Thanks to a recent press release from Canada, another repurposed COVID-19 drug is making news worldwide: colchicine. Like chloroquine, it is cheap, generic and even “natural”, prepared from the plants (here, autumn crocus or glory lilly). Colchicine is an anti-inflammatory drug which interacts with mitosis (eg, of immune cells), and was applied as gout medicine for thousands of years, later for other autoimmune diseases. It also has a use in prevention of atrial fibrillation, which is why it was previously proposed to use in combination with the two magic COVID-19 drugs chloroquine and azithromycin, to prevent heart complications.
But now colchicine is a miracle drug of its own. The idea being, if immunosuppressive corticosteroid hormones like dexamethasone help severely ill ventilated COVID-19 patients, it stands to reason that colchicine will help SARS2-CoV positive patients who are not even hospitalised. It can be even the COVID-19 crusher, a miracle preventive drug, there to succeed where chloroquine and ivermectin succeeded failed succe.., science has not decided yet, you know.
Science per Press Release
Hence, a cardiologist in Montreal, Canada, ran a clinical trial and here is the recent press release.
MONTREAL, January 22, 2021 – The Montreal Heart Institute (MHI) announced today that the COLCORONA clinical trial has provided clinically persuasive results of colchicine’s efficacy to treat COVID-19. The study results have shown that colchicine has reduced by 21% the risk of death or hospitalizations in patients with COVID-19 compared to placebo. This result obtained for the global study population of 4488 patients approached statistical significance. The analysis of the 4159 patients in whom the diagnosis of COVID-19 was proven by a naso-pharyngeal PCR test has shown that the use of colchicine was associated with statistically significant reductions in the risk of death or hospitalization compared to placebo. In these patients with a proven diagnosis of COVID-19, colchicine reduced hospitalizations by 25%, the need for mechanical ventilation by 50%, and deaths by 44%. This major scientific discovery makes colchicine the world’s first oral drug that could be used to treat non-hospitalized patients with COVID-19.
“Our research shows the efficacy of colchicine treatment in preventing the ‘cytokine storm’ phenomenon and reducing the complications associated with COVID-19,” said Dr. Jean-Claude Tardif, Director of the MHI Research Center, Professor of Medicine at the Université de Montréal and Principal Investigator of the COLCORONA trial. “We are pleased to offer the first oral medication in the world whose use could have a significant impact on public health and potentially prevent COVID-19 complications for millions of patients.”
You might want to see the paper now? There is none. Not even a preprint, just this press release, plus various forms of excited news coverage, especially in Canada. Actually, the drug’s envisioned success was celebrated in the media long before the clinical trial started. As proof of Dr Tardif’s genius, the initial expectations in May 2020 proved to match exactly the now announced final results of the clinical trial:
““Our experience with colchicine suggests that early use of this drug may help dampen inflammation that the body produces in response to the (COVID-19) virus and enable patients to recover without being hospitalized,” said Dr. Binita Shah, an interventional cardiologist and assistant professor in the Department of Medicine, and the study’s principal investigator at NYU Langone.”
One year after the pandemic began, we are still doing science per press releases, and the media and politicians are following unquestioningly.
STAT News brought a critical article though, where sceptical experts unimpressed by press release alone were quoted. There was also this:
“But here’s a caveat: the press release said these results were not statistically significant, although the numbers are close. When the researchers excluded 329 patients who were diagnosed with Covid-19 based on family contacts or clinical symptoms, but who did not have positive PCR tests, there was a 25% reduction in hospitalization, and substantial reductions in the need for mechanical ventilation and deaths.
Outside experts view those results as less trustworthy because the trial did not meet its main goal. They also agreed that the number of patients who needed mechanical ventilation or who died is likely to be small, making it difficult to draw firm conclusions. The press release does not include absolute numbers on the number of people who became hospitalized, needed to be put on ventilators, or died.“
Now, let’s see what other information we can gather if Dr Tardif can’t be arsed with releasing it. Here is that COLCORONA clinical trial, registered as NCT04322682. Its principal investigator is indeed the cardiologist Jean-Claude Tardif of Montreal Heart Institute, who has a Wikipedia page since 2013 because he knew already back then that he will that noteworthy.
The COLCORONA trial was supposed to recruit 6000 patients, in fact it’s still listed as “recruiting”. On 29 November 2020, the Montreal heart Institute issued a press release that they are still recruiting, Tardif was quoted:
“We aim at completing patient recruitment in this large study within the next month and hope that colchicine will reduce the risk of COVID-19 complications.“
But one month later, he had the final results and issued the above press release.
As of the time of writing, the trial’s own website, COLCORONA.net, still invites you on the homepage to participate in the clinical trial, from your home in specific locations in USA, Canada, Spain, South Africa and Brazil (update: when this article went online minutes later, Greece was added, see below). So the recruitment continues then or what, after the trial’s results were publicised?

Only that the results were announced with 4488 participants, of which only 4159 were included in the final analysis. The trial was triple blinded: participants, care providers and Investigators did not know if the patient received colchicine or placebo.
“at least one high-risk criteria”?
It was designed as a telemedicine trial from home: patients are distance-recruited, submit their virus-testing and other clinical results remotely and receive colchicine medicine per mail. Afterwards, “Follow-up phone or video assessments will occur at 15 and 30 days following randomization“.
This is the kind of patients the trial sought to recruit:
- “Patient must possess at least one of the following high-risk criteria: 70 years or more of age, obesity (BMI ≥ 30 kg/m2), diabetes mellitus, uncontrolled hypertension (systolic blood pressure ≥150 mm Hg), known respiratory disease (including asthma or chronic obstructive pulmonary disease), known heart failure, known coronary disease, fever of ≥38.4°C within the last 48 hours, dyspnea at the time of presentation, bicytopenia, pancytopenia, or the combination of high neutrophil count and low lymphocyte count;
- “Female patient is either not of childbearing potential, defined as postmenopausal for at least 1 year or surgically sterile, or is of childbearing potential and practicing at least one method of contraception […]”
Also the May 2020 trial announcement spoke of participants: “They must be 40 years or older, have at least one high-risk criteria“. Yet all of last week’s press releases about COLCORONA’s “Major Breakthrough” mention nothing about those risk-factor inclusion requirements. Only that the participants had to be PCR positive for the coronavirus. Even the recruiting website says you only need to be “40 years old or over“. So what happened with the trial criteria for risk factors like age, diabetes, obesity, cardiovascular issues or asthma? Abandoned mid-trial? Did they at least stick to recruiting people over 39 years old? I asked Dr Tardif on Twitter, but he did not reply.
Recall that STAT News mentioned that some participants were enlisted in the trial because they “were diagnosed with Covid-19 based on family contacts“. This means, entire families participated, which creates heavy confounding issues. First of all, the genetic background for certain illnesses like diabetes, asthma or hypertension can skew trial results when entire families are included. Then, because it was a telemedicine trial from home: are the investigators sure that the family members were not swapping their individually assigned colchicine and placebo pills, especially since they all assumed to have received same drug?
And what about women of childbearing age, supposedly excluded from the trial as per registration? It seems women aged 40 (or maybe even younger?) were not excluded after all, and colchicine is a potent anti-mitotic drug, which safety in pregnancy is everything but sure.
Finally, excluding telemedicine participants with COVID-19 symptoms but negative PCR tests to make your results statistically significant is not really kosher. What if these people got tested positive days later, as it happens often enough with the COVID-19 disease? From press releases and recruitment page, it rather seems the telemedicine trial relied on a single one-time positive coronavirus test.
The Greeks were first!
The Greek media, when reporting about the Montreal results, proudly reminded that Greeks discovered the colchicine COVID-19 cure first and even published it as a peer reviewed paper:
Deftereos et al Effect of Colchicine vs Standard Care on Cardiac and Inflammatory Biomarkers and Clinical Outcomes in Patients Hospitalized With Coronavirus Disease 2019The GRECCO-19 Randomized Clinical Trial JAMA Network Open. (2020) doi: 10.1001/jamanetworkopen.2020.13136
The study, published in June 2020, discovered:
“In this randomized clinical trial of 105 patients, the rate of the primary clinical end point (clinical deterioration) was higher in the control group than in the colchicine group, and the time to clinical deterioration was shorter in the control group than in the colchicine arm. No difference was observed in the primary biochemical end point (high-sensitivity troponin concentration), but patients in the colchicine group had a smaller increase in dimerized plasma fragment D compared with patients in the control group.“
The trial NCT04326790 was supposed to recruit 180 patients and is in fact still listed as “recruiting”. Its registration and the published paper declare regarding masking: “None (Open Label)”, which means the patients knew if they received colchicine or not. The published paper mentions:
“Patient recruitment started on April 3, 2020, and was terminated on April 27, 2020, because of slow enrollment as a result of the rapid flattening of the curve of COVID-19 cases in Greece. Overall, 105 patients (61 [58.1%] men; median [IQR] age, 64 [54-76] years) fulfilled the admission criteria and were included and randomized at 16 clinical sites (Figure 1). Their baseline characteristics are summarized in Table 1; most patients received chloroquine or hydroxychloroquine (103 [98.1%]) and azithromycin (97 [92.4%]).“
So basically they were studying if colchicine can prevent heart complications from the unnecessary chloroquine and azithromycin treatment. Good to know it helps, but that is neither novel nor useful knowledge these days, since Didier Raoult‘s magic drug combo has been debunked by now, and warned against exactly because of its adverse effects on the heart. In fact, that was also exactly the reason AHEPA doctors administered colchicine to their patients in the first place, that was even mandated by the Greek medical authorities already in March 2020, to counteract the side-effects of chloroquine and azithromycin. What is clear, that the results of AHEPA study are heavily confounded and have little to do with COVID-19 itself.
As aside: that clinical trial was coordinated by the AHEPA hospital in Thessaloniki. This is the same heart clinic which previously collaborated with Celixir, the British company owned by Nobelist Martin Evans and his criminally-minded partner, Ajan Reginald. For the clinical trial with Celixir “stem cell” product, AHEPA clinicians were sanctions by the Greek regulator and then destroyed the patient data, as one of them even admitted (read details here). The Greeks were suddenly listed on COLCORONA website as trial site, hours or even minutes after I last checked.

And now colchicine was approved as a standard care COVID-19 drug in Greece, while they simultaneously became official trial site partners of Tardif’s COLCORONA:
“Greek health experts at the committee of the Health Ministry gave the green light for the administration of colchicine, a well-known and cheap cardiological drug, to be included in the protocol of oral treatment in patients with Covid – 19. The approval was reportedly given following the results of a big Canadian study in which also Greece participated.
The Canadian study COLCORONA showed that the administration of colchicine to covid-19 patients “reduced mortality by 44%, hospitalization by 25% hospitalization and the need for mechanical ventilation by 50%” Cardiology Professor and National Coordinator for the Greek part of the study Spyros Defteraios said”
National heath policies via science per press release!
Oh, and since Greece was declared a COLCORONA trial site, does it mean the 105 AHEPA patients, treated in a non-blinded manner with colchicine on top of chloroquine and azithromycin, were included among the ~4500 participants in Tardif’s press release? What kind of trial design is that and what are we supposed to believe now?
Update 26.01.2021. Tardiff was interviewed in Canadian media, which mentioned that “Researchers are preparing to publish the findings in a scientific journal.” Tardiff called upon doctors to prescribe colchicine off-label for non-hospitalised patients right now:
“So any doctor, tomorrow, who reads this can definitely decide to prescribe if he wants.”
Also, Tardiff advocated for colchicine as COVID-19 drug in December 2002, in this BMJ article: Reyes et al 2020. There, he mentioned common adverse side effects of colchicine (which affects dividing cells): diarrhoea and… pneumonia:
“However, in contrast to most available data, one retrospective and one prospective study did report increased pneumonia risk with colchicine“

Tardif forgot to say that one of these irrelevant studies which found a significant increase in pneumonia in colchicine-treated heart patients was his own: Tardif et al NEJM 2019, he was even the trial’s (NCT02551094) principal investigator!
Update 27.01.2021
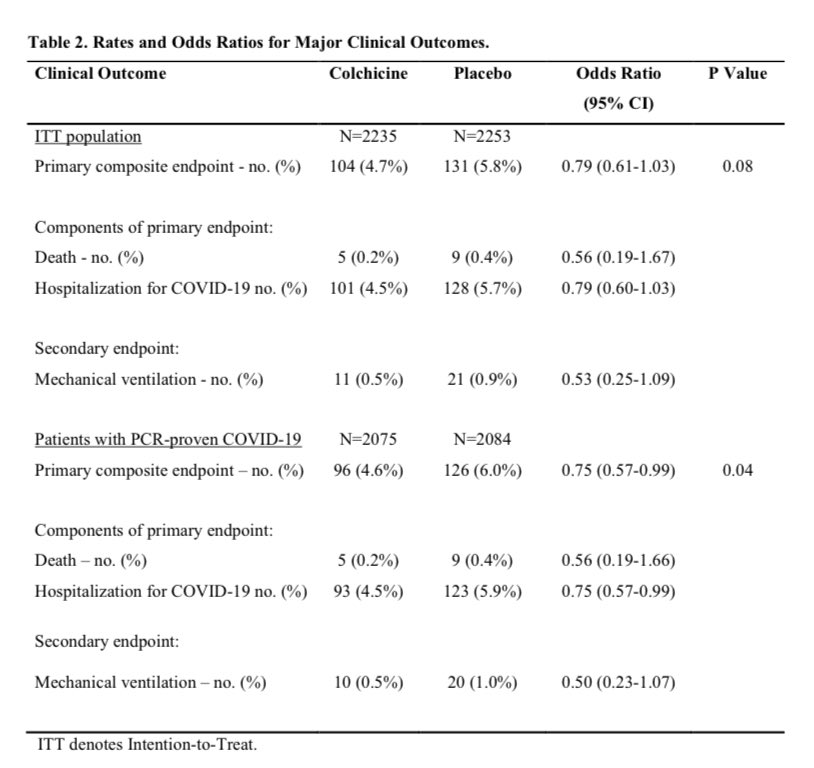
And the manuscript is out! From the preprint Tardif et al medRxiv 2020, abstract:
“A total of 4488 patients were enrolled. The primary endpoint occurred in 4.7% of the patients in the colchicine group and 5.8% of those in the placebo group (odds ratio, 0.79; 95.1% confidence interval (CI), 0.61 to 1.03; P=0.08). Among the 4159 patients with PCR-confirmed COVID-19, the primary endpoint occurred in 4.6% and 6.0% of patients in the colchicine and placebo groups, respectively (odds ratio, 0.75; 95% CI, 0.57 to 0.99; P=0.04).”
p-value of 0.04 for primary endpoint, and this AFTER removing 329 patients as unqualified. Oh dear. Tardif’s earlier pneumonia results (Tardif et al NEJM 2019,) had p=0.03, but now the effect is reversed and colchicine switched from causing to preventing pneumonia (p=0.02).
This is interesting:
“On December 11, 2020, the steering committee chairman informed the data safety monitoring board that the investigators had decided to terminate the study once 75% of the planned patients were recruited and had completed the 30-day follow-up. This decision was made due to logistical issues related to maintaining the central study call center active 24 hours per day for a prolonged period of time, as well as the need to provide healthcare systems with study results in a timely fashion given the state of the COVID-19 pandemic.”
Ah, so this is why the actual results don’t match the “major breakthrough” of the press release.
So here is an alternative from Germany, and you need no prescription for that. Just pop over to your local confectionery store.
Liquorice!
A preprint from the University of Duisburg-Essen in Germany was published:
L. van de Sand, M. Bormann, M. Alt, L. Schipper, C.S. Heilingloh, D. Todt, U. Dittmer, C. Elsner, O. Witzke, A. Krawczyk Glycyrrhizin effectively neutralizes SARS-CoV-2 in vitro by inhibiting the viral main protease bioRxiv (2020) doi: 10.1101/2020.12.18.423104
Its abstract said:
“In the present study, we investigated aqueous licorice root extract for its neutralizing activity against SARS-CoV-2 in vitro, identified the active compound glycyrrhizin and uncovered the respective mechanism of viral neutralization. We demonstrated that glycyrrhizin, the primary active ingredient of the licorice root, potently neutralizes SARS-CoV-2 by inhibiting the viral main protease. Our experiments highlight glycyrrhizin as a potential antiviral compound that should be further investigated for the treatment of COVID-19.”
The study’s lead author Adalbert Krawczyk is a virologist at the University Clinic Essen and he did not reply to my email. So I can’t tell you how he came upon licorice, and his preprint is not telling either, just a brief mention of “traditional herbal medicine substances” without any reference. The only clue I have is that the licorice extract is popular in Traditional Chinese Medicine (TCM) and Ayurveda, in fact TCM concoctions containing licorice, like Yinqiao or Qingfei Paidu, were already touted as promising COVID-19 cures.
The preprint declares the licorice extract to be safe and effective:
“The aqueous licorice root extract showed neutralizing effects even at a subtoxic concentration of 2 mg/ml, (Figure 1A and B). This concentration is lower than the normal consuming dilution e.g. in tea (12.5 mg/ml). Although licorice root tea may represent a good candidate for complementary use, the identification and characterization of the active compound is of great importance for a potential clinical application.”
For glycyrrhizin itself, the authors determined:
No cytotoxic effect could be observed even at a concentration of 4 mg/ml (Figure 1B). […] Complete virus neutralization was achieved at subtoxic concentrations of 0.5 mg/ml under pre- and 1 mg/ml under post-entry conditions (Figure 1A and B). […] The EC50 was calculated with 0.44 mg/ml, uncovering glycyrrhizin as a potent compound effective against SARS-CoV-2 (Figure 1C).“
The university issued a press release which was promptly picked up by the local news throughout the state of Northrhine-Westfalia:
“Do the Essen researchers now recommend consuming more liquorice or liquorice root tea? “You can’t say that generally. You shouldn’t overdo it: The maximum daily dose of glycyrrhizin is 100 milligrams, which corresponds to around 50 grams of liquorice, depending on the variety ”, says PD Dr. Krawczyk. Keeping a sufficient distance, observing the hygiene rules and getting vaccinated if possible offers the best protection based on current knowledge. “Consuming liquorice or liquorice root extract as a tea could possibly has a positive effect on the course of the disease,” says the specialist virologist.”
Licorice root can contain anything between 2% and 25% glycerrhizin and the licorice extract contains 10-20% of that drug (Omar et al 2012). The daily maximum of 100 mg glycyrrhizin at the concentration of 12.5 mg/ml licorice extract in a tea translates into as little as 40-80 millilitres of licorice tea, according to Dr Krawczyk’s own estimates. To attempt achieving anything near 1 mg/ml glycyrrhizin concentration in your blood would leave you dead for sure, especially if you have any cardiovascular problems. But free from COVID-19, so there is that.
It does remind me of how another German scientist announced to save the world from COVID-19 with artemisia extracts. Those also work on the coronavirus in vitro at concentrations which are, shall we say, not physiologically feasible. But never mind, the Max Planck Institute director Peter Seeberger even owns a company selling such teas, clinical trials with which were announced in USA.
Turkmenistan

Now, in Turkmenistan, a Central-Asian post-soviet country ruled since decades by an insane kleptocratic dictator with an absurd personality cult (after the founding dictator Niyazov died in 2006, his personal doctor Berdymukhamedov took over), this research from University of Duisburg-Essen was met with official blessing. As Coda reported:
“At a Cabinet meeting on December 25, President Gurbanguly Berdymukhamedov spoke about the abundant medicinal properties of the [licorice] plant and pointed to one of its main compounds as a possible cure. “Today, scientists note that the glycyrrhizic acid contained in this plant prevents the development of a new coronavirus, which the whole world is fighting against. Moreover, even a small concentration of an aqueous extract of licorice root has a neutralizing effect,” he said, according to the state news agency TDH.”
Officially, Turkmenistan has no COVID-19 at all. Not a single case. In fact, just saying these words, “coronavirus” or “COVID” will get you arrested as per governmental order. In parallel, face masks are mandated to be worn at all times because of “dust”, burial gatherings are forbidden and secret mass graves are made as flat as possible as not to be seen from space. State-controlled media limitedly began extolling the virtues of licorice plant. Coda writes:
“Another piece in Neutral Turkmenistan singled out Berdymukhamedov for praise. “The wisdom and foresight of the head of state, who identified the experience of ancestors and modern technologies as a priority in the development of the domestic medical industry, is confirmed by the research results of not only Turkmen, but also foreign scientists.”
The impact of the president’s recommendation can already be seen in schools across the country. Radio Free Europe’s Turkmen service reported that children have been ordered to include licorice syrup in their mandatory first aid kits, along with masks, disinfectants and rubber gloves, when attending school.”
So much for the University of Duisburg-Essen to be proud of. Another press release maybe, featuring praise from Berdymukhamedov?
But now, dear readers, science has spoken again, what shall we all be taking as COVID-19 preventive? Licorice bonbons? Or colchicine? Or both?
With ivermectin, hydroxychloroquine and Vitamin D in combination? And don’t forget zinc?
Or how about this, some actual shit as COVID-19 medicine:

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). I need to stock up on licorice candy.
€5.00


I will have a laugh if it works.
LikeLike
Which one, colchicine or licorice?
LikeLike
Colchicine, there is a biological plausibility since COVID seems to kill by an excess late immune response (hence Il 6 inhibitors and steroids are helpful.) Colchicine is an immune modulator which does reduce Il 6 amongst other things. If it is beneficial it will not be because of its anti COVID actions but its immune modulatory ones.
LikeLike
They are not planning to use it on ventilated patients at ICU (where dexamethasone was already proven), but on everyone PCR+. Possibly even as preventive.
Colchicine can kill, very painfully. Meaning, use at home brings a great danger of overdose.
LikeLike
Colchicine has a narrow therapeutic index and has known interactions. Almost all deaths are due to overdose or ignorance of the potential interactions. Deciding it is too toxic before trying it would not meet the criteria for “better science”
Dexamethasone is used pre ITU in all patients meeting criteria for admission to hospital so not just on the ventilated and is beneficial even pre ITU.
I agree it will all come down to study design.
I am not advocating this treatment but for the “better science ” principals. Deciding the answer before you have it is as bad as arriving at a conclusion with no or manipulated evidence.
LikeLike
Since you work at NHS, you should know about dangers of drug overdose at home. Colchicine is being pushed for at home use for NON-hospitalised virus-positive patients.
I checked about dexamethasone in UK:
“In England the WHO guidance is likely to apply primarily to patients with COVID-19 who are hospitalised and receiving supplemental oxygen.”
Click to access C0870_COVID-19-Therapy-Corticosteroids-including-dexamethasone-and-hydrocortisone-letter_131120.pdf
So you say everyone is receiving it who enters the hospital as COVID+?
LikeLike
Sorry not clear, I meant everyone who is admitted with COVID infection being the reason for admission. Those are the ones who get steroids. We had been giving prednisolone because it is easier (and no less effective) but we are back to dex. because there is a national shortage of pred. Admission is almost always for supplemental oxygen otherwise they are at home.
I do agree that giving drugs to everyone with just a COVID positive test is not a good idea since most will get better and so any beneficial effect of your intervention will be harder to see and side effects will be more of an issue.
LikeLike
Exactly, you don’t send people home with a bag of steroid hormones to take ad libitum.
Now, have a look at the update I inserted in the text above.
Interesting paper, pneumonia proved associated with colchicine. Guess who the author is.
https://www.nejm.org/doi/full/10.1056/NEJMoa1912388
LikeLike
Hi Adrian,
did you see the preprint? Impressed with the data?
LikeLike
The Colchicine paper is difficult to judge without the data. There is a small effect in a small subgroup group.
LikeLike
I think this is the begin of a wonderful friendship.
LikeLike
Colchicine: Too toxic to use in cancer chemotherapy. What more can one say?
LikeLike
You are a Big Licorice shill.
LikeLiked by 1 person
One can say quite a lot actually. There are obvious reasons why you should not mix 2 immunosuppressive therapies. I’m not sure it has been explored much because it has seemed so obvious that why would one try?
There are in vitro studies and it was used in the 1950’s but not in real patients.
LikeLike
French version here, by Alexander Samuel:
https://rechercheindependante.blogspot.com/2021/01/colchicine-ou-reglisse.html
LikeLike
Small effect in small subgroup and not able to assess study on preprint. And the outcome of admission to hospital may not translate to survival
LikeLike
Pingback: France’s Ugly Brown Derriere – For Better Science
Pingback: Why not chemical castration (to escape COVID-19)? – For Better Science
Pingback: Die with a smile: antidepressants against COVID-19 – For Better Science
LikeLike
Dr Krawczyk is back, this time with curcumin, to prove every Ayurvedic quack right.
Maren Bormann , Mira Alt , Leonie Schipper , Lukas Van De Sand , Vu Thuy Khanh Le-Trilling , Lydia Rink , Natalie Heinen , Rabea Julia Madel , Mona Otte , Korbinian Wuensch , Christiane Silke Heilingloh , Thorsten Mueller , Ulf Dittmer , Carina Elsner , Stephanie Pfaender , Mirko Trilling , Oliver Witzke , Adalbert Krawczyk Turmeric Root and Its Bioactive Ingredient Curcumin Effectively Neutralize SARS-CoV-2 In Vitro Viruses (2021) doi: 10.3390/v13101914
The authors mention curcumin works against SARS-CoV2 even better than chloroquine! News Medical explains:
It is difficult to reach this concentration of curcumin orally, what with the safety profile the authors themselves declare: “European Food Safety Authority (EFSA) panel concluded that evidence supports an acceptable daily intake (ADI) of 3 mg/kg bodyweight per day for curcumin“. But curcumin can be injected into bloodstream, and it sure will prevent COVID-19 with 100% efficiency, by killing you.
LikeLike