France is the place where research integrity truly happens, as a circus clown show. In other countries, fraud is a dirty business dealt in the dark alleys of academia, but in France, it is the prime time entertainment, with all the elites participating, playing victim of persecution, denouncing traitors of the Republic, invoking Nazis and Vichy regime, and issuing legal threats, all on the internet and in national newspapers. Let me sum up so far:
- Olivier Voinnet, star of plant sciences, went from being a fraudulent pariah to a Dreyfus figure, a champion of research integrity who single-handedly uncovered a huge fraud conspiracy committed by his right-hand man Patrice Dunoyer, but the main research findings remain unaffected.
- Anne Peyroche, former CNRS president, was found guilty of having published research fraud and faces a sack by Commissariat à l’énergie atomique.
- Catherine Jessus, until recently chief biologist at CNRS, was defended by all the French elites of Academie de Sciences and CNRS against malicious slander committed by anonymous critics, myself and Le Monde. Turned out, exactly same kind of data manipulations can be fraudulent in Voinnet papers but perfectly scientific in papers by Jessus.
- Francis-Andre Wollmann, the dialectic investigator of both Voinnet and Jessus, Academie de Sciences member and Knight of the Honour Legion, himself coauthored papers with manipulated data.
- Guido Kroemer, another French star scientist and Photoshop artist, used to host in his Paris lab his Spanish cancer research colleague Carlos-Lopez-Otin, a fugitive persecuted by retractions, ridicule and mysterious disappearance of mice. It is not known if Carlos’ friends from Opus Dei came to visit or not, but they sure helped him return back to Oviedo in early March 2019.
- Frédérique Vidal, Minister for Research in the current Macron government, apparently interfered with the misconduct investigations in both affairs of Peyroche and Jessus. It becomes progressively clearer why Vidal tends to side with dishonest scientists: she herself published some manipulated data as university professor. And in this article, I present even more material on this topic, in two new papers authored by Vidal.
The Minister for Research Frederique Vidal is a specialist in molecular genetics of germ cell development. She received her PhD from University of Nice and was appointed assistant professor there in 1995, and full professor in 2004. In 2009, Vidal rose to the rank of Head of Department of Life Sciences, and in 2012, President of the University of Nice. Vidal remained in this position until her appointment as Minister of Higher Education, Research and Innovation in 2017. Like Wollman, she is also Knight of the Honour Legion.
I previously reported a duplicated gel in this Vidal paper:
F. Vidal, P. Lopez, L.A. Lopez-Fernandez, F. Ranc, J.C. Scimeca, F. Cuzin, M. Rassoulzadegan
Gene trap analysis of germ cell signaling to Sertoli cells: NGF-TrkA mediated induction of Fra1 and Fos by post-meiotic germ cells
Journal of Cell Science 2001 Jan;114(Pt 2):435-43.
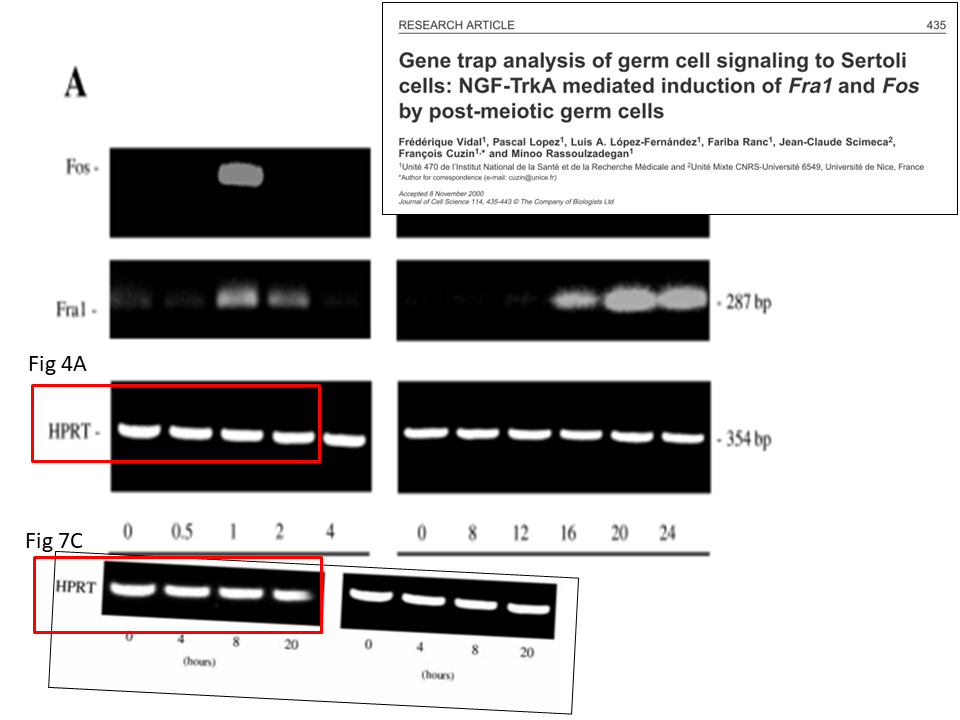
A certain expert then provided additional evidence on PubPeer, it turned out the gel featured in the same paper not twice, but trice. The left gel in Figure 7C is namely same as the right one, just as in Figure 4A, only artistically blurred and rotated:
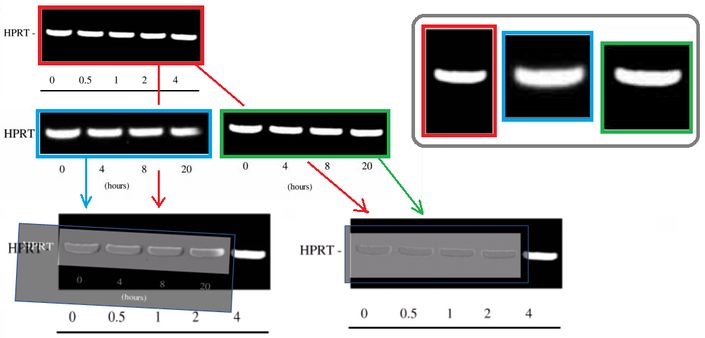
It also was pointed out that the fifth gel band in Figure 4A looks suspiciously too similar to the first band in the same gel (and in its two other incarnations). Hoya camphorifolia explained:
“the PCR results for HPRT in Fig 7C (left-hand panel) are more similar than one would expect to the first four lanes of the results in Fig 4A, slightly defocussed and rotated through 2deg. See, for instance, the enlargements of the first lane(s), at the upper right.
It is certainly true that the respective lanes (and the spacings between them) allow the two panels to overlap and cancel out, when one is black-white reversed and superimposed on the other (at lower left).
But this should not blind us to the observation that the HPRT band from Fig 4A also closely resembles the right-hand band from 7C, without blurring or rotation, as shown in the superimposition at lower right.”
The Ministry of of Higher Education, Research and Innovation reacted swiftly and mercilessly. Same month my article appeared, in October 2018, it issued this statement to the French higher education news service News Tank:
“Charges have been laid against a scientific publication published 17 years ago and co-authored by seven authors, including the current Minister of Higher Education, research and innovation. These unacceptable accusations are totally unfounded. The goal sought by their author is clear: to destabilize the minister. Faced with these accusations, the Minister is studying the possibility to institute legal proceedings.”
I sent my two-fingered salute back to the Government of France, yet nothing happened since. But now a letter arrived in my mailbox. Not an email, a real letter, in an envelope from France. The unsigned missive presented evidence of more duplicated gel bands in Vidal papers. These being:
Pascal Lopez, Frédérique Vidal, Luc Martin, Luis A. Lopez-Fernandez, Jean-François Rual, Barry S. Rosen, François Cuzin, Minoo Rassoulzadegan
Gene Control in Germinal Differentiation: Rnf6, a Transcription Regulatory Protein in the Mouse Sertoli Cell
Molecular and Cellular Biology (2002) DOI: 10.1128/MCB.22.10.3488-3496.2002
Received November 19, 2001; Returned for modification January 7, 2002; Accepted January 25, 2002; Published online May 15, 2002.
and
Pascal Lopez, Ruken Yaman, Luis A. Lopez-Fernandez, Frédérique Vidal, Daniel Puel, Philippe Clertant, François Cuzin and Minoo Rassoulzadegan
A Novel Germ Line-specific Gene of the Phosducin-like Protein (PhLP) Family
Journal of Biological Chemistry (2002) doi: 10.1074/jbc.M207434200
Received for publication, July 24, 2002, and in revised form, October 28, 2002. Published, November 6, 2002
This is the problem, in overview, the duplicated gel bands are highlighted in colour:
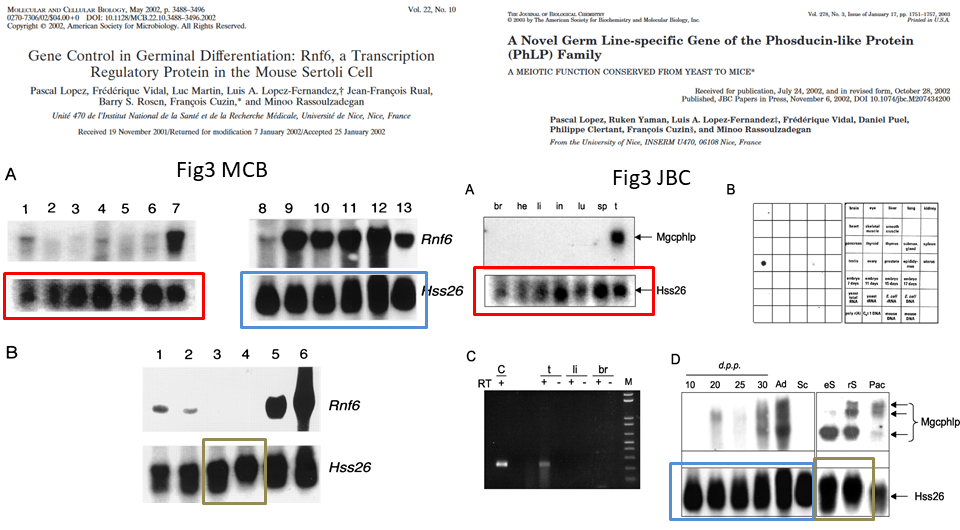
Northern Blot is the method to analyse RNA expression. RNA extracts from cells or tissues are run on a gel and transferred to a blot membrane, which is then incubated with a radioactively labelled specific probe, in this case to detect individual messenger RNAs (mRNA). The images in question show the loading control for the Hss26 mRNA, and these are obviously duplicated between the two papers. However, for the red and blue framed cases, one must say the samples are the same. The MCB paper provides this legend, and it matches the legend in the JBC paper:
“Northern blot analysis of total RNA prepared from brain (lane1), heart (lane 2), liver (lane 3), intestine (lane 4), lung (lane 5), spleen (lane 6), adult testis (lane 7), testis of a 10-day-old mouse (lane 8), testis of a 20-day-old mouse (lane 9), testis of a 25-day-old mouse (lane 10), testis of a 30-day-old mouse (lane 11), testis of a 2-month-old mouse (lane 12), and freshly isolated Sertoli cells from 3-week-old mice (lane 13)”
In both cases, the authors declare respectively: “Hybridization was performed in succession with probes for cDNAs of the Rnf6 and S26 proteins” and “Hybridization was performed in succession with a complete Mgcphlp cDNA probe and with a cDNA probe for the ubiquitous ribosomal S26 protein“. This means that the images for Rnf6 and corresponding Hss26 mRNA signals in MCB come from the same Northern blot membrane, and same is declared for the Mgcphlp and Hss26 signals in JBC.
The authors might be tempted to explain that it was the same Northern blot membrane in both papers, stripped with chemical solutions to remove the previously bound radioactive probe; it would resemble the defence which Lopez-Otin used for his Perennial Northern Blot. Only that is a rather weird scientific approach. Both papers report namely novel studies of Rnf6 and Mgcphlp mRNA expression, in these cases scientists generally prepare new gels and do not reuse old blot membranes, simply because they will not know which signal is correct and which might be a residue from the previously applied probe. They do not know in advance how strong the signal from a new probe would be, while the stripping process flushes out not just the old probe, but also some of the blotted RNA material also. Basically, stripping is something one does not do for new explorative analyses, and this is likely why a blot membrane reuse was not declared in the two papers.
But why the same loading control then? Maybe the authors decided not to probe for loading control again when they repeated the gel for the Mgcplp study, using same samples in same order. That is not really a good practice, because we can’t know how well the samples preserved, entered the gel and transferred to membrane in this repeated experiment.
Another issue highlighted above is the gel lane splicing between lanes 12 and 13 in MCB Figure 3A and corresponding lanes in Figure 3D in JBC paper. It is important because the authors did highlight the splicing one lane further in same figure 3D in JBC, with a clear vertical dividing line. Here it is in close-up:
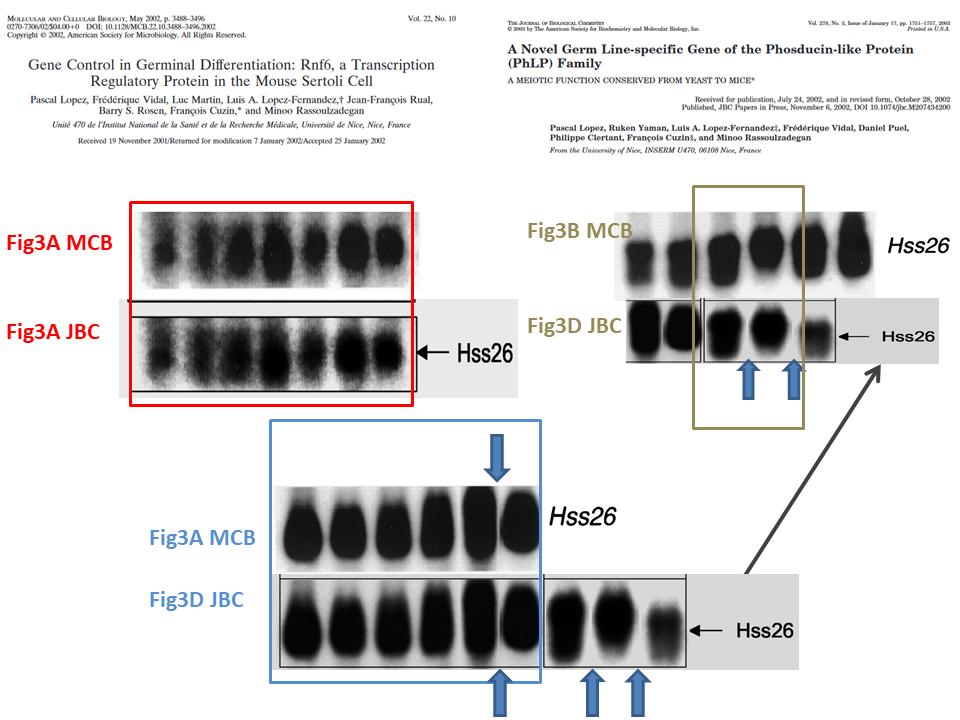
The authors knew splicing of gels must be clearly indicated, and did so in that one instance. But in other instances, they chose to hide gel splicing from peer reviewers and readers, which I indicate above with the blue arrows. And this is where we arrive to a case of intentional data manipulation, highlighted in green.
The two penultimate bands in Hss26 gel in Figure 3D JBC are spliced in, and one sees where they originally come from: the Hss26 gel in MCB paper. But the samples are not same, according to the figure legend. The original MCB legend goes: “Northern blot analysis of total RNA prepared from germinal fractions purified (≥90% pure) by elutriation centrifugation. Fractions: […] 3 and 4, round spermatids“. Compare to the legend of JBC paper for these cloned bands: “eS, rS […], 80–90% pure elutriated preparations of, respectively, elongated spermatids, round spermatids“. Hence, different preparation methodology and different samples. The reuse cannot be explained by mistake, what is presented in Figure 3D JBC as continuous gel is actually a collage of 3 different gel bands which originally showed something else than described.

The shoddy attitude to indicating gel splicing only where one feels safe to do so, is visible again in Figure 7B of the MCB paper:
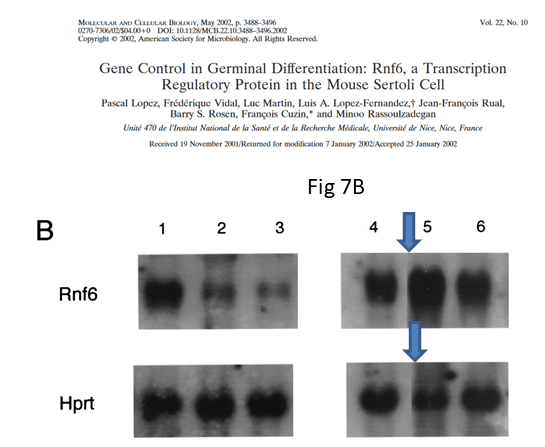
The authors declared “The same membrane was hybridized in succession with probes for the Rnf6 (top) and Hprt (bottom) mRNAs” but now we cannot be sure about that. When scientists start passing off old data as new and even stealthily mixing it into a collage to generate new results, the trust is gone. It might be not a minor issue that one of the authors is a government minister for research, whose ministry’s only reaction so far was a threat of legal action against those who reported the evidence.
Now we know why you protect Peyroche and Jessus so much, Professor Vidal.

Update 25.05.2019. On 15 May an Expression of Concern for the paper Vidal et al 2001 was published:
“Journal of Cell Science was alerted to duplicated HPRT blots in Fig. 4A and Fig. 7C of this paper. The authors state that the conclusions of the paper are not affected by the duplicated control blots, but were unable to locate the original data from almost 20 years ago. Without the original full blots, the journal is unable to determine whether the results and conclusions reported in the paper are compromised.
The journal is publishing this Expression of Concern to make readers aware of these issues. The authors offer cell lines used in the paper for replication by any interested investigators and apologise to readers for any inconvenience caused.”
Another article in News Tank followed on 24.05.2019.
We learn that it was Vidal responsible for the original lab book which now cannot be found. There seems to be a CD-ROM around, but it is not readable with modern technology, according to last author Minoo Rassoulzadegan. But now comes the kicker, in News Tank, the target being again yours truly:
“Frédérique Vidal “always reserves the right” to institute legal proceedings. […] “Frédérique Vidal, as a minister, has not deemed it necessary to prosecute at this time. However, she still reserves the right to do so, should personal accusations be brought against her”
Crazy clowns. Anyway, what is this, from Université de Nice Sophia Antipolis, featuring err… or dear… does this count as a “personal accusation”?
Isabelle Gillot, Cédric Matthews, Daniel Puel, Frédérique Vidal, and Pascal Lopez
Ret Finger Protein: An E3 Ubiquitin Ligase Juxtaposed to the XY Body in Meiosis
Int J Cell Biol. (2009) doi: 10.1155/2009/524858

Update 5.06.2019.
In today’s News Tank article (Actualité n° 148672), the senior author Minoo Rassoulzadegan explained everything. Here some translated quotes from the news piece, and please also note her reply on yet another Photoshop-train-wreck figure below.
“Leonid Schneider asserts that the authors “chose to hide the splicing” between bands of gel with results of their research in some figures of these articles. According to Minoo Rassoulzadegan, this selection of information is “necessary” for understanding the results. “Car radios are easy to handle, but not biological results,” she says.“

I was called many things, but never a car radio mechanic. Regarding the reused loading control, Rassoulzadegan explains:
“According to Minoo Rassoulzadegan, “indeed it is the same filter (nitrocellulose membrane) that has been used several times to hybridize with different probes”. “The two candidate genes came out of so-called “gene-trap” experiments. We have identified more than 400 clones and then mass characterized them, carried out gene expression analyses in different tissues, and for this the same filters have been used several times and, I repeat, with different probes.
It is quite normal that the loading controls are identical for both experiments. It is routine to use the same nitrocellulose membrane loaded with different tissues for different analyses, the same filter can be used more than ten times. Research is expensive in terms of time and resources and researchers must avoid waste.“
She doesn’t go into why loading controls were reused between different samples. But then again, Professor Rassoulzadegan is on a roll. Guess how she explained this:
Lopez P, Yaman R, Lopez-Fernandez LA, Vidal F, Puel D, Clertant P, Cuzin F, Rassoulzadegan M.
A novel germ line-specific gene of the phosducin-like protein (PhLP) family. A meiotic function conserved from yeast to mice.
J Biol Chem. 2003 DOI:10.1074/jbc.M207434200
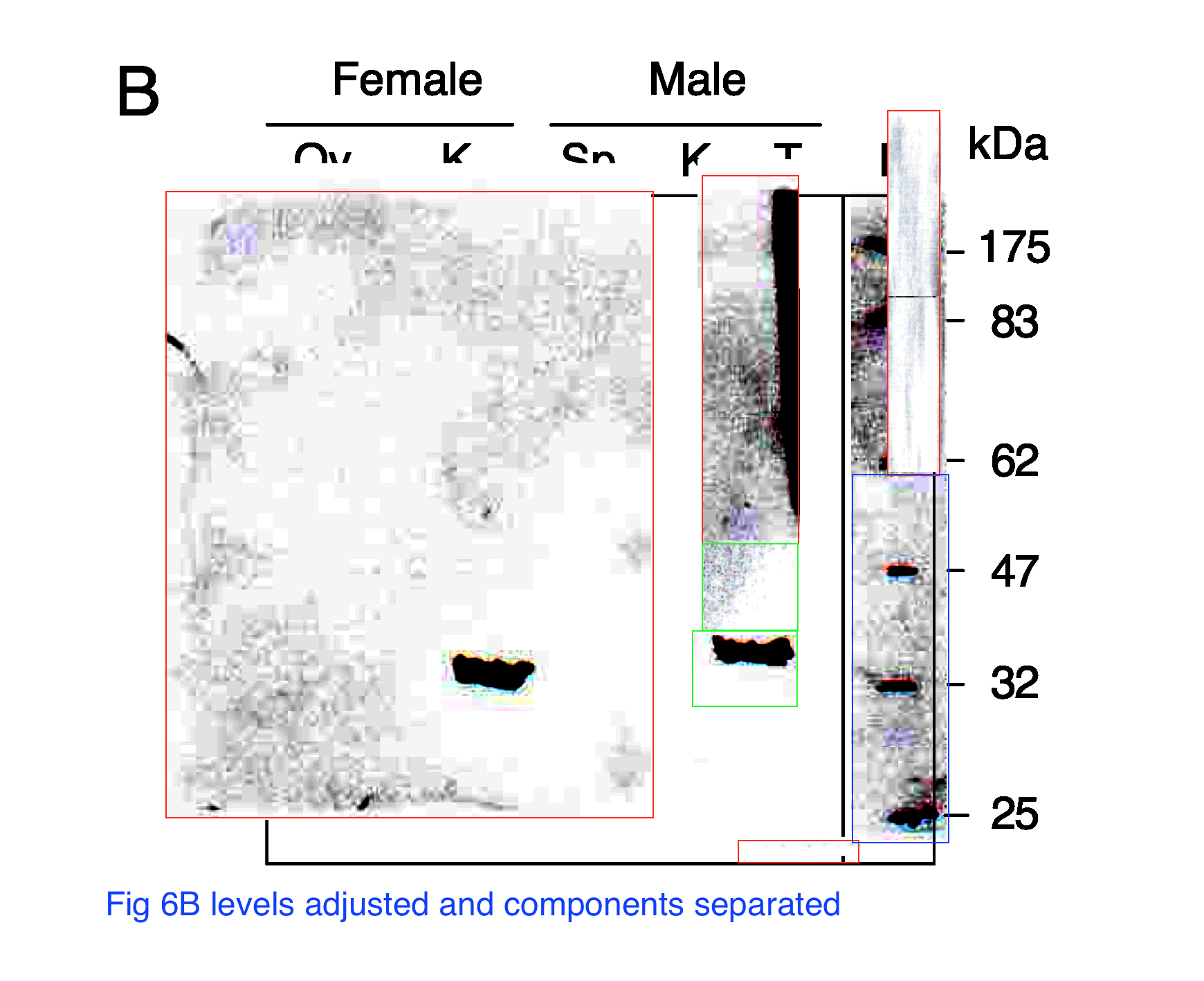
“According to Minoo Rassoulzadegan, “it is clear that in the lane T (testicles), there is much more signal than for the samples in the other lanes and it was necessary to adjust the signal, otherwise we would have seen nothing for example in the Sp lane (sperm) or somatic lanes like K (kidney) “.
Jessus wept.

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00


Pingback: Démission de Frédérique Vidal : la pression monte ! – Université Ouverte
Pingback: Remember Charles-Henri Lecellier? – For Better Science