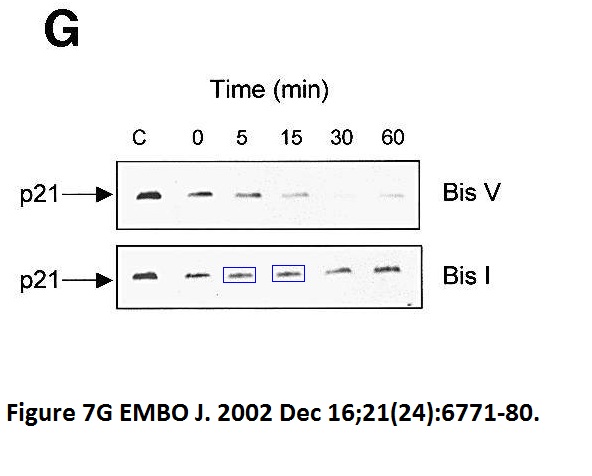
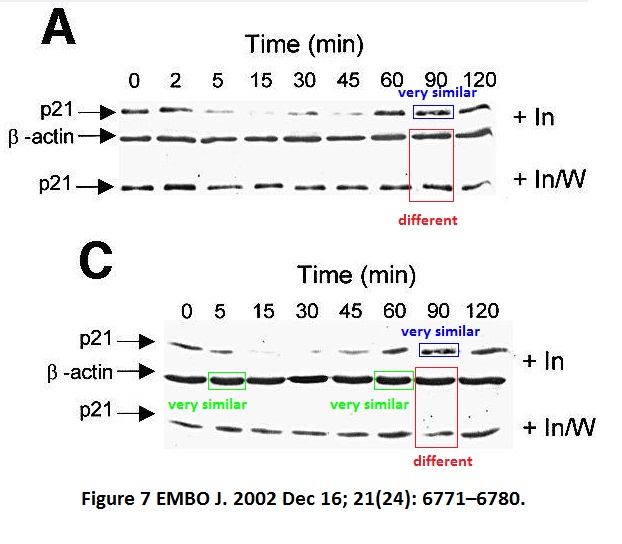
Maura Wallace used to head a unit at University of Edinburgh, a local newspaper once celebrated her receiving a charity donation for her cancer research. The charity money was put to good use.
Maura Wallace, Kathryn L. Ball Docking-dependent regulation of the Rb tumor suppressor protein by Cdk4 Molecular and Cellular Biology (2004) doi: 10.1128/mcb.24.12.5606-5619.2004

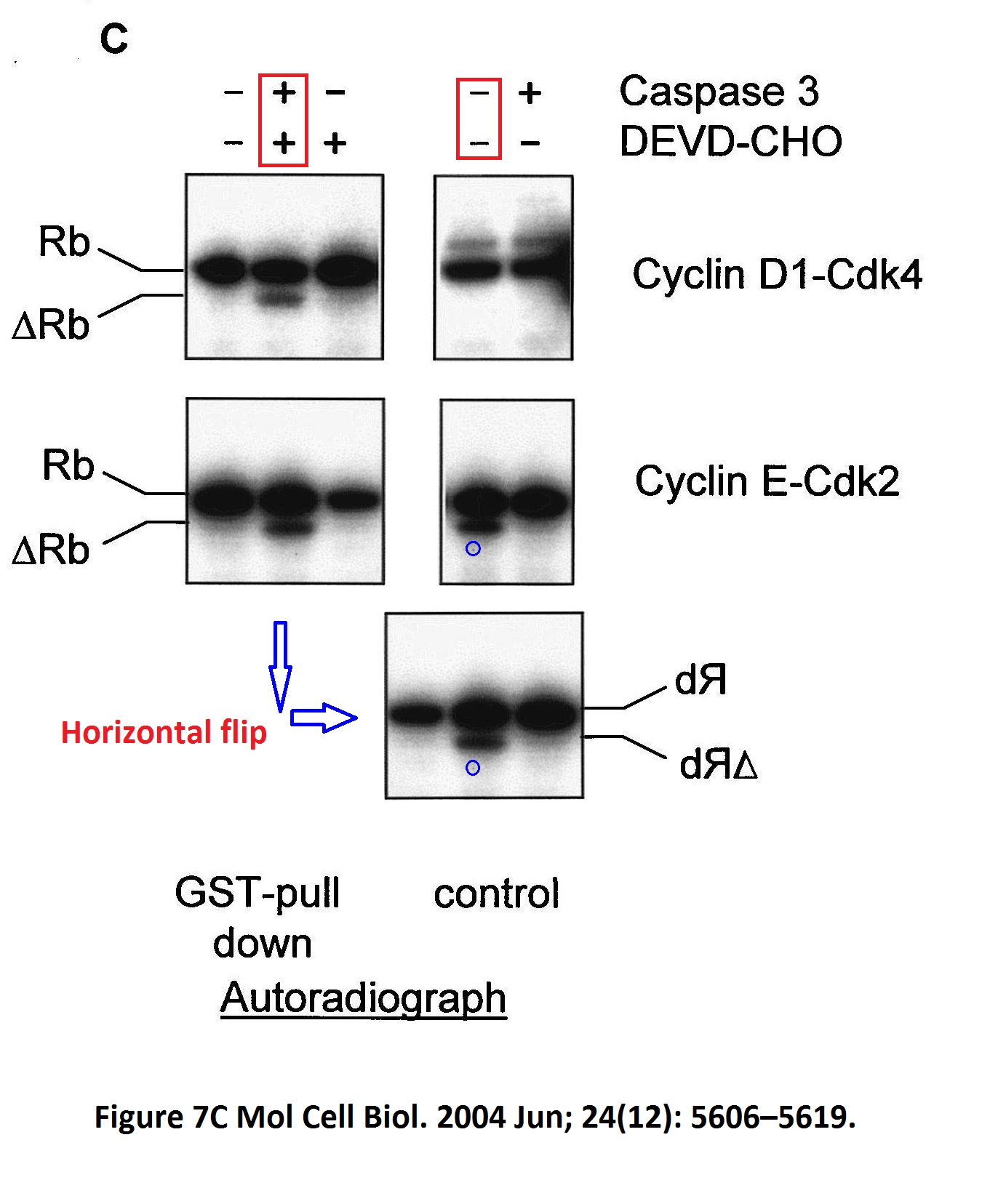


Well, at least we were spared Ball explaining on PubPeer that none of that data fabrication affected any conclusions, as her husband used to. Here a bit more of the Hupp and Ball game, courtesy of Clare Francis:




Now, you might say, well this all looks very serious. The University of Edinburgh sure won’t like it, a research misconduct investigation is probably already under way in full steam.
What are you, born yesterday and living the Moon? University of Edinburgh is not investigating anyone, in fact they refuse to admit any notifications of suspected research misconduct so they won’t have to open any investigations. Not on the dean of the veterinary faculty, the bully Argyle, and most certainly not on Hupp and Ball. Although, Edinburgh did investigate someone once, because the external pressure got too high and because the main perpetrator was a Bulgarian woman.
The senior lecturer Irina Stancheva was dismissed. All her partners in data forgery were absolved in full and enjoy their prestigious professorships at prestigious British universities on the virtue of being British males. Even Stancheva’s retractions were coordinated as not to hurt these important gentlemen.
The research integrity guidelines at Edinburgh state:
“Professor Jonathan Seckl, Vice-Principal Planning, Resources and Research Policy, is the main responsible contact for Research Misconduct. Professor Seckl delegates responsibility for research misconduct for each College.”
Seckl never replied to my notifications of course which bureaucratically means I never submitted anything. It is worth mentioning that Seckl is a collaborator of Moshe Szyf (here a questionable paper Weaver et al 2004), and also, the Edinburgh Vice-Principal can sympathise: his brother and Imperial College London professor Michael Seckl has a nice PubPeer record, including a retraction.
That retraction Michael Seckl earned together with Julian Downward, a somewhat controversial professor at The Crick. On the occasion of his other retraction, Downward explained to Retraction Watch that, let me check, yes, “Following a series of exhaustive London Research Institute (LRI) internal and external investigations, it was concluded” that also Dr Downward became a victim of a fraudulent foreigner he nourished at his bosom. A sad and tragic fate befalling many white British men of science.
There are no dirty foreigners to blame in our Hupp and Ball game (except yours truly), so you must understand why Michael Seckl’s brother Jonathan and his Edinburgh colleagues keep disregarding my email notifications. Plus, the Institute of Genetics and Molecular Medicine where Hupp and Ball work is part of College of Medicine and Veterinary Medicine, and its head Moira Whyte has vigorously defended Argyle from bullying complaints, including from a faculty member dying of cancer.

So you see, we up some hardened and ruthless opposition in Edinburgh. Don’t get fooled by the pictures of cuddly animals they deploy. These people will bite your head off if you get too close.
But evil as I am, I found a way in.
Edinburgh may refuse to admit my notification of suspected research misconduct, but I also contacted the University of Dundee, where the Hupp and Ball game originally began in the lab of Sir David Lane. I did this recalling how in 2016 Dundee cracked down on the science fraud by their former lecturer Robert Ryan. Ryan was made to resign, his funding withdrawn, and that despite Ryan’s alma mater University College Cork in Ireland aggressively defending his research, determined to protect at all costs their professor and Ryan’s mentor (and co-author) Maxwell Dow.
So I sent the PubPeer evidence to the University of Dundee, Right away, my notification was acknowledged with this message from Paul Davies, one of university’s research integrity leads:
“Thank you very much for raising this with the School of Life Sciences Research Integrity Group. As an Institution, we are proactively engaged in Research Integrity matters: we deal with the underlying causes of reproducibility issues in research and thoroughly evaluate all concerns and allegations of research misconduct involving our current and former researchers. We subscribe to PubPeer alerts (in fact we worked with PubPeer to enable searching by institution) and react to each one involving the University of Dundee. We are aware of the published comments you highlight and we are currently looking at these and will evaluate them thoroughly.“
Davies then announced to inform “all stakeholders, including the Journals, to establish the facts.” Which will first and foremost include the University of Edinburgh. Maybe Jonathan Seckl will have to grudgingly allow an investigation after all. Expect no misconduct findings though!

Nobody in Edinburgh is answering emails, and neither does Hupp’s and Ball’s mentor Sir David Lane in Stockholm. He should though, even one of his own papers is affected.
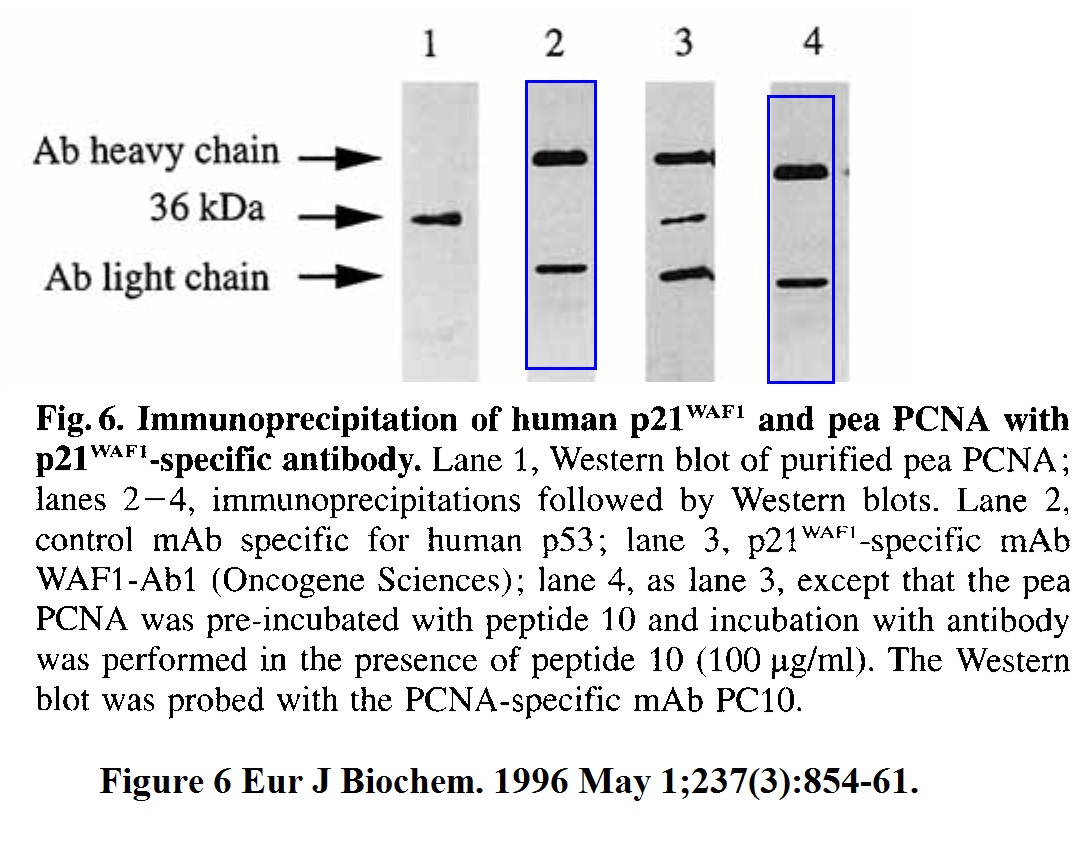
Kathryn L. Ball, David P. Lane Human and plant proliferating-cell nuclear antigen have a highly conserved binding site for the p53-inducible gene product p21WAF1 European Journal of Biochemistry (1996) doi: 10.1111/j.1432-1033.1996.0854p.x


https://www.hindawi.com/post/defending-science-and-defending-scientists/
“Outrage about outrage is less productive than reflecting on what led to the outrage in the first place.
This can be seen too when the criticism is coming from within science itself. Problems arise when we are not self-critical and we characterise questioning as attacks. It is not pleasant to be on the receiving end of what some have dubbed “data thugs”, but there is a reason why some researchers have had to retract articles and step down from posts – and it is not personal animosity. It is not only those outside science who may have agendas, but also scientists themselves such as those who have been co-opted by industry.”
LikeLike
https://www.beatson.gla.ac.uk/About/governance.html
Governance
The Beatson Institute for Cancer Research is a Registered Scottish Charity (no. SCO06106) and a company limited by guarantee (registered in Scotland no. 84170).
The Institute is governed by its Board of Directors who are the trustees of the charity. The Board is ultimately responsible for all aspects of the Institute, including its scientific strategy, operational policies, regulatory compliance and financial stewardship and accountability. On a day-to-day basis, many of these responsibilities are delegated to the Institute’s Management Team.
Board of Directors
Prof John Iredale (Chair)*
Pro Vice Chancellor, Health and Life Sciences, University of Bristol
https://research-information.bris.ac.uk/en/persons/john-p-iredale
“John Iredale is Pro Vice Chancellor Health at the University of Bristol. He holds the chair of Experimental Medicine, and his areas of leadership and responsibility are for the research, teaching and management strategy for the Schools of Medicine, Veterinary Medicine, Dentistry and Biomedical Sciences across the relevant Faculties at the University of Bristol. He also holds Honorary Consultant contracts with the North Bristol NHS Trust and the University Hospitals Bristol Foundation Trust. Previously Professor Iredale was the Regius Chair of Medical Science, Dean of Clinical Medicine and Vice Principal Health Services at the University of Edinburgh where he led the Medical School. He has previously held the Chair of Medicine in Edinburgh (2006-2013), the chair of Medicine at the University of Southampton (2004-2006) and a Personal Chair in Hepatology (2000-2004).”
Expression of Concern
https://pubpeer.com/publications/AAFF7A25AFBCD1D9A029D471EFB3EA
https://ajp.amjpathol.org/article/S0002-9440(17)30974-4/fulltext#relatedArticles
Much more similar than expected
https://pubpeer.com/publications/61F294FC188B16FA980CB03704FEAD
LikeLike
https://www.beatson.gla.ac.uk/About/scientific-advisory-board.html
Scientific Advisory Board
The Institute’s Scientific Advisory Board meets every 18 months to discuss our strategy and progress with the Director, Deputy Director and other members of the senior management team.
Scientific Advisory Board
Margaret Frame (Chair)
University of Edinburgh, UK
https://pubpeer.com/search?q=margaret+frame
LikeLike
It’s the U.K. so you mustn’t say anything. Spread of problematic publications during the period you become established.
LikeLike
When I read Margaret Frame’s replies to comments on Pubpeer I get the impression that Margaret Frame
has a poor grasp of physics, for example not understanding that the same blot for one protein cannot be compared with more than one blot of another another protein, and does not understand the rules which forbid publishing the same data more than once. Not understanding physics (the real world) and not understanding the rules help you get on as you can publish more than than your data would ordinarily allow. One of the benefits of being thick and ignorant is that you fly upwards.
LikeLike
https://pubpeer.com/publications/F9B2C5E784EBA863D4574002FAADD6#11
“Thank you for identifying these data discrepancies in our study. The University of Dundee research integrity team is analyzing the data at this time and we will post a conclusion of the review, and possible actions that will be taken, once the evaluation has been completed.”
https://pubpeer.com/publications/41D51C3BFAEC7E3A69D1A04C382C8B#6
“Thank you for identifying these data discrepancies. The University of Dundee research integrity team is now analyzing the data and we will post a conclusion of the review, and possible additional actions, once the evaluation has been completed.”
https://pubpeer.com/publications/A57F4F29D56A29C25C82CFB9E721FE#7
“Thank you for highlighting this data duplication query in fig 3C. The University of Dundee research integrity team is now analyzing the data and we will post a conclusion of the review, and possible additional actions, once the evaluation has been completed.”
LikeLike
Do we misjudge William McGonagall? The worst poet in the English language? Perhaps he was right all along.
“Good people of Dundee, your voices raise,
And to Miss Baxter give great praise;
Rejoice and sing and dance with glee,
Because she has founded a college in Bonnie Dundee.”
LikeLike
https://pubpeer.com/publications/5B3B8BBC36B54171AB1675DC9672FF#6
Thank you for highlighting your concerns. We have taken this seriously and have asked the University of Dundee research integrity adviser’s for their opinion. We will post a response on PubPeer once an evaluation has been completed.
https://pubpeer.com/publications/237D18C38399F408830D40F3E21FD1#8
Thank you for highlighting your concerns. We have taken this seriously and have asked the University of Dundee research integrity adviser’s for their opinion. We will post a response on PubPeer once an evaluation has been completed.
https://pubpeer.com/publications/363971ABAA64CA2D9EF537B6764C99#2
Thank you for highlighting your concerns. We have taken this seriously and have asked the University of Dundee research integrity adviser’s for their opinion. We will post a response on PubPeer once an evaluation has been completed.
https://pubpeer.com/publications/16180CE2482CFEBD624AEF605B1D20#7
Thank you for highlighting your concerns. We have taken this seriously and have asked the University of Dundee research integrity adviser’s for their opinion. We will post a response on PubPeer once an evaluation has been completed.
https://pubpeer.com/publications/8D0C92051F0A0567B4E409C1F05707#2
Thank you for highlighting your concerns. We have taken this seriously and have asked the University of Dundee research integrity adviser’s for their opinion. We will post a response on PubPeer once an evaluation has been completed.
LikeLike
https://www.beatson.gla.ac.uk/Cancer-Metabolism-Growth-and-Survival/martin-bushell.html
Prof Martin Bushell – RNA and Translational Control in Cancer
Appointments
2018-present: Senior Group Leader, CRUK Beatson Insitute, Glasgow
2018-present: Honorary Professor, University of Leicester
2011-2018: Deputy Director, MRC Toxicology Unit
2014-2018: Professor, Biochemistry Department, University of Leicester
2010-2015: MRC Senior Fellow, Group Leader MRC Toxicology Unit, Leicester
2011-2014: Reader, Biochemistry Department, University of Leicester
2005-2010: BBSRC David Phillips Fellow, School of Pharmacy, Nottingham University
2004-2004: Wellcome Trust ViP Fellow, University of Leicester
2003-2003: Wellcome Trust Travelling Fellow, University of Leicester
2001-2002: Wellcome Trust Travelling Fellow, Stanford University, CA, USA
1999-2000: Postdoctoral Fellow, Prof. M. Clemens, London
Problematic data:-
https://pubpeer.com/publications/DF38BCCD8F6B4A7908450F9032EB4F#2
https://pubpeer.com/publications/41E163998740C2E66E3B0D40D5B3A5
https://pubpeer.com/publications/7619B2B66C8F65BBEF61729ABD5165#4
https://pubpeer.com/publications/DC4763FA02B946FB7E468CBACAA0D9#9
https://pubpeer.com/publications/DC4763FA02B946FB7E468CBACAA0D9#11
https://pubpeer.com/publications/DC4763FA02B946FB7E468CBACAA0D9#12
https://pubpeer.com/publications/DC4763FA02B946FB7E468CBACAA0D9#13
https://pubpeer.com/publications/DC4763FA02B946FB7E468CBACAA0D9#14
https://pubpeer.com/publications/095D933A79348FD1D3983C75FA67C5#5
https://pubpeer.com/publications/D721B8C45393121D7A7AAA827AD2AC#1
https://pubpeer.com/publications/7D2A6E7510046A12DF9444C3974BF5#4
https://pubpeer.com/publications/7FB269E1C7F53ED9093676859657A0#2
https://pubpeer.com/publications/B619088AB412E4B323ADEDAE286F6E#19
LikeLike
https://www.beatson.gla.ac.uk/About/history.html
“Prof John Wyke became Director in 1987 and worked to develop links between the Beatson Institute and the University of Glasgow – in particular with the departments of Medical and Radiation Oncology.”
https://rse.org.uk/fellowship/john-wyke/
Professor John Anthony Wyke FRSE, FMedSci
Former Director, Beatson Institute for Cancer Research
The senior author here: https://pubpeer.com/publications/8BC5726A6DFA89EE59A39D886A0435
LikeLike
“Nobody in Edinburgh is answering emails, and neither does Hupp’s and Ball’s mentor Sir David Lane in Stockholm.”
David P. Lane failing to notice image duplication.
https://pubpeer.com/publications/C2C201CB09FBB254623C87F88B6546
Quick plug for Kanaga Sabapathy.
https://medicine.nus.edu.sg/bch/faculty/kanaga-sabapathy/
https://pubpeer.com/search?q=kanaga+sabapathy
LikeLike
“Both work on cell cycle regulation, he [Ted Hupp] on p53, she on the p21 protein.
Another person “working on p53” and its big bother p73. Similar trade practices.
https://renaissance.stonybrookmedicine.edu/pathology/faculty/moll
https://pubpeer.com/search?q=ute+moll
LikeLike
“Nobody in Edinburgh is answering emails, and neither does Hupp’s and Ball’s mentor Sir David Lane in Stockholm.”
Waiting for a Nobel prize to be awarded by people in the same institute?
Sir David Lane is recognised as one of the 3, or 4, group leaders who “discovered” p53 in 1979, but he did not discover its wild-type function. Of course without discovering a protein which was often, but not always, abundant in cancers there could to no discovery of its wild-type function, but nevertheless Sir David Lane did not discover its wild-type function. The discovery of its wild-type function is now credited to a group in Toronto led by S Benchimol.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2966958/
“…the p53 gene could be inactivated in several models of human or murine cell transformation (Mowat et al, 1985).”
Nature. 1985 Apr 18-24;314(6012):633-6. doi: 10.1038/314633a0.
Rearrangements of the cellular p53 gene in erythroleukaemic cells transformed by Friend virus
M Mowat, A Cheng, N Kimura, A Bernstein, S Benchimol
PMID: 3990796 DOI: 10.1038/314633a0
“The high frequency of p53 gene inactivation and rearrangement observed in Friend erythroleukaemic cells is intriguing and unexpected. Moreover, it suggests that inactivation of the p53 gene is not a random event, but is closely associated with the induction of erythroleukaemia by Friend virus complex or its helper virus.”
LikeLike
Another problem for the Nobel prize committee is going to be the readily observable, frequent, image manipulation by Arnold J Levine, one of the 3, or 4 group leaders who “discovered” p5 without understanding its true function.
https://pubpeer.com/search?q=arnold+levine
LikeLike
Sir David Lane might be able to confirm that Robert “5 retractions” Weinberg will not be receiving a Nobel prize. Hint: ask along the corridor.
https://forbetterscience.com/2020/01/20/the-wizard-men-curing-breast-cancer/
http://retractiondatabase.org/RetractionSearch.aspx#?auth%3dWeinberg%252c%2bRobert%2bA
LikeLike
That’s exactly why Arnie Levine will never get the Nobel Prize.
LikeLike
LikeLike
“Discovering” something without understanding it is the scientific equivalent of Imperialism. Go forth and claim vast tracts without understanding them. Sir David Lane will forever be remembered for his “discovery of p53”, but he didn’t really get it.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2966958/
Thierry Soucci’s thesis is that “p53 and the various interpretations of its function in cells serves as an example of how scientific paradigms can influence research fields”.
“The p53 gene has spent 30 years oscillating chaotically between classification as an oncogene and as a tumour suppressor.”
Do you have to explain that away by “paradigm shifts”, or that people, until S Benchimol, ignored logic?
To put it bluntly, is Sir David Lane a bit thick?
LikeLike
Remember that it is the U.K. and we mustn’t say anything.
The British scientific establishment is extremely autocratic, and many are crushed under the jackboot.
Incestuous and self-serving are other words that come to mind. Those words are not without evidence.
https://journals.plos.org/plosone/article/file?id=10.1371/journal.pone.0211460&type=printable
Most UK scientists who publish extremely highly-cited papers do not secure funding from major public and charity funders: A descriptive analysis
“..we searched for UK-based authors of peer reviewed papers that were published between January 2006 and February 2018 and received over 1000 citations in Scopus. We explored whether these authors have held a grant from the National Institute for Health Research (NIHR), the Medical Research Council
(MRC) and the Wellcome Trust and compared the results with UK-based researchers who serve currently on the boards of these bodies.
From the 1,370 papers relevant to medical, biomedical, life and health sciences with more than 1000 citations in the period examined, we identified 223 individuals from a UK institution at the time of publication who were either first/last or single authors. Of those, 164 are still in UK academic institutions, while 59 are not currently in UK academia (have left the country, are retired, or work in other sectors). Of the 164 individuals, only 59 (36%; 95% CI: 29–43%) currently hold an active grant from one of the three funders. Only 79 (48%; 95% CI: 41–56%) have held an active grant from any of the three funders between 2006–2017.
Conversely, 457 of the 664 board members of MRC, Wellcome Trust, and NIHR (69%; 95% CI: 65–72%) have held an active grant in the same period by any of these funders. Only 7 out of 655 board members (1.1%) were first, last or single authors of an extremely highly-cited paper.”
UK-based researchers who serve currently on the boards of these bodies serve themselves first is the simplest explanation. Why go against Occam’s razor? Occam was an Englishman after all.
LikeLike
Why doesn’t Sir David Lane return his knighthood?
https://www.iarc.who.int/friends-of-iarc/professor-sir-david-lane/
Sir David Lane is a “friend of IARC” so why doesn’t he do it a favour and help expose fake data within the IARC?
It’s there for all to see: https://forbetterscience.com/2018/10/11/who-cures-cancer-in-photoshop/
LikeLike
https://www.iarc.who.int/friends-of-iarc/professor-sir-david-lane/
Professor Sir David Lane made the landmark discovery of the p53 cancer gene in 1979.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2966958/
“History mainly remembers the virological approach that led to the discovery of p53. However, Lloyd Old’s team at the Ludwig Institute for Cancer Research in New York, USA took an immunological approach and published their findings in the same year (DeLeo et al, 1979). Old showed that the humoral response of mice to some induced tumour cells was directed against a 53-kDa protein. His team found that in animals, SV40 tumours elicited an immune response specific to this protein. Crawford also described antibodies in human serum that target the 53-kDa protein in 9% of breast cancer patients. This immunological research was just as original, but the compartmentalization of research meant that scientists did not immediately recognize the link between the two sets of observations (DeLeo et al, 1979). Several years later, it was established that the protein was the same and it was imaginatively baptized p53 (Crawford, 1983).”
“…131 articles published on the topic during this period did not integrate p53 into the family of oncogenes. Among these studies was the finding that both p53 alleles were inactivated in murine erythroleukaemia induced by the Friend virus (Mowat et al, 1985).”
To say that Sir David Lane is the discoverer of p53 when Lionel Crawford had discovered it years before, and the fact that Sir David Lane did not understand its wild-type function is stretching it a bit.
Sir David Lane might argue that Lionel Crawford did not appreciate its true function in 1983, but neither did Sir David Lane in 1979.
To have imaginatively baptized it may be Sir David Lane’s true contribution.
LikeLike
https://news.cancerresearchuk.org/2009/10/04/high-impact-science-p53/
“A recent review by Professor Carol Prives and Cancer Research UK’s Professor Karen Vousden highlights how far we have come – and how far we still have to go – in understanding this complicated molecule.”
They should review their own data (take a closer look).
Karen Vousden: https://pubpeer.com/search?q=vousden
Carol Prives:https://pubpeer.com/search?q=prives
https://forbetterscience.com/2017/09/21/carol-prives-innocent-victim-of-susana-gonzalez-data-manipulations/
LikeLike
https://www.nature.com/articles/nrc2723
A few paragraphs, but worth the read.
“It was while studying these SV40-derived tumor antigens that several groups independently stumbled on p53. This happened in 1979, thirty years ago (Timeline). Working at the ICRF (now London Institute for Cancer Research), David Lane and Lionel Crawford realized that when sera from animals bearing SV40-induced tumors were employed to immunoprecipitate SV40 large T-antigen, a non-viral protein with an apparent molecular mass of about 53 kDa came along for the ride 3. Further analysis established that this cellular protein was physically complexed with SV40 large T-antigen. Thus the viral protein, previously shown to be largely responsible for the transforming and tumorigenic activity of the SV40 virus, had selected this hitherto unknown cellular protein as its partner for an intimate, specific interaction. At the same time, Daniel Linzer and Arnold Levine applied a similar immunological approach to SV40 transformed cells, and came up with essentially the same observations, namely that such cells harbored a complex between the SV40 large T-antigen and the cellular 53 kDa protein 4. Three other groups, those of Alan Smith in the UK, Robert Carroll in New York and Pierre May in France, simultaneously made very similar findings, all published in 1979 5-7.
Interestingly, Linzer and Levine also found that their antisera precipitated the same 53 kDa protein from teratocarcinoma (a germ cell tumor)-derived cells, despite the fact that the latter did not harbor any SV40 proteins; this indicated that a subset of the antibodies raised against the viral-induced tumor were capable of interacting directly with this cellular protein 4. In parallel, Lloyd Old and coworkers demonstrated that animals immunized with non-virally transformed cells produced antibodies to the same 53 kDa protein 8, rightfully qualifying it as a cellular tumor antigen. Moreover, Varda Rotter, working in the lab of David Baltimore, was able to identify the same protein being produced in excess in cells transformed by a retrovirus, the Abelson murine leukemia virus 9. Hence, very high levels of this new cellular protein were present not only in SV40-transformed cells but also in other types of cancer cells, but little or no p53 protein could be detected in non-transformed cells.
As is often the case with independent discoveries of the same protein, each lab gave it a different name and continued to publish subsequent papers using their favorite name, creating quite a bit of confusion in this very young field. It was only in 1983, during the first p53 Workshop in Oxted, UK (Fig. 1), that representatives of the different p53 groups got together to discuss a common nomenclature. After a reasonable deal of inevitable debate, the term “p53” emerged as the winner and has stayed with us ever since. Ironically, p53 is actually a misnomer. When coined, it purportedly related to the molecular mass of the protein, which on the basis of its migration in SDS-polyacrylamide gels was estimated to be about 53 kDa. As realized later, this was a gross overestimate, presumably due to the presence of a proline-rich region that slows down the migration of the protein in such gels. In fact, the correct molecular mass of the human p53 protein is only 43.7 kDa, and that of the mouse protein is even less. But who would dare change a winning name?”
My emphasis:
“As is often the case with independent discoveries of the same protein, each lab gave it a different name and continued to publish subsequent papers using their favorite name, creating quite a bit of confusion in this very young field. It was only in 1983, during the first p53 Workshop in Oxted, UK (Fig. 1), that representatives of the different p53 groups got together to discuss a common nomenclature. After a reasonable deal of inevitable debate, the term “p53” emerged as the winner and has stayed with us ever since.”
The discoverer’s of p53 seem to be:-
David Lane and Lionel Crawford, U.K.
Alan Smith. U.K.
Robert Carroll, U.S.A.
Daniel Linzer and Arnold Levine, U.S.A.
Pierre May, France.
Varda Rotter and David Baltiore, U.S.A.
With time we learn that much, if not all research is the result of collective effort, yet the system is still operating in the days of Imperialism, where one person is credited will everything (.
Why do organisations still put out such mindless, and truncated propaganda? Is the propaganda just truncated and inaccurate so it can fit on the page, or a blatant attempt to ride rough-shod over the reality?
https://ki.se/en/mtc/david-lane-group
“The protein p53, widely known as the guardian of the genome, was discovered by Prof. Sir David Lane in 1979 and has over the years been recognized as a tumor suppressor gene mutated in more than half of all malignant tumors occurring in adults.”
No mention of anybody else.
The Karolinska probably runs courses on DEI (diversity, equity, and inclusion), but still runs on the old Imperial model when it matters, perhaps with the eye to collecting another Nobel prize itself.
https://www.iarc.who.int/friends-of-iarc/professor-sir-david-lane/
“Professor Sir David Lane made the landmark discovery of the p53 cancer gene in 1979. ”
No mention of anybody else.
LikeLike
Hupp and Ball publications suggest the question:
does working under the patronage of a famous person (Sir David Lane) lead to bad science?
A case in point.
https://forbetterscience.com/2020/10/07/gregg-semenza-real-nobel-prize-and-unreal-research-data/
Daniele Gilkes working under patronage of Gregg Semenza (Nobel Prize 2019).
https://www.nobelprize.org/prizes/medicine/2019/semenza/facts/
https://forbetterscience.com/2020/10/07/gregg-semenza-real-nobel-prize-and-unreal-research-data/
https://pubpeer.com/search?q=semenza
https://www.hopkinsmedicine.org/profiles/details/daniele-gilkes
https://pubpeer.com/search?q=gilkes
LikeLike
“Nobody in Edinburgh is answering emails, and neither does Hupp’s and Ball’s mentor Sir David Lane in Stockholm. He should though, even one of his own papers is affected.”
Does being a prolific author lead to bad science? Are hundreds of papers triaging for cutting corners?
Does working under the patronage of a prolific author lead to bad science? Great men spreading themselves too thinly, not knowing what is really going on?
Lane DP
479 papers
https://pubmed.ncbi.nlm.nih.gov/?term=lane%20dp&sort=date&ac=yes
Vogelstein B
601 papers
https://pubmed.ncbi.nlm.nih.gov/?term=vogelstein+b&sort=date
Somebody well known for being prolific in “cancer research” and “p53 world” is Bert Vogelstein.
https://en.wikipedia.org/wiki/Bert_Vogelstein
” his studies on colorectal cancers revealed that they result from the sequential accumulation of mutations in oncogenes and tumor suppressor genes.”
https://en.wikipedia.org/wiki/Alfred_G._Knudson
https://en.wikipedia.org/wiki/Two-hit_hypothesis
“Knudson suggested that two “hits” to DNA were necessary to cause the cancer. In the children with inherited retinoblastoma, the first mutation in what later came to be identified as the RB1 gene, was inherited, the second one acquired. In non-inherited retinoblastoma, instead two mutations, or “hits”, had to take place before a tumor could develop, explaining the later onset.”
The two hit hypothesis in not limited to inherited cancers. The principal of more than one hit had already been established by Alfred Knudsen. What does Bert Vogelstein add to that? Bert Vogelstein did not discover the first oncogenes, or the first tumor suppressor genes either. He did not discover the numbers 3 or 4 either.
Some may say that Bert Vogelstein has bludgeoned the “cancer results result from the sequential accumulation of mutations in oncogenes and tumor suppressor genes” paradigm to death”. It certainly it not a novel idea.
https://www.science.org/doi/10.1126/science.40.1041.857
https://www.biozentrum.uni-wuerzburg.de/zeb/research/topics/theodor-boveri/a-virtual-boveri-library/
When it comes to venturing outside “cancer world”, and “omics” into more basic and precise (genetic engineering) biology Bert Vogelstein and those under his patronage seem to fall over their own feet, and be very clumsy.
Nature . 1999 Oct 7;401(6753):616-20. doi: 10.1038/44188.
14-3-3Sigma is required to prevent mitotic catastrophe after DNA damage
T A Chan 1, H Hermeking, C Lengauer, K W Kinzler, B Vogelstein
Affiliation
1The Johns Hopkins Oncology Center, Program in Human Genetics, The Johns Hopkins University School of Medicine, Baltimore, Maryland 21231, USA.
PMID: 10524633 DOI: 10.1038/44188
https://pubpeer.com/publications/876EA56A4549BCAA87455EA00E5A19
Many theories why the duplications don’t affect the results.
https://pubpeer.com/publications/876EA56A4549BCAA87455EA00E5A19#3
Figure 4a.
https://imgur.com/l6OojJ0
https://pubpeer.com/publications/876EA56A4549BCAA87455EA00E5A19#76
Figure 2a.
https://imgur.com/AA3pX4L
LikeLike
What is it about p53 that they cannot correct their mistakes?
Nat Med . 2001 Oct;7(10):1111-7. doi: 10.1038/nm1001-1111.
Ferredoxin reductase affects p53-dependent, 5-fluorouracil-induced apoptosis in colorectal cancer cells
P M Hwang 1, F Bunz, J Yu, C Rago, T A Chan, M P Murphy, G F Kelso, R A Smith, K W Kinzler, B Vogelstein
Affiliation
1Howard Hughes Medical Institute, Johns Hopkins University School of Medicine, Baltimore, Maryland, USA.
PMID: 11590433 PMCID: PMC4086305 DOI: 10.1038/nm1001-1111
https://pubpeer.com/publications/DFE8C59F221AB16981586ACEFE366A
Correction is not about figure 4d.
Figure 4d. Much more similar than expected.
LikeLike
iScience. 2021 Jul 17;24(8):102878. doi: 10.1016/j.isci.2021.102878. eCollection 2021 Aug 20.
CHIP-dependent regulation of the actin cytoskeleton is linked to neuronal cell membrane integrity
Catarina Dias 1 2, Erisa Nita 1, Jakub Faktor 3 4, Ailish C Tynan 1, Lenka Hernychova 3, Borivoj Vojtesek 3, Jesper Nylandsted 5, Ted R Hupp 1 4, Tilo Kunath 2, Kathryn L Ball 1
Affiliations
1Institute of Genetics and Cancer, University of Edinburgh, Edinburgh EH4 2XU, UK.
2Centre for Regenerative Medicine, Institute for Stem Cell Research, School of Biological Sciences, The University of Edinburgh, Edinburgh EH16 4UU, UK.
3Research Centre for Applied Molecular Oncology, Masaryk Memorial Cancer Institute, 656 53 Brno, Czech Republic.
4University of Gdansk, International Centre for Cancer Vaccine Science, 80-822 Gdansk, Poland.
5Membrane Integrity Group, Danish Cancer Society Research Center, Strandboulevarden 49, 2100, Copenhagen, Denmark.
PMID: 34401662 PMCID: PMC8350547 DOI: 10.1016/j.isci.2021.102878
22 Feb 2022 erratum.
https://www.cell.com/iscience/fulltext/S2589-0042(22)00191-2?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS2589004222001912%3Fshowall%3Dtrue
Stylistic changes to the manuscript were made for clarity (and involved the addition of a single vertical black line to Figure 1K). This indicates that although the two samples were run on the same gel, they were not in adjacent lanes (they were originally separated by a CHIP heterozygous cell sample). In addition, the legend for Figure 2I is modified, showing that the ANXA2 data came from re-probing the membrane shown in Figure 1K (in this case including the CHIP heterozygous cell sample). As a result, some of the β-actin and CHIP controls are common to both Figures 1K and 2I.
In the supplemental information, white lines were added to Figure S1. The PCR data were taken from a larger screen, and the samples may not have been in adjacent lanes on the agarose gel. Further, Figures S7A and S7B have been consolidated into a single figure (Figure S7A) containing all the original data. The figure legends have been edited to reflect this change.
All the authors agree that the changes have no impact on the outcome or conclusions of the study, as no data has been removed or added post publication of the original manuscript. The authors apologize for any confusion caused to the readers.
LikeLike
Good. That paper was not flagged on PubPeer, meaning the university does investigate.


What kind of figure is this anyway:
Also, in the corrected Fig S7, ANXA2 is most definitely not from the same gel as the rest.
LikeLike
PLoS One . 2011;6(12):e27806. doi: 10.1371/journal.pone.0027806. Epub 2011 Dec 14.
FAK deletion promotes p53-mediated induction of p21, DNA-damage responses and radio-resistance in advanced squamous cancer cells
Kathryn Graham 1, Kim Moran-Jones, Owen J Sansom, Valerie G Brunton, Margaret C Frame
Affiliation
1
The Beatson Institute for Cancer Research, Garscube Estate, Bearsden, Glasgow, Scotland.
PMID: 22194793 PMCID: PMC3237418 DOI: 10.1371/journal.pone.0027806
Problematic data figures 2D and 4C. Much more similar than expected.
LikeLiked by 1 person
Virus Res. 2005 Mar;108(1-2):1-14. doi: 10.1016/j.virusres.2004.07.004.
Human papillomavirus 16 L2 inhibits the transcriptional activation function, but not the DNA replication function, of HPV-16 E2
A Okoye 1, P Cordano, E R Taylor, I M Morgan, R Everett, M S Campo
Affiliations collapse
Affiliation
1Division of Pathological Sciences, Institute of Comparative Medicine, Glasgow University, Garscube Estate, Glasgow G61 1QH, Scotland, UK.
PMID: 15681049
DOI: 10.1016/j.virusres.2004.07.004
https://pubpeer.com/publications/596DAC3CAB42F5794FF1A7F11BF4DC
Figures 2B and 4. Much more similar than expected.
Figure 2A. Vertical splices in L2 and E2 panels, but not in Actin panel.
Figure 4. Vertical splices in E2 panel between L2 minus and L2 plus lanes. but not splices apparent in the accompanying Actin panel.
LikeLiked by 1 person
J Biol Chem. 2001 Sep 7;276(36):33861-8. doi: 10.1074/jbc.M100958200. Epub 2001 Jul 11.
Cell transformation by the E5/E8 protein of bovine papillomavirus type 4. p27(Kip1), Elevated through increased protein synthesis is sequestered by cyclin D1-CDK4 complexes
V O’Brien 1, G J Grindlay, M S Campo
Affiliation
1Beatson Institute for Cancer Research, CRC Beatson Laboratories, Garscube Estate, Glasgow G61 1BD, Scotland, United Kingdom. vobrien@beatson.gla.ac.uk
PMID: 11448948
DOI: 10.1074/jbc.M100958200
https://pubpeer.com/publications/615A5D738EC13B39E234A7BC551115
Figure 1. Much more similar than expected.
LikeLiked by 1 person
https://www.gla.ac.uk/researchinstitutes/icams/newsandevents/februarynewsletter2018/headline_572941_en.html
“ICAMS Professor George Baillie has been appointed as Editor-in-Chief of Cellular Signalling, an internationally respected Elsevier journal. Professor Baillie had served as co-editor for 6 years before being chosen by Elsevier to lead the modernisation and rejuvenation of the Journal.
He has appointed a new team of Co-editors and refreshed the Editorial Board. Novel features of the journal will included themed special issues, faster times to publication and decision on acceptance after a single review. Professor Baillie would like to extend an invitation to ICAMS researchers to submit any paper that has a obvious “cell signalling” component for fast, fair and efficient review.”
Special favours? “Professor Baillie would like to extend an invitation to ICAMS researchers to submit any paper that has a obvious “cell signalling” component for fast, fair and efficient review”.
Problematic data galore! Makes me think of the classic comedy “Whisky galore!”.
https://en.wikipedia.org/wiki/Whisky_Galore!_(1949_film)
2019 remake.
J Biochem . 2019 Jul 1;166(1):97-106. doi: 10.1093/jb/mvz016.
Phosphorylation of PDE4A5 by MAPKAPK2 attenuates fibrin degradation via p75 signalling
K F Houslay 1, B A Fertig 2, F Christian 2, A J Tibbo 2, J Ling 2, J E Findlay 3, M D Houslay 3, G S Baillie 2
Affiliations collapse
Affiliations
1Department of Respiratory, Inflammation and Autoimmunity, MedImmune, Granta Park, Cambridge, UK.
2Institute of Cardiovascular and Medical Sciences, University of Glasgow, Glasgow, UK.
3Institute of Cancer Studies and Pharmaceutical Science, King’s College, 150 Stamford Street, London, UK.
PMID: 30859186
PMCID: PMC6607969
DOI: 10.1093/jb/mvz016
Problematic data figure 2. Much more similar than expected.
Figure 3. Much more similar than expected.
See:-
https://pubpeer.com/publications/4500EAFC5C40497347E63F88238BF6#1
https://pubpeer.com/publications/4500EAFC5C40497347E63F88238BF6#2
https://pubpeer.com/publications/4500EAFC5C40497347E63F88238BF6#3
Anybody looking to hire an hilarious director?
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6607969/
G. S. Baillie, Institute of Cardiovascular and Medical Science, College of Medical, Veterinary and Life Sciences, University of Glasgow, Office 534, Wolfson-Link Building, Glasgow G12 8QQ, UK. Tel: 01413301662, Fax: 01413304328, email: george.Baillie@glasgow.ac.uk
LikeLiked by 1 person
26 April 2022 retraction.
https://www.pnas.org/doi/full/10.1073/pnas.2205198119
Biochemistry Retraction for “β-Arrestin-mediated PDE4 cAMP phosphodiesterase recruitment regulates β-adrenoceptor switching from Gs to Gi,” by George S. Baillie, Arvind Sood, Ian McPhee, Irene Gall, Stephen J. Perry, Robert J. Lefkowitz, and Miles D. Houslay, which was first published January 27, 2003; 10.1073/pnas.262787199 (Proc. Natl. Acad. Sci. U.S.A. 100, 940–945).
The undersigned authors note the following,
“We were informed by the University of Glasgow Research Integrity Council that, as part of a larger investigation of anomalies in a series of papers from one of their laboratories, it was found that lanes 2 and 3 of Fig. 4C are identical. While the data in this figure panel were obtained in myocardial cells, which are the sole focus of Fig. 4, the University of Glasgow did not identify irregularities in any other data presented in the paper, including the data obtained with HEK cells, which constitute the remainder of the data in the paper (Figs. 1–3). Nonetheless at the request of the University of Glasgow the paper is being retracted.
We offer our apologies to the scientific community for any inconvenience this may have caused.” George S. Baillie, Arvind Sood, Ian McPhee, Stephen J. Perry, and Robert J. Lefkowitz
” the University of Glasgow did not identify irregularities in any other data presented in the paper, including the data obtained with HEK cells, which constitute the remainder of the data in the paper (Figs. 1–3).”
Problematic data figures 3a and 3c. Much more similar than expected.
Pubpeer comments. https://pubpeer.com/publications/82986F4A4D89EA465E822A9809F154
LikeLiked by 1 person