In the medieval Flemish town, there is a Catholic University (KU) of Leuven, and inside this Belgian university there is a stem cell institute. The queen of this stem cell institute is the zombie scientist Catherine Verfaillie, whose once amazingly groundbreaking stem cells discoveries were long since debunked as phony garbage full of data manipulation.
But this story is not about the zombie queen Verfaillie, but about a Roman troll she hosts in her institute. Maurilio Sampaolesi is full professor at the KU Leuven, and he once cured Duchenne muscular dystrophy, together with his Roman mentor, Giulio Cossu. This story is about these two Italian stem cell magicians.
Just some years ago, stem cells and regenerative medicines were the hope of humanity. All diseases looked curable, organs were being grown in the lab like tomatoes, even the Catholic Church rejoiced when Verfaille discovered in early noughties that cells inside your bone marrow were pluripotent just like the cells in the embryonic blastocyst. These magic bone marrow cells could regenerate and reconstitute every tissue in the body, Verfaille was a star and even celebrated on a Belgian stamp. The surgeon Paolo Macchiarini applied this preclinical discovery into clinical breakthroughs, many lives were claimed to have been saved with his trachea transplants where bone marrow cells were seeded on an artificial scaffold.
Now we know that it all was fake.
These days, nobody is talking about pluripotent cells inside the bone marrow or anywhere else in the human body, except the usual criminally-inclined quack doctors running fraudulent stem cell clinics in former USSR, Latin America, or Germany.
But back then, when Verfaillie claimed in Nature to have found stem cell magic in the bone marrow, Cossu and his trainee Sampaolesi claimed in Nature to have harnessed stem cell magic of adult tissues to cure muscular dystrophy, in dogs. The two Italians and their Golden Retrievers were celebrated by the scientific community and the public alike, as the many sufferers of this incurable muscle-wasting degenerative disease saw hope on the horizon.
In November 2006, The Telegraph wrote:
“A stem-cell treatment for the degenerative and fatal disease muscular dystrophy, which mostly affects boys and young men, could be available for testing on patients within two years, following a remarkable series of experiments reported today. […]
The dramatic results of stem-cell therapy on the animals are reported in the journal Nature by Prof Giulio Cossu, the director of the Stem Cell Research Institute of San Raffaele Scientific Institute of Milan. The work shows that it is possible to halt this devastating disease and possibly even reverse it to a degree. […]
The professor estimated that another year or two will be necessary to get a patient’s own genetically modified stem cells to work. “Even though by itself it may not lead to a complete cure, it would ameliorate the condition, then step by step we could work on this to the point of getting a real cure.
Prof Cossu has already discussed human transplants with an Italian charity and is planning clinical trials. The first attempt is likely to use stem cells from a donor with a similar tissue type, the method that succeeded with the dogs.”
An esteemed Oxford professor, Dame Commander of the British Empire and expert on Duchenne muscular dystrophy (DMD) opined:
“Prof Kay Davies, of Oxford University, commented: “The use of stem cells to treat human disease holds great promise but the actual delivery of such therapy is thought to be many years away.
“The data presented in the paper from Cossu’s group, using a particular type of stem cell, a mesoangioblast stem cell, changes that view. . . this is a major step forward and suggests that this approach may be effective for use in DMD patients.”
Thing is, Davies’ own dystrophy research is full of data manipulations, and she is also extremely welcoming towards research misconduct of others.
And sure enough, also the scientific discoveries by Cossu and Sampaolesi were apparently not the breakthroughs we were told they were. Nobody is talking about these results and their discoverers anymore, and nobody wants to correct or retract their old papers despite the data irregularities therein. The spotlight is now taken by new bullshitters of stem cell research, preferably American.
This was that groundbreaking Nature paper by Sampaolesi and Cossu from 14 years ago:
Maurilio Sampaolesi, Stephane Blot , Giuseppe D’Antona , Nicolas Granger , Rossana Tonlorenzi , Anna Innocenzi , Paolo Mognol , Jean-Lauren Thibaud , Beatriz G. Galvez , Ines Barthélémy , Laura Perani , Sara Mantero , Maria Guttinger , Orietta Pansarasa , Chiara Rinaldi , M. Gabriella Cusella De Angelis , Yvan Torrente , Claudio Bordignon , Roberto Bottinelli , Giulio Cossu Mesoangioblast stem cells ameliorate muscle function in dystrophic dogs Nature (2006) doi: 10.1038/nature05282
The Nature study gave hope to many dystrophy sufferers, but it was trash. And it was debunked right away in a 2007 letter to editor by Allan Bretag:
“My first concern is that control and test dogs were not matched for disease characteristics at the start of treatment to avoid inadvertently biased groupings in small experimental cohorts, where extensive individual variations exist. Also, the authors’ evaluations were not blinded. […]
Second, Fig. 5a of Sampaolesi et al.1 indicates that muscle strength declined, rather than was maintained as they imply, in the treated legs of all dystrophic dogs in which it was assessed. […]
Control dystrophic dogs lived, on average, 129 days longer than six out of ten treated dogs (123 days longer than those treated with autologous mesoangioblasts). Three of the four remaining treated dogs, described as “well” 400 days post natal, “rapidly lost walking ability” when immunosuppression ceased (257 days post natal for two of these).
Altogether, the evidence presented by Sampaolesi et al.1 does not convince me that the dogs benefited from mesoangioblast treatment; a rigorous demonstration correlating muscle function, dystrophin expression and mesoangioblast infusion, with adequate controls, would have been helpful in this regard. It is therefore premature to consider a clinical trial in humans as a justifiable extension of this study.”
The first correction came in 2013:
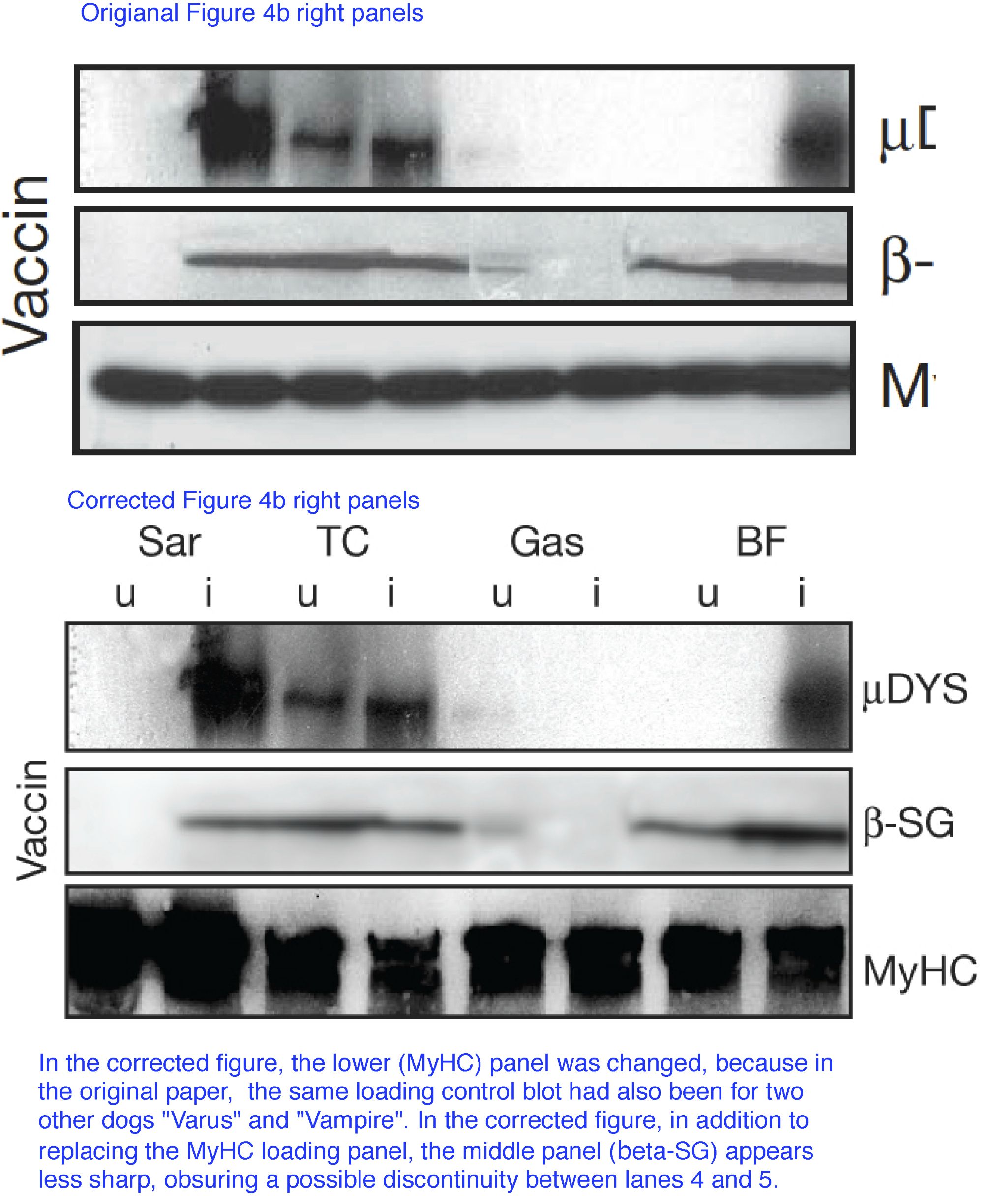
That same figure was then corrected again in 2014:
“In Fig. 4b of this Article, the gel for the loading control MyHC for the dog Varus was an unintentional duplication of the loading controls for the dog Vampire (which is correct). The correct gel is shown below in Fig. 1. The error does not affect any of our results and a University College London (UCL) committee appointed by the Vice Provost for Research has investigated the data presented in this Article and is satisfied of its authenticity.”
Indeed, there was an investigation at UCL where Cossu was professor at that time. UCL is very good in whitewashing research misconduct. The elite London university proved it twice, bigtime, for example in the Macchiarini affair:
But UCL’s greatest achievement was to overrule and to suppress the findings of their own investigation in order to declare their Birkbeck College master and major donor David Latchman as an innocent victim of some foreigner’s fraud:
Hence, no wonder that in 2013, UCL officially found absolutely nothing wrong with Cossu’s papers, as Times Higher Education reported:
“A University College London investigation has found that the research practice of a senior molecular biologist “did not always meet the standard of good practice expected of UCL researchers”.
Suspicions of image manipulation were raised last November concerning eight papers published between 2006 and 2012 by the laboratory of Giulio Cossu, who joined UCL in May 2011 as professor of human stem cell biology.
The allegations were made by an anonymous whistleblower known as “Clare Francis”. UCL’s formal procedure for investigating misconduct allegations requires a named accuser, but, in a statement, the university says that its “commitment to good practice in research” obliged it to examine the allegations.
The investigating panel, which included an external expert, concluded that no research misconduct had occurred. It cleared Professor Cossu of any “deliberate intention to mislead” and found no substance to the “large majority” of the allegations.
But it found that in a “few” cases “the conduct and presentation of the research in question did not always meet the standard of good practice expected of UCL researchers”.
“Where specific errors were detected, the panel recommended that, where possible, corrections should be made to the published papers,” the statement adds. But the investigation “found no indication that the errors altered the fundamental conclusions of these studies, many of which have been confirmed by independent research groups”.
It says that Professor Cossu “has acknowledged the spirit and details of the report” and “sincerely regrets” the errors.
“He will…implement processes to ensure to the best of his ability that such errors do not occur in the future,” the statement adds.”
Just before that decision, a muscle stem cell paper co-authored by Cossu was retracted:
Addolorata Pisconti , Silvia Brunelli , Monica Di Padova , Clara De Palma , Daniela Deponti , Silvia Baesso , Vittorio Sartorelli , Giulio Cossu , Emilio Clementi Follistatin induction by nitric oxide through cyclic GMP: a tightly regulated signaling pathway that controls myoblast fusion The Journal of Cell Biology (2006) doi: 10.1083/jcb.200507083
The retraction notice from January 2013 stated:
“After concerns were raised by a reader, the Editors of The Journal of Cell Biology detected the following issues with the data in the above article:
(1) The top panel in Fig. 1 F (NOS-1μ) is identical to the top panel in Fig. 2 B (α-sGC).
(2) The bottom panel in Fig. 1 F (NOS-III) is identical to the bottom panel in Fig. 2 B (GAPDH).
(3) Lanes 2 and 3 of the Follistatin panel for satellite cells in Fig. 4 A are identical to lanes 1 and 2 of the Myostatin panel for satellite cells in the same figure.
(4) A band appears to have been erased from lane 4 of the Follistatin panel for satellite cells in Fig. 4 A.
(5) Lane 4 of the Myostatin panel for satellite cells in Fig. 4 A is identical to lane 1 of the Follistatin panel for E12.5/E15.5 in the same figure.
(6) The E9.5 explants and E12.5/E15.5 panels for GAPDH in Fig. 4 A are identical.
Given the above issues with the experimental data in Fig. 4 A, the quantification data in Fig. S1 A cannot be validated.
No issues were detected with the other figures in the paper nor with the other parts of the figures listed above (Fig. 1, A–E, G, and H; Fig. 2, A and C–E; Fig. 4 B; and Fig. S1 B). […]
As a result of this retraction, no data in this paper should be cited in the scientific literature.
The Authors and Editors have informed the University of Milan of this retraction.”

Cossu just joined UCL in 2012 as full professor, having left home country, where he ha da full professorship and led a stem cell centre at the San Raffaele research hospital in Milan. After that 2013 investigation, Cossu left UCL, officially innocent of misconduct. But he didn’t return to Italy, but travelled north to become professor at the University of Manchester.
Cossu’s previous collaboration at the University of Milan with Emilio Clementi‘s lab there has been truly productive:

Clara Sciorati , Beatriz G. Galvez , Silvia Brunelli , Enrico Tagliafico , Stefano Ferrari , Giulio Cossu , Emilio Clementi Ex vivo treatment with nitric oxide increases mesoangioblast therapeutic efficacy in muscular dystrophy Journal of Cell Science (2006) doi: 10.1242/jcs.03300
We can assume it was Clementi’s lab fault because some other copied gel bands derived from a 2006 Clementi paper without Cossu’s contribution:
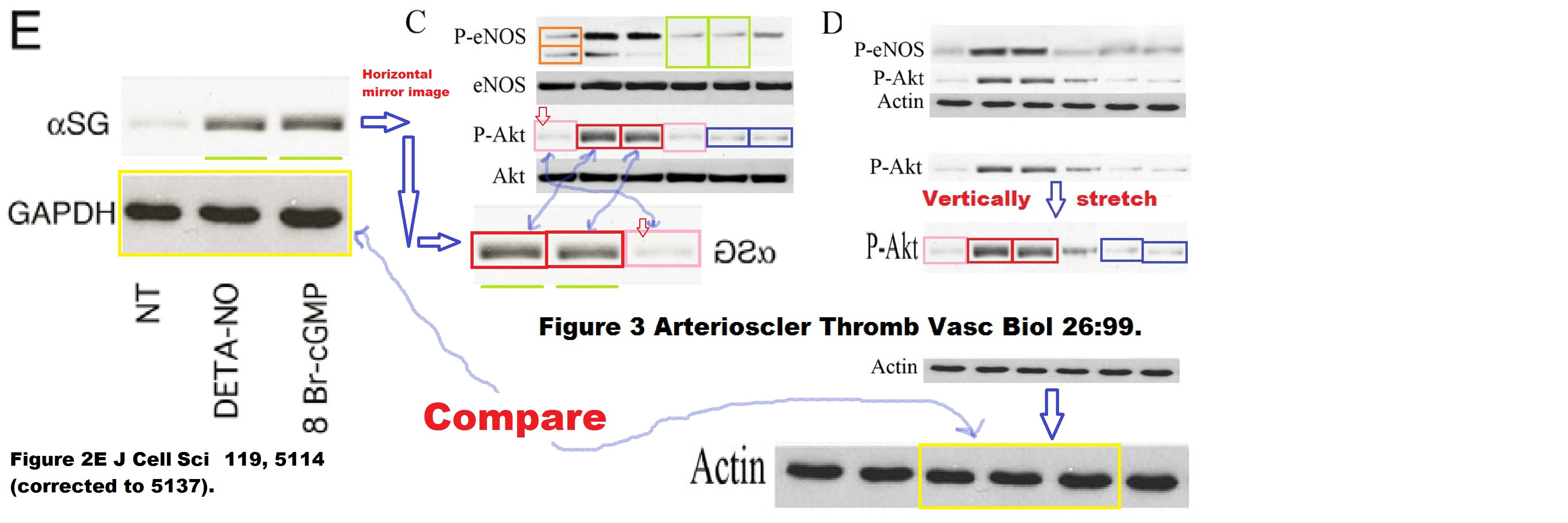
No correction for that paper, maybe that journal had enough of Cossu.
You would expect someone under research misconduct investigation to be particularly careful which data he submits for a new publication, but…
Ornella Cappellari , Sara Benedetti , Anna Innocenzi , Francesco Saverio Tedesco , Artal Moreno-Fortuny , Gonzalo Ugarte , Maria Grazia Lampugnani , Graziella Messina , Giulio Cossu Dll4 and PDGF-BB convert committed skeletal myoblasts to pericytes without erasing their myogenic memory Developmental Cell (2013) doi: 10.1016/j.devcel.2013.01.022
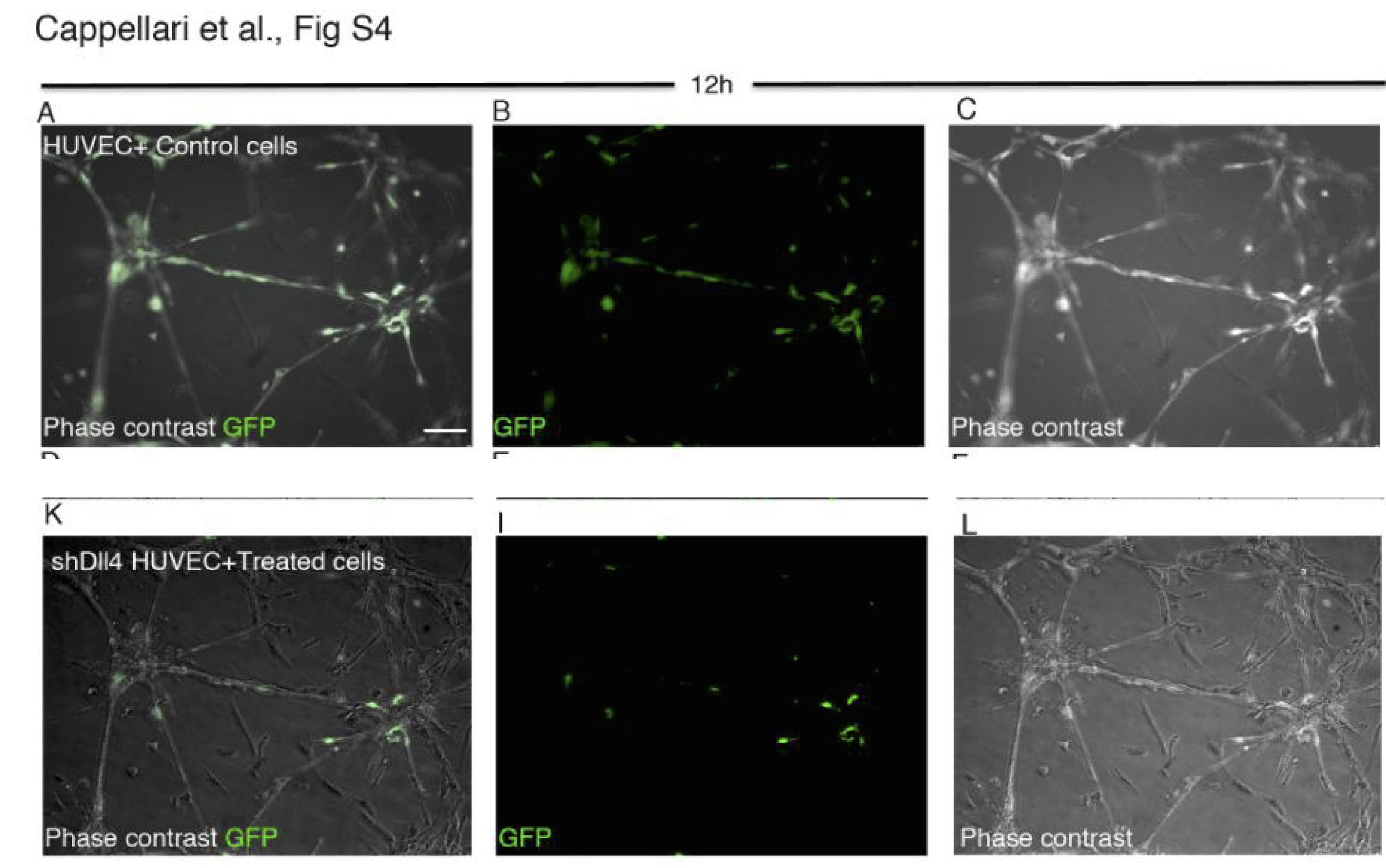
Cell Press issued in 2014 a correction:
“In the originally published version of this paper, Figures S4A–S4C correctly showed control cells; however, these same cells erroneously also appeared in Figures S4K–S4L. We have now replaced the latter three panels with the correct data….”
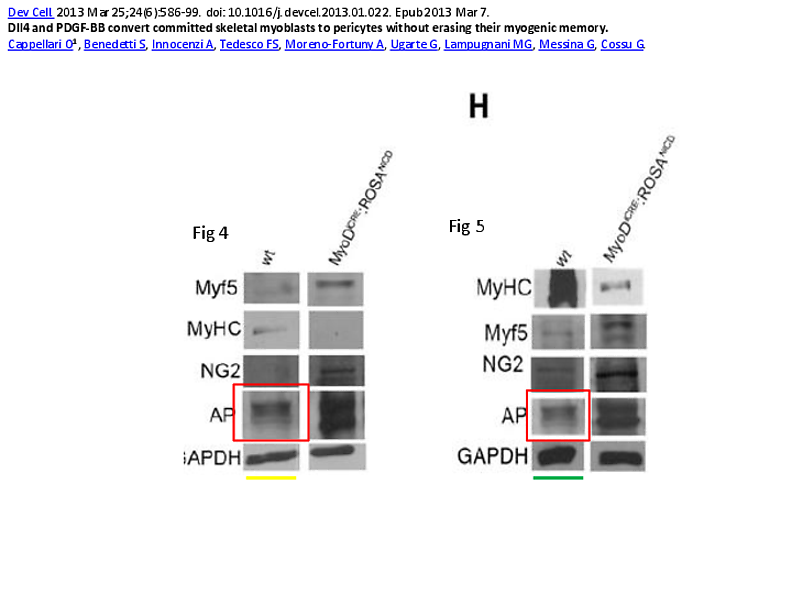
But apparently Cell Press has a rule forbidding second corrections, so a duplicated gel band is to remain:
Cossu discovered a lot in his time. In those golden years of regenerative medicine, scientific community accepted it as a proven fact that heart stem cells exist. Only after Piero Anversa was exposed as a mega-fraudster, masses of his papers retracted, did this huge field of cardiac stem cells collapse. But a funny thing is that a number of scientists managed to somehow build on and to extend Anversa’s fraudulent results, which either proves that Anversa was a prophet, or that the field of degenerative regenerative medicine is full of cheaters perpetuating each other’s fraud. E.g. what to make of the heart stem cell research by David Argyle, head of Veterinary School at the University of Edinburgh?
Also Cossu and Sampaolesi proved in 2008, in Nature no less, that cardiac mesangioblasts were self-renewing and differentiate into beating heart muscle, those being true heart stem cells. Which presumably makes it a science fact, everyone must apologise to Anversa.
Here the follow-up paper by Cossu and Sampaolesi about cardiac stem cells:
Beatriz G Gálvez, Diego Covarello , Rosanna Tolorenzi , Silvia Brunelli , Arianna Dellavalle , Stefania Crippa , Salman Afroze Azmi Mohammed , Ludovica Scialla , Ivan Cuccovillo , Fabiola Molla , Lidia Staszewsky , Francesco Maisano , Maurilio Sampaolesi, Roberto Latini , Giulio Cossu Human cardiac mesoangioblasts isolated from hypertrophic cardiomyopathies are greatly reduced in proliferation and differentiation potency Cardiovascular Research (2009) doi: 10.1093/cvr/cvp159
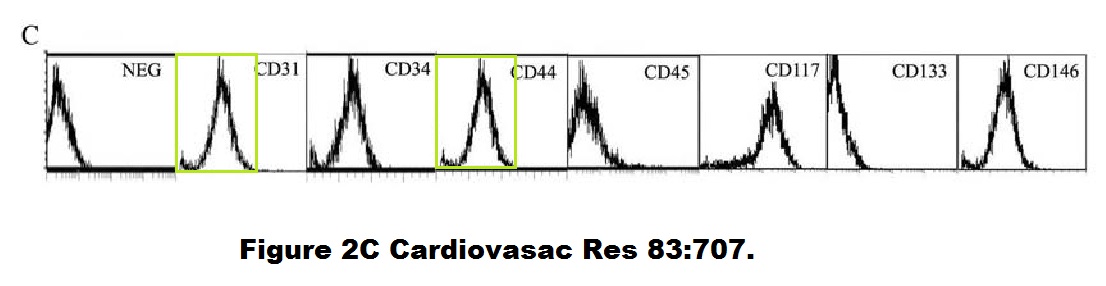
Small thing, a duplicated flow cytometry histogram, but maybe a clue of how reliable all these stem cell discoveries by Cossu and Sampaolesi really were.
Before dogs, muscular dystrophy was cured by Cossu’s and Sampaolesi’s stem cell therapy in mice:
Maurilio Sampaolesi , Yvan Torrente, Anna Innocenzi , Rossana Tonlorenzi , Giuseppe D’Antona , M. Antonietta Pellegrino , Rita Barresi , Nereo Bresolin , M. Gabriella Cusella De Angelis , Kevin P. Campbell , Roberto Bottinelli , Giulio Cossu Cell therapy of alpha-sarcoglycan null dystrophic mice through intra-arterial delivery of mesoangioblasts Science (2003) doi: 10.1126/science.1082254
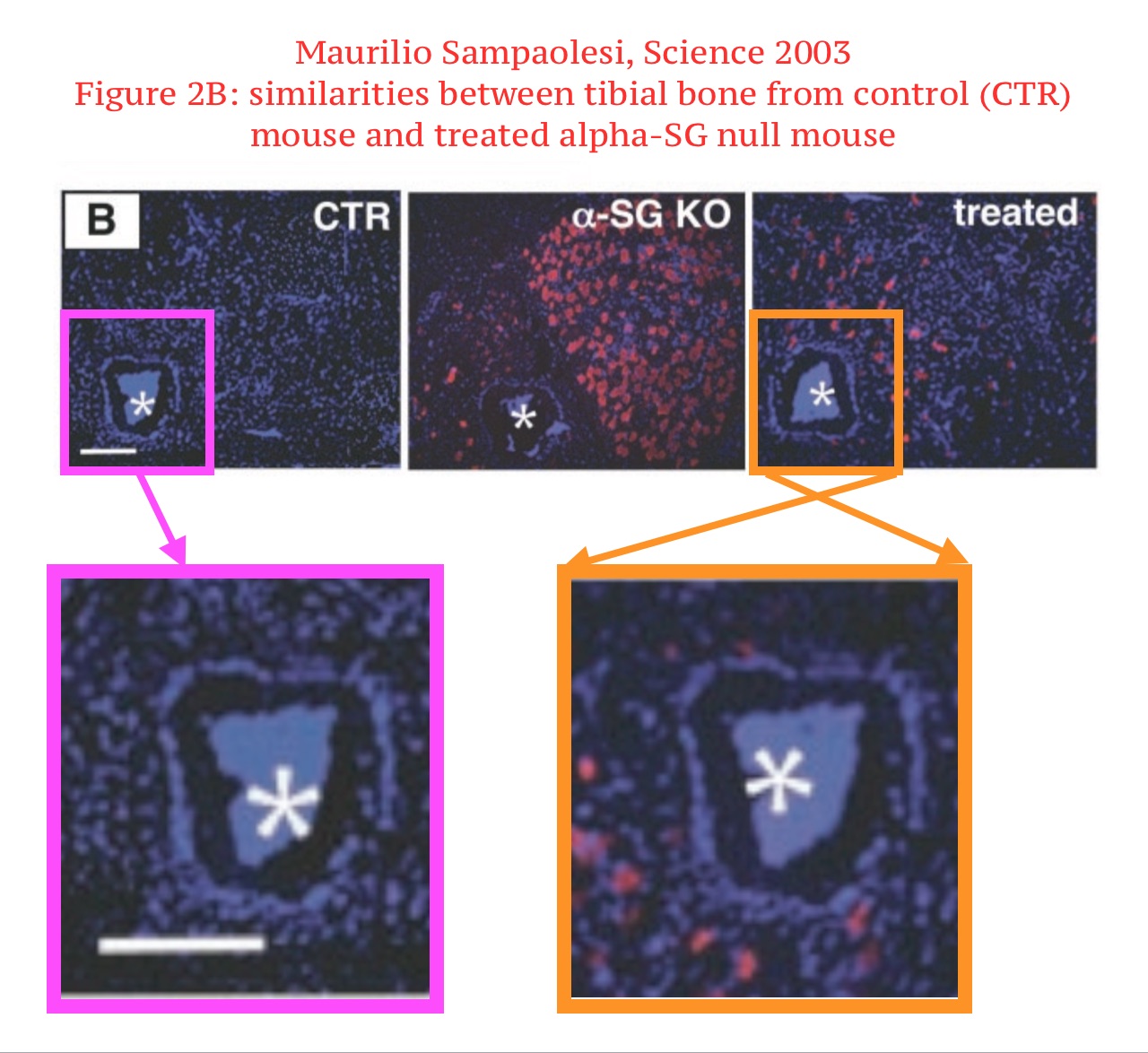
This is not just an innocent duplication. First, the red signal is very different between the control and the treated mice. But it also seems, the microscopy image was digitally rearranged by some mysterious fraudulent crook.
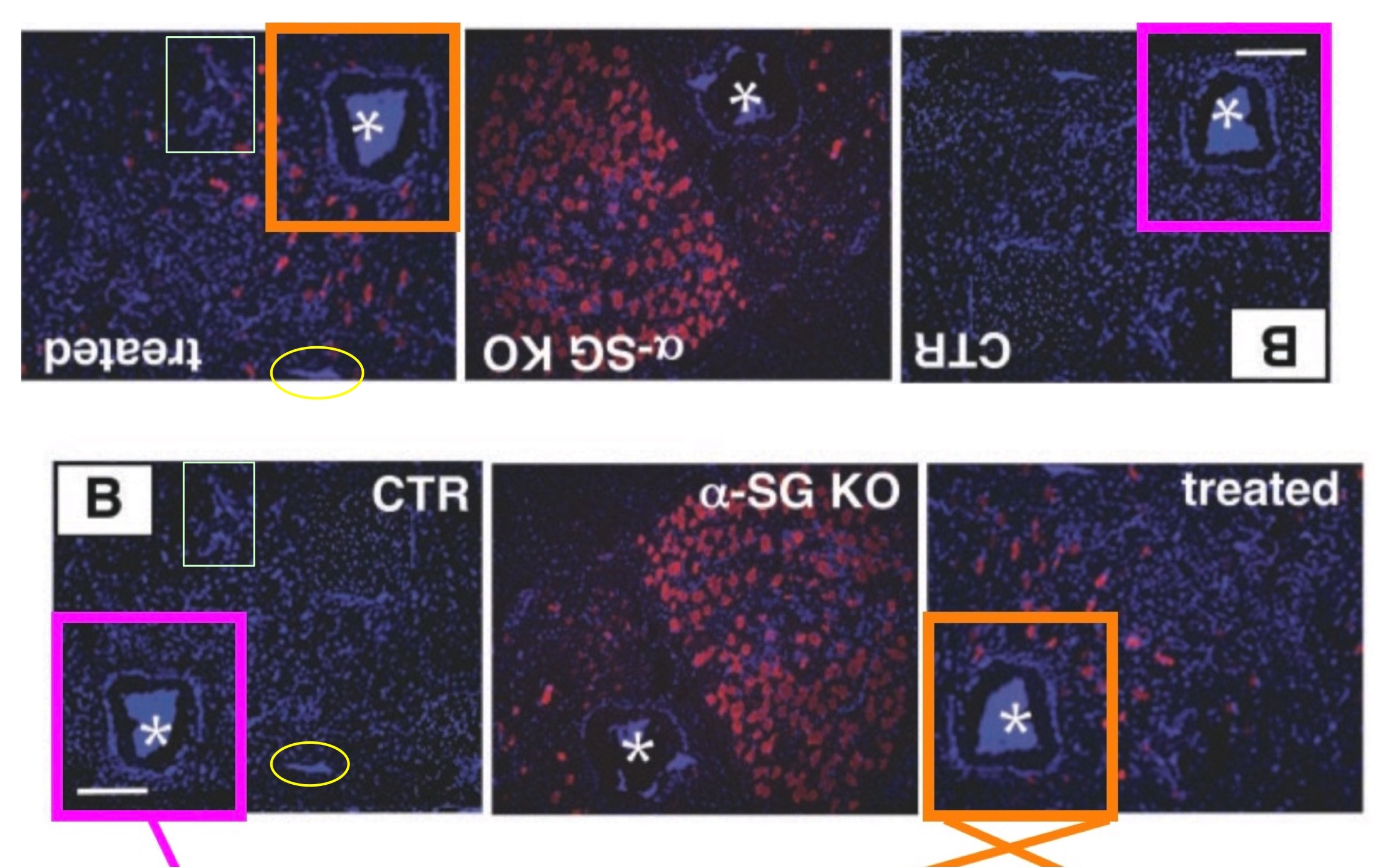
Cossu and Sampaolesi know of this problem with their paper for years, but neither they not the editor of Science have found time so far to do anything about it. But then again, Science editors are busy with ignoring many other things.
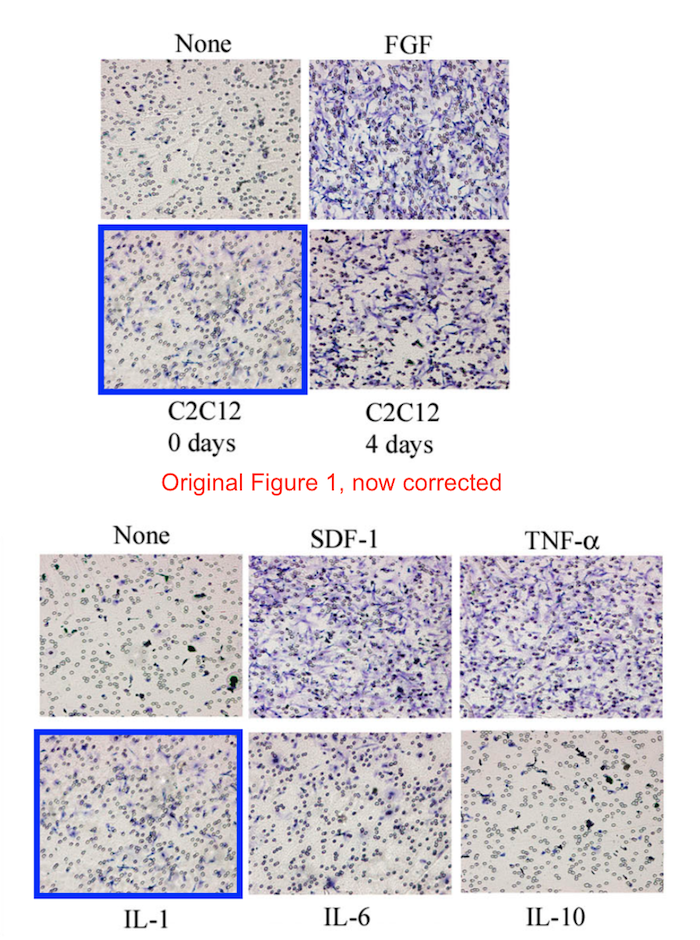
Soon after that breakthrough cure of mice, and before the breakthrough in dogs, a potential cure for human disease suffers was established. Cossu and Sampaolesi even found the right stem cells in the blood of healthy donors. Just sort these “mesangioblasts” by FACS for the famous surface protein market CD133 (AC133), which, science concurred back then, was definitely established to be THE universal stem cell marker of all stem cells:
Yvan Torrente, Marzia Belicchi , Maurilio Sampaolesi , Federica Pisati , Mirella Meregalli , Giuseppe D’Antona , Rossana Tonlorenzi , Laura Porretti , Manuela Gavina , Kamel Mamchaoui , Maria Antonietta Pellegrino , Denis Furling , Vincent Mouly , Gillian S. Butler-Browne , Roberto Bottinelli , Giulio Cossu , Nereo Bresolin Human circulating AC133(+) stem cells restore dystrophin expression and ameliorate function in dystrophic skeletal muscle Journal of Clinical Investigation (2004) doi: 10.1172/jci20325
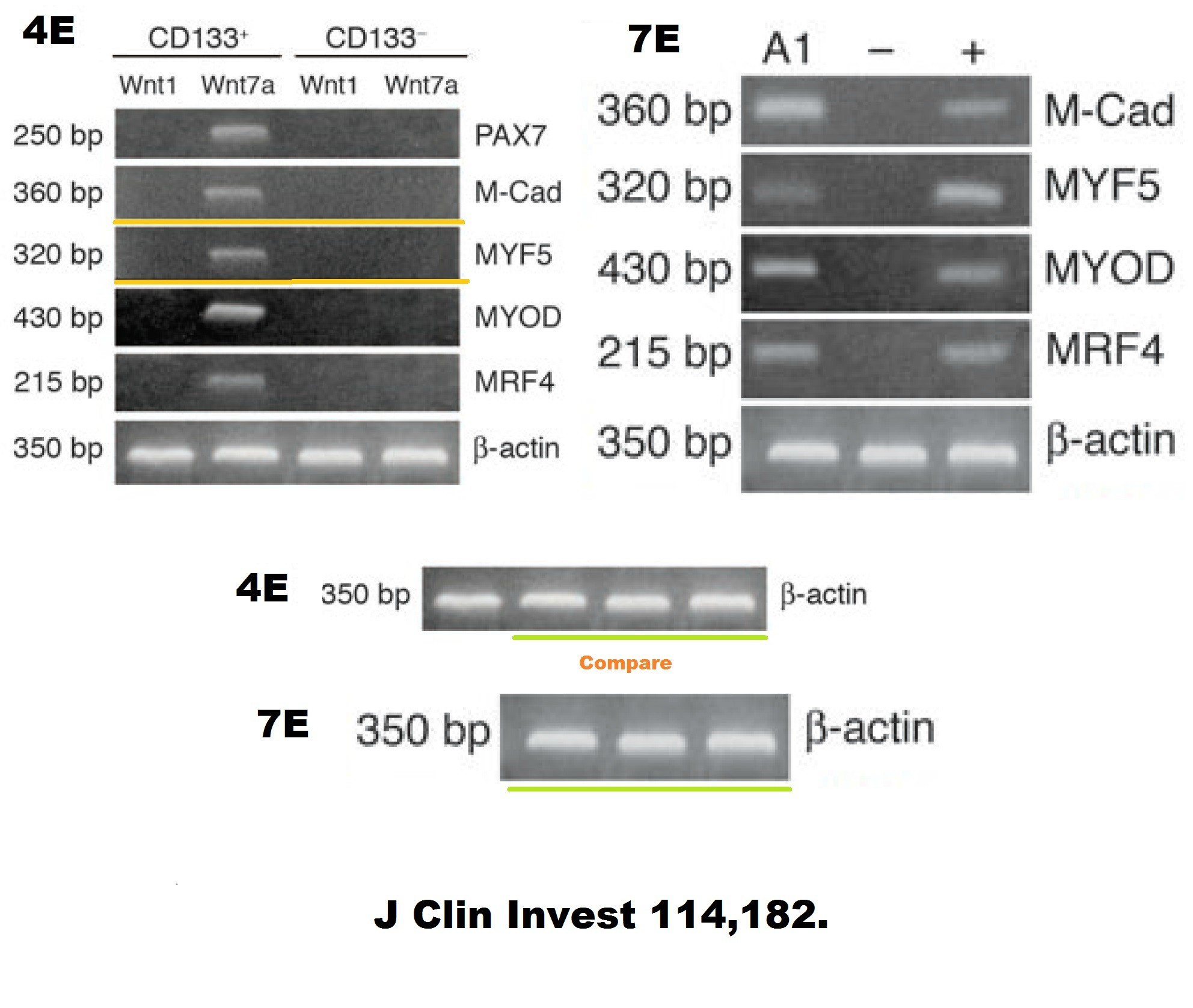
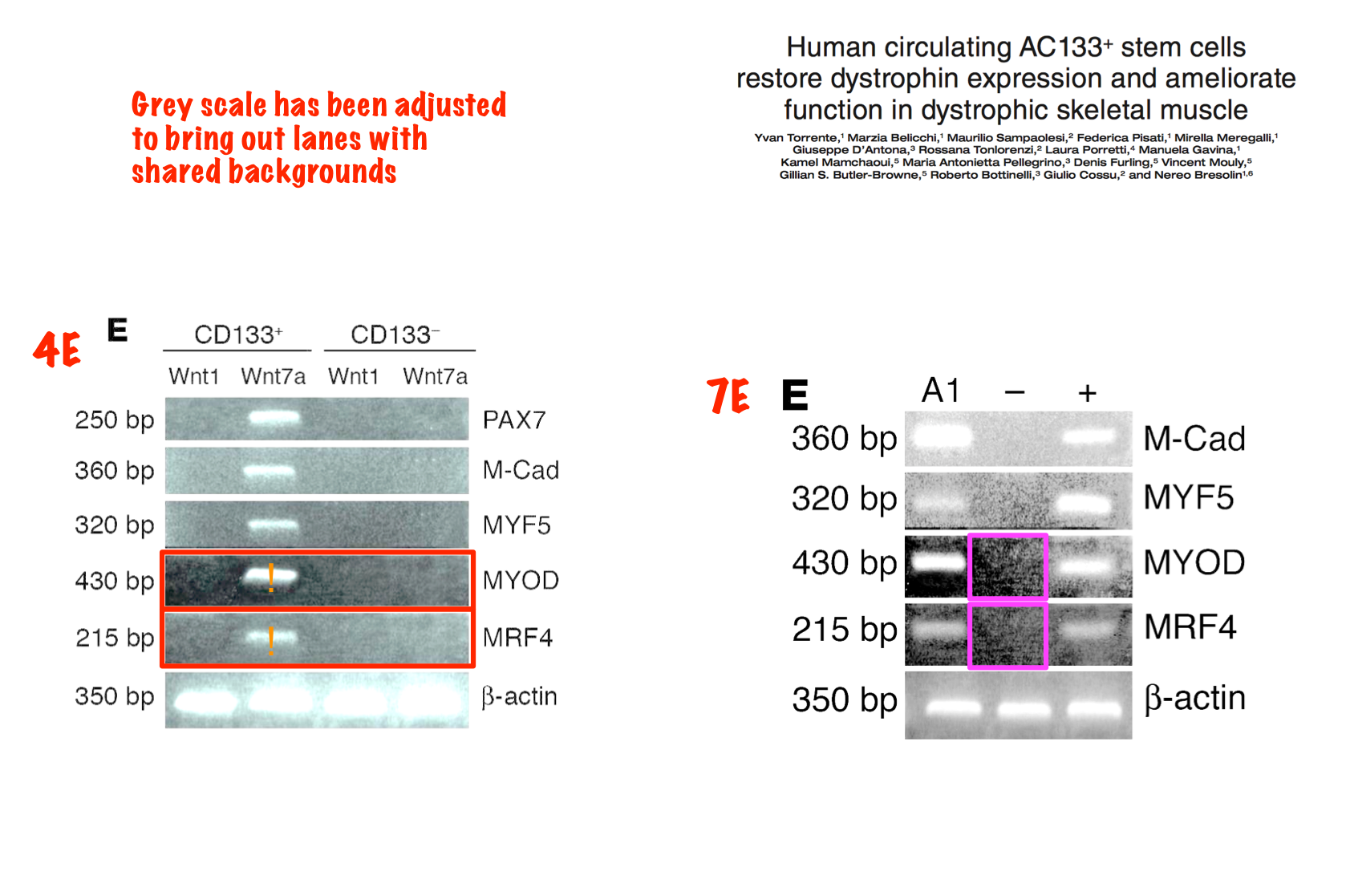
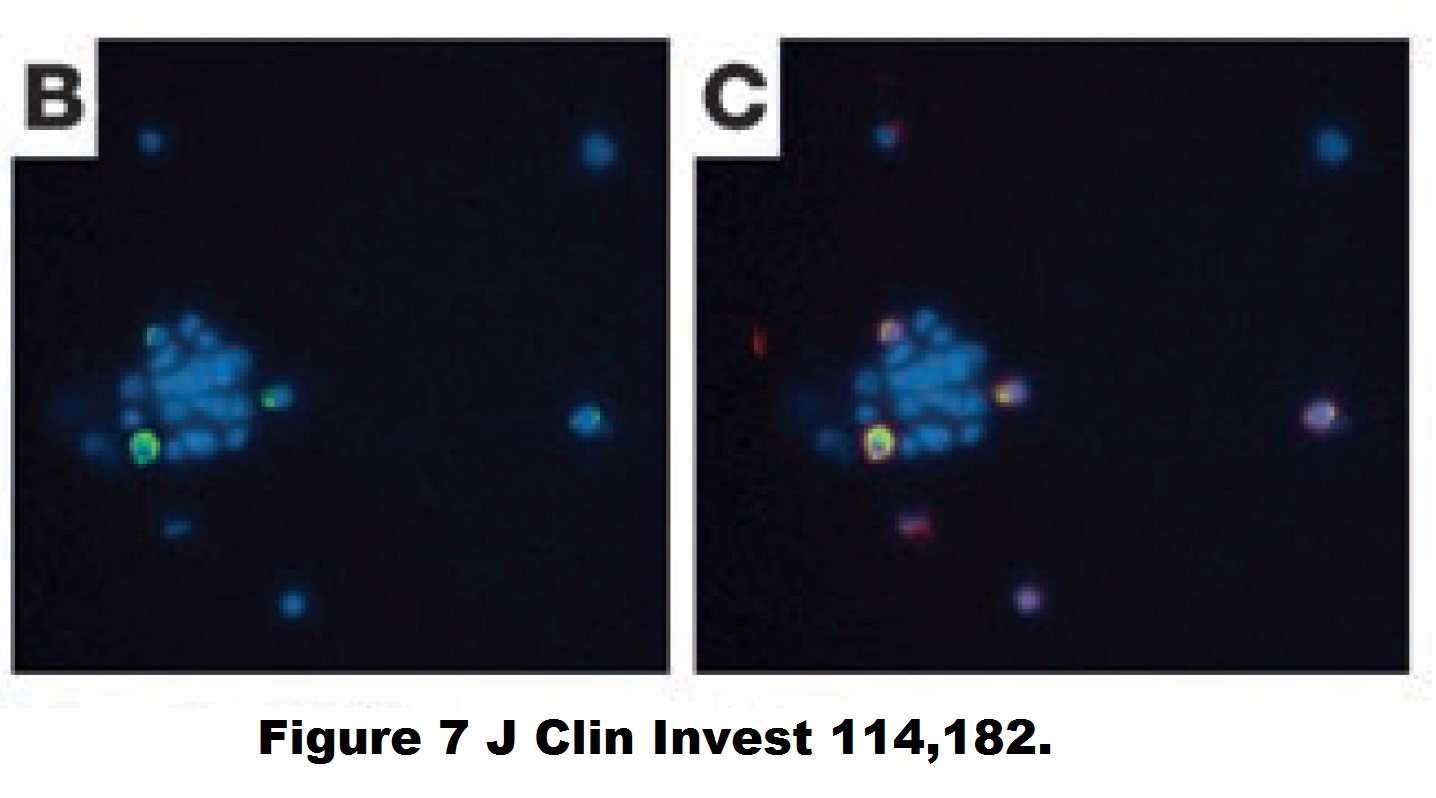
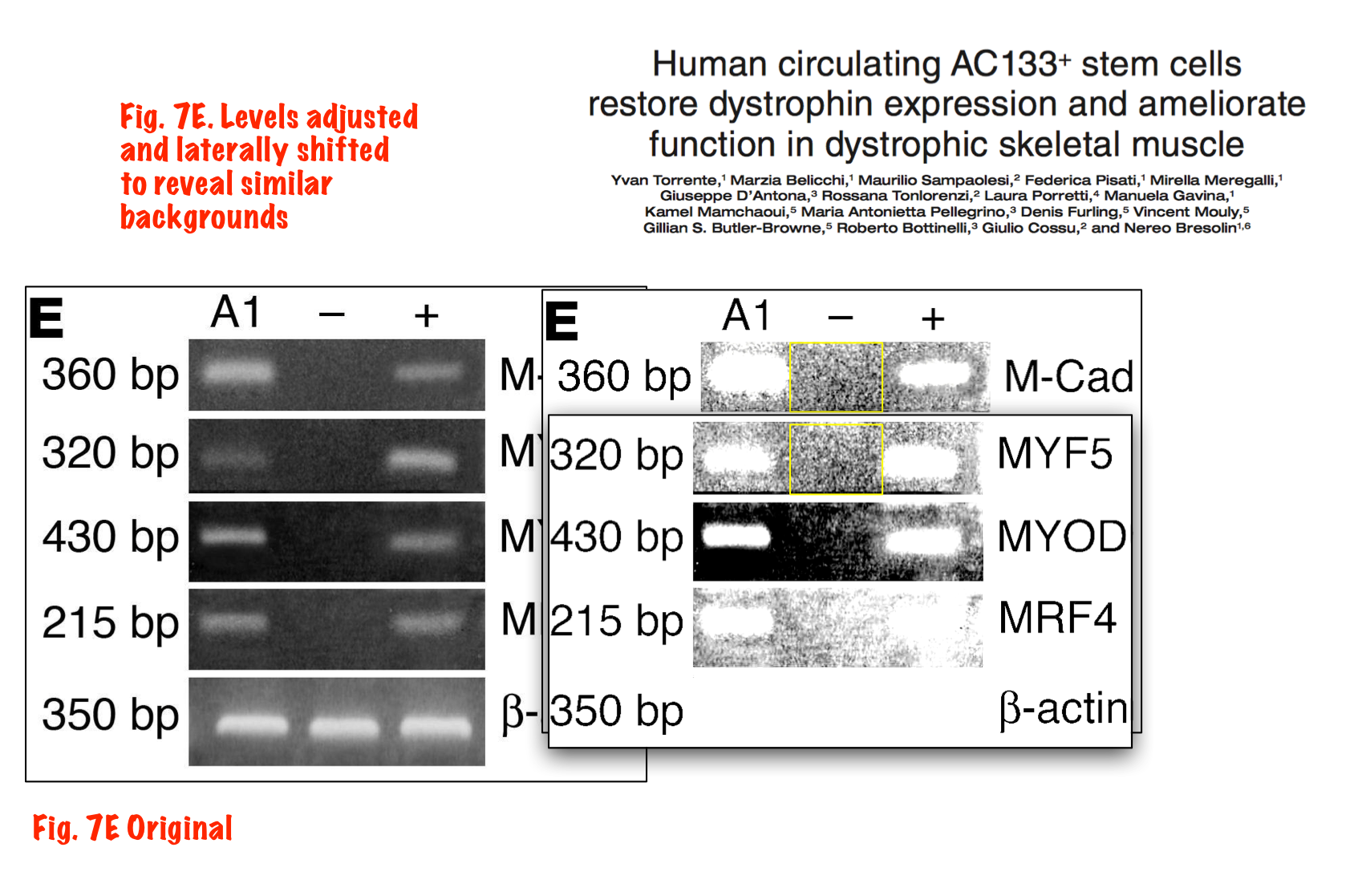
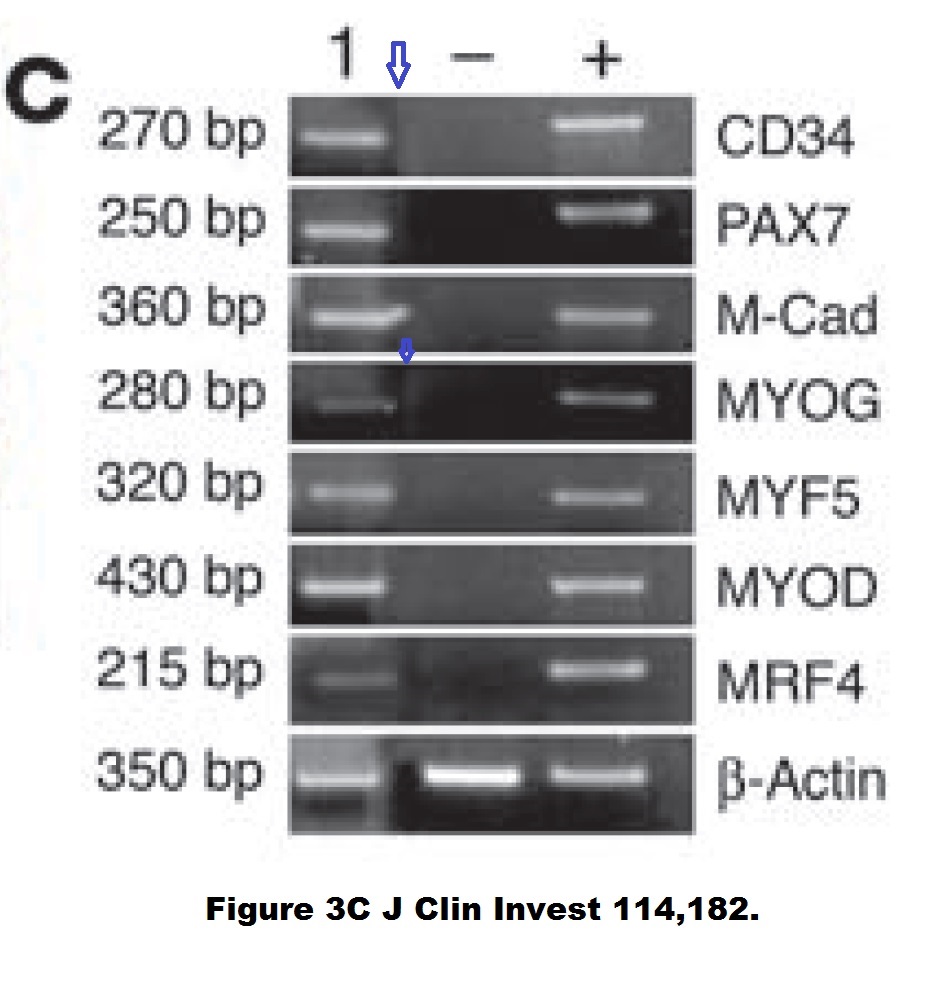
Judging form the issues above, this study is obviously fraudulent. But its authors made fine careers in academia, and anyway, nobody cares about those miraculous CD133 claims of the past now. Like heart stem cells, the CD133 claims have all been long forgotten and are never mentioned again, not even by their original discoverers. I personally think all past research worldwide about amazing stem cell successes like these should be checked for fraud.
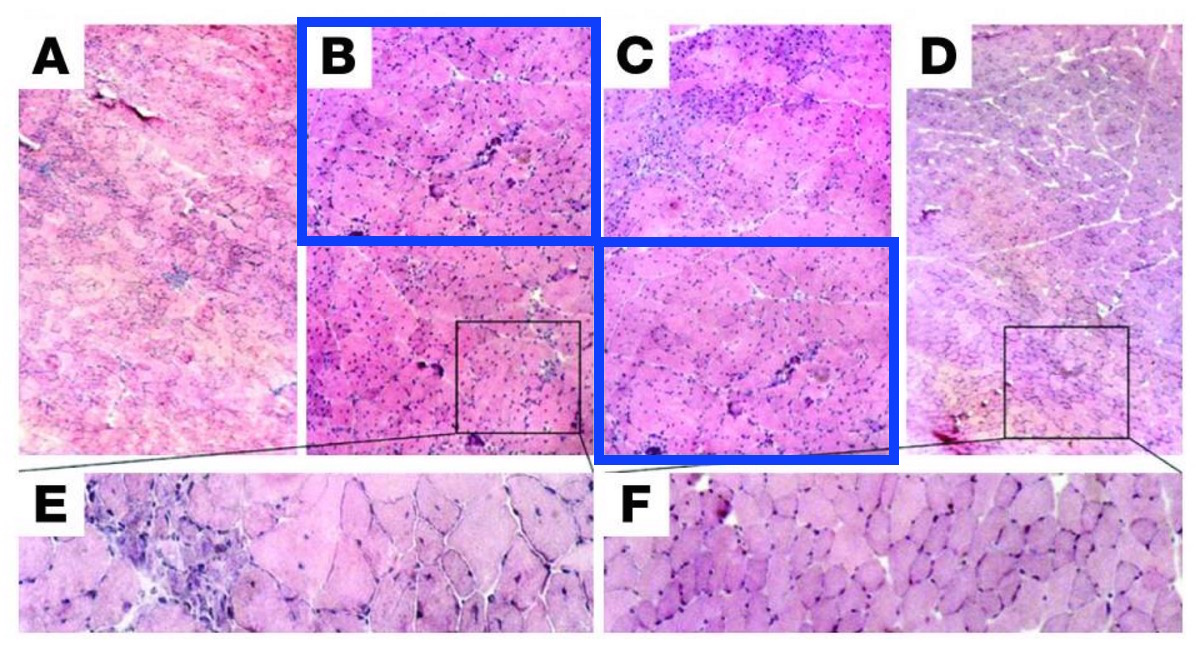
2006 was a very productive year for Cossu and Sampaolesi. The two men from Rome achieved impressive progress on muscle dystrophy in dogs, and even a “complete repair” of dystrophic muscle in mice!
Beatriz G. Galvez , Maurilio Sampaolesi , Silvia Brunelli , Diego Covarello , Manuela Gavina , Barbara Rossi , Gabriela Constantin , Gabriela Costantin , Yvan Torrente , Giulio Cossu Complete repair of dystrophic skeletal muscle by mesoangioblasts with enhanced migration ability The Journal of Cell Biology (2006) doi: 10.1083/jcb.200512085
The paper was subjected to corrections trice, 2x in 2006 and once in 2013, also in the same year concerns were expressed (and apparently assuaged since). In 2006, Figure 9B was replaced, because “the top left (Control) and top middle (Treated) panels under the “120 days” heading of Fig. 9 B were duplicated.“

The linked image diagrams the most obviously shared features, though everything in the lower left image can be mapped to both of the upper two images.“
The last correction was a biggie and mentioned “a clerical error during figure preparation”
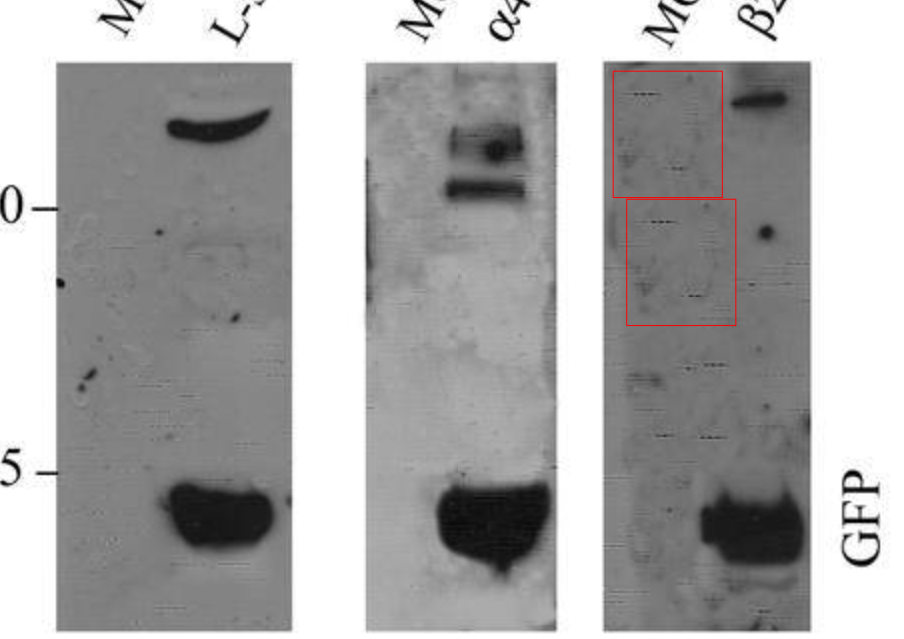
Those are simply forgeries. Elisabeth Bik, going through Sampaolesi’s publications in the wake of the affair of his current superior Verfaille, commented on PubPeer:
- “Correction 1 addressed the duplication in Figure 9B, but I agree with Peer 4 in comment #7 that the new figure 9 “Control” panel appears to be a composite of the “Treated” panel – making this replacement even worse than the original simple duplication.
- Correction 2 addressed a name misspelling.
- Correction 3 addressed duplications in Figures 1, 5, and 6, that are worth visualizing here.“
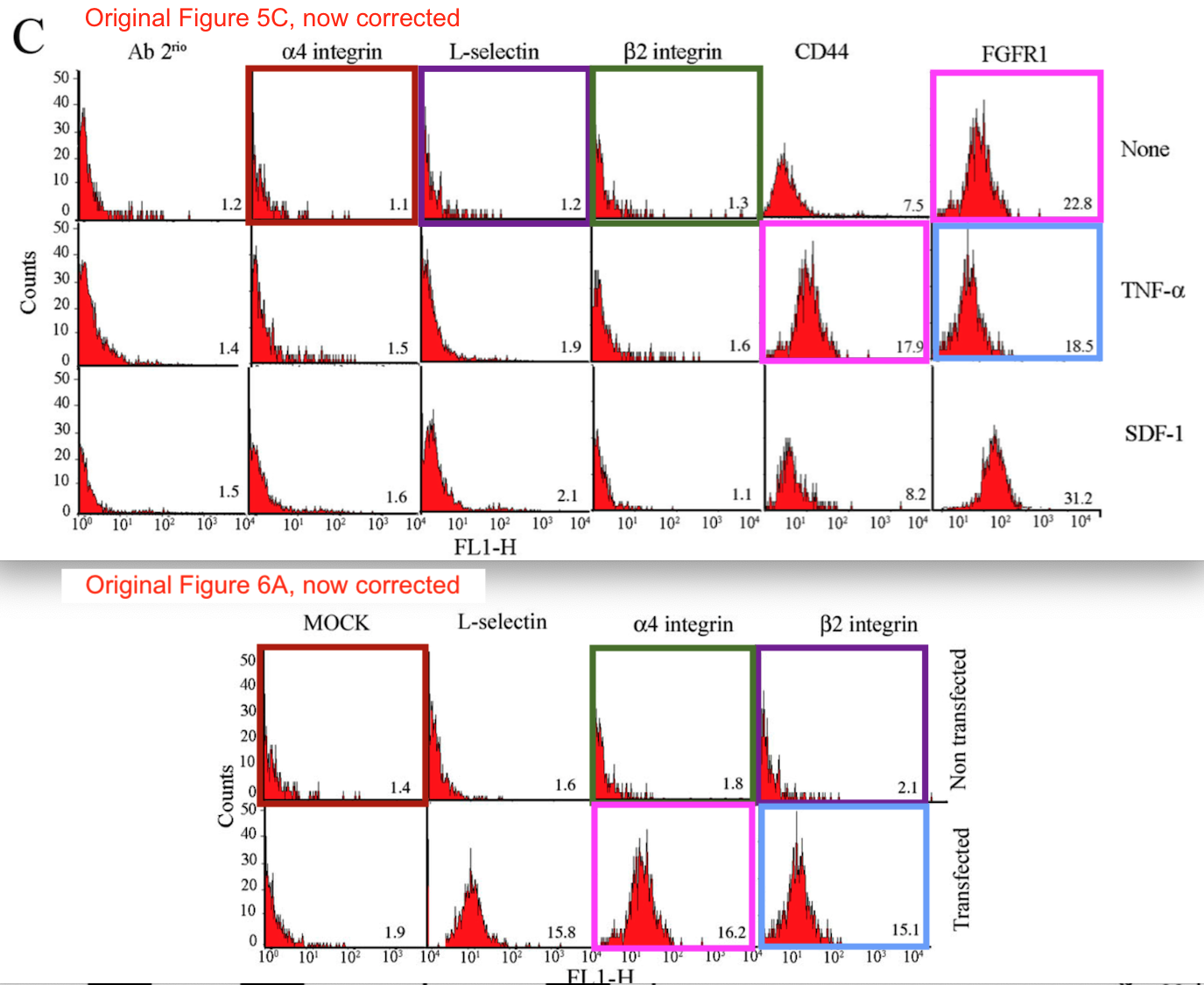
Bik noted:
“These are also not simple duplications, but appear to be duplicated FACS plots with altered numbers.”
But because this paper received all these corrections, you must trust the J Cell Biology editors that all conclusions remain unaffected and the corrected results are 100% reliable and trustworthy.
Cossu and Sampaolesi are really skilled issuers of corrections:
Marco Cassano , Stefano Biressi , Amanda Finan , Laura Benedetti , Claudia Omes , Renata Boratto , Frank Martin , Marcello Allegretti , Vania Broccoli , Gabriella Cusella De Angelis , Paolo M. Comoglio , Cristina Basilico , Yvan Torrente , Paolo Michieli , Giulio Cossu , Maurilio Sampaolesi Magic-factor 1, a partial agonist of Met, induces muscle hypertrophy by protecting myogenic progenitors from apoptosis PLoS ONE (2008) doi: 10.1371/journal.pone.0003223
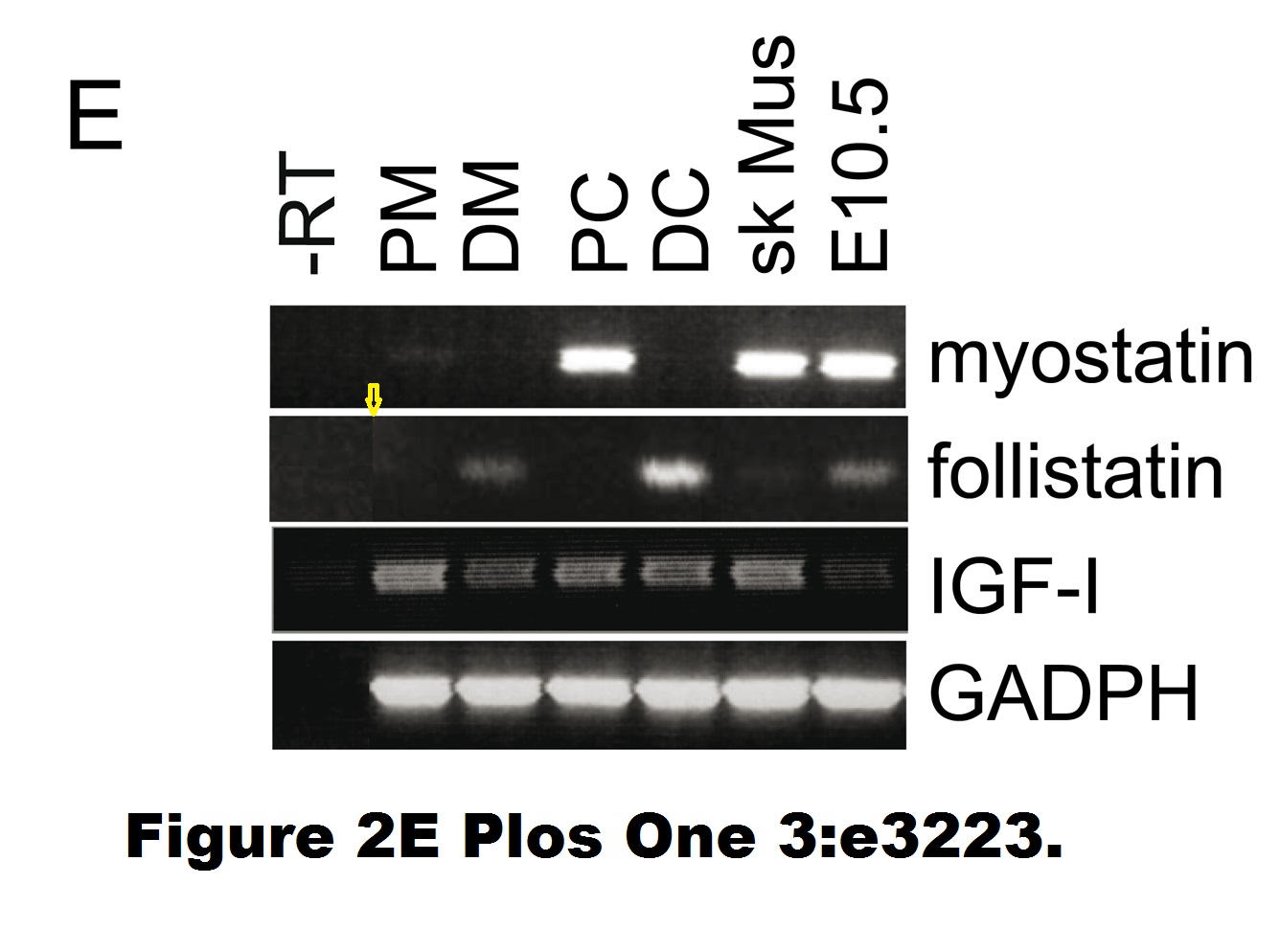
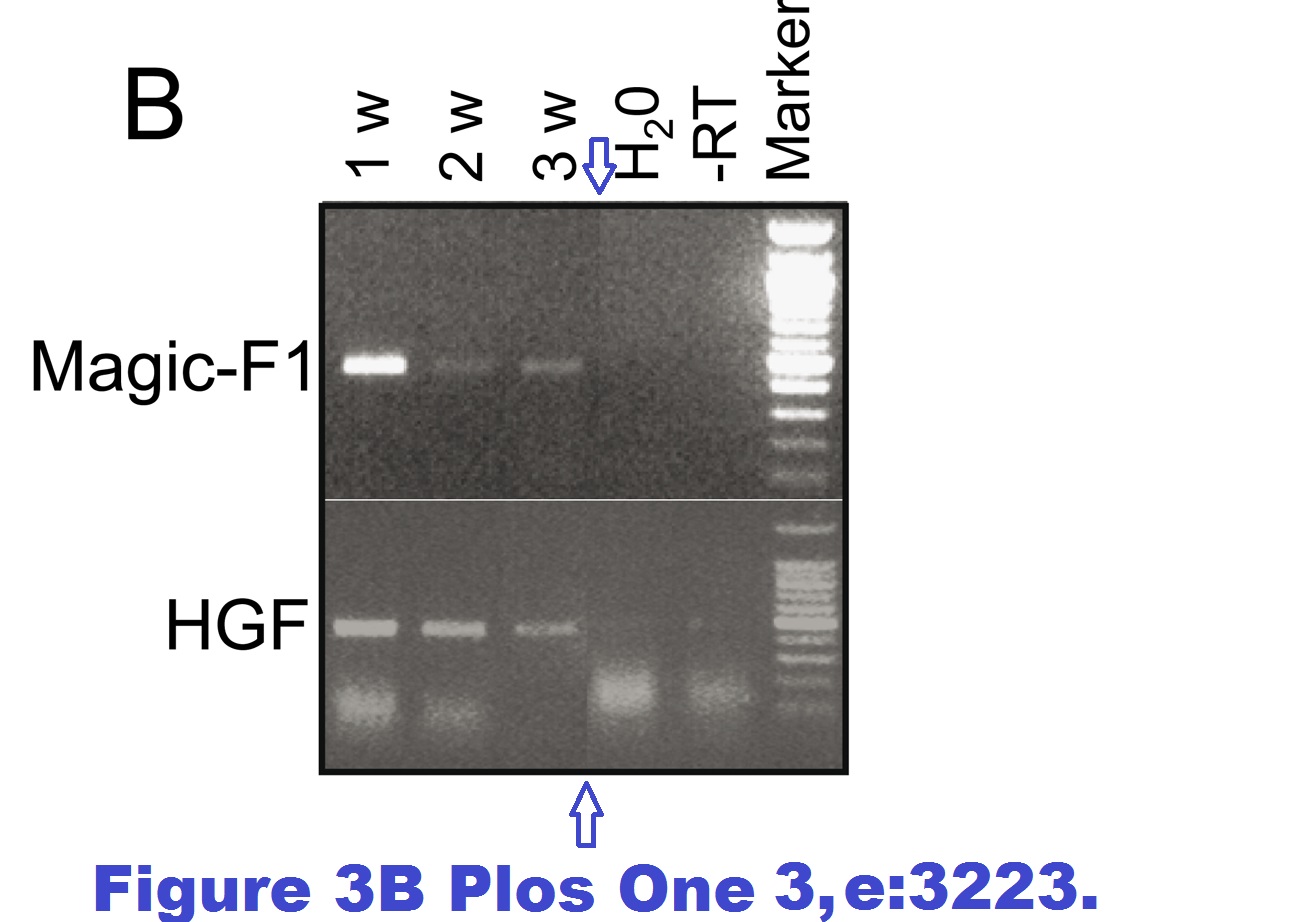
Also, an immunofluorescence image was misplaced. In 2019, Plos One published a correction:
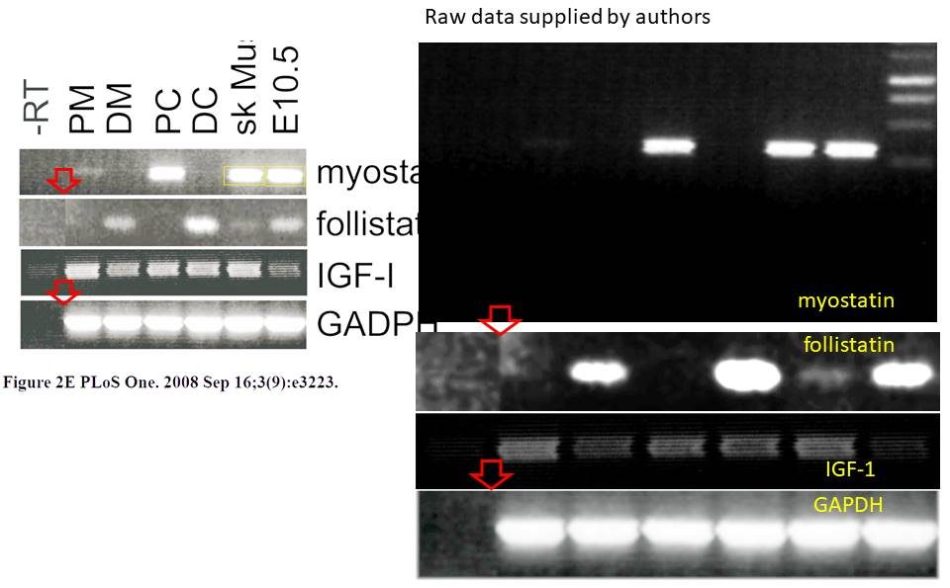
“The authors have provided the original images used in Figures 2E and 3B. The original images do not contain background irregularities and the source of the image artifact is unclear.”
Sure, the images the authors provided for Figure 3B show no splicing tracks anymore. But those for Figure 2E still do, maybe the authors forgot to fix it? Also, in January 2022 someone alleged on PubPeer a band duplication in this figure:
At some point, nobody was interested in correcting anything anymore:
Y. Torrente , M. Belicchi , C. Marchesi , G. D’antona , F. Cogiamanian , F. Pisati , M. Gavina , R. Giordano , R. Tonlorenzi , G. Fagiolari , C. Lamperti , L. Porretti , R. Lopa , M. Sampaolesi, L. Vicentini , N. Grimoldi , F. Tiberio , V. Songa , P. Baratta , A. Prelle , L. Forzenigo, M. Guglieri, O. Pansarasa, C. Rinaldi, V. Mouly, G. S. Butler-Browne, G. P. Comi, P. Biondetti, M. Moggio, S. M. Gaini, N. Stocchetti, A. Priori, M. G. D’angelo, A. Turconi, R. Bottinelli, G. Cossu, P. Rebulla, N. Bresolin Autologous transplantation of muscle-derived CD133+ stem cells in Duchenne muscle patients Cell Transplantation (2007) doi: 10.3727/000000007783465064
In 2013, Cossu left UCL for the University of Manchester, maybe he didn’t want to return to Milan where he was such a bigwig. In 2008, long before the trouble arrived, Sampaolesi used his impressive publication record of regenerative medicine successes to get a professorship at Verfaillie’s stem cell institute at KU Leuven. He is still there, and even if he is not an international big star of stem cell research who publishes in the big journals anymore, Sampaolesi seems to have grown bigger in other ways.
This is the kind of science Sampaolesi’s lab contributes to:
Riccardo D’Aquino , Virginia Tirino , Vincenzo Desiderio , Michèle Studer , Gabriella Cusella De Angelis , Luigi Laino , Alfredo De Rosa , Diego Di Nucci , Sabata Martino , Francesca Paino , Maurilio Sampaolesi, Gianpaolo Papaccio Human neural crest-derived postnatal cells exhibit remarkable embryonic attributes either in vitro or in vivo European Cells and Materials (2011) doi: 10.22203/ecm.v021a23
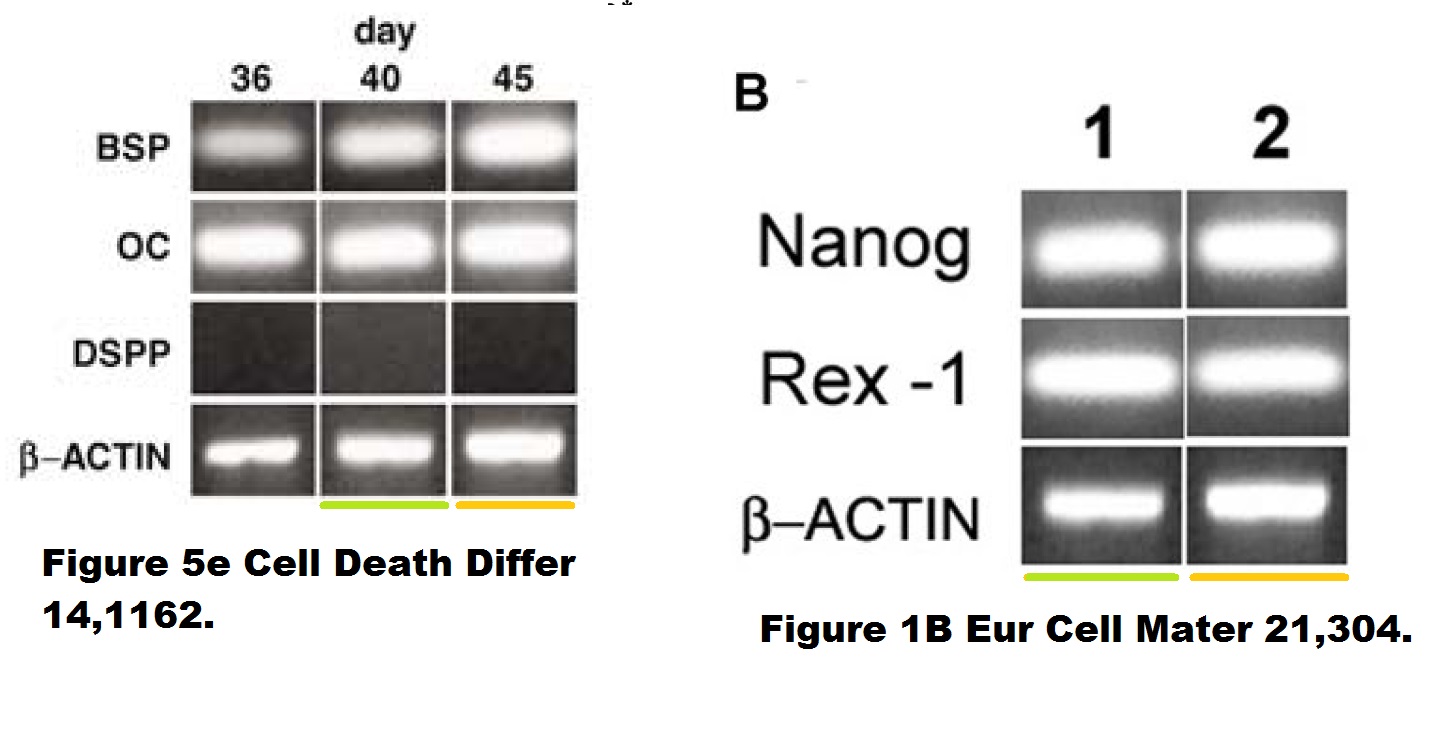
Sampaolesi never replied to my email (also Cossu remained silent). Here another paper by the KU Leuven professor:
T. Martinello , I. Bronzini , L. Maccatrozzo , A. Mollo , M. Sampaolesi, F. Mascarello , M. Decaminada , M. Patruno Canine adipose-derived-mesenchymal stem cells do not lose stem features after a long-term cryopreservation Research in Veterinary Science (2011) doi: 10.1016/j.rvsc.2010.07.024
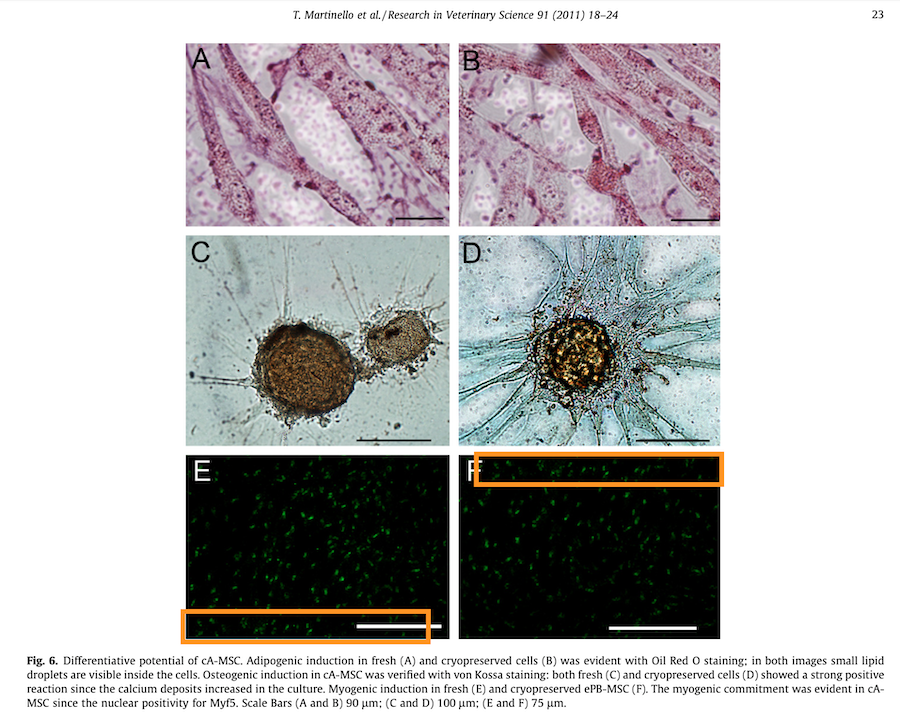
A mistake of oversight? Those are collaborative papers, and maybe the problematic figures were not done at KU Leuven. But there is a saying about your choice of friends showing who you really are. And every author is co-responsible, especially those senior authors in professorship position who knowingly decide to do nothing about the evidence.
Francesco D’Angelo , Ilaria Armentano , Ilaria Cacciotti , Roberto Tiribuzi , Mattia Quattrocelli , Costantino Del Gaudio , Elena Fortunati , Enrica Saino , Auro Caraffa , Giuliano Giorgio Cerulli , Livia Visai , Josè Maria Kenny , Maurilio Sampaolesi , Alessandra Bianco , Sabata Martino, Aldo Orlacchio Tuning multi/pluri-potent stem cell fate by electrospun poly(L-lactic acid)-calcium-deficient hydroxyapatite nanocomposite mats Biomacromolecules (2012) doi: 10.1021/bm3000716
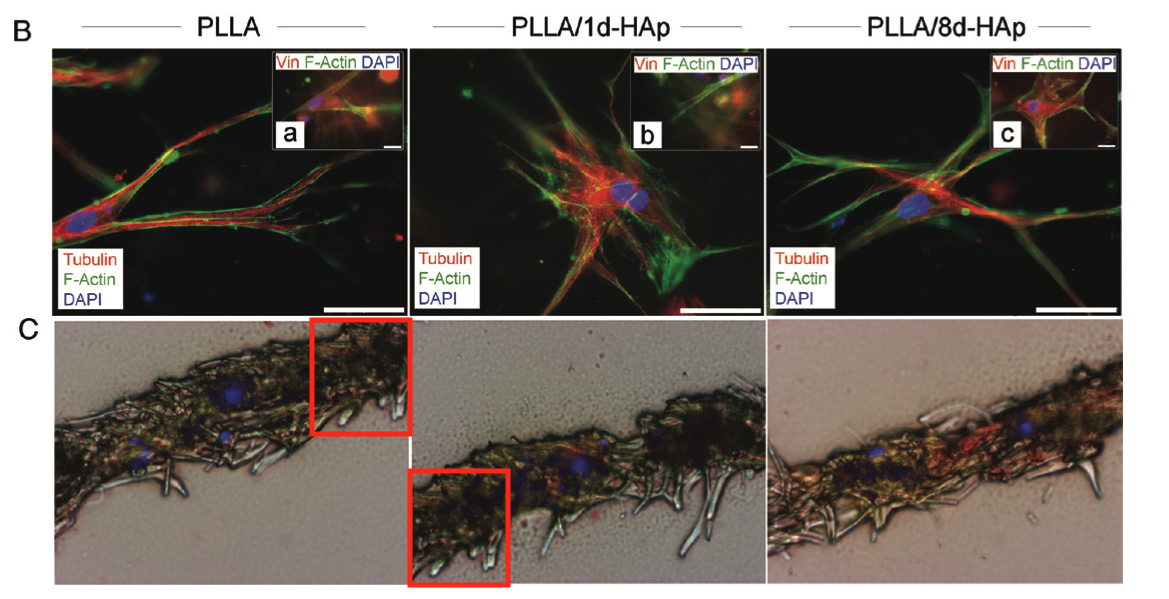
Aldo Orlacchio is a special case, look what this University of Perugia professor published together with Sampaolesi, uncovered by Elisabeth Bik:
Sabata Martino, Roberto Tiribuzi , Elisa Ciraci , Georgia Makrypidi , Francesco D’Angelo , Ilaria Di Girolamo , Angela Gritti , Gabriella M. Cusella De Angelis , Gianpaolo Papaccio , Maurilio Sampaolesi, Anna Concetta Berardi, Alessandro Datti, Aldo Orlacchio Coordinated involvement of cathepsins S, D and cystatin C in the commitment of hematopoietic stem cells to dendritic cells The International Journal of Biochemistry & Cell Biology (2011) doi: 10.1016/j.biocel.2011.02.001
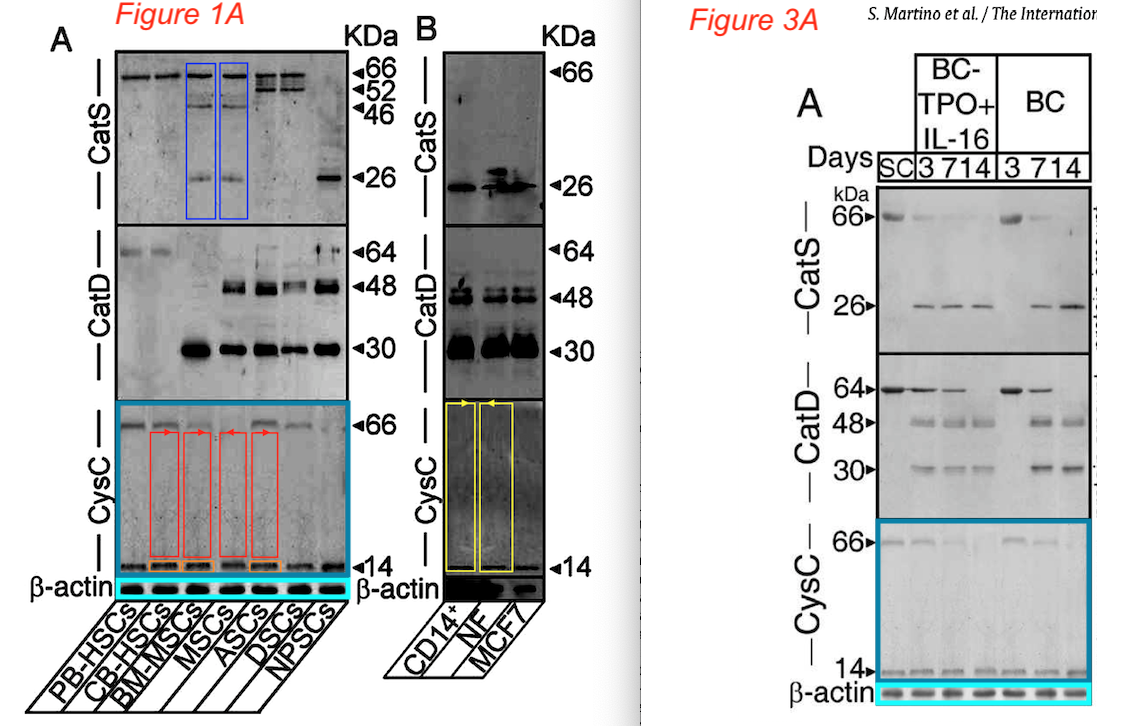
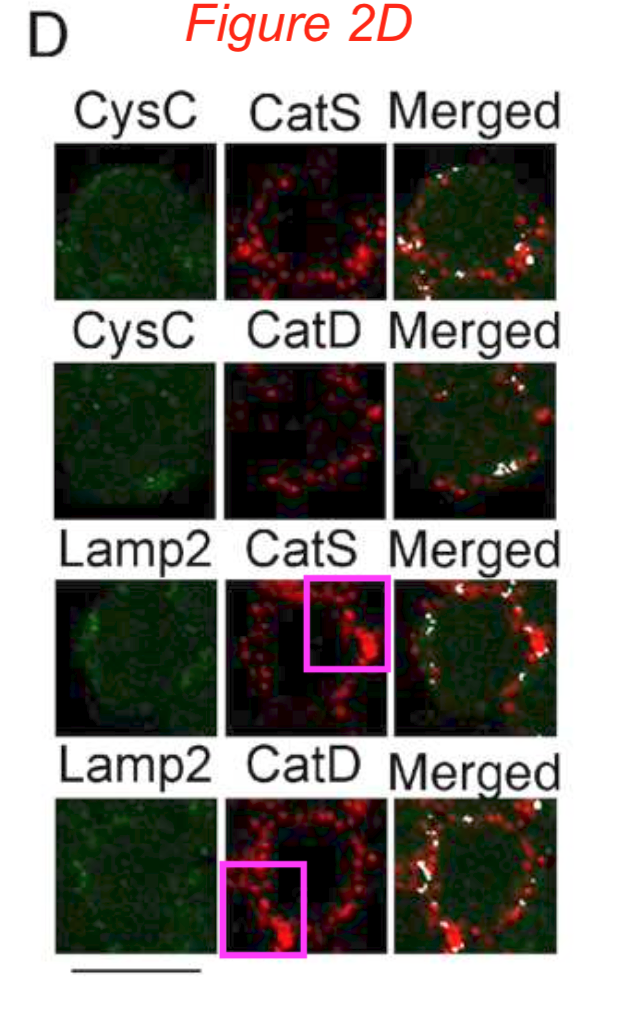
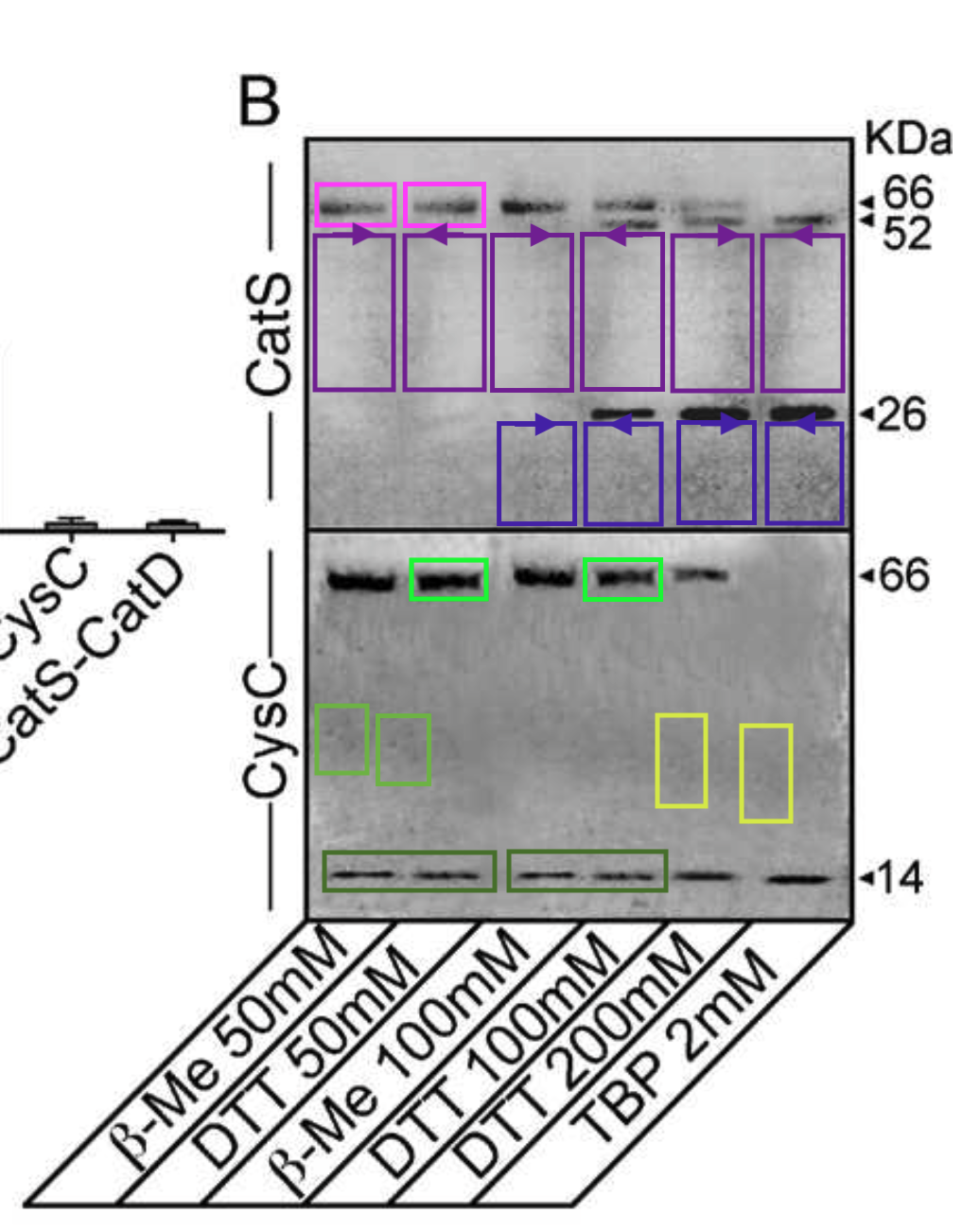
It’s an Elsevier journal, so exactly nothing at all was done. Not much is happening also with this study on “multipotent cells” from horse blood:
Tiziana Martinello , Ilaria Bronzini , Lisa Maccatrozzo , Ilaria Iacopetti , Maurilio Sampaolesi, Francesco Mascarello , Marco Patruno Cryopreservation does not affect the stem characteristics of multipotent cells isolated from equine peripheral blood Tissue Engineering Part C Methods (2010) doi: 10.1089/ten.tec.2009.0512
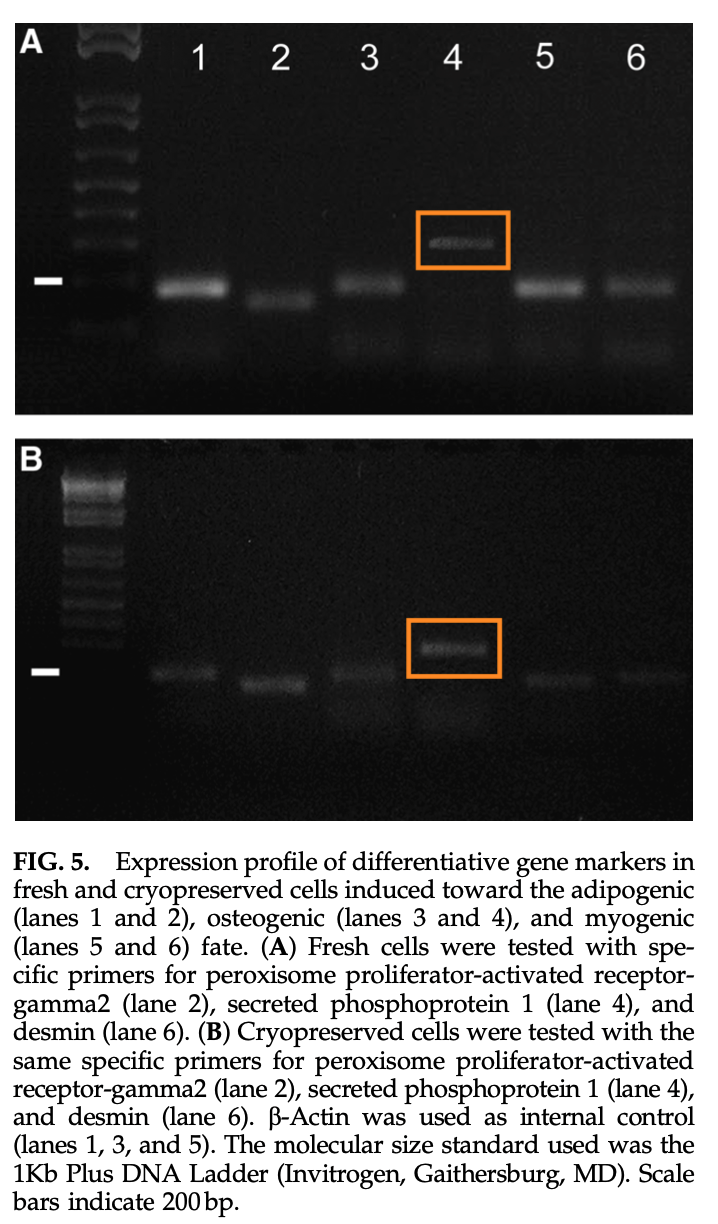

Academia is full of zombie scientists, fallen stars whom nobody should touch with a barge pole, yet they all are perfectly safe in their cushy well-paid faculty jobs, raising in their labs new generations of cheaters, occasionally angling a big grant or placing a paper in a fancy journal. Their networks hold, and while academia has no jobs for whistleblowers, there is always a warm place for a well-connected cheater.
Sampaolesi’s case is somewhat reminiscent of the story of another former young star of stem cell research, Jens Schwamborn, who is safely occupying a professorship in Luxembourg despite past research misconduct findings in Germany.
Yet Sampaolesi’s past papers with Cossu were whitewashed by UCL, and KU Leuven only investigates the work done on their campus. Thus, there is nothing to investigate, think of these amazing muscular dystrophy breakthroughs as vintage science fiction, or as jokes at the costs of the Duchenne patients.
The End.
Contact Form
I thank all my donors for supporting my journalism. You can be one of them!
Make a one-time donation:
I thank all my donors for supporting my journalism. You can be one of them!
Make a monthly donation:
Choose an amount
Or enter a custom amount
Your contribution is appreciated.
Your contribution is appreciated.



Pingback: Survival bias – ocasapiens
This might be an interesting question: what has become of major science related claims, in particular those discussed in mass media, from five, ten, twenty, fifty years ago? I have no systematic analyses for this, but I suspect the results might be less than stellar. Which then raises then leads to the question: is there any reason to think widely publicised results today are more trustworthy?
LikeLike
You could never work as a science journalist with such an attitude, tsk tsk.
LikeLike
Hi Leonid
Marvelous work.
That Stem cell lab tweet with the picture immediately reminded me of those dancing students in that french lab you wrote about a few years ago. (no not the one about IHU “il pleut de la merde”, the other one)
LikeLike
It’s a Nobel Prize lab!
https://forbetterscience.com/2018/10/02/the-alpha-males-of-physics/
LikeLike
“In the medieval Flemish town, there is a Catholic University (KU) of Leuven…”
J Clin Invest . 2005 Jan;115(1):118-27. doi: 10.1172/JCI19189.
Revascularization of ischemic tissues by PDGF-CC via effects on endothelial cells and their progenitorsXuri Li 1, Marc Tjwa, Lieve Moons, Pierre Fons, Agnes Noel, Annelii Ny, Jian Min Zhou, Johan Lennartsson, Hong Li, Aernout Luttun, Annica Pontén, Laetitia Devy, Ann Bouché, Hideyasu Oh, Ann Manderveld, Silvia Blacher, David Communi, Pierre Savi, Françoise Bono, Mieke Dewerchin, Jean-Michel Foidart, Monica Autiero, Jean-Marc Herbert, D Collen, Carl-Henrik Heldin, Ulf Eriksson, Peter Carmeliet
Affiliation1The Center for Transgene Technology and Gene Therapy, Flanders Interuniversitary Institute for Biotechnology (VIB), University of Leuven, Leuven, Belgium.
PMID: 15630451 PMCID: PMC535797 DOI: 10.1172/JCI19189
Problematic data figure 5A. Much more similar than expected.
FYI: Peter Carmeliet’s Pubpeer record: https://pubpeer.com/search?q=peter+carmeliet
According to Pubmed Peter Carmeliet has 731 publications.
https://pubmed.ncbi.nlm.nih.gov/?term=carmeliet%20p&sort=date&ac=yes
How is it possible to perform 731 separate pieces of scientific work?
LikeLike
Note that 5th author J Clin Invest . 2005 Jan;115(1):118-27. doi: 10.1172/JCI19189, Agnes Noel, has received a chunky 2022 Editor’s Note for:.
Cancer Res. 2014 Dec 1;74(23):6758-70. doi: 10.1158/0008-5472.CAN-13-2994. Epub 2014 Oct 15.
EGFR activation and signaling in cancer cells are enhanced by the membrane-bound metalloprotease MT4-MMP
Alexandra Paye 1, Alice Truong 1, Cassandre Yip 1, Jonathan Cimino 1, Silvia Blacher 1, Carine Munaut 1, Didier Cataldo 1, Jean Michel Foidart 1, Erik Maquoi 1, Joelle Collignon 2, Philippe Delvenne 3, Guy Jerusalem 2, Agnès Noel 4, Nor Eddine Sounni 4
Affiliations
1Laboratory of Tumor and Developmental Biology, Groupe Interdisciplinaire de Génoprotéomique Appliquée-Cancer (GIGA-Cancer), University of Liège, Liège, Belgium.
2Department of Medical Oncology, Centre Hospitalier Universitaire (CHU), Sart Tilman, Liège, Belgium.
3Department of Pathology, University of Liège, Liège, Belgium.
4Laboratory of Tumor and Developmental Biology, Groupe Interdisciplinaire de Génoprotéomique Appliquée-Cancer (GIGA-Cancer), University of Liège, Liège, Belgium. nesounni@ulg.ac.be agnes.noel@ulg.ac.be.
PMID: 25320013 DOI: 10.1158/0008-5472.CAN-13-2994
https://pubpeer.com/publications/8E9164E5AA9BE110BF5EFBDA87951B
2022 Editor’s Note.
https://aacrjournals.org/cancerres/article-abstract/82/4/734/678081/Editor-s-Note-EGFR-Activation-and-Signaling-in?redirectedFrom=fulltext
The editors are publishing this note to alert readers to concerns about this article (1). In Fig. 2, the actin loading control bands for cyclin D1 and cyclin D2 are identical–the authors clarified that the Western blots for cyclin D1 and cyclin D2 were performed on the same samples, however, this was not indicated in the figure legend. Additionally, in Fig. 6C, the p-EGFR bands in MDA-MB-231 cells showing stimulation by TGFα treatment are identical to the p-EGFR bands showing stimulation by EGF treatment. In the original submission of this manuscript, a correct version of this figure was used to show both TGFα and EGF could stimulate p-EGFR in control vector (CTR)– and MT4-MMP–expressing (MT4) MDA-MB-231 cells, but these panels were mistakenly duplicated in the revised and final versions of the manuscript.
LikeLike
J Clin Invest. 2005 Feb 1; 115(2): 237–246.
Published online 2005 Jan 13. doi: 10.1172/JCI22079
PMCID: PMC544035
PMID: 15650770
Role of Gas6 receptors in platelet signaling during thrombus stabilization and implications for antithrombotic therapy
Anne Angelillo-Scherrer,1 Laurent Burnier,1 Nathalie Flores,1 Pierre Savi,2 Maria DeMol,3 Paul Schaeffer,2 Jean-Marc Herbert,2 Greg Lemke,4 Stephen P. Goff,5 Glenn K. Matsushima,6 H. Shelton Earp,7 Christian Vesin,8 Marc F. Hoylaerts,3 Stéphane Plaisance,3 Désiré Collen,3 Edward M. Conway,3 Bernhard Wehrle-Haller,8 and Peter Carmeliet3
Author information
1 Division of Angiology and Hemostasis, Department of Internal Medicine, Faculty of Medicine and University Hospitals of Geneva, Geneva, Switzerland.
2 Department of Cardiovascular Research, Sanofi-Synth–labo Recherche, Toulouse, France.
3 Center for Transgene Technology and Gene Therapy, Flanders Interuniversity Institute for Biotechnology, and Center for Molecular and Vascular Biology, University of Leuven, Leuven, Belgium.
4 The Salk Institute for Biological Studies, San Diego, California, USA.
5 Department of Biochemistry and Molecular Biophysics, Columbia University, New York, New York, USA.
6 University of North Carolina Neuroscience Center, and
7 Lineberger Comprehensive Cancer Center, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA.
8 Department of Cellular Physiology and Metabolism, University of Geneva Medical School, Geneva, Switzerland.
Address correspondence to: Anne Angelillo-Scherrer, Division of Angiology and Hemostasis, University Medical Center, 1 rue Michel-Servet, CH-1211 Geneva 14, Switzerland. Phone: 41-22-379-55-67; Fax: 41-22-372-92-99;
E-mail: Anne.Angelillo@medecine.unige.ch
Problematic data figure 4. Much more similar than expected.
LikeLike
Additional problematic data . J Clin Invest. 2005 Feb 1; 115(2): 237–246.
Problematic data figure 5. Resting FACS profiles much more similar than expected for different genotypes (different samples).
LikeLike
Proc Natl Acad Sci U S A. 2005 Jul 26; 102(30): 10587–10592.
Published online 2005 Jul 18. doi: 10.1073/pnas.0502752102
PMCID: PMC1180776
PMID: 16027358
Immunology
Gene targeting of VEGF-A in thymus epithelium disrupts thymus blood vessel architecture
Susanna M. Müller,* Grzegorz Terszowski,* Carmen Blum,* Corinne Haller,†‡ Viviane Anquez,†§ Stephen Kuschert,† Peter Carmeliet,¶ Hellmut G. Augustin,∥ and Hans-Reimer Rodewald*†**
Author information
*Department of Immunology, University of Ulm, D-89081 Ulm, Germany; †Basel Institute for Immunology, CH-4005 Basel, Switzerland;
¶Flanders Interuniversity Institute for Biotechnology, Katholieke Universiteit, B-3000 Leuven, Belgium; and
∥Department of Vascular Biology and Angiogenesis Research, Tumor Biology Center, D-79106 Freiburg, Germany
** To whom correspondence should be addressed. E-mail: hans-reimer.rodewald@uni-ulm.de
‡Present address: Universität Basel, 4056 Basel, Switzerland.
§Present address: F. Hoffmann La Roche, CH-4070, Basel, Switzerland.
Edited by Diane Mathis, Harvard Medical School, Boston, MA
Figure 3F. Much more similar than expected.
LikeLike
Am J Pathol. 2005 Sep; 167(3): 721–732.doi: 10.1016/S0002-9440(10)62046-9
PMCID: PMC1698739PMID: 16127152
Fibrin-Induced Skin Fibrosis in Mice Deficient in Tissue Plasminogen Activator
Alexander de Giorgio-Miller, Steve Bottoms, Geoffrey Laurent, Peter Carmeliet,† and Sarah Herrick
Author information
From the Department of Medicine,* University College London, Rayne Institute, London, United Kingdom; and
the Centre for Transgene Technology and Gene Therapy,† Flanders Interuniversity Institute for Biotechnology, Katholieke Universiteit Leuven, Leuven, Belgium
Supported by Johnson and Johnson Medical Limited and the MedicalResearch Council, UK.Accepted for publication June 2, 2005.Address reprint requests to Dr. Sarah Herrick, Faculty of Life Sciences,3.239 Stopford Bldg., The University of Manchester, Oxford Rd.,Manchester M13 9PT. E-mail: sarah.herrick@manchester.ac.uk
Problematic data figures 4A and 4C. Much more similar than expected.
LikeLike
How are they related?
True joker is on the left.
https://www.vibconferences.be/speaker/peter-carmeliet
Dr. Peter Carmeliet is head of the Laboratory of Angiogenesis and Vascular Metabolomics and former director of the VIB — Vesalius Research Center, at the University of Leuven in Belgium. He graduated as Doctor in Medicine in 1984, and completed his Ph.D. in Medicine in 1989. During his Postdoctoral work at the Whitehead Institute, MIT in Cambridge USA, he acquired the knockout technology. After his return to Leuven in 1992, Dr. Carmeliet started his own research group with a focus on how blood vessels grow (angiogenesis) in health and disease. By developing transgenic mice lacking VEGF, he discovered that VEGF is a key player in angiogenesis. Dr. Carmeliet’s findings have led to the (pre)clinical development of novel therapeutic strategies for angiogenic diseases. He also showed that PlGF is a disease—restricted candidate in many angiogenic disorders and tested the therapeutic potential of anti-PlGF.
Dr. Carmeliet published over 662 research articles, reviews and book chapters (> 75.650 citations, h-index: 129). He is recognized as one of the top 400 most influential scientists in biomedical research. He has been elected member of the Royal Netherlands Academy of Arts and Sciences (KNAW), the European Molecular Biology Organization (EMBO) and the German Academy of Science Leopoldina and the European Academy of Sciences (eurasc). In 2010 he was awarded Doctor Honoris Causa from the Johann Wolfgang Goethe University, Germany. His achievements have been recognized by several honors and awards, such as the the Heineken Prize for Medicine (2018), ARC Foundation Léopold Griffuel Award (2018), the Blaise Pascal Medal in Medicine and Life Sciences by the European Academy of Sciences (2011), Ernst Jung Medical Award (2010), the FWO Joseph Maisin Prize for Excellence in Fundamental Biomedical Research (2010), the ERC Advanced Research Grant (2010), the Francqui Chair, Brussels (2007, 2006), the Interbrew Baillet Latour Health Prize (2005), the Outstanding Investigator Award from the International Society of Heart Research (2002) and the Marion Barnhart Memorial Award (1995). In 2015 he was granted the noble title of Baron by King Filip of Belgium.
https://en.wikipedia.org/wiki/Fernandel
Fernand Joseph Désiré Contandin (8 May 1903 – 26 February 1971), better known as Fernandel, was a French actor and singer. Born near Marseille, France, to Désirée Bedouin and Denis Contandin, originating in Perosa Argentina, an Occitan town located in the province of Turin, Italy.[1] He was a comedy star who first gained popularity in French vaudeville, operettas, and music-hall revues. His stage name originated from his marriage to Henriette Manse, the sister of his best friend and frequent cinematic collaborator Jean Manse. So attentive was he to his wife that his mother-in-law amusingly referred to him as Fernand d’elle (“Fernand of her”).[2][3]
LikeLike
Who advises the Belgian Royal Family on science?
King Filip has made Peter Carmeliet a Baron, and Queen Mathilde gave the 2013 Artois-Baillet Latour Foundation prize to Carlo Croce. It is quiet embarrassing that their advisers were unable to spot the flawed science.
http://retractiondatabase.org/RetractionSearch.aspx#?auth%3dCroce%252c%2bCarlo%2bM
10 retractions.
https://en.wikipedia.org/wiki/Carlo_M._Croce
https://retractionwatch.com/2019/07/19/osu-cancer-researcher-carlo-croce-loses-appeal-of-new-york-times-libel-suit/
LikeLike
J Biol Chem . 2001 Oct 19;276(42):39192-6. doi: 10.1074/jbc.C100428200. Epub 2001 Aug 23.
Hypoxia-inducible factor-2alpha (HIF-2alpha) is involved in the apoptotic response to hypoglycemia but not to hypoxia
K Brusselmans 1, F Bono, P Maxwell, Y Dor, M Dewerchin, D Collen, J M Herbert, P Carmeliet
Affiliation
1
Center for Transgene Technology and Gene Therapy, Flanders Interuniversity Institute for Biotechnology, KU Leuven, Leuven B-3000, Belgium.
PMID: 11546756 DOI: 10.1074/jbc.C100428200
Problematic data figure 1D. Multiple vertical changes in signal (splicing) blue arrows. HIF-2 alpha panel.
Two vertical strips much more similar (repeated) than expected (red rectangles). The data are not integral, but highly composite.
LikeLike
J Clin Invest. 2006 Sep 1; 116(9): 2344–2355.
Published online 2006 Aug 24. doi: 10.1172/JCI28123
PMCID: PMC1551932
PMID: 16932810
Critical function of Bmx/Etk in ischemia-mediated arteriogenesis and angiogenesis
Yun He,1,2 Yan Luo,1,3 Shibo Tang,1,3 Iiro Rajantie,4 Petri Salven,4 Matthias Heil,5 Rong Zhang,1 Dianhong Luo,1 Xianghong Li,1 Hongbo Chi,1 Jun Yu,1 Peter Carmeliet,6 Wolfgang Schaper,5 Albert J. Sinusas,1 William C. Sessa,1 Kari Alitalo,4 and Wang Min1
Author information
1Interdepartmental Program in Vascular Biology and Transplantation, Yale University School of Medicine, New Haven, Connecticut, USA.
2School of Public Health, and
3Zhongshan Ophthalmic Center, Sun Yat-Sen University, Guangzhou, People’s Republic of China.
4Molecular and Cancer Biology Program and Ludwig Institute for Cancer Research, Haartman Institute, Biomedicum Helsinki and Helsinki University Hospital, University of Helsinki, Helsinki, Finland.
5Department of Experimental Cardiology, Max-Planck-Institute for Heart and Lung Research, Bad Nauheim, Germany.
6Center for Transgene Technology and Gene Therapy, Flanders Interuniversity Institute for Biotechnology (VIB), Leuven, Belgium.
Address correspondence to: Wang Min, Interdepartmental Program in Vascular Biology and Transplantation, Department of Pathology, Yale University School of Medicine, BCMM 454, 295 Congress Avenue, New Haven, Connecticut 06510, USA. Phone: (203) 785-6047; Fax: (203) 737-2293; E-mail: wang.min@yale.edu
Problematic data figure 6A. Splice in IB: Bmx , but no splicing in IB:beta-Tubulin.
LikeLike
Additional problematic data . J Clin Invest. 2006 Sep 1; 116(9): 2344–2355.
Problematic data figure 1A. Splice in Bmx panel, but no splice in beta-Tubulin panel.
LikeLike
Development . 2007 Dec;134(24):4381-93. doi: 10.1242/dev.000026.
Ttrap is an essential modulator of Smad3-dependent Nodal signaling during zebrafish gastrulation and left-right axis determination
Camila V Esguerra 1, Luc Nelles, Liesbeth Vermeire, Abdelilah Ibrahimi, Alexander D Crawford, Rita Derua, Els Janssens, Etienne Waelkens, Peter Carmeliet, Desiré Collen, Danny Huylebroeck
Affiliation1Center for Transgene Technology and Gene Therapy, VIB, Herestraat 49, B-3000 Leuven, Belgium. camila.esguerra@med.kuleuven.be
PMID: 18039968 DOI: 10.1242/dev.000026
Problematic data figure 5C.
Straight, vertical change in signal between left and right lanes Smad4 lysate panel,
but no vertical change in signal TTRAP (lysate) panel.
LikeLike
J Biol Chem. 2009 Jul 31; 284(31): 20708–20717.
Published online 2009 Jun 1. doi: 10.1074/jbc.M109.018804
PMCID: PMC2742836
PMID: 19487690
Plasminogen Activator Inhibitor-1 Regulates Integrin αvβ3 Expression and Autocrine Transforming Growth Factor β Signaling*
Benjamin S. Pedroja,‡ Leah E. Kang,‡ Alex O. Imas,‡ Peter Carmeliet,§ and Audrey M. Bernstein‡,1
Author information
From the ‡Department of Ophthalmology, Mount Sinai School of Medicine, New York, New York 10029 and
the §Vesalius Research Center, Vlaams Instituut voor Biotechnologie and Katholieke Universiteit Leuven, 3000 Leuven, Belgium
1 To whom correspondence should be addressed: Mount Sinai School of Medicine, Box 1183, 1 Gustave L. Levy Place, New York, NY 10029., Fax: 212-289-5945; E-mail: audrey.berstein@mssm.edu
Figure 3. Much more similar than expected.
LikeLike
Data EMBO J . 2005 Sep 7;24(17):3093-103 very similar to data in Oncogene. 2005 Feb 17;24(8):1461-6,
yet some conditions are different.
EMBO J . 2005 Sep 7;24(17):3093-103. doi: 10.1038/sj.emboj.7600769. Epub 2005 Aug 18.
G1 checkpoint failure and increased tumor susceptibility in mice lacking the novel p53 target Ptprv
Gilles Doumont 1, Alain Martoriati, Chantal Beekman, Sven Bogaerts, Patrick J Mee, Fabrice Bureau, Emanuela Colombo, Myriam Alcalay, Eric Bellefroid, Francesco Marchesi, Eugenio Scanziani, Pier Giuseppe Pelicci, Jean-Christophe Marine
Affiliation
1
Laboratory for Molecular Cancer Biology, Flanders Interuniversity Institute for Biotechnology (VIB), University of Ghent, Ghent, Belgium.
PMID: 16107883 PMCID: PMC1201350 DOI: 10.1038/sj.emboj.7600769
Oncogene . 2005 Feb 17;24(8):1461-6. doi: 10.1038/sj.onc.1208256.
dapk1, encoding an activator of a p19ARF-p53-mediated apoptotic checkpoint, is a transcription target of p53
Alain Martoriati 1, Gilles Doumont, Myriam Alcalay, Eric Bellefroid, Pier Giuseppe Pelicci, Jean-Christophe Marine
Affiliation
1
Laboratory for Molecular Cancer Biology, Flanders Interuniversity Institute for Biotechnology (VIB), Technologiepark, 927, Ghent B-9052, Belgium.
PMID: 15608685 DOI: 10.1038/sj.onc.1208256
LikeLike
Additional Data EMBO J . 2005 Sep 7;24(17):3093-103 very similar to data in Oncogene. 2005 Feb 17;24(8):1461-6, yet some timings are different.
LikeLike
Cell Death Differ. 2010 Apr; 17(4): 633–641.Published online 2009 Dec 18.
doi: 10.1038/cdd.2009.202PMCID: PMC2892162NIHMSID: NIHMS181956PMID: 20019750
Monoallelic but not bialleleic loss of Dicer1 promotes tumorigenesis in vivo
Irina Lambertz,1 David Nittner,1 Pieter Mestdagh,2 Geertrui Denecker,1 Jo Vandesompele,
2 Mike A. Dyer,3 and Jean-Christophe Marine1,
*Author information
1Laboratory for Molecular Cancer Biology, VIB-UGent, B9052 Ghent, Belgium
2Center for Medical Genetics, Ghent University Hospital Hospital, B9000 Ghent, Belgium
3Department of Developmental Neurobiology, St Jude Children’s Research Hospital, Memphis, TN 38105, USA
1Laboratory for Molecular Cancer Biology, VIB-UGent, B9052 Ghent, Belgium
2Center for Medical Genetics, Ghent University Hospital Hospital, B9000 Ghent, Belgium
3Department of Developmental Neurobiology, St Jude Children’s Research Hospital, Memphis, TN 38105, USA
*Correspondence should be addressed to: Pr. Dr. Jean-Christophe Marine, VIB-UGent-KULeuven, Laboratory For Molecular Cancer Biology, Technologiepark, 927, B-9052 Ghent, Belgium,
Telephone: +32 (0)9 33 13640, Fax: +32 (0)9 33 13516, chris.marine@dmbr.ugent.be
Problematic data figure 2A. Much more similar than expected.
LikeLike
Mol Cell Biol. 2010 Nov;30(22):5394-405. doi: 10.1128/MCB.00330-10. Epub 2010 Sep 20.
Widespread overexpression of epitope-tagged Mdm4 does not accelerate tumor formation in vivo
Sarah De Clercq 1, Agnieszka Gembarska, Geertrui Denecker, Marion Maetens, Michael Naessens, Katharina Haigh, Jody J Haigh, Jean-Christophe Marine
Affiliation
1Laboratory for Molecular Cancer Biology, Department of Biomedical Molecular Biology, VIB-UGent, Technologiepark, B-9052 Ghent, Belgium.
PMID: 20855528 PMCID: PMC2976380 DOI: 10.1128/MCB.00330-10
Figure 2E. Vertical splicees in vinc panels, but not in myc panels.
LikeLike
EMBO J. 2010 Aug 4; 29(15): 2538–2552.Published online 2010 Jun 29. doi: 10.1038/emboj.2010.140PMCID: PMC2928696PMID: 20588255
MDM2 recruitment of lysine methyltransferases regulates p53 transcriptional outputLihong Chen,1 Zhenyu Li,1 Aleksandra K Zwolinska,2 Matthew A Smith,3 Brittany Cross,1 John Koomen,1 Zhi-Min Yuan,4 Thomas Jenuwein,5 Jean-Christophe Marine,2 Kenneth L Wright,3 and Jiandong Chena,1
Author information
1Molecular Oncology Department, Moffitt Cancer Center, Tampa, FL, USA
2VIB-KULeuven, Laboratory for Molecular Cancer Biology, Leuven, Belgium
3Immunotherapy Department, Moffitt Cancer Center, Tampa, FL, USA
4Department of Radiation Oncology, University of Texas Health Science Center, San Antonio, TX, USA
5Department of Epigenetics, Max-Planck Institute of Immunobiology, Freiburg, GermanyaMolecular Oncology Department, Moffitt Cancer Center, MRC3057A, 12902 Magnolia Drive, Tampa, FL 33612, USA.
Tel.: +1 813 745 6822; Fax: +1 813 745 6817; E-mail: jiandong.chen@moffitt.org
Problematic data figure 7D. Much more similar than expected.
LikeLike
Cell Death Differ. 2013 Jul; 20(7): 888–897.Published online 2013 Mar 1. doi: 10.1038/cdd.2013.12PMCID: PMC3679451PMID: 23449391
p53 promotes VEGF expression and angiogenesis in the absence of an intact p21-Rb pathway
M Farhang Ghahremani,1,2 S Goossens,1,2 D Nittner,3,4 X Bisteau,5 S Bartunkova,1,2 A Zwolinska,3,4 P Hulpiau,1,2 K Haigh,1,2 L Haenebalcke,1,2 B Drogat,1,2,7 A Jochemsen,6 P P Roger,5 J-C Marine,3,4,8 and J J Haigh1,2,8,*
Author information
1Vascular Cell Biology Unit, Department for Molecular Biomedical Research, VIB, Ghent, Belgium
2Department for Biomedical Molecular Biology, Ghent University, Ghent, Belgium
3Center for the Biology of Disease, VIB-KU Leuven, Leuven, Belgium
4Laboratory for Molecular Cancer Biology, Center for Human Genetics, Faculty of Medicine, KU Leuven, Leuven, Belgium
5Interdisciplinary Research Institute en Biologie Humaine et Moleculaire, Université Libre de Bruxelles, Campus Erasme, Brussels, Belgium
6Leiden University Medical Centre, Department of Molecular Cell Biology, Leiden, The Netherlands
6Leiden University Medical Centre, Department of Molecular Cell Biology, Leiden, The Netherlands
*Department for Biomedical Molecular Biology, Ghent University, Technologiepark 927, Ghent (Zwijnaarde), 9052 Belgium.
Tel: +32 9 33 13 661; Fax: +32 9 33 13 609; E-mail: Jody.haigh@dmbr.vib-UGent.be
7Current address: Interdisciplinary Research Institute en Biologie Humaine et Moleculaire, Université Libre de Bruxelles, Brussels, Belgium
8These authors contributed equally to this work.
Problematic data figure 1d. Vertical splice p53 panel, but not in beta-tubulin panel.
LikeLike
Eur J Cancer . 2016 Mar;55:98-110. doi: 10.1016/j.ejca.2015.12.002. Epub 2016 Jan 17.
p53 Reactivation by PRIMA-1(Met) (APR-246) sensitises (V600E/K)BRAF melanoma to vemurafenib
Mohammad Krayem 1, Fabrice Journe 1, Murielle Wiedig 1, Renato Morandini 1, Ahmad Najem 1, François Salès 1, Leon C van Kempen 2, Catherine Sibille 3, Ahmad Awada 4, Jean-Christophe Marine 5, Ghanem Ghanem 6
Affiliations
1Laboratory of Oncology and Experimental Surgery, Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium.
2Department of Pathology, McGill University and Lady Davis Institute for Medical Research, Jewish General Hospital, 3755 Chemin de la Côte-Sainte-Catherine, H3T 1E2 Montreal, QC, Canada.
3Department of Pathology, Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium.
4Medical Oncology Clinic, Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium.
5Laboratory for Molecular Cancer Biology, Center for Human Genetics, KUleuven, Leuven, Belgium; Laboratory for Molecular Cancer Biology, Center for the Biology of Disease, VIB-KULeuven, Belgium.
6Laboratory of Oncology and Experimental Surgery, Institut Jules Bordet, Université Libre de Bruxelles, Brussels, Belgium.
Electronic address: gghanem@ulb.ac.be
PMID: 26790143 DOI: 10.1016/j.ejca.2015.12.002
Data much more similar than expected.
https://pubpeer.com/publications/BFC9C3EF61AEB27C46BC3473003342#1
https://pubpeer.com/publications/BFC9C3EF61AEB27C46BC3473003342#3
https://pubpeer.com/publications/BFC9C3EF61AEB27C46BC3473003342#4
LikeLike
Cancers (Basel). 2019 Aug; 11(8): 1093.
Published online 2019 Aug 1. doi: 10.3390/cancers11081093
PMCID: PMC6721382
PMID: 31374895
The Benefit of Reactivating p53 under MAPK Inhibition on the Efficacy of Radiotherapy in Melanoma
Mohammad Krayem,1,2,* Malak Sabbah,1 Ahmad Najem,1 An Wouters,3 Filip Lardon,3 Stephane Simon,2 François Sales,1 Fabrice Journe,1,4 Ahmad Awada,1,5 Ghanem E. Ghanem,1 and Dirk Van Gestel2
Author information
1Laboratory of Oncology and Experimental Surgery, Institut Jules Bordet, Université Libre de Bruxelles, Rue Héger-Bordet 1, 1000 Brussels, Belgium
2Department of Radiation Oncology, Institut Jules Bordet, Université libre de Bruxelles, 1000 Brussels, Belgium
3Center for Oncological Research (CORE), University of Antwerp, 2610 Wilrijk, Belgium
4Department of Human Anatomy and Experimental Oncology, Université de Mons (UMons), Research Institute for Health Sciences and Technology, 7000 Mons, Belgium
4Department of Human Anatomy and Experimental Oncology, Université de Mons (UMons), Research Institute for Health Sciences and Technology, 7000 Mons, Belgium
*Correspondence: eb.tedrob@meyark.dammahom or eb.ca.blu@meyarkm; Tel.: +32-25413674; Fax: +32-25413349
Problematic data figure 2A. Much more similar than expected.
LikeLike
The email addresses of the corresponding author Cancers (Basel). 2019 Aug; 11(8): 1093
have become scrambled.
They should read:-
*Correspondence: mohammad.krayem@bordet.be or mkrayem@ulb.ac.be
LikeLike
Problematic data Clin Epigenetics . 2012 Jun 15;4(1):9. doi: 10.1186/1868-7083-4-9.
Problematic data Editor-in-Chief, Lucia Altucci, Clin Epigenetics.
Problematic data Clin Epigenetics . 2012 Jun 15;4(1):9. doi: 10.1186/1868-7083-4-9.
Clin Epigenetics . 2012 Jun 15;4(1):9. doi: 10.1186/1868-7083-4-9.
Hypermethylation of the 5′ CpG island of the p14ARF flanking exon 1β in human colorectal cancer displaying a restricted pattern of p53 overexpression concomitant with increased MDM2 expression
Christine Nyiraneza 1, Christine Sempoux, Roger Detry, Alex Kartheuser, Karin Dahan
Affiliation
1
Center for Human Genetics, Université Catholique de Louvain, Cliniques universitaires Saint-Luc, Avenue Hippocrate 10, Brussels, B-1200, Belgium.
karin.dahan@uclouvain.be
PMID: 22703554 PMCID: PMC3470942 DOI: 10.1186/1868-7083-4-9
Problematic data figure 1. Much more similar than expected.
Problematic data Editor-in-Chief, Lucia Altucci, Clin Epigenetics.
See: https://pubpeer.com/search?q=altucci
https://clinicalepigeneticsjournal.biomedcentral.com/about/editorial-board
Editorial Board
Editors-in-Chief
Lucia Altucci, Università degli Studi della Campania, Italy
Marianne Rots, University Medical Center Groningen, The Netherlands
Founding Editor
Ulrich Mahlknecht, St Lukas Clinic, Solingen, Germany
LikeLike
https://clinicalepigeneticsjournal.biomedcentral.com/about/editorial-board
Editorial Board
Editors-in-Chief
Lucia Altucci, Università degli Studi della Campania, Italy
http://retractiondatabase.org/RetractionSearch.aspx#?auth%3dAltucci%252c%2bLucia
1 retraction and 1 expression of concern to date for Lucia Altucci.
LikeLike
J Biol Chem. 2000 Jul 28;275(30):23227-33. doi: 10.1074/jbc.M000916200.
Cloning of two human thyroid cDNAs encoding new members of the NADPH oxidase family
X De Deken 1, D Wang, M C Many, S Costagliola, F Libert, G Vassart, J E Dumont, F Miot
Affiliation
1Institut de Recherche Interdisciplinaire, Bruxelles, Belgium.
PMID: 10806195
Figure 3. much more similar than expected.
LikeLike
Oncogene. 2005 Aug 25;24(36):5543-51. doi: 10.1038/sj.onc.1208735.
CBX7 controls the growth of normal and tumor-derived prostate cells by repressing the Ink4a/Arf locus
David Bernard 1, Juan F Martinez-Leal, Sian Rizzo, Dolores Martinez, David Hudson, Tapio Visakorpi, Gordon Peters, Amancio Carnero, David Beach, Jesus Gil
Affiliation
1Laboratory of Molecular Virology, Faculty of Medicine, Free University of Brussels, Brussels, Belgium.
PMID: 15897876
DOI: 10.1038/sj.onc.1208735
How to explain the apparent image duplication, and data from an earlier paper being used to represent a different experiment?
https://pubpeer.com/publications/15897876
LikeLike