Now that we are going through the worst pandemic anyone alive today can remember, maybe it is a good time to reconsider our attitude to research fraud. It is not just an academic issue, falsified data does actually very much affect the conclusions, and, believe it or not, bad science can kill. On the occasion of the coronavirus, this post by Smut Clyde will show you some awfully fake vaccine research.
COVID-19 is presently not curable, there is no pharmacological intervention available which could deal with this novel coronavirus. This unpleasant situation already led many national governments to follow the easy promises of a chloroquine-cure by the narcissistic French microbiologist Didier Raoult. Whose approach to science seems to be mostly about ignoring scientific evidence, bending all possible rules, and terrorizing his subordinates into delivering him the results he likes to see. No wonder Raoult is the favourite scientist of all dictatorially inclined politicians, starting with the US president Donald Trump.
What else is on offer? The chloroquine cure was originally applied in China, which Communist Party also suggests the Traditional Chinese Medicine (TCM) as the ultimate solution to the coronavirus. Indian Hindu nationalist government (who are big fans of chloroquine) combined both ideas by recommending an Ayurvedic malaria drug as COVID-19 therapy. In US, one university put forward Thomas Webster, a scientist with record of dishonesty, to seriously suggest nanoparticles as a possible solution. Then there are of course very predictably “stem cells“, peddled by Trump’s lawyer Rudy Giuliani and already approved by the US FDA.
Those of us who suspect being bullshitted, hope for a coronavirus vaccine to arrive soon. Problem is, vaccine development takes long time, it is unlikely an effective and safe vaccine can be delivered before the end of 2020 using established technology. Faced with the global COVID-19 disaster, politicians turned to other biotech solutions, which promise to deliver a different kind of a vaccine in a matter of months or even weeks, using mRNA approach.
Two companies are dominating the news: the German CureVac received an offer from Trump to buy their as of yet non-existent coronavirus vaccine and reserve it exclusively for USA, MAGA-style. German government had to interfere, CureVac’s CEO resigned and the company received a huge public investment, with the promise to deliver a vaccine by autumn 2020. Let us hope, because so far CureVac’s promises for a different vaccine ended up with nothing to show for but a failed phase 2 clinical trial.
In USA, CureVac’s competitor Moderna already opened recruitment to its human clinical trials for COVID-19 mRNA-based vaccine, without any previous preclinical testing for efficacy or safety. The safety testing would happen straight in human volunteers in a phase 1 trial. That however was put on hold on 30 March, maybe it is a good opportunity to recall this secretive company’s past now: Moderna (which also has yet to show a single success for any of its products in phase 2 clinical trial) was founded by the controversial former Harvard researcher Kenneth Chien (now at Karolinska). The research attitude at Moderna, as described by a STAT investigation, dampens the hope for their mRNA vaccines:
“Failed experiments have been met with reprimands and even on-the-spot firings. They recalled abusive emails, dressings down at company meetings, exceedingly long hours, and unexplained terminations.“
Vaccine research is a sensitive subject, because of the problems created by the worldwide antivaxxer movement, which even seems to find its supporters in the Israeli Academy of Sciences. Fraud in vaccine research would be definitely not helpful. And yet, this is what the following story by Smut Clyde is about: fake research into vaccine and cancer medicine delivery, in this case by some privileged Brahmin university crooks from Sagar, Madhya Pradesh, India. They offer fake nanotechnology approaches for fake vaccine delivery against real diseases like hepatitis B, tetanus and malaria.
Why don’t we all take this global coronavirus disaster as the opportunity to crack down on research fraud and bad science, nationally and worldwide? Do we really have much use for phony cures and false promises to deal with real diseases, especially now? Is it so difficult to understand that manipulated or outright fabricated research kills real people? No disease and no patient was ever cured by Photoshop, even if WHO itself seems to be on the fence.
Here is the spreadsheet with 75 fraudulent papers on vaccine and cancer research which Smut Clyde assembled. And now, meet the vaccine fraudsters:
Halo Payload: Why should we explain this to you?
By Smut Clyde

Just when you were getting used to ‘liposomes’ and ‘exosomes’ entering the advertising lexicon of cosmetics and health quackery as signifiers of Sciencyness, along came niosomes and transfersomes and vesosomes and emulsomes. Anyone who identified these as “The cows from Cold Comfort Farm” has failed the test (the judges’ decision is final).
In fact these are all additions to the menagerie of Drug Delivery research: different ways of sequestering a drug within a microscopic or nanoscopic vesicle with the idea of delivering the molecules to a specific tissue instead of wasting them all over the body. The drug might be encapsulated within cubosomes, for instance, in a form that can be snorted, then transported to the brain along olfactory neurons, thus dodging the blood-brain barrier. Because Drug Delivery straddles the boundary between nanotechnology and molecular biology, papers can feature faked diagrams from both fields – photoshopped electron microscopy and immunohistochemistry. So far “noisome”, “handsome”, “toothsome”, “quarrelsome” and “meddlesome” are not recognised as Drug Delivery terms, and now my ambition is to publish a paper introducing them all.
With that as context, here to pique the readers’ interest are Figures 7 and 6 respectively from Jain et al (2012) [A2] and Choudhary et al (2016) [B12].


They are delicate pastel crystals, yet somehow woven or crocheted as well. One can only be sure that there is nothing biological about them, even though the figure captions identify them as the stomach mucosa of rats, bearing alcohol-induced peptic ulcers: treated in [A2] with ranitidine bismuth citrate and amoxicillin trihydrate (delivered by concentric nested liposomes) and in [B12] with rabeprazole and amoxicillin (delivered by “polymeric low-density microballoons”). The two papers have Govind P. Agrawal as a shared author.
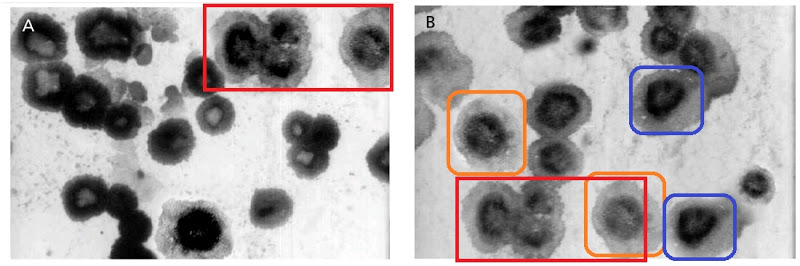
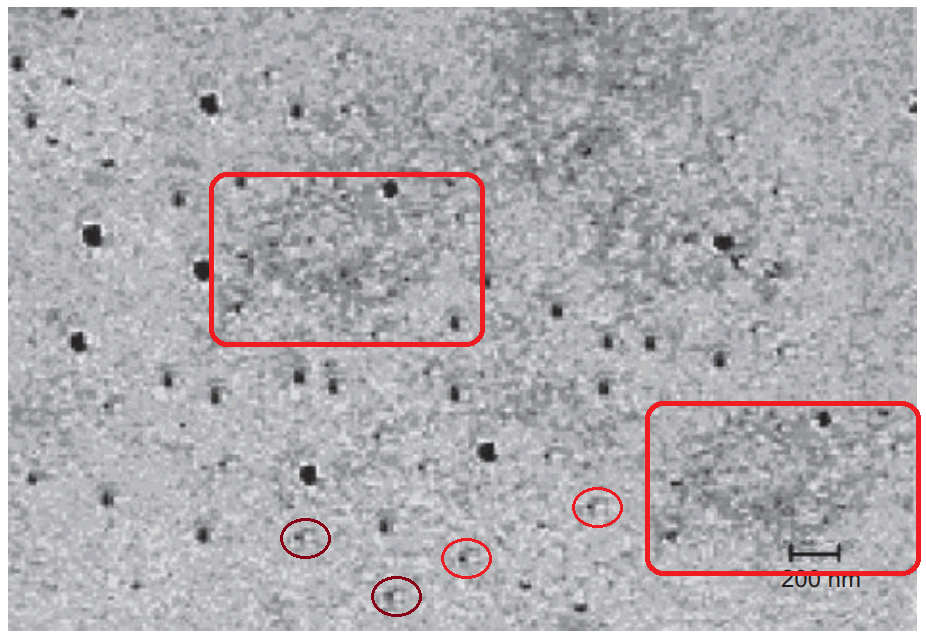
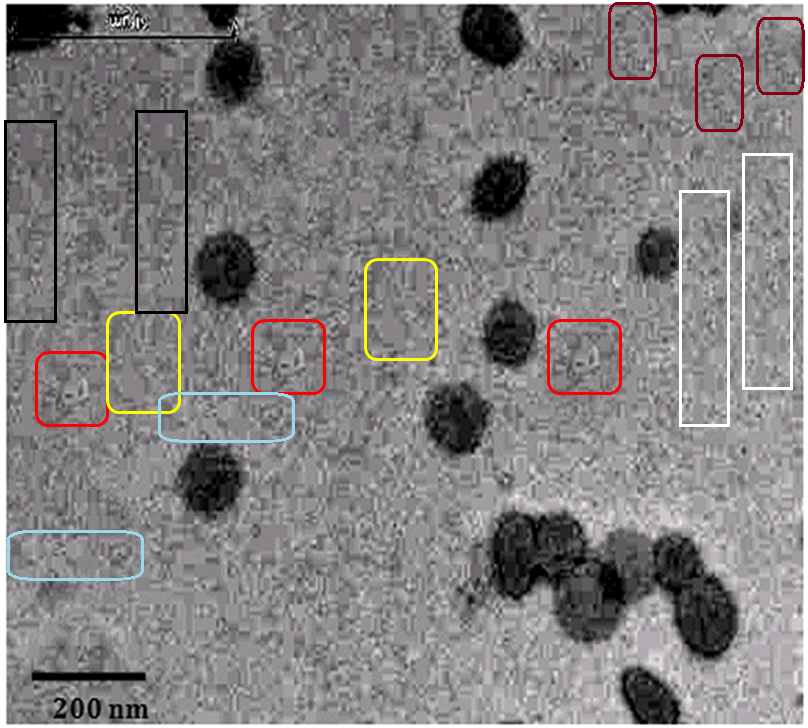
What is one to make of Figures 1 of Rai et al (2016) [B11] and 1 of Sudheesh et al (2011) [C17]? The former depicts “Eudragit-coated dextran microspheres of 5-fluorouracil” (targeting the colon) while the latter are “tetanus toxoid-loaded aminated gelatin nanoparticles” (for vaccines). Close inspection reveals the 2011 version to be a swarm of replicated pairs and triplets of spheres. Yet these Bubbles Congeries [Lovecraft 1931] have enough in common besides the background that their single origin is undeniable.

The authors of both versions are affiliated to the Department of Pharmaceutical Sciences at Dr. Hari Singh Gour University (Sagar, Madhya Pradesh, India) but in different laboratories.
As the head of the latter laboratory, a Dr S. P. Vyas responded on the PubPeer thread for [C17]. Alas, his answers revealed little, except that he is not accustomed to questions… not even to questions of a rhetorical nature, that are asked into the void and not directly at him. Also, it is below his dignity to haggle with anonymous critics, although onymous critics are equally beneath his contempt.
All the above is what is called “foreshadowing” in literary circles, and we will return to it. I feel drawn to examine Dr Vyas’ research oeuvre at Dr. H.S.G. University where advanced drug-delivery -somes promise cancer cures and next-generation oral or nasal vaccines. But a too-rapid ascent to his lofty eminence carries the risk of altitude sickness, and I prefer a more gradual approach. We begin with the ADINA Institute of Pharmaceutical Sciences (also of Sagar), headed by Dr Ashish Kumar Jain and source of the first of those crystallised / crocheted stomach linings [A2].
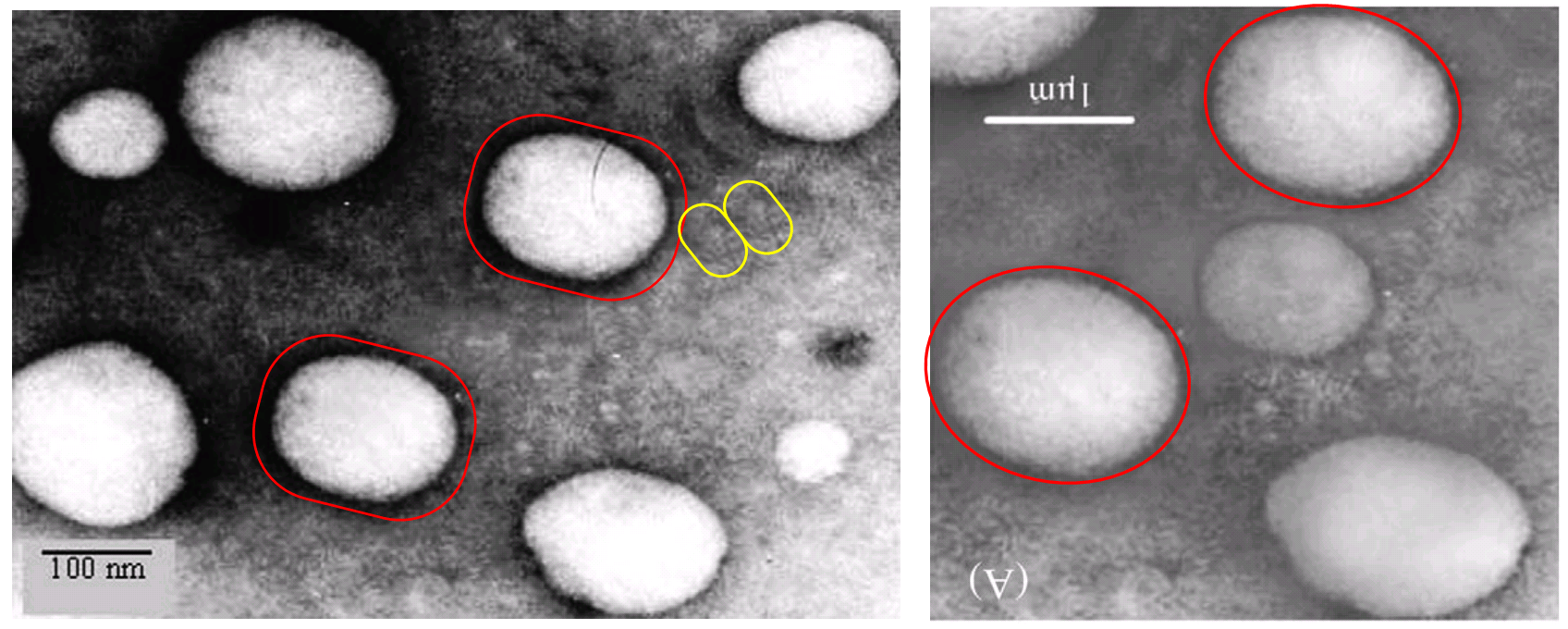
No-one can accuse the ADINA Institute of wasting research funds. Once they have created an image it receives maximum mileage. In a trilogy of papers, a Scanning Electron Microscopy (SEM) Photoshop variously depicts microballoons with payloads of Famotidine for the upper small intestine (Fig 3B, Choudan et al 2013 [A3]), or of Mesalamine chloride (1B, Jain & Jain 2013a [A4]); or perhaps they are phenylalanine-anchored liquid nanocarriers to treat AIDS-related encephalopathy (3B, Vyas et al 2015 [A9]). SEMs like this will be a recurring theme through the rest of this post. TEMs will be another.
These multiwalled carbon nanotubes conjugated with hyaluronic acid (to target colon cancer) were only used once, in Prajapati et al (2019) [A10]. But one cannot question the team’s Photoshop skills. I recognise 3(a) as a homage to the back-cover album art for “Dance of the Lemmings“.

Figures 7 from Bilthariya et al (2015) [A8] and 8 from Jain et al (2014) [A6] show tissue fluorescing after permeation with FITC. The only uncertainty is whether the tissue came from “inflamed joint sac” or “liver sac”, and whether its entry into the cells was facilitated by particles of folate-conjugated albumin or lactosaminated-N-succinyl chitosan, laden with etoricoxib or acyclovir.

At left, in Fig 6 of Mishra et al (2014) [A7] the tissue was liver again, and the facilitating nanoparticles were comprised of chitosan and “glycyrrhizin conjugated low molecular weight chitosan”. That is not definitive, though, as the images had lost focus so that (a) and (b) were barely recognisable.
[A6] and [A7] are also a convenient gateway to Figs 5(b) and 4(b), where the reason for manipulating one or other image (or both!) is unclear; perhaps it was for practice, or to create a Spot-the-Difference puzzle

The corresponding Figs 5(a) and 4(a) are identical, but they contain their own Spot-the-Embedded-Figures puzzle.
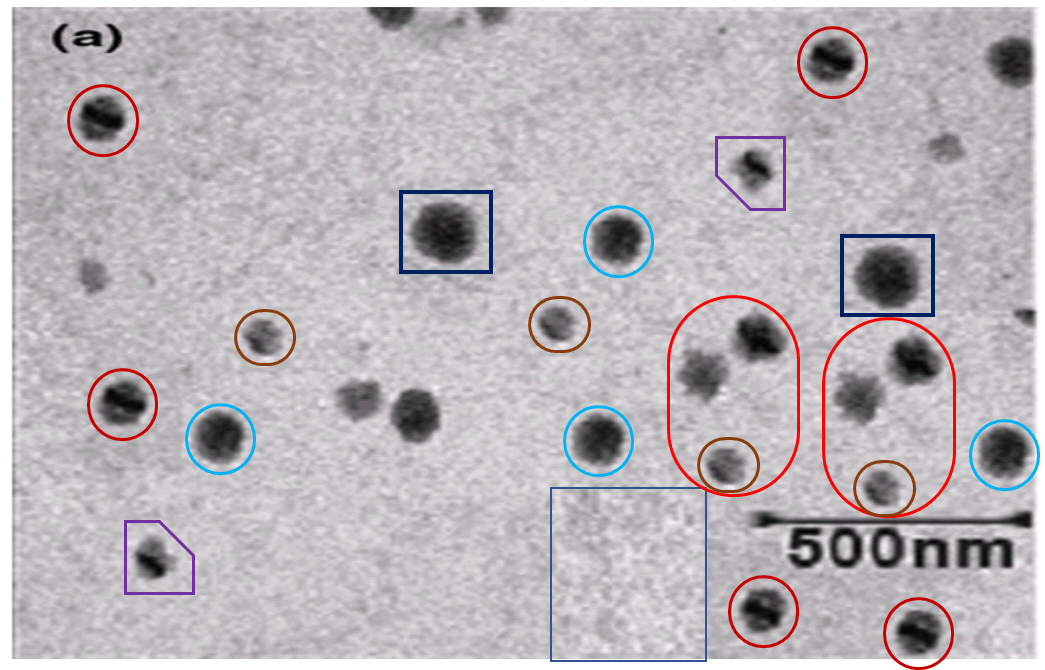
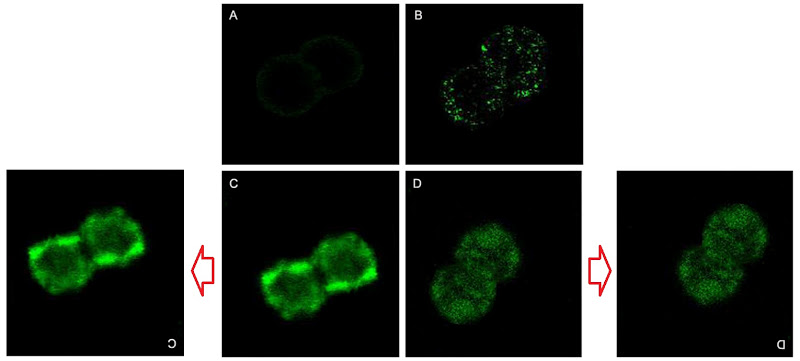
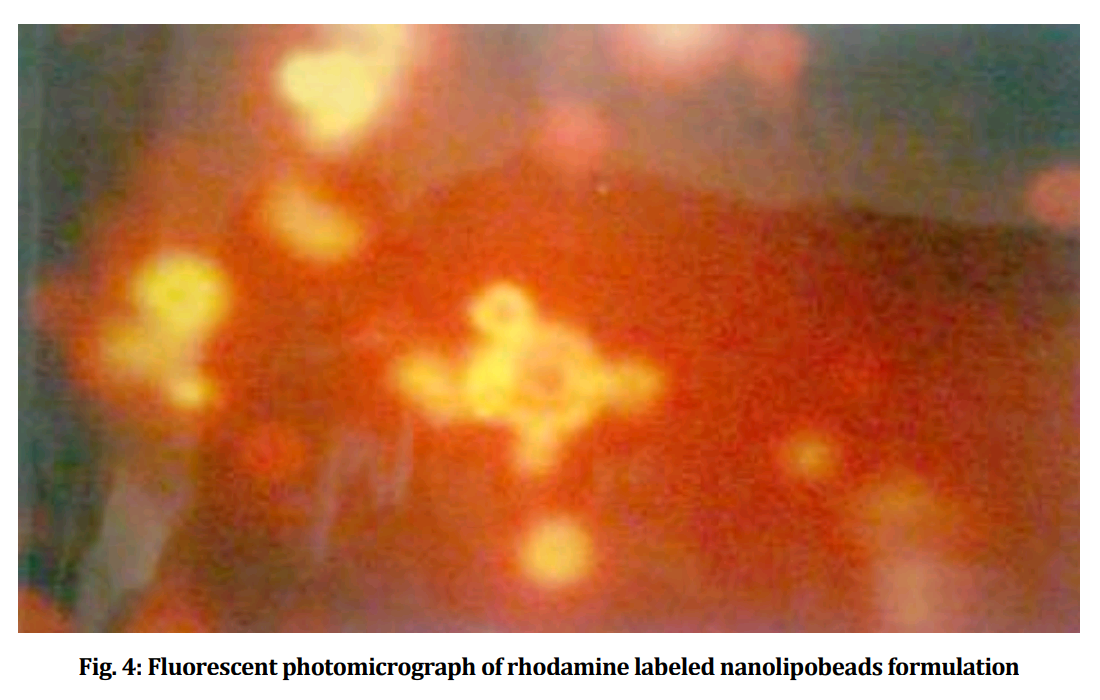
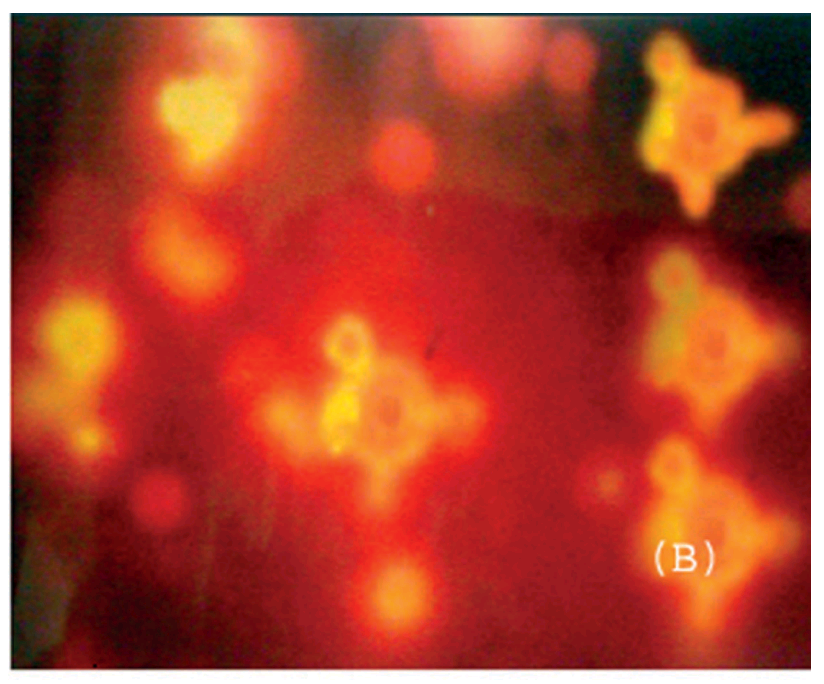
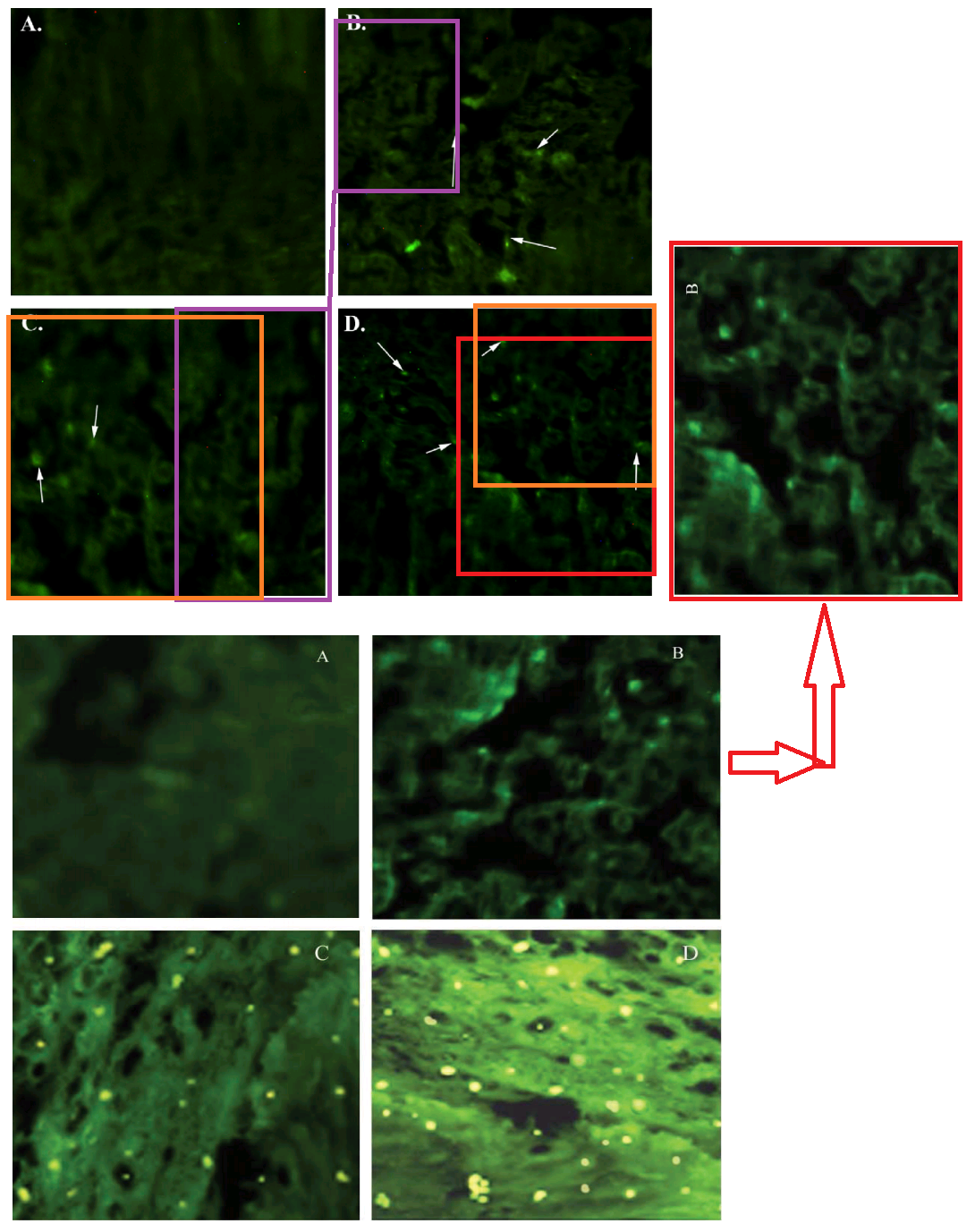
Fluorescent tissue samples is becoming another recurring theme in this post. Below, Figs 4 from Jain, Agrawal & Agrawal (2011) [A1], and 3B from Jain & Jain (2013b) [A5]. Both are a “fluorescence photomicrograph of rhodamine-123 labeled nanolipobeads formulation”, designed to cure stomach ulcers by eliminating H. Pylori. The 2013 version was photoshopped with the triplicated nanolipobeads at the right either to defuse accusations of self-plagiarism, or because the authors really like the design aesthetic of 1970s Krautrock album art.
It is almost time to move on. To include everything covered in the relevant PubPeer threads would be exhaustive even by my standards, so out of great-souled mettā I have organised the haul from trawling those threads into a spreadsheet for interested readers to consult. The spreadsheet (like the Sources section at the bottom of this post) is divided for convenience into three sections, but boundaries are permeable: A.K. Jain was still affiliated to Dr H.S. Gour University for the first five of the 13 papers grouped together under his name, not yet having assumed leadership of the ADINA institute, and G.P. Agrawal (Jain’s colleague or mentor, I assume) was co-author on those five.
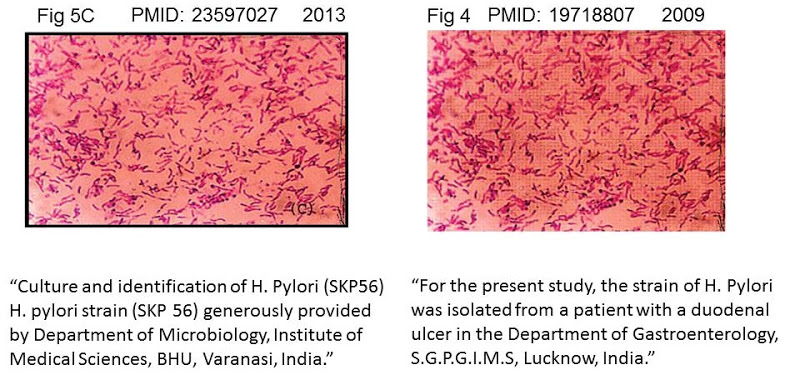
The two institutions enjoy a close, image-sharing association. There is no better way to illustrate this collegiality than Fig 5C of [A5], which had earlier appeared as Fig 4 of Jain et al (2009a) [C7] – filed below in the “Suresh P. Vyas” section.
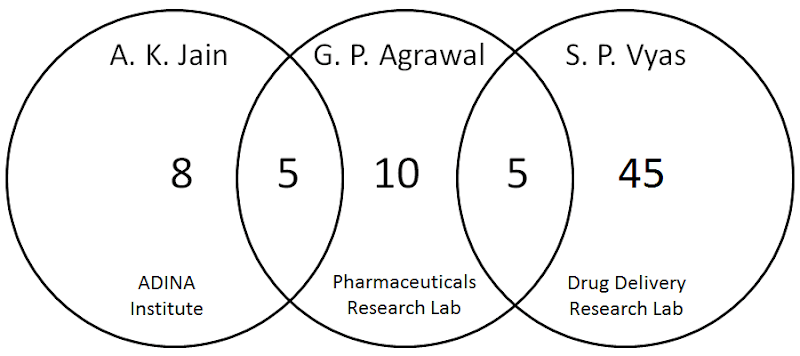
The pedants and blackboard monitors and incorrigible scolds who hang out at PubPeer have devoted 15 threads to Dr Agrawal’s papers, excluding the five co-authored with Jain, but including five co-authored with S. P. Vyas. My generosity is boundless and I created a diagram to help keep track of the co-authorship situation.
We have already seen one repurposed / modified image, the “Bubbles Congeries” shared between [B11] and [C17]. To continue in this vein, here’s a SDS-PAGE blot (a form of protein-separating electrophoresis) showing how a nasal vaccine against Hep-B works by wrapping up HBsAg surface antigens within liposomes. More specifically, within “Immunoglobulin immobilized liposomal constructs” in one incarnation (Fig 4 from B. Tiwari et al 2011 [B7]) and within “Viral protein complexed liposomes” in the other (Fig 3 from S. Tiwari et al 2011 [B8]).

These fluorescing cell clusters demonstrate how HNGC1 tumor-cell clusters absorb Methotrexate better (or perhaps doxorubicin) when the drug is concealed inside Cationized Albumin Conjugated Solid Lipid Nanoparticles (or perhaps in Cationic ligand appended nanoconstructs), thus curing brain cancer. In Fig 13 of Agrawal et al (2011a) [B4], the cells are multiplied by zooming in on them at three different sizes.
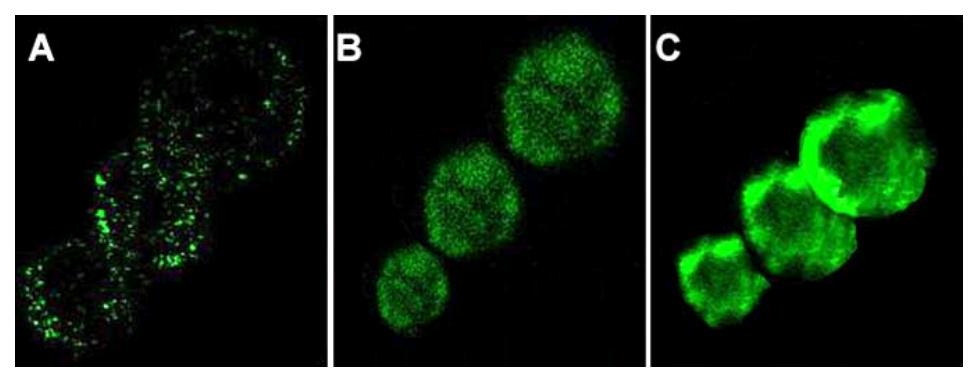
In Fig 11 of Agrawal et al (2011b) [B3], the same cells pair up with 180°-flipped copies of themselves.
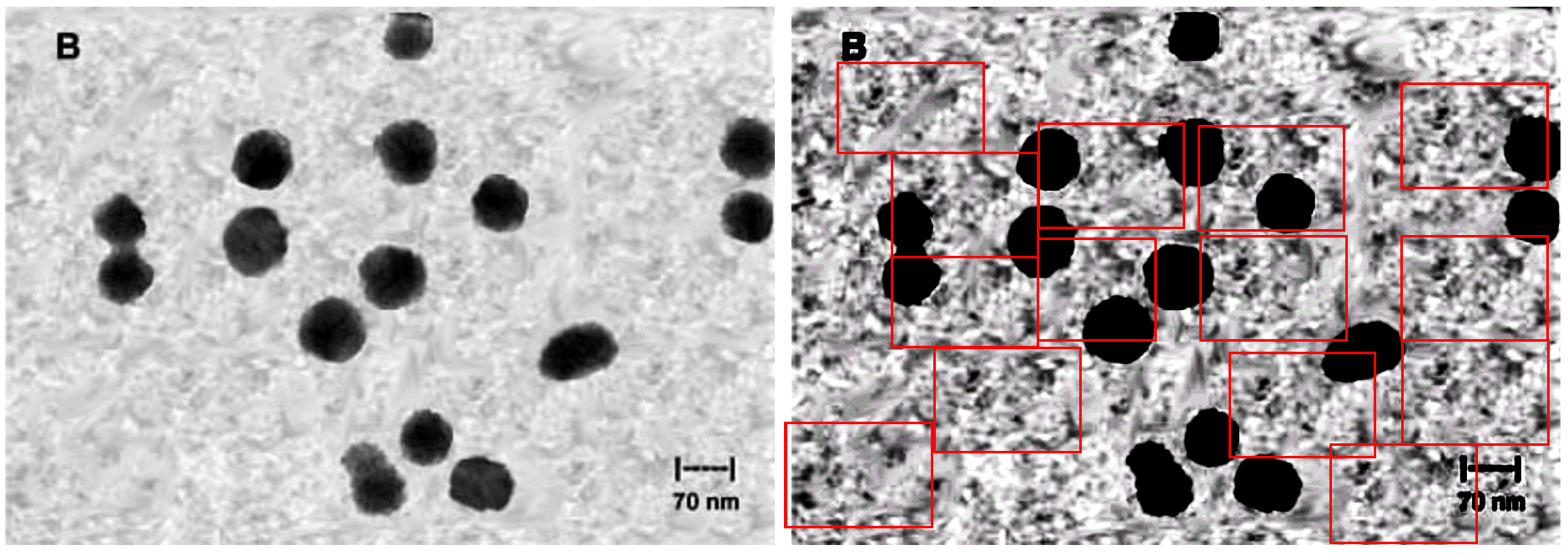
In general the Pharmaceuticals Research Laboratory staff are profligate with digital resources and their photoshops are not recycled so thoroughly. The next examples are one-offs. Boxes mark the cloned sections in Fig 7(a) of [B3], a TEM photomicrograph of SLN-DOX.

Much could be said about the thumb-print Solid-Lipid Nanoparticles (plain and CBSA conjugated) in Fig 5 of [B4]. It is easier, though, to bring up the wallpapered backgrounds with contrast enhancement:

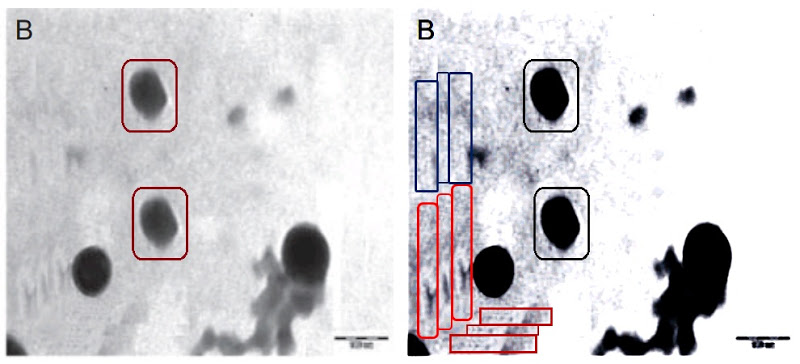
In Fig 1(b) of [B7], the cluster at lower right reminds me of a styled figure fleeing the scene after duplicating the liposomal constructs at centre. Enhanced contrast calls attention to glitched repeating margins around the lower-left corner.
Figs 2 of Yadav et al (2010) [B2] and 2 of Jain et al (2014) [B10] fell victim to extreme image compression at some stage, turning them into seas of JPG artefacts. The lack of resolution makes it difficult to comment on any similarities among the nanoparticles (“hyaluronic acid decorated PLGA nanoparticles for delivery of 5-fluorouracil” and “Adapalene loaded solid lipid nanoparticles gel” for acne treatment respectively), but areas of background have been copy-pasted.
Please be patient for one more example. In Jain et al (2010) [B1] the Solid Lipid Nanoparticles were mannosylated for better delivery of doxorubicin to brain tumours. The backgrounds of Figs 1(a) and (b) are surprisingly similar, while a ‘T’ motif — a kind of watermark, perhaps — repeats across 1(b).

The outlines of the NPs themselves only repeat in a few pairs and triplets. Still, it is clear from similarities among their internal structures that they were cut out from a handful of templates, as exercises in decoupage worthy of Matisse.

The PubPeer thread for [B1] came to the attention of the authors, with the lead / corresponding author replying first, before responsibility shifted to a minor co-author.
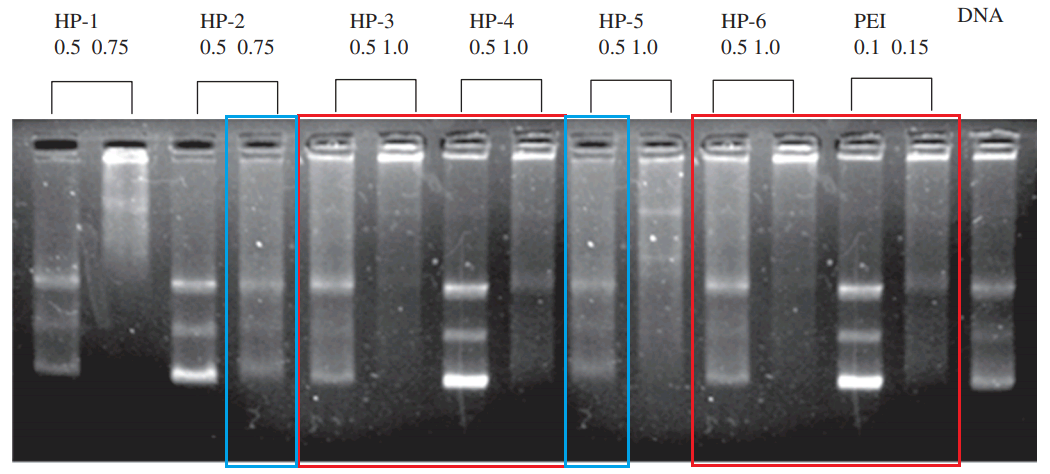
We are now sufficiently acclimatised, I think, for the final ascent from the “Agrawal Plateau” to “Peak Vyas”. A ladder will help, by which I mean a molecular ladder bridging the gap between Mishra et al (2011) [B5] and Mishra et al (2014) [C23]. Both papers addressed the need for an mucosa-absorbed oral vaccine against Hep.B, but the trojan-horse nanoparticles concealing HBsAg proteins were constructed from Lectin-anchored PGLA in [B5] and from LTA-appended Chitosan in [C23]. Anyway, the two papers’ SDS-PAGE blots have much in common – not just the molecular-weight lane.

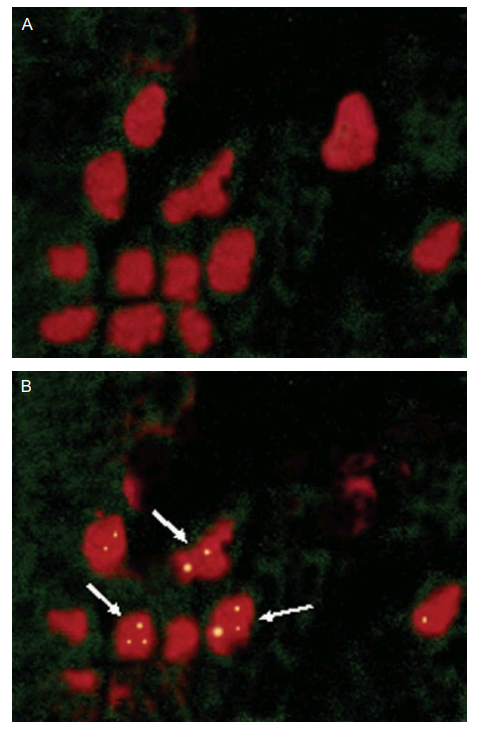
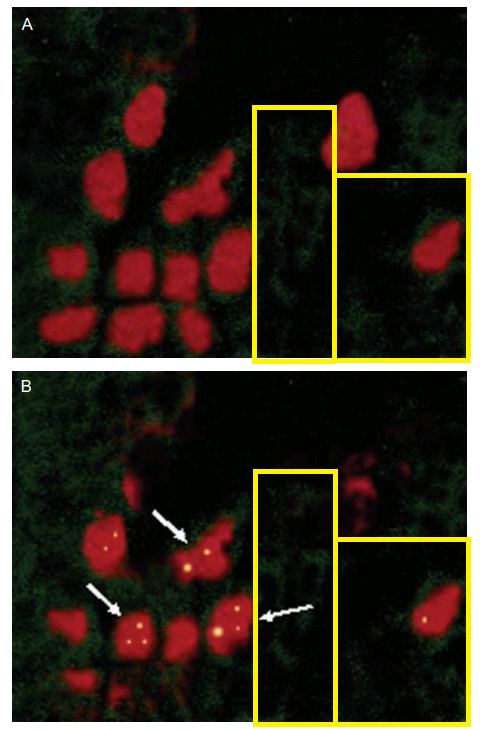
Fig 5 of [B5] featured these views of the NPs adhering to Peyer’s Patches in mouse intestine, though a comparison of the green nebulae of fluorescing NPs reveals A and B to be tailored versions of a single original. They are not false-color x-rays of ichthyosaur flipper bones.
Thus the reappearance of these frames in Fig 7 of [C23], now as chitosan NPs, are four uses of a single image. The Indian Council of Medical Research must be pleased by such thrift in the use of their research grants.
… Fig 1A of Tiwari & Vyas (2011) [C18] leaves us no wiser as to the nature of the NPs, which is not specified, though it does count as a fifth use (flipped through 180°).
At this point the comparison between Figures 1 of Paliwal et al (2011) [B6] and 1D of Paliwal et al (2013) [C8] is invidious, but I cannot resist the temptation to flog a dead horse with another dead horse. In [B6] the Solid-Lipid NPs are biomimetic and laden with a payload of lipid-conjugate low-molecular-weight heparin; in [C8] they are glyceryl behenate-cored and laden with methotrexate; perhaps that is why there are fewer of them.

Anyway, on to Dr Sureth P. Vyas. As one mark of his productivity, his H-Index is 64, and those 50 papers addressed in PubPeer threads span only some 10 years of his career.
Three of those papers were from a veritable scientific supergroup: the coauthors included Kailish C. Gupta, erstwhile Director of the CSIR-Indian Institute of Toxicology Research and no stranger to PubPeer in his own right. The last I checked, an inquiry into the research methods of Dr Gupta and others at CSIR-IITR had finished and a report was awaiting action… that was in January but people can be forgiven if events have distracted them. My point is that we have reached the highest levels in the Indian research establishment.
Pathak et al (2007) [C4]:
Now [C10] and Pathak et al (2009b) [C9] addressed, respectively, the tumor targeting of “polysaccharide decked polyethylenimine based nanocomposites” and tumor gene therapy mediated by “Chondroitin Sulfate−PEI Nanoconstructs“, with surprisingly similar results.
Anyway, Dr Vyas has progressed to the career stage where his energies and attention are divided among the usual senior academic roes of journal editing, and preparing Review Papers and Editorials and book chapters… not to forget his religious duties, shared with the ADINA Institute (as part of the collegial relationship foreshadowed above). This would leave little time for monitoring and mentoring, and it could easily be that the systematic image fabrication collected in the third section of the spreadsheet is all the work of the colleagues and students and outside contractors who contributed to the papers that Dr Vyas signed. This is consistent with the variations in the quality of forgery. It is also possible that the papers are sound other than their digitally constructed illustrations, and that the parade of cutting-edge game-changing vaccines, antivirals and cancer treatments promised therein are about to cut the game and change the edge any time now.
https://www.dovepress.com/journal-editor-integrated-pharmacy-research-and-practice-eic135
https://www.journals.elsevier.com/international-journal-of-pharmaceutics/editorial-board
https://www.future-science.com/journals/tde/editors
https://scienceforecastoa.com/Journals/Pages/JournalEditorialBoard.aspx/SJNN
I’ll pick a handful of examples at random. These look like more microballoons but according to Fig 15(b) from Asthana et al (2014) [C22], they are “Mannosylated chitosan nanoparticles for delivery of antisense oligonucleotides for macrophage targeting”.

Mangal et al (2011) [C16] and (2014) [C21] were yet another breakthrough in vaccine development, this time constructing mucoadhesive nanoparticles from (respectively) tri-methylated chitosan OR alginate-coated chitosan to conceal the proteins of Hep-B OR anthrax for administration by nose OR mouth. But like the scenery in a cheap video game, if you scan across the SEM images of Fig 1 from [C16], you find yourself back where you started. Much like reading this post. Moreover, the scenery from 1B also appears – with a longer cycle – as 1A in [C21].
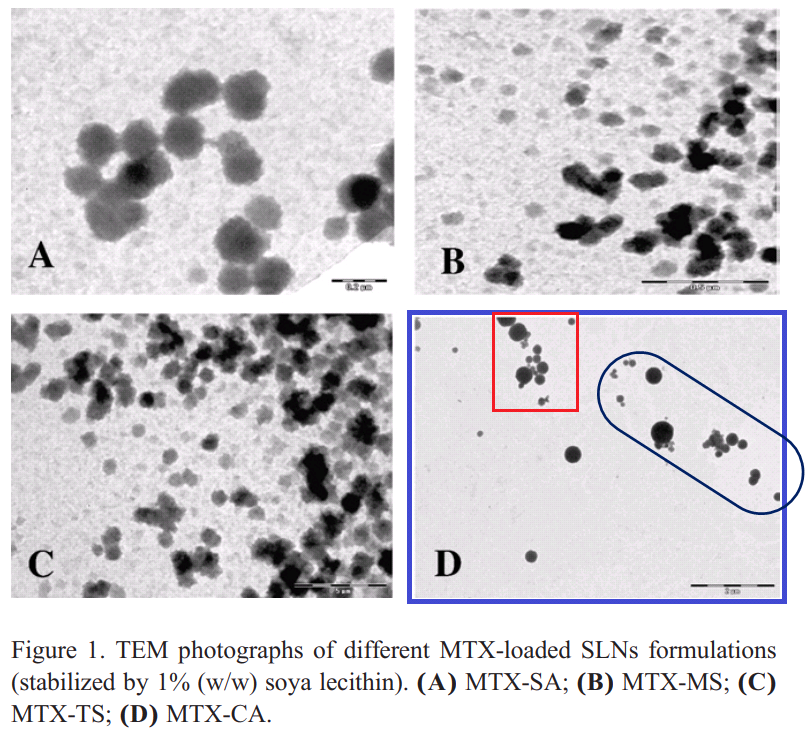
In Fig 4 of Gupta et al (2012) [C20], “tristearin-based solid lipid nanoparticles stabilized by soya phosphatidylcholine” were coated with O-palmitoyl mannan, so they can arm macrophages with their payload of Amphotericin B and help them defeat leishmaniasis. The five examples of OPM-SLNs are identical, though rotated and resized.


After all this you may be wondering what Niosomes look like. Fig 2B of Jain et al (2006) [C1] contains a few, encapsulating chitosan nanoparticles, and repeating themselves. Disappointingly, they are hard to distinguish from the bog-standard liposomes in 2A.
A general suspicion emerges from all this that when anyone in the Drug Delivery Laboratory needs a Western Blot or a microphotograph to illustrate some narrative, the standard practice is to fake it in Photoshop rather than conduct an experiment. Or you remix an existing image (real or forged) and relabel it… rotating it through 180° if you’re dedicated to research integrity.
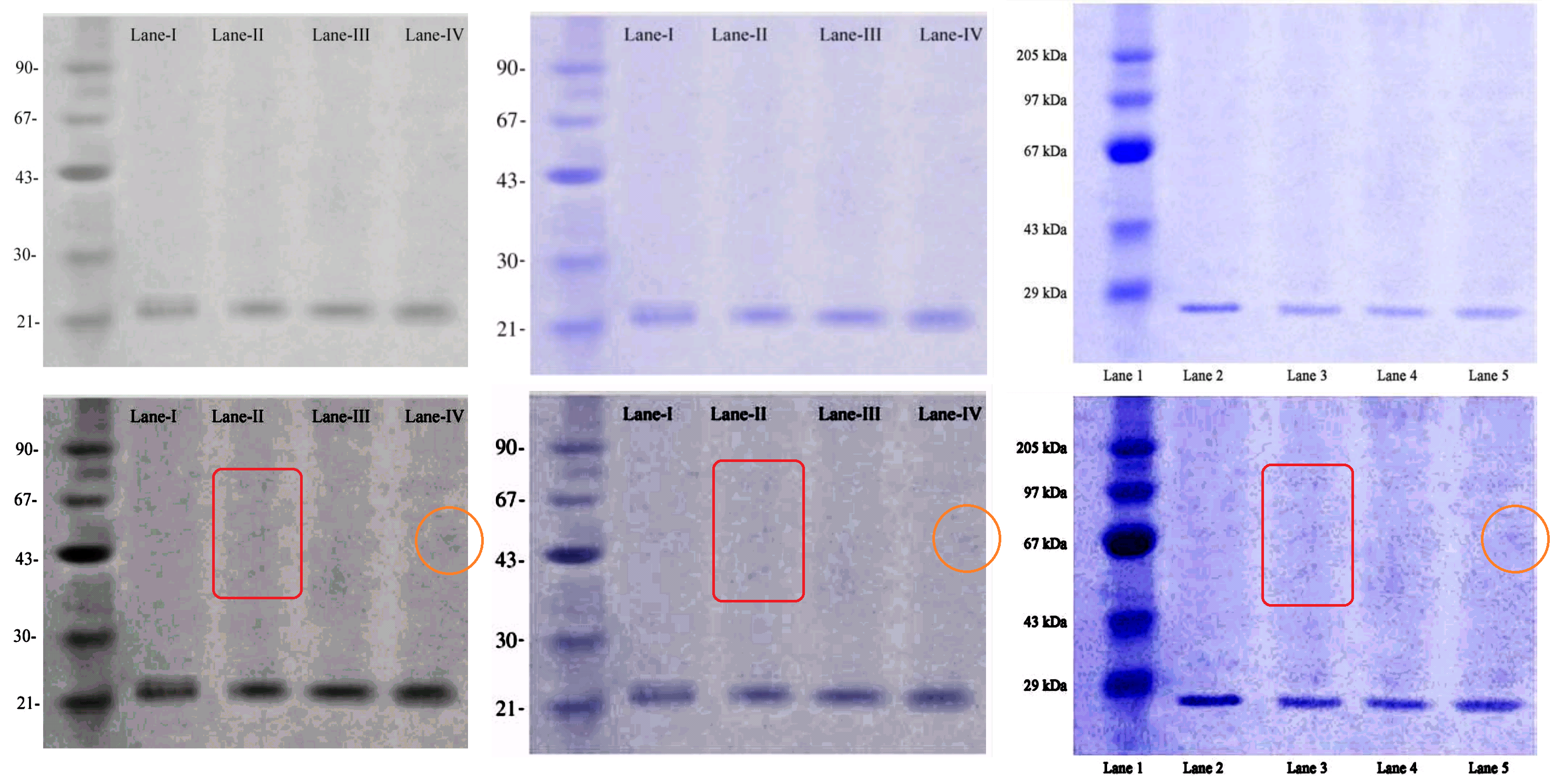
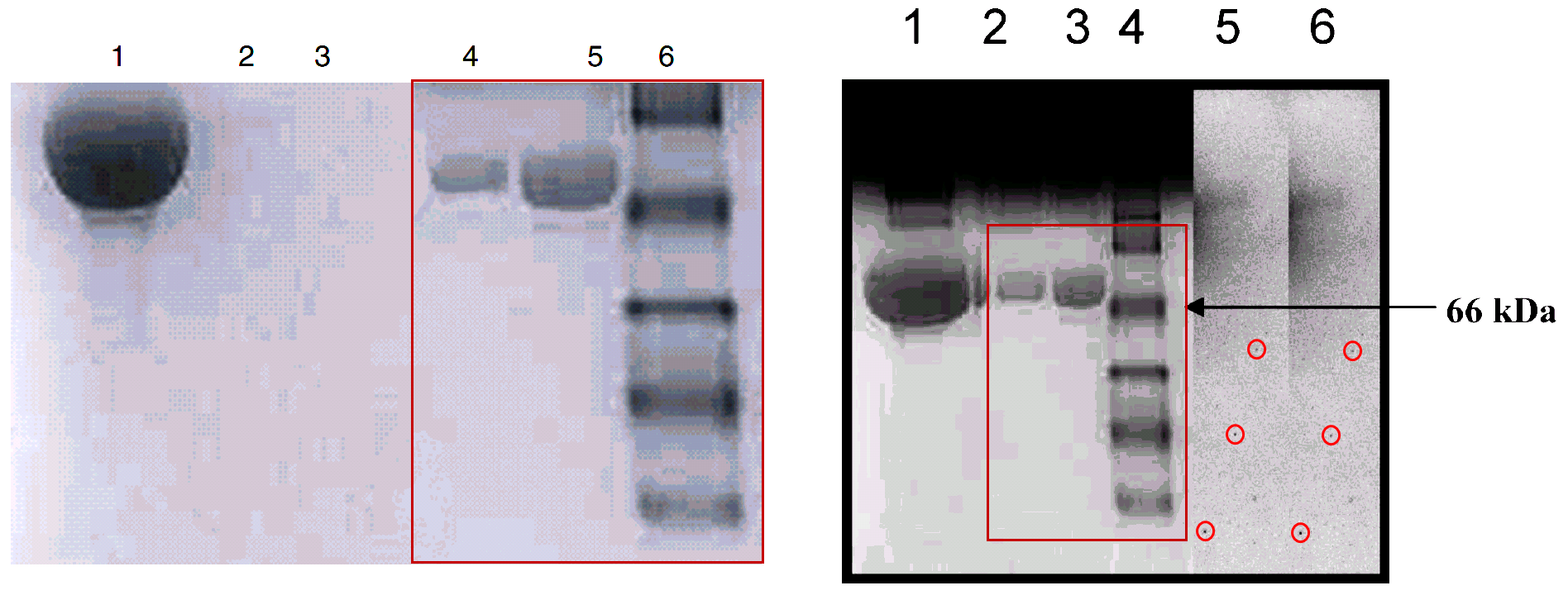
This post is already too long but I want to finish on a high-note, having saved the best until last. A thread from [C12] leads to this Identity Line-up of SDS-PAGE blots, with enhanced-contrast versions below:
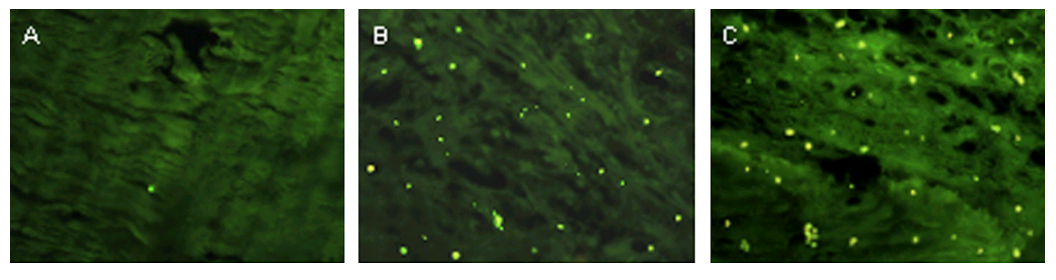
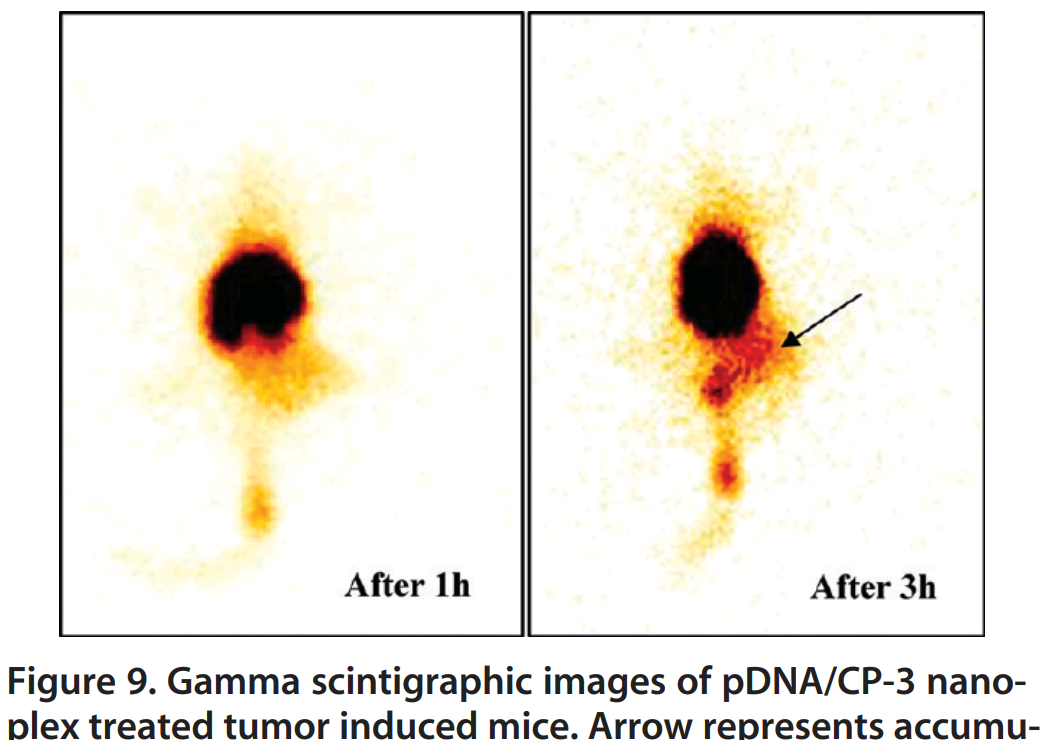
Hold that in your mind while we examine these three enlargements of rat nasal mucosa samples, fluorescing because the rats were snorting FITC-labeled “glycol chitosan coated liposomes” for a DNA vaccine, which the cells absorbed. Fig 4C, Khatri et al (2008) [C5].
The two strands quickly meet in [C11], where Panel (C) reappeared twice in Fig 5(C/D) — with one version rotated through 90 — to illustrate the absorption of Gel-Core and LIGS (liposomes in situ gelling system).
From [C11], things get complicated… Fig 5(B) is variously rotated, chopped up and rearranged to create three of the four panels in Fig 5 of Jain et al (2010) [C14]. The team had moved on by then and were promising an oral vaccine so the tissues have become rat gastric mucosa, while the NPs are now composed of “PEG–PLA–PEG block copolymers”.
Pressure of space means that one diagram must suffice to show some further metamorphoses of Fig 5, but the papers themselves are old friends by now. At left, a 2011 appearance in Fig 5 of [C16], home of that “cyclic scenery” SEM. Middle, a 2009 appearance in Fig 4 of [C6]. At right, Fig 3 from Pawar et al (2010) [C15], where poly(lactic-co-glycolic acid) microparticles have been coated in chitosan for extra mucoadhesion and vaccinivity.
 And as coda: don’t forget [C21], the 2014 oral anthrax vaccine with the other cyclic-scenery images! By then the fluorescing tissues had morphed into Peyer’s Patches from murine intestine, tagged with alginate-coated chitosan microparticles.
And as coda: don’t forget [C21], the 2014 oral anthrax vaccine with the other cyclic-scenery images! By then the fluorescing tissues had morphed into Peyer’s Patches from murine intestine, tagged with alginate-coated chitosan microparticles.
I have no idea how much funding has poured into ADINA and the Drug Delivery Lab to sustain their digital activities but the funders shouldn’t hold their breaths waiting for cancer treatments or oral vaccines in return. Nor would I advise all those people who read and cite Jain’s, Agrawal’s and Vyas’ papers to try replicating or extending their results.
Sources
A:
- “NANOLIPOBEADS BASED DRUG DELIVERY SYSTEM FOR EFFECTIVE MANAGEMENT OF PEPTIC ULCER”, ASHISH K.JAIN*, ABHINAV AGRAWAL AND GOVIND P. AGRAWAL. International Journal of Current Pharmaceutical Research (2011)
- “Double-Liposome – Based Dual-Drug Delivery System as Vectors for Effective Management of Peptic Ulcer“, Ashish K. Jain, Abhinav Agarwal, Himanshu Agrawal, Govind P. Agrawal. Journal of Liposome Research (2012) doi: 10.3109/08982104.2012.655284
- “Design of Buoyant Famotidine Loaded Microballoons Directed for Upper Small Intestinal Absorption Window”, Mahendra Chouhan, GP Agrawal, Ashish Jain , Sandeep Patidar. International Journal of Research in Pharmacy and Science (2013)
- “DEVELOPMENT AND CHARACTERIZATION OF MICROBALLONS BASED DRUG DELIVERY SYSTEM OF MESALAMINE HYDROCHLORIDE“, ASHISH KUMAR JAIN, Sunil Kumar Jain. Journal of Drug Delivery and Therapeutics (2013) doi: 10.22270/jddt.v3i3.503
- “Development and characterization of nanolipobeads-based dual drug delivery system for H. pylori targeting“, Ashish Kumar Jain, Sunil Kumar Jain. Journal of Drug Targeting (2013) doi: 10.3109/1061186x.2013.784978
- “Lactosaminated-N-succinyl chitosan nanoparticles for hepatocyte-targeted delivery of acyclovir“, Nivrati Jain, Vaibhav Rajoriya, Prateek Kumar Jain, Ashish Kumar Jain. Journal of Nanoparticle Research (2014) doi:10.1007/s11051-013-2136-x
- “Glycyrrhizin conjugated chitosan nanoparticles for hepatocyte-targeted delivery of lamivudine“, Deepak Mishra, Nivrati Jain, Vaibhav Rajoriya, Ashish K. Jain. The Journal of pharmacy and pharmacology (2014) doi: 10.1111/jphp.12235
- “Folate-conjugated albumin nanoparticles for rheumatoid arthritis-targeted delivery of etoricoxib“, Upasana Bilthariya, Nivrati Jain, Vaibhav Rajoriya, Ashish Kumar Jain. Drug Development and Industrial Pharmacy (2015) doi: 10.3109/03639045.2013.850705
- “Targeting of AIDS related encephalopathy using phenylalanine anchored lipidic nanocarrier“, Anil Vyas, Ankit Jain, Pooja Hurkat, Ashish Jain, Sanjay K. Jain. Colloids and Surfaces B Biointerfaces (2015) doi: 10.1016/j.colsurfb.2015.04.049
- “Hyaluronic acid conjugated multi-walled carbon nanotubes for colon cancer targeting“, Shiv Kumar Prajapati, Ankit Jain, Chirag Shrivastava, Ashish Kumar Jain. International Journal of BiologicalMacromolecules (2019) doi: 10.1016/j.ijbiomac.2018.11.116
B
- “Mannosylated solid lipid nanoparticles as vectors for site-specific delivery of an anti-cancer drug“, Ashay Jain, Abhinav Agarwal, Saikat Majumder, Narendra Lariya, Anil Khaya, Himanshu Agrawal, Subrata Majumdar, Govind P. Agrawal. Journal of Controlled Release (2010) doi: 10.1016/j.jconrel.2010.09.003
- “Development and characterization of hyaluronic acid decorated PLGA nanoparticles for delivery of 5-fluorouracil“, Awesh K. Yadav, Abhinav Agarwal, Gopal Rai, Pradeep Mishra, Sanyog Jain, Anil K. Mishra, Himanshu Agrawal, Govind P. Agrawal. Drug Delivery (2010) doi: 10.3109/10717544.2010.500635
- “Cationic ligand appended nanoconstructs: a prospective strategy for brain targeting“, Abhinav Agarwal, Himanshu Agrawal, Shailja Tiwari, Sanyog Jain, Govind P. Agrawal. International Journal of Pharmaceutics (2011) doi: 10.1016/j.ijpharm.2011.09.039
- “Cationized Albumin Conjugated Solid Lipid Nanoparticles as Vectors for Brain Delivery of an Anti-Cancer Drug“, Abhinav Agarwal, Saikat Majumder, Himanshu Agrawal, Subrata Majumdar, Govind P. Agrawal. Current Nanoscience (2011) doi: 10.2174/157341311794480291
- “Lectin anchored PLGA nanoparticles for oral mucosal immunization against hepatitis B“, Neeraj Mishra, Shailja Tiwari, Bhuvaneshwar Vaidya, Govind P. Agrawal, Suresh P. Vyas. Journal of Drug Targeting (2011) doi: 10.3109/10611861003733946
- “Biomimetic solid lipid nanoparticles for oral bioavailability enhancement of low molecular weight heparin and its lipid conjugates: in vitro and in vivo evaluation“, Rishi Paliwal, Shivani R. Paliwal, Govind P. Agrawal, Suresh P. Vyas. Molecular Pharmaceutics (2011) doi: 10.1021/mp200109m
- “Immunoglobulin immobilized liposomal constructs for transmucosal vaccination through nasal route“, Brajesh Tiwari, Abhinav Agarwal, Anil K. Kharya, Narendra Lariya, Gauravkant Saraogi, Himanshu Agrawal, Govind P. Agrawal. Journal of Liposome Research (2011) doi: 10.3109/08982104.2010.498003
- “Viral protein complexed liposomes for intranasal delivery of hepatitis B surface antigen“, Shailja Tiwari, Sunil Kumar Verma, Govind P. Agrawal, Suresh P. Vyas. International Journal of Pharmaceutics (2011) doi: 10.1016/j.ijpharm.2011.04.029
- “Dual targeted polymeric nanoparticles based on tumor endothelium and tumor cells for enhanced antitumor drug delivery“, Madhu Gupta, Gousia Chashoo, Parduman Raj Sharma, Ajit Kumar Saxena, Prem Narayan Gupta, Govind Prasad Agrawal, Suresh Prasad Vyas. Molecular Pharmaceutics (2014) doi: 10.1021/mp400404p
- “Adapalene loaded solid lipid nanoparticles gel: an effective approach for acne treatment“, Amit K. Jain, Ashay Jain, Neeraj K. Garg, Abhinav Agarwal, Atul Jain, Som Akshay Jain, Rajeev K. Tyagi, Rakesh K. Jain, Himanshu Agrawal, Govind P. Agrawal. Colloids and Surfaces B Biointerfaces (2014) doi: 10.1016/j.colsurfb.2014.05.041
- “Eudragit-coated dextran microspheres of 5-fluorouracil for site-specific delivery to colon“, Gopal Rai, Awesh K. Yadav, Narendra K. Jain, Govind P. Agrawal. Drug Delivery (2016) doi: 10.3109/10717544.2014.913733
- “Stomachspecific polymeric low density microballoons as a vector for extended delivery of rabeprazole and amoxicillin for treatment of peptic ulcer“, Sandeep Choudhary, Ashay Jain, Mohd Cairul Iqbal Mohd Amin, Vijay Mishra, Govind P. Agrawal, Prashant Kesharwani. Colloids and Surfaces B Biointerfaces (2016) doi: 10.1016/j.colsurfb.2016.01.048
C
- “Chitosan nanoparticles encapsulated vesicular systems for oral immunization: preparation, in-vitro and in-vivo characterization“, Sanyog Jain, Rakesh Kumar Sharma, S. P. Vyas. The Journal of pharmacy and pharmacology (2006) doi: 10.1211/jpp.58.3.0003
- “Targeted brain delivery of AZT via transferrin anchored pegylated albumin nanoparticles“, Vivek Mishra, Sunil Mahor, Amit Rawat, Prem N. Gupta, Praveen Dubey, Kapil Khatri, Suresh P. Vyas. Journal of Drug Targeting (2006) doi: 10.1080/10611860600612953
- “Cationic transfersomes based topical genetic vaccine against hepatitis B“, Sunil MAHOR, Amit RAWAT, Praveen K DUBEY, Prem N GUPTA, Kapil KHATRI, Amit K GOYAL, S P VYAS. International Journal of Pharmaceutics (2007) doi: 10.1016/j.ijpharm.2007.03.006
- “Engineered polyallylamine nanoparticles for efficient in vitro transfection“, Atul Pathak, Anita Aggarwal, Raj K. Kurupati, Soma Patnaik, Archana Swami, Yogendra Singh, Pradeep Kumar, Suresh P. Vyas, Kailash C. Gupta. Pharmaceutical Research (2007) doi: 10.1007/s11095-007-9259-7
- “Surface modified liposomes for nasal delivery of DNA vaccine“, Kapil Khatri, Amit K. Goyal, Prem N. Gupta, Neeraj Mishra, Abhinav Mehta, Suresh P. Vyas. Vaccine (2008) doi: 10.1016/j.vaccine.2008.02.058
- “Synthesis, characterization and evaluation of novel triblock copolymer based nanoparticles for vaccine delivery against hepatitis B“, Arvind K. Jain, Amit K. Goyal, Prem N. Gupta, Kapil Khatri, Neeraj Mishra, Abhinav Mehta, Sharad Mangal, Suresh P. Vyas. Journal of Controlled Release (2009) doi: 10.1016/j.jconrel.2009.02.010
- “Polyelectrolyte coated multilayered liposomes (nanocapsules for the treatment of Helicobacter pylori infection“, Parul Jain, Sanyog Jain, K. N. Prasad, S. K. Jain, Suresh P. Vyas. Molecular Pharmaceutics (2009) doi: 10.1021/mp8002539
- “Effect of lipid core material on characteristics of solid lipid nanoparticles designed for oral lymphatic delivery“, Rishi Paliwal, Shivani Rai, Bhuvaneshwar Vaidya, Kapil Khatri, Amit K. Goyal, Neeraj Mishra, Abhinav Mehta, Suresh P. Vyas. Nanomedicine Nanotechnology Biology and Medicine (2009) doi: 10.1016/j.nano.2008.08.003
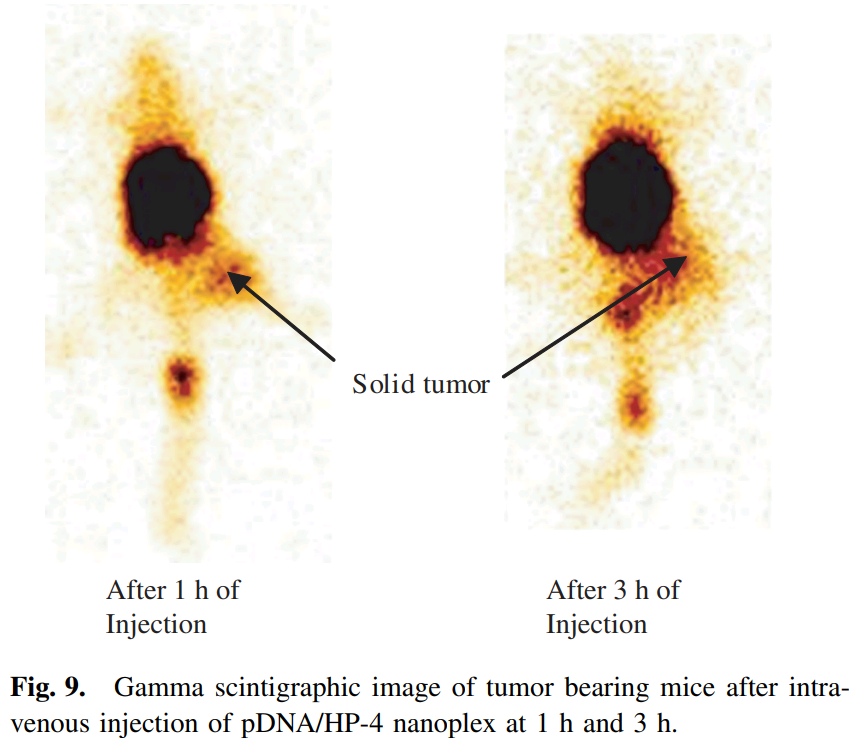
- “Gene Expression, Biodistribution, and Pharmacoscintigraphic Evaluation of Chondroitin Sulfate−PEI Nanoconstructs Mediated Tumor Gene Therapy“, Atul Pathak, Pradeep Kumar, Krishna Chuttani, Sanyog Jain, Anil K. Mishra, Suresh P. Vyas, Kailash C. Gupta. ACS Nano (2009) doi: 10.1021/nn900044f
- “Efficient tumor targeting by polysaccharide decked polyethylenimine based nanocomposites“, Atul Pathak, Archana Swami, Soma Patnaik, Sanyog Jain, Krishna Chuttani, Anil K. Mishra, Suresh P. Vyas, Pradeep Kumar, Kailash C. Gupta. Journal of Biomedical Nanotechnology (2009) doi: 10.1166/jbn.2009.1031
- “Liposome in situ gelling system: Novel carrier based vaccine adjuvant for intranasal delivery of recombinant protein vaccine“, Shailja Tiwari, Amit K. Goyal, Neeraj Mishra, Bhuvaneshwar Vaidya, Abhinav Mehta, Devyani Dube, Suresh P. Vyas. Procedia in Vaccinology (2009) doi: 10.1016/j.provac.2009.07.027
- “Development and characterization of novel carrier gel core liposomes based transmission blocking malaria vaccine“, Shailja Tiwari, Amit K. Goyal, Neeraj Mishra, Kapil Khatri, Bhuvaneshwar Vaidya, Abhinav Mehta, Yimin Wu, Suresh P. Vyas. Journal of Controlled Release (2009) doi: 10.1016/j.jconrel.2009.08.004
- “Polymeric nanospheres modified with YIGSR peptide for tumor targeting“, Praveen K. Dubey, Deepak Singodia, S. P. Vyas. Drug Delivery (2010) doi: 10.3109/10717544.2010.490249
- “PEG–PLA–PEG block copolymeric nanoparticles for oral immunization against hepatitis B“, Arvind K. Jain, Amit K. Goyal, Neeraj Mishra, Bhuvaneshwar Vaidya, Sharad Mangal, Suresh P. Vyas. International Journal of Pharmaceutics (2010) doi: 10.1016/j.ijpharm.2009.12.013
- “Evaluation of mucoadhesive PLGA microparticles for nasal immunization“, Dilip Pawar, Amit K. Goyal, Sharad Mangal, Neeraj Mishra, Bhuvaneshwar Vaidya, Shailja Tiwari, Arvind K. Jain, Suresh P. Vyas. The AAPS Journal (2010) doi: 10.1208/s12248-009-9169-1
- “Pharmaceutical and immunological evaluation of mucoadhesive nanoparticles based delivery system(s) administered intranasally“, Sharad Mangal, Dilip Pawar, Neeraj K. Garg, Arvind K. Jain, S.P. Vyas, D.S.V. Raman Rao, K.S. Jaganathan. Vaccine (2011) doi: 10.1016/j.vaccine.2011.04.112
- “Nanoparticle-based immunopotentiation via tetanus toxoid-loaded gelatin and aminated gelatin nanoparticles“, M. S. Sudheesh, S. P. Vyas, D. V. Kohli. Drug Delivery (2011) doi: 10.3109/10717544.2010.549525
- “Novel approaches to oral immunization for hepatitis B“, Shailja Tiwari, Suresh P. Vyas. Current Infectious Disease Reports (2011) doi: 10.1007/s11908-010-0152-x
- “Development, characterization and in vivo assessment of effective lipidic nanoparticles for dermal delivery of fluconazole against cutaneous candidiasis“, Madhu Gupta, Suresh P. Vyas. Chemistry and Physics of Lipids (2012) doi: 10.1016/j.chemphyslip.2012.01.006
- “Development and Characterization of Amphotericin B Loaded Solid Lipid Nanoparticles Against Experimental Visceral Leishmaniasis“, Swati Gupta, Anuradha Dube, Suresh P. Vyas. Pharmaceutical Nanotechnology (2012) doi: 10.2174/2211738511301010054
- “Evaluation of mucoadhesive carrier adjuvant: toward an oral anthrax vaccine“, Sharad Mangal, Dilip Pawar, Udita Agrawal, Arvind K. Jain, Suresh P. Vyas. Artificial Cells Nanomedicine and Biotechnology (2014) doi: 10.3109/21691401.2013.769447
- “Mannosylated Chitosan Nanoparticles for Delivery of Antisense Oligonucleotides for Macrophage Targeting“, Gyati Shilakari Asthana, Abhay Asthana, Dharm Veer Kohli, Suresh Prasad Vyas. BioMed Research International (2014) doi: 10.1155/2014/526391
- “Development and characterization of LTA-appended chitosan nanoparticles for mucosal immunization against hepatitis B“, Neeraj Mishra, Kapil Khatri, Madhu Gupta, Suresh P. Vyas. Artificial Cells Nanomedicine and Biotechnology (2014) doi: 10.3109/21691401.2013.809726

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00
















Smut: Although your repeated misuse of horse carcasses is disturbing, your exposé on these Photoshop artists is, once again, masterful.
LikeLiked by 1 person
Professor Vyas is probably not causing much damage to the scientific literature, now that he has risen to the pinnacle of the research establishment (getting there through a well-funded career of delivering nothing but made-up results that will waste the time of anyone who tries to replicate them) and is mainly publishing Review Papers. I am more worried about the activities of the students he mentored and taught his methods.
LikeLiked by 1 person
Here is a handy tutorial for his students, and it’s free:
LikeLike
The case for fraud made in this post is strong. These papers and the so-called ‘scientists’ who published them deserve ridicule, perhaps even criminal charges. They are indeed crooks. However, painting with a broad brush using casual, unreflective terms such as “privileged Brahmin university crooks” detracts from the message. Some thoughts:
(a) While brahmins in India are indeed privileged, and even if all authors on all publications are brahmins, no connection has been established here between the authors’ ‘Brahminism’ or their ‘privilege’ and the fraud.
(b) Assuming all coauthors bear responsibility for a publication, it is not clear that every author is brahmin. Moreover, relatively speaking, there are privileged castes other than the brahmins. Although it remains a work in progress, India has affirmative action policies to redress the negative impact of casteism. It would be no surprise to find non-brahmins among the authors.
(c) Re the last name ‘Jain’ for example: Jainism arose in opposition to Brahminism. In general, Jains, as a sect, are opposed to the caste system and do not regard themselves as brahmins although some rituals are observed. Like most things in India, there is great variety and complexity.
I could go on. It is easy to imagine substitute terms for groups in the West, privileged or otherwise, that would raise a furore or at the very least leave a bad taste in your mouth.
This is Dr. Schneider’s blog, Smut Clyde’s guest post. You are free to make posts on your blog in your own way, any way you choose. I am merely pointing out that offhand comments like that add nothing to the matter under discussion. It is best to keep the focus on the science or, as in this egregious case, the complete lack thereof.
LikeLike
Thanks for your comment. I would however would be interested to know if being Brahmin provides a certain protection when massive fraud like this is exposed. What are your views?
LikeLike
It does – like any mutual backscratching network of privilege. However, note that such protection and advantage do not extend exclusively to Brahmins. In some states in southern India for example, university fraud and corruption – scientific or otherwise – is rampant. The networks are mostly non-brahmin, for sociopolitical/historical reasons. It would take too long to explain here. These have been reported extensively in the newspapers. The recent cases from Annamalai University reported by Elisabeth Bik and covered widely in India is a case in point – many of the powers that be are not brahmin.
LikeLike
Oops. That should have read: “… are rampant”!
LikeLike
Pingback: Bullshitter Mauro Ferrari out as ERC President – For Better Science
Pingback: Niyaz Ahmad’s technician did it – For Better Science
Pingback: How David Ojcius and Jie Yan solved Leptospirosis – For Better Science
well, Mr Smut, seems you were all wrong slandering Professor Suresh Vyas and must now apologise.

Correction 5 May 2020:
“In the original publication, we reported Figure 1 as a transmission electron microphotograph (TEM) of low molecular weight heparin (LMWH)-lipid conjugate loaded solid lipid nanoparticles (SLNs). The image was inadvertently wrongly provided. The corrected TEM image is appended below. This correction does not affect the main conclusion of the manuscript. We sincerely apologize for this unfortunate incidence and inconvenience caused to the readers due to this error.”
Figure 1. A representative TEM image of LMWH-lipid conjugate loaded SLNs.
https://pubpeer.com/publications/738C358DE744A499F67DBB3F76801F
LikeLike
This retraction is even better – for paper c22.
LikeLike
But on the other hand there was this retraction, for paper C4:
LikeLike
There was no need to target an entire caste here as “Brahmin university crooks.” It detracts from your objectivity and the focus of the article and this site- unless you can prove causation, there is no point making inflammatory claims about Brahmins being crooks. Scientific frauds as such goes undetected in India and often does not result in firings, whether or not one is Brahmin or not- such is the state of the country’s scientific temper. Detection only occurs at higher levels. Brahmins are not the only ones to blame, and various factors are at play. Moreover, correlation is not causation- as a scientific writer you should know better and be more responsible.
LikeLike
Not an expert on India, but I daresay Brahmin fraudsters get away with more than lower castes.
LikeLike
Maybe, maybe not. I am far from unbiased in my assessment of this. But it you’re not an expert, why make generalzing and potentially inflammatory comments on it? India is a powder keg. Something you’ll take as absurd, but the level of targeting Brahmins receive is crazy, and while I was skeptical of their ability to be targeted, lose jobs, and be victims of worse things as other marginalized groups are, recent reading has changed my outlook. Lower castes also have high levels of legal protections and affirmative action programs in India, something you would know if you were Indian. Also, until causation and correlation are linked, no point making inflammatory statements. There is no evidence linking Brahmins to higher levels of fraud or higher incidence of getting away with it- these are mere accusations as of now. Again, a scientist or scientific writer should know better than to go off of speculation, as indicated by statements like “I daresay.”
LikeLike
Yeah right. Under Hindu fascist Modi male Brahmin must be the most persecuted group of all. Got it. Thanks for your advice on how to do journalism.
LikeLike