Schneider Shorts of 25 November 2022 – how money trumps basic medical ethics for elite London universities, a Belgian failed scientist doing business in Korea, retractions overdue, forgiven or unavoided, plus horse racing, an Israeli image integrity software doing what it should, and a cool research integrity workshop in Liverpool.
Table of Discontent
Science Elites
- Imperial Standards – Melico’s trial without ethics approval, nobody minds
- Proofig in action – watch Dror Koldkin-Gal’s software absolve Didier Raoult!
Retraction Watchdogging
- Those are my pictures, and if you don’t like them… well, I have others – Dr Liu tried everything
- Whitelisted by MDPI – Munawar Iqbal didn’t give up after retraction
Science Breakthroughs
- Brain cancer scam – King’s College and Northwest Biotherapeutics present rigged trial data in JAMA
- Alzheimer’s Hibiscus – Johan Auwerx’ Korean company found a cure!
- Smart gambling – Finnish researchers prove amazing things about horse racing
News in Tweets
Science Elites
Imperial Standards
We all know how fancy the Imperial College London is, but did you know they are so elite that British national regulations on human experimenting do not apply to them? Imperial scientists do not need an ethics approval for their clinical trials, that’s who elite they are. And Imperial’s Research Integrity Officer Jon Hancock even silently confirmed to me that yes, they do not accept the authority of Human Research Authority (HRA). Also the Springer Nature journal, Nature Food, bows to the Imperial might and apparently agrees, that yes, these Imperial geniuses indeed need no ethics approvals to run their business.
It is indeed a business because we are talking about the urine-analysis start-up Melico, owned by the Imperial trio Elaine Holmes, Gary Frost and Isabel Garcia-Perez, as well as by the former Imperial professor and Holmes’ life partner, Jeremy Nicholson. I previously wrote about them.
Imperial Piss-Take
“Having reviewed the Conflict of Interest disclosures made by Professor Frost, Professor Holmes and Dr Garcia-Perez, and having also reviewed additional information concerning their company, Melico, […] the College is satisfied that they have no undisclosed or unmanageable conflicts of interests” – Arts Bachelor (Honours)
It is about this paper:
Isabel Garcia-Perez , Joram M. Posma , Edward S. Chambers , John C. Mathers , John Draper , Manfred Beckmann , Jeremy K. Nicholson , Elaine Holmes , Gary Frost Dietary metabotype modelling predicts individual responses to dietary interventions Nature Food (2020) doi: 10.1038/s43016-020-0092-z
Like in all other Melico-related publications, the Melico-owning authors declared “no competing interests“ which was endorsed as good scientific practice by their employees, the Imperial College London and the Murdoch University in Australia. As Imperial’s Hancock educated me, for Imperial professors publishing a paper advertising the business model of their own unmentioned private company can never constitute a conflict of interests.
So let’s see what else was wrong in that paper. The authors declared there (typos theirs, highlight mine):
“Study protocol, ethics and consent. Te study was approved by the London–Brent Research Ethics Committee and carried out in accordance with the Declaration of Helsinki (13/LO/0078). Te study protocol is available from ref. 21. All participants provided written informed consent. An external cohort (NutriTech32) was used to validate associations with health-related markers (London–Brent Ethics Committee reference number: 12/LO/0139). Tis dataset contained 65 participants (31 female), with a mean BMI of 29.0±2.9 kgm−2 at baseline, for which urine samples were available from baseline measurements before starting the clinical trial (NCT01684917).We used our metabolite discovery study, which uses a semicontrolled four-day feeding study. Ethical approval for this study is subsidiary to the main study ‘Dietary biomarker discovery using metabonomics’. The study was approved by the London–Brent Research Ethics Committee and carried out in accordance with the Declaration of Helsinki (13/LO/0078).”
I never heard of the concept of subsidiary ethics approvals, so I decided to investigate.
The authors name in the published pdf the clinical trial NCT01684917, but link to a different clinical trial registration, which took place in USA. The correct NCT01684917 trial by Imperial College however was also very different from the older one (ISRCTN43087333) from which they derive their “subsidiary” ethics approval.
The protocol of the main study, which completed in 2015/2016, had just one trial arm with 19 participants and was about “four 3-day interventions during 4 consecutive weeks, with the administration of specific diets differing in amounts of specific foods deemed to have high public health importance (oily fish, wholegrain foods, fruits and vegetables)“. The Nature Food study, which completed in 2020, had 36 participants and 3 arms: “Experimental: Life style advice“, “Active Comparator: UK background diet” and:
“Metabolomic inquiry- This inquiry took place prior to the randomized controlled trial and include 50 volunteers who will then be asked to volunteers of the weight loss study. Were randomly assigned to one of five different diets; red meat, fish, poultry, processed meat or a supplement and vegetarian option.”
Basically, two totally different and unrelated trials. To use an old ethics approvals for unrelated future clinical studies is illegal, in fact this is what Didier Raoult is being criminally investigated for in France.
Didier Raoult fraud: “Je ne regrette rien”
One year on: more fake data, financial fraud and illegal and falsified clinical trials by the chloroquine guru Didier Raoult.
But maybe in UK this is legal? I asked HRA, who informed me on 1 November 2022 (highlights mine):
“We can confirm that we are aware of the matter that you have raised, which has been investigated under the HRA breaches policy and procedure. As an outcome of this investigation, in July 2022 the Research Ethics Committee (REC) identified that a breach had occurred due to the additional feeding study, which is referred to in the Nature Foods publication, being conducted without a favourable ethics opinion from the REC in place. We have been informed that the research team had believed that the study had ethical approval under the favourable opinion of the main study (REC reference: 13/LO/0078), however the REC had not been informed that a further study would be undertaken and the sub-study documentation had not been approved for use.
In response, the sponsor has informed us that they intend to implement a number of preventative actions to ensure that a similar situation does not arise again. These actions include: a four monthly section ethics meeting for staff dealing with human studies, an audit of the use of consent forms and patient information sheets in all studies to ensure alignment with good clinical practice and all staff involved in a clinical study will be brought together with the Chief Investigator before the study begins to discuss roles and responsibilities, including study management, safety reporting and any required regulatory submissions and approvals throughout the lifecycle of the study.
Finally, the term ‘subsidiary’ ethics approval is not one that is used or recognised by the HRA. We have recommended that the sponsor contacts the journal to notify them that the sub-study did not have ethical approval and that the reference to subsidiary ethical approval should be removed. The sponsor has informed us that the research team have contacted the journal editor to discuss this matter. Following this, we are satisfied that the appropriate corrective action will have taken place.”
The sponsor being the Imperial College. Who indeed seem to have contacted the journal to negotiate with them the proper action which is specifically NOTHING AT ALL.
I forwarded this HRA statement to the Imperial’s Research Integrity Officer Jonathan Hencock and to the London-based Nature Food chief editor Anne Mullen, with this note:
“Please understand that if you don’t reply I will have to build on what you previously told me and inform my readers that the Imperial College does not recognise the authority and opinion of HRA and NHS.
I also understand that the Nature Publishing Group doesn’t recognize HRA as a qualified public authority, at least not qualified to inform the editorial decisions.”
Neither side replied. Where money is at stake, national laws on human research cease to apply to elite universities, and national medical authorities set up to protect patients can f*** right off.
Now you can understand why The Lancet (also in London) is free to continue its support for Paolo Macchiarini and Martin Birchall. UCL still wants to make money with trachea transplants.
Time to retract Lancet paper on tissue engineered trachea transplants
Paolo Macchiarini affair: I reproduce the letter Patricia Murray, Raphael Levy, Peter Wilmshurst and myself published in The BMJ on 2 March 2022. I also publish Wilmshurst’s appeal to the UCL leadership.
But! On 14 November 2022, two weeks after my emails, Nature Foods issued this Editor’s Note:
“Readers are alerted that the statement of ethical approval for the semi-controlled four-day feeding study utilised in this Article, granted as subsidiary to the main study ‘Dietary biomarker discovery using metabonomics’ approved by the London–Brent Research Ethics (13/LO/0078), has been called into question. A further editorial response will follow the resolution of this issue for this Article.”
I am pretty sure this is how they think they closed the case for good. We’ll see.
Proofig in action!
We know that Ilana Koldkin-Gal‘s husband Dror is spamming fellow cheater scientists offering his pre-submission screening services for their fraudulent papers. We know Kolodkin-Gal’s software Proofig is being used by various scholarly publishers to screen problematic papers in order to officially dismiss evidence of image data forgery.
But so far we never saw Proofig in action. Until now, applied to a paper by none other but the French chloroquine quack and patient-abusing sociopath, Didier Raoult. It was flagged by Elisabeth Bik in October 2021:
Zhongxing Liang , Bernard La Scola, Hubert Lepidi , Didier Raoult Production of Bartonella Genus-Specific Monoclonal Antibodies Clinical and diagnostic laboratory immunology (2001) doi: 10.1128/cdli.8.4.847-849.2001
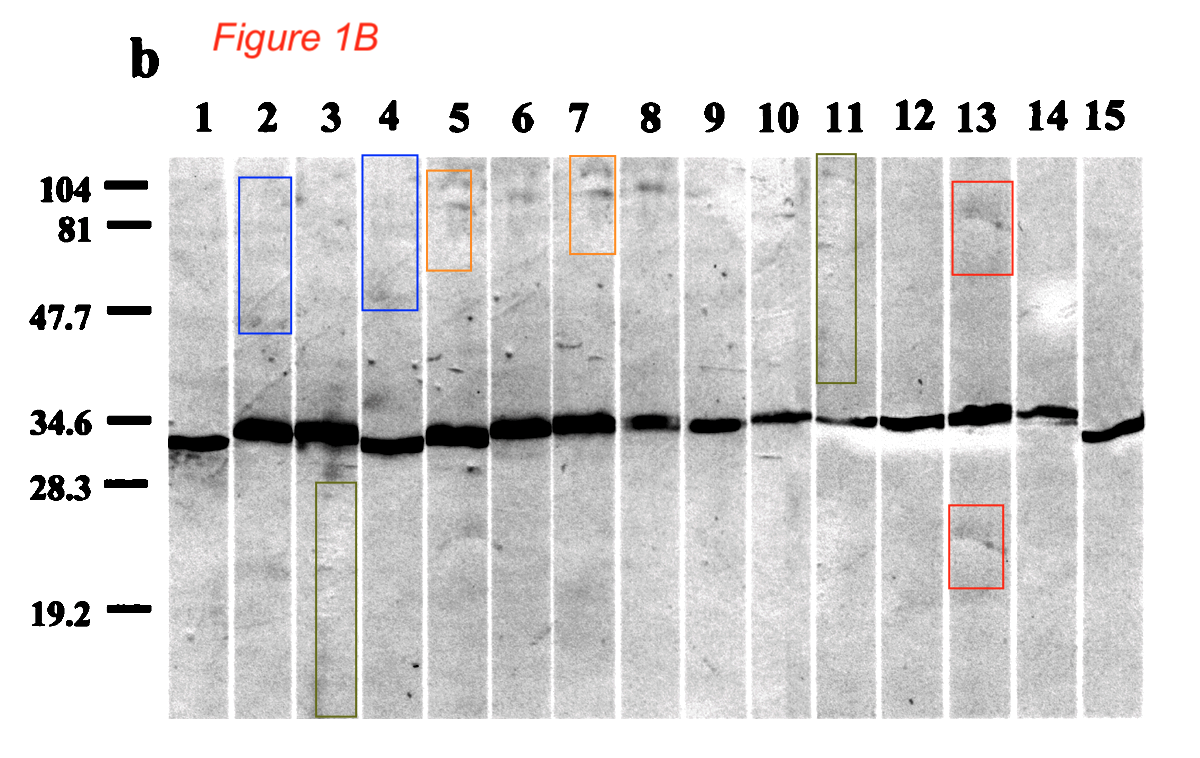
The journal is published by the American Society for Microbiology (ASM) which once blacklisted Raoult for fraud, read here:
Chloroquine genius Didier Raoult to save the world from COVID-19
As COVID19 pandemic rages on, French microbiologist Didier Raoult offers a cure. President Trump is convinced, but is Raoult’s research reliable, here and in general?
Another PubPeer user found even more instances of data forgery and inappropriate data recycling in this publication. But then, in November 2022, someone arrived to declare:
“Ran Figure 1B sub images through Proofig and no suspected regions mentioned by #1 were found.“
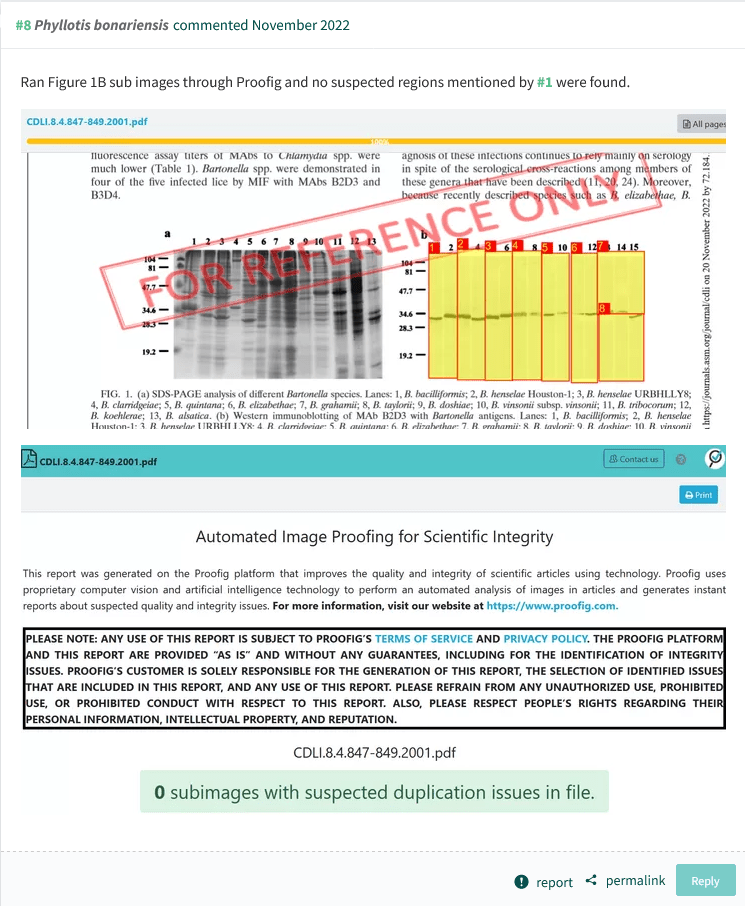
ASM is not using Proofig, so it wasn’t them commenting anonymously. Neither is Proofig generally available to others (Bik previously tried to test it, request denied). But it is not entirely unlikely that Raoult himself (or one of his IHU Marseille allies) bought Kolodkin-Gal’s “expert” services to negate all the PubPeer evidence of data forgery found by Bik and others in Raoult’s papers (note: much of Raoult’s PubPeer record concerns absence of ethics approvals or hidden COI).
In any case, Proofig authoritatively says there are no duplications in that figure. But of course there are, look again:

Now you see how Proofig works. Its fraudster customers will probably give five stars.
Retraction Watchdogging
Those are my pictures, and if you don’t like them… well, I have others
Who could ever expect any fraud in such a venerable scholarly journal, dedicated to TCM and Ayurveda? This important ginseng study was flagged in July 2022 (Hattip Tiger BB8):
Ming-wei Liu , Rui Wei , Mei-xian Su , Hui Li , Tian-wen Fang, Wei Zhang
Effects of Panax notoginseng saponins on severe acute pancreatitis through the regulation of mTOR/Akt and caspase-3 signaling pathway by upregulating miR-181b expression in rats BMC Complementary and Alternative Medicine (2018) doi: 10.1186/s12906-018-2118-8
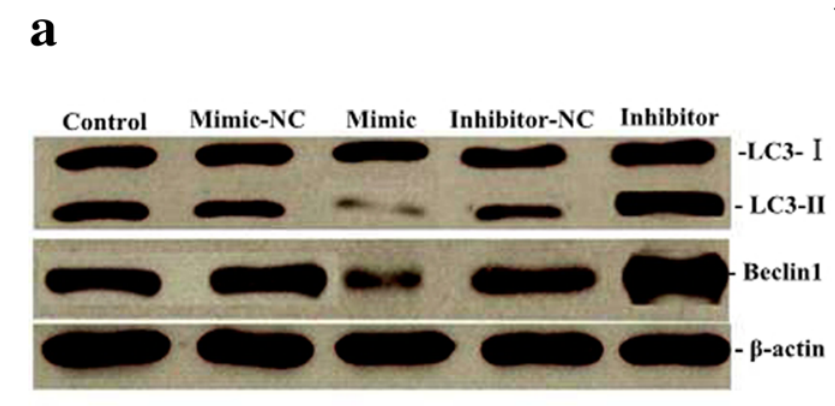
Groucho Marx, uhm, Ming-wei Liu, professor of emergency medicine from Kunming, immediately posted raw data on PubPeer to dispel the concerns:
“Dear PubPeer I thank you so much for hearing from you! I am very sad and sorry that this happened. Based on your comment, I am providing you with the original, uncropped, images, and our corrected image.“
It may have been raw data, but there was a catch, as another PubPeer user noted:
“Your posted images have nothing to do with the issue raised in comment #1, none of the bands in the images that you posted, and claimed to be original images, look even close to the Western Blotting in question.”
Again, Liu had images to share:
“We found that the raw data we provided to you in the last time was wrong, and we are very sorry.“
M. arachidis made a comparison:

Please see my side by side comparison of your original figure, compared to the blot you have now provided. I hope you can see why this is still confusing…“
Liu of course has yet another set of raw data:
“For the wrong data, the reason is that we missubmitted the results that tested in the pre-experiment to you. We have made the pre-experiment of the test of Beclin1 for protein expression before this research. Due to the different instruments used in the pre-experiment and this experiment, there are great difference in the results. […] Because the band of Beclin1 has been edited in the published papers, and compared to the unedited data, there still some differences remain. We have done everything we can do...”
Of course also this new set of raw data didn’t fit. Cheshire had enough and turned attention to other figures:
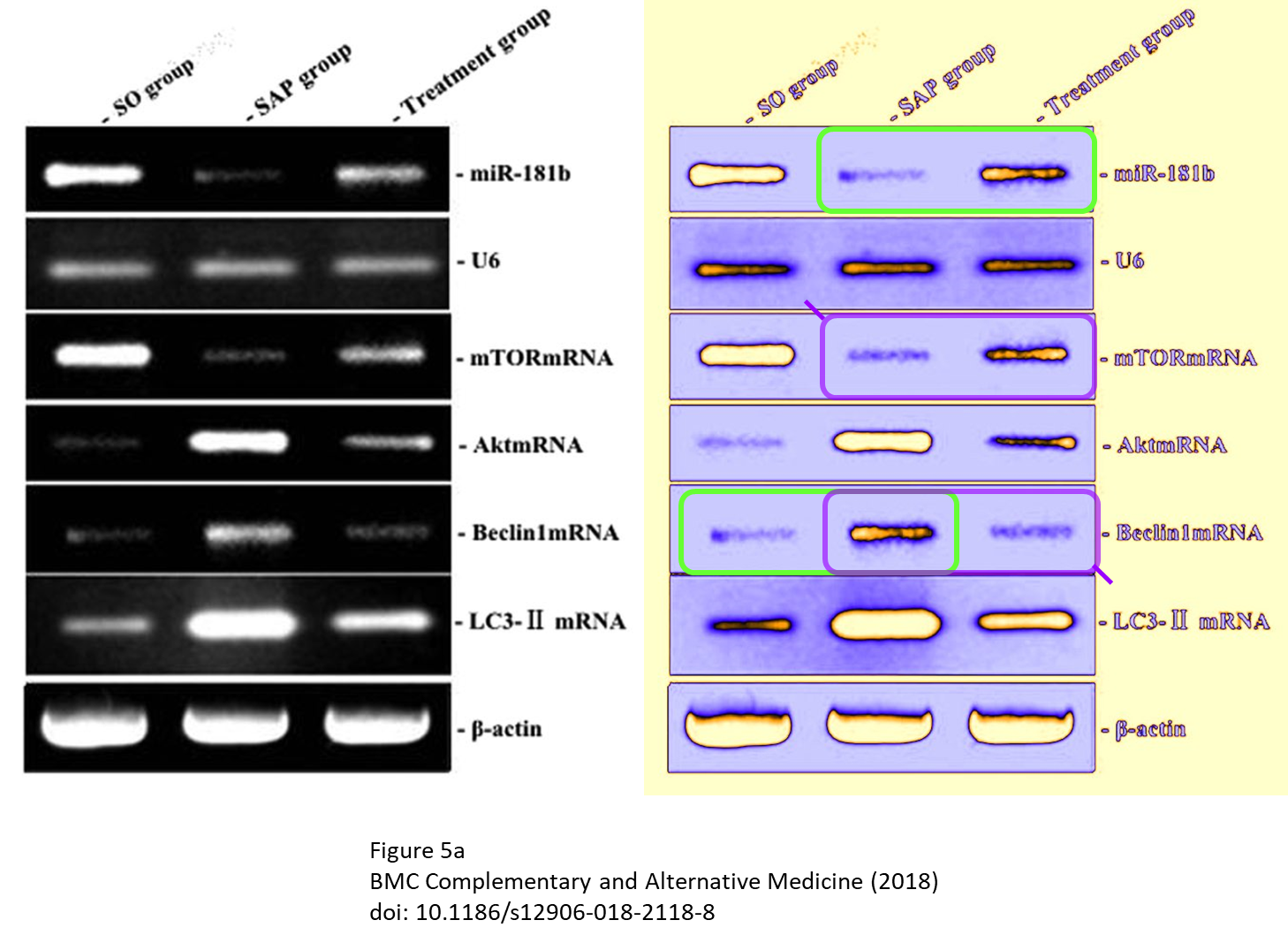

Liu was very helpful and shared replacement figures:
” we supplemented a lot of new experimental data and re-organized the figures including Figure 5A and 7A. in purpose to improve the image quality, which led to the misuse of the image..
Fortunately, this individual mistake doesn’t influence the statistical results of the study […] I will contact the Editor-in-Chief of BMC Complement Altern Med to correct this mistake.”
That was in September 2022. On 23 November 2022 Liu’s paper was retracted:
“The Editor has retracted this article because, after publication, concerns were raised with respect to a number of the figures, in particular:
- Panel Beclin1 in Figure 2A appears to contain visual anomalies
- In Figure 5A panel miR-181b overlaps with panel Beclin1mRNA
- In Figure 5A panel mTORmRNA overlaps with panel Beclin1mRNA
- The microscopy images in Figure 7 are also present in Figure 4E of [1]
The Editor therefore no longer has confidence in the veracity of the data presented.
In addition, the rats in this study were euthanized by cervical dislocation which is not an appropriate method for rats in excess of 200 g in mass.
None of the authors have responded to any correspondence from the editor/publisher about this retraction.”
Prof Liu must have run out of pictures.
Whitelisted by MDPI
In 2017, the Pakistani scholar Munabar Iqbal retracted a paper, in MDPI no less.
Adeela Nairan , Usman Khan , Munawar Iqbal , Maaz Khan , Khalid Javed , Saira Riaz , Shahzad Naseem , Xiufeng Han Structural and Magnetic Response in Bimetallic Core/Shell Magnetic Nanoparticles Nanomaterials (2016) doi: 10.3390/nano6040072
“It has come to our attention that Figure 3 of the title paper [1] contains unacceptable levels of image manipulation and thus does not provide firm evidence of the particles reported. In particular, particles have been copied and pasted to make multiple copies in a single image, and the high contrast between the inner and outer shells of the particles are not plausible given their supposed composition. Further images provided by the authors exhibited the same flaws in contrast. As such, the conclusions of the paper are no longer supported and the paper will be marked as retracted.”
It was very fake, as visualised by Elisabeth Bik in 2021 on PubPeer:

Question to you: were Iqbal and his friends banned from MDPI after this? Did they at least screen his published and newly submitted papers? No and no.
Flagged in 2021:
Adeela Nairan , Maaz Khan , Usman Khan , Munawar Iqbal , Saira Riaz , Shahzad Naseem Temperature-Dependent Magnetic Response of Antiferromagnetic Doping in Cobalt Ferrite Nanostructures Nanomaterials (2016) doi: 10.3390/nano6040073

In 2022, Iqbal became really productive, and MDPI was more than happy to take his money and his fraud:
Saddam Hussain , Norah Alwadai , Muhammad I. Khan , Muhammad Irfan , Ikram-ul-Haq , Hind Albalawi , Aljawhara H. Almuqrin , Maha M. Almoneef , Munawar Iqbal The Effect of 600 keV Ag Ion Irradiation on the Structural, Optical, and Photovoltaic Properties of MAPbBr3 Films for Perovksite Solar Cell Applications Materials (2022) doi: 10.3390/ma15155299
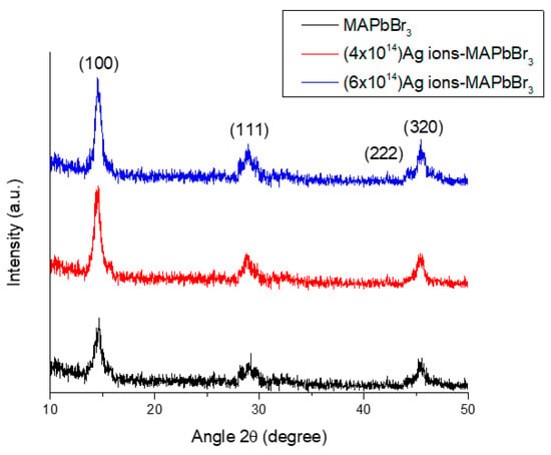
Also in MDPI:
Muhammad Amjad , Muhammad Iftikhar Khan , Norah Alwadai , Muhammad Irfan , Ikram-ul-Haq , Hind Albalawi , Aljawhara H. Almuqrin , Maha M. Almoneef , Munawar Iqbal Photovoltaic Properties of ZnO Films Co-Doped with Mn and La to Enhance Solar Cell Efficiency Nanomaterials (2022) doi: 10.3390/nano12071057
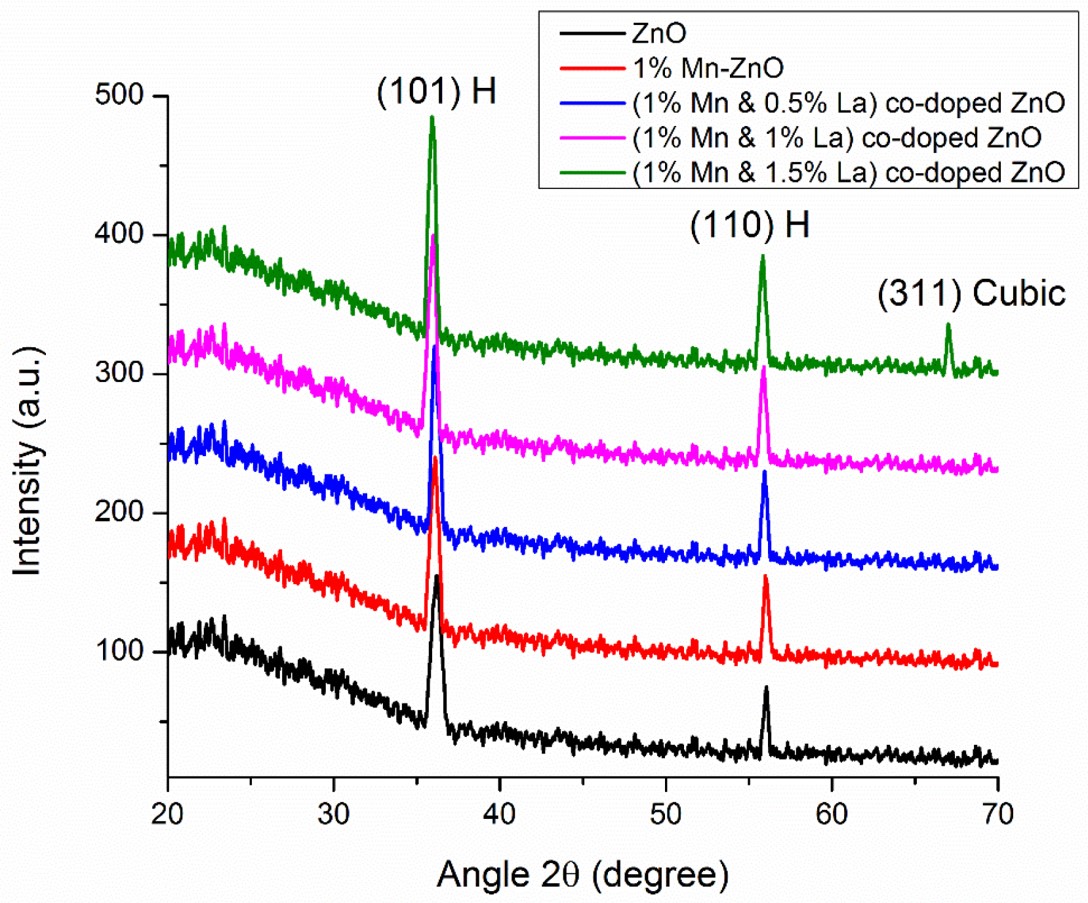
How about these cloned and forged spectra, in two more MDPI papers:

Iqbal has of course more on PubPeer, with other publishers, especially Elsevier. Here an example, revealing his hobby of torturing small animals to produce fake science:
Mazhar Abbas , Tariq Hussain , Muhammad Arshad , Abdur Rahman Ansari , Asma Irshad , Jan Nisar , Fida Hussain , Nasir Masood , Arif Nazir , Munawar Iqbal Wound healing potential of curcumin cross-linked chitosan/polyvinyl alcohol International Journal of Biological Macromolecules (2019) doi: 10.1016/j.ijbiomac.2019.08.153

And in March 2021, Iqbal’s University of Lahore celebrated that their assistant professor of chemistry made it to the “51st position in the list World’s Top 2% Scientists” in a Stanford University list by John Ioannidis.

You see how little Ioannidis’ fetish of citation metrics is worth. In fact, even some of Ioannidis’ own citations stem from papermills:
The Rise of the Papermills
“Is it possible that through no fault of Zintzaras & Ioannidis, their work was incorporated into a papermill template, accruing hundreds of spurious citations?” – Smut Clyde
Science Breakthroughs
Brain cancer scam
A hot new press release by King’s College London announced a breakthrough in glioblastoma therapy:
“Results of a phase three clinical trial of a novel brain cancer vaccine, DCVax-L, that uses a patient’s stem cells to target cancer cells extended survival by many months to years for glioblastoma patients. […]
The vaccine is created for each patient individually by isolating specific immune cells, known as dendritic cells, from their blood. These cells are then primed with biomarkers from a sample of the patient’s tumor. When the vaccine containing the cells is injected back into the patient, it shares that information so that the body’s entire immune system recognizes and attacks the target.
The vaccine was trialed for eight years, and involved more than 300 patients from the U.K., U.S., Canada and Germany”
This was the trial NCT00045968, run by the company Northwest Biotherapeutics (NWBT) and this is the paper:
Linda M. Liau , Keyoumars Ashkan , Steven Brem , Jian L. Campian , John E. Trusheim , Fabio M. Iwamoto , David D. Tran , George Ansstas , Charles S. Cobbs , Jason A. Heth , Michael E. Salacz , Stacy D’Andre , Robert D. Aiken , Yaron A. Moshel , Joo Y. Nam , Clement P. Pillainayagam , Stephanie A. Wagner , Kevin A. Walter , Rekha Chaudary , Samuel A. Goldlust , Ian Y. Lee, Daniela A. Bota, Heinrich Elinzano, Jai Grewal, Kevin Lillehei, Tom Mikkelsen, Tobias Walbert, Steven Abram, Andrew J. Brenner, Matthew G. Ewend, Simon Khagi, Darren S. Lovick, Jana Portnow, Lyndon Kim, William G. Loudon, Nina L. Martinez, Reid C. Thompson, David E. Avigan, Karen L. Fink, Francois J. Geoffroy, Pierre Giglio, Oleg Gligich, Dietmar Krex, Scott M. Lindhorst, Jose Lutzky, Hans-Jörg Meisel, Minou Nadji-Ohl, Lhagva Sanchin, Andrew Sloan, Lynne P. Taylor, Julian K. Wu, Erin M. Dunbar, Arnold B. Etame, Santosh Kesari, David Mathieu, David E. Piccioni, David S. Baskin, Michel Lacroix, Sven-Axel May, Pamela Z. New, Timothy J. Pluard, Steven A. Toms, Victor Tse, Scott Peak, John L. Villano, James D. Battiste, Paul J. Mulholland, Michael L. Pearlman, Kevin Petrecca, Michael Schulder, Robert M. Prins, Alton L. Boynton, Marnix L. Bosch Association of Autologous Tumor Lysate-Loaded Dendritic Cell Vaccination With Extension of Survival Among Patients With Newly Diagnosed and Recurrent Glioblastoma: A Phase 3 Prospective Externally Controlled Cohort Trial JAMA Oncology (2022) doi: 10.1001/jamaoncol.2022.5370
There are no positive results actually, the DCVax drug is not just trash, it’s dangerous.
Here is the debunking by Adam Feuerstein in STAT News:
“The data involving the treatment called DCVax might appear to be encouraging, particularly for patients with newly diagnosed glioblastoma, an aggressive form of brain cancer. But the survival benefit was derived from an untrustworthy, post-hoc analysis that was implemented and conducted after the study had already been completed. […]
Originally, the study incorporated the gold-standard design for robust clinical trials. Investigators recruited 331 patients with newly diagnosed glioblastoma and randomized them in a blinded fashion to receive either DCVax or a matching placebo. In a pre-specified analysis of its primary efficacy goal, the DCVax group went a median 6.2 months without their brain tumors returning compared to a median of 7.6 months for patients offered a placebo.
DCVax performed worse than a placebo. The study failed. A subsequent survival analysis comparing DCVax to placebo was also pre-specified, but was never performed.
After the study was completed, Northwest Bio threw out the negative results, replacing the original placebo group with survival data collected from 1,366 patients who participated in five entirely separate and previously completed clinical trials.
When the 233 patients treated with DCVax were compared to the 1,366 patients taken from “external controls,” Northwest Bio said it found a 20% relative reduction in the risk of death.”
Feuerstein also notes:
“But on Thursday, alongside the main DCVax study paper, the study’s protocol — essentially an instruction manual for how the study was conducted — was also published as a supplement in JAMA Oncology. And in it, the study authors disclosed that an interim analysis of the original tumor-progression endpoint was conducted in March 2015, well before the study had been completed.
The outcome of the interim analysis, however, was not disclosed in the protocol. […]
In August 2015, five months after the now-confirmed interim analysis took place, the Food and Drug Administration placed a clinical hold on the DCVax study, barring the company from enrolling any new patients. The hold wasn’t lifted until February 2017, and then only on the condition that the company close the study, falling short of its planned patient enrollment.
Northwest Bio has never offered a public explanation for the FDA clinical hold.”
Would you like who a key collaborator of Northwest Bio in London is? Mark Lowdell, UCL professor with a regmed lab at Royal Free who made the deadly trachea transplants for Martin Birchall and more recently was caught on selling umbilical cord blood to anti-aging quacks. Lowdell is the business partner of the Northwest Bio CEO Linda Power, here are details. For example the duo are the sole officers of Revimune, a dormant company that holds patents for Lowdell’s company INmune Bio and for Power’s Northwest Bio. Both made many millions with this DCVax drug already, both with investment money and patient payments.
Lowdell’s snout is in every through of money it seems.
Martin Birchall’s two dead pigs to prove trachea transplants are safe
Imagine you are Martin Birchall, laryngologist and ENT surgeon, star of regenerative medicine at UCL and trachea transplant enthusiast. You and your business partner Videregen need to explain to EU bureaucrats why your technology of decellurised cadaveric trachea is perfectly safe, what with all the dead patients of yours and your former best friend Paolo…
Alzheimer’s Hibiscus
An article in Medical News Today informs:
“A recent study conducted in mice, whose results appear in the journal Alzheimer’s Research & Therapy, suggests that gossypetin, a compound derived from the flower of the roselle plant, a hibiscus species, may hold promise as a therapeutic agent for Alzheimer’s.
The study found that gossypetin helped reduce cognitive deficits and lower beta-amyloid levels in brain regions involved in cognitive function in a mouse model of Alzheimer’s disease. […]
In the present study, the researchers used the 5xFAD mouse model to examine the effects of gossypetin. The 5xFAD mouse model expresses two human genes carrying a total of five mutations that are common in familial Alzheimer’s disease. […]
Treatment of 5xFAD mice with gossypetin also reduced the size and number of beta-amyloid plaques in the hippocampus and cortex, brain regions involved in cognition. Moreover, the administration of gossypetin also led to a decrease in beta-amyloid monomers and oligomers in these brain regions.“
The lead author Kyong-Tai Kim from the Pohang University of Science and Technology in South Korea is quoted:
“Gossypetin from hibiscus will contribute to the development of a safe and affordable drug for patients suffering from [Alzheimer’s disease].”
This is the study:
Kyung Won Jo , Dohyun Lee , Dong Gon Cha , Eunji Oh , Yoon Ha Choi , Somi Kim , Eun Seo Park , Jong Kyoung Kim , Kyong-Tai Kim Gossypetin ameliorates 5xFAD spatial learning and memory through enhanced phagocytosis against Aβ Alzheimer’s Research & Therapy (2022) doi: 10.1186/s13195-022-01096-3
The authors declare no conflicts of interest except one who is an employee of Novmetapharma. We are also informed:
“This research was mainly supported by the Novmetapharma Co.”
So far, so useless, so boring. But look who this Korean’s company’s very un-Korean research director is?
It’s the Belgian cell biologist Johan Auwerx, now professor at Swiss Federal Institute of Technology Lausanne (EPFL) in Switzerland and the man with a fat PubPeer record of almost 30 papers. Here an example:
Elisabeth Fayard , Kristina Schoonjans , Jean-Sébastien Annicotte , Johan Auwerx Liver receptor homolog 1 controls the expression of carboxyl ester lipase Journal of Biological Chemistry (2003) doi: 10.1074/jbc.m302370200

Or this:
Lluis Fajas , Viviane Egler , Raphael Reiter , Jacob Hansen, Karsten Kristiansen, Marie-Bernard Debril , Stéphanie Miard , Johan Auwerx The retinoblastoma-histone deacetylase 3 complex inhibits PPARgamma and adipocyte differentiation Developmental Cell (2002) doi: 10.1016/s1534-5807(02)00360-x


Another one:
Lluis Fajas , Kristina Schoonjans , Laurent Gelman , Jae B. Kim , Jamila Najib , Genevieve Martin , Jean-Charles Fruchart , Michael Briggs , Bruce M. Spiegelman , Johan Auwerx Regulation of peroxisome proliferator-activated receptor gamma expression by adipocyte differentiation and determination factor 1/sterol regulatory element binding protein 1: implications for adipocyte differentiation and metabolism Molecular and Cellular Biology (1999) doi: 10.1128/mcb.19.8.5495

Or this:
Lluis Fajas , Rebecca L. Landsberg , Yolande Huss-Garcia , Claude Sardet, Jacqueline A. Lees, Johan Auwerx E2Fs regulate adipocyte differentiation Developmental Cell (2002) doi: 10.1016/s1534-5807(02)00190-9



In this regard, the only reaction so far was Auwerx’ first author and now his fellow EPFL professor Lluis Fajas bullshitting his critics on PubPeer:
Lluis Fajas , Stephanie Miard , Michael R. Briggs , Johan Auwerx Selective cyclo-oxygenase-2 inhibitors impair adipocyte differentiation through inhibition of the clonal expansion phase Journal of Lipid Research (2003) doi: 10.1194/jlr.m300248-jlr200


Fajas explained in June 2020:
“I feel very uncomfortable not knowing with whom I am discussing. […] Please let me insist, all the blots shown in the different papers are the same experiment. This is the reason why the PPARg blot (common to all) is shown several times. Have a nice day“
When asked by PubPeer users “why are the days of differentiation different in each paper?“, Fajas retorted: “This is a mistake.” And that was it. No retractions, no corrections, no nothing. The two failed scientists Fajas and Auwerx continued stuffing their pockets with fat Swiss professorial salaries, and even that was not enough. There had to be industry money of course:
Auwerx previously was spotted next to David Sinclair on the advisory board of MetroBiotech, an anti-aging scam sponsored by the Pentagon. And now he went to Korea to sell hibiscus for Alzheimer’s.
In April 2020 I informed Auwerx and his EPFL about the falsified data in his papers. Auwerx never replied, but the scientific ombudsman of the EPFL, emeritus professor Winship Herr, announced to have a look. I haven’t heard from him since and used this new occasion to send a reminder, two and a half years later. Herr told me:
“Please note that my job description as EPFL Scientific Ombudsperson does not include performing autonomous investigations. I aid concerned individuals to resolve issues of scientific integrity with the EPFL. I decide whether an accusation is admissible and if so transmit the accusation to the EPFL president’s office for investigation.“
Most obviously Herr did nothing it all with my notification of suspected research misconduct in those two and a half years. Because he must have decided it was inadmissible. Herr did not protest to this supposition.
Smart gambling
A press release by the University of Eastern Finland:
“According to recent study from researchers at the University of Eastern Finland and the University of Liverpool, men with higher IQs are more inclined to bet on horse races.
Men with higher numerical IQs were shown to be more likely to partake in skill-based gambling, such as horse racing, choose more complicated betting options, and spend more money, according to a study published in the Journal of Behavioral Decision Making.
More than 15,000 Finnish males who had taken an IQ test as part of their mandatory military duty while enlisting in the Finnish Defense Forces participated in the research. This was cross-referenced with thorough records on the men’s online gambling behavior as well as statistics from Statistics Finland on their socioeconomic standing, income, and level of education.”
This is the paper:
Niko Suhonen, Jani Saastamoinen, David Forrest and Tuomo Kainulainen “Does IQ predict engagement with skill-based gambling? Large-scale evidence from horserace betting” Journal of Behavioral Decision Making. (2022) DOI: 10.1002/bdm.2300
Obviously the Betteridge’s law of headlines applies here. The press release quotes two supremely intelligent men sharing wisdom:
“Commenting on the findings, Professor David Forrest from the University of Liverpool Management School said: “Our research found a strong correlation between men with a high IQ and those who take part in skilled gambling, such as betting on horse racing. It is important to note that our findings can’t necessarily be generalised to chance-based gambling, such as gaming machines.” […]
Associate Professor Jani Saastamoinen of the University of Eastern Finland said: “Betting could be compared to solving crossword puzzles. Perhaps, bettors like to crunch numbers and find the winning horses – even though they know they’ll end up losing money in the long run.”
With science like this, who needs parody.
News in Tweets
- Wiley now retracted at least two papers for which the publisher previously declared “The data remain reliable & we view the matter closed.” But in between the Chinese authorities, acting on PubPeer evidence by Smut Clyde, determined those papers to be fraudulent, i.e. sourced from papermills; their “authors” were sanctioned and retractions were requested. Wiley eventually teeth-gnashingly obliged. Both retractions are in BioFactors, I wrote about Ma et al 2020 in earlier Shorts here (October 2022 retraction here), and here is Bao et al 2019, its undated retraction here. Both state “The retraction has been agreed following an investigation based on allegations raised by a third party.”
- Following a previous Expression of Concern, a Retraction for Wang et al Bioscience Reports 2020 (typo theirs, highlight mine): “This article is being retracted from Bioscience Reports at the request of the Editorial Board following receipt of a notification from a reader […] The authors had provided replacement images for Figure 5A, however as the original data could not be validated, a Correction could not procced. The Editor-in-Chief, Editorial Board and the authors do not agree to the Retraction.”
- HHR-ORI report from “ORI found that Douglas D. Taylor, Ph.D. (Respondent), former Professor and Vice Chair for Research, Department of Obstetrics & Gynecology, UL, engaged in research misconduct […] ORI found based on a preponderance of the evidence that Respondent intentionally, knowingly, or recklessly used falsely labeled images to falsely report data in figures, and in one finding, intentionally, knowingly, or recklessly plagiarized, reused, and falsely labeled an image to falsely report data in a figure. Respondent’s research misconduct occurred in one (1) funded PHS grant application, twelve (12) unfunded PHS grant applications, and two (2) PHS-supported published papers.” Taylor is barred from applying for NIH grants and reviewer activities for 3 years, four retractions are requested. Taylor only has one paper listed on PubPeer.
- A research integrity workshop organised by Patricia Murray featuring Peter Wilmshurst and David Sanders! At the University of Liverpool, Monday, 5th December 2022 from 2pm-4.30pm, also online via Zoom. I strongly recommend attending!
I thank all my donors for supporting my journalism. You can be one of them!
Make a one-time donation:
I thank all my donors for supporting my journalism. You can be one of them!
Make a monthly donation:
Choose an amount
Or enter a custom amount
Your contribution is appreciated.
Your contribution is appreciated.








“Please let me insist, all the blots shown in the different papers are the same experiment.” (Fajas)…..Well, that is a big problem; isn’t publishing the same stuff in different journals copyright violation??!!
LikeLike
Count the f***s EPFL gives.
http://cigreport.genomyx.ch/marriage-de-raison-the-interplay-between-cell-cycle-control-and-metabolism/
LikeLike
NWBO … Can’t King’s College press release do a bit more to confuse people about DC Vax?
“Unlike chemotherapy and radiotherapy, the vast majority of patients reported no side-effects from the immunotherapy vaccine. Only five of the 331 patients who took part reported any significant adverse effects, which may have been related to their treatment.”
The vaccine was given after patients had 6 weeks of radiotherapy and temozolomide, a combination which most certainly does produce significant adverse effects. While it’s good news that few experienced side effects from the vaccine, NO DATA suggests the vaccine alone is effective to treat cancer in humans.
JAMA Oncology should be ashamed to publish a paper in which only the sponsor has full access to the study data.
LikeLike