Following is the new investigative study by the Liverpool biologist and professor for stem cells and regenerative medicine, Patricia Murray. It is again about the Welsh company Celixir, founded by the Nobel Prize winner and former Cardiff University chancellor Sir Martin Evans and his partner, the struck-off dentists and pathological liar, Ajan Reginald.
In brief: Celixir sells magic iMP cells which they inject into patients’ hearts. It seems those iMP cells are nothing other than blood from some other heart attack patients, but the company denies what they themselves admitted elsewhere. The cells were first tested in clinical trial in Greece (published in 2016, with all patient data “transferred to the trash“), after the trial apparently proved illegal and brought Celixir in serious trouble with Greek authorities. But not in UK. The more Celixir lies, the more determined the British Medicines and Healthcare products Regulatory Agency (MHRA) gets for Celixir to experiment on 50 British patients in London.
I am not making this up: as internal emails revealed, MHRA was initially inclined to stop the clinical trial and to investigate Celixir, but as Murray exposed more lies and fraud, MHRA threw its full weight behind Celixir’s business interests and declared Murray a “nuisance” who expects MHRA to do what they think is her job.
20.10.2020 Please see update at the very end about a very relevant Expression of Concern issued by Springer Nature for Anastasiadis et al 2016.
This is how Brexit Britain, finally free of EU shackles, will now operate. With European Medicines Agency (EMA) oppressive oversight overthrown and EMA itself chased out of London, MHRA is free to sell the entire British population for medical experiments. I predicted this already in 2016, and now we have first solid evidence.

Ajan Reginald is a very vain man, who likes to play victim while physically incapable of honesty. Patricia Murray even made this unofficial Ajan Reginald and Celixir biography.
Look at what he writes about himself in third person:
“Ajan, who had to retire from practicing dentistry following a horrific sports injury that left his eyesight permanently damaged, who has since dedicated himself to pioneering stem cell research that has gone on to save countless lives, and which will continue to save lives, is the sole target of these vicious attacks.“
And yet, Sir Martin fully trusts Ajan. As a reliable source who spoke to Evans in October 2019 mentioned, the Nobelist brushes off most of the allegations. Sir Martin maintains his confidence in Ajan, but has even advised him not to parade himself as a doctor (which the struck-off dentist repeatedly did nevertheless). In any case, according to Evans the phase 2b clinical trial in London was about to begin next week, i.e. in late October 2019.
It seems the Nobelist Sir Martin Evans is not a bumbling old genius loon, befuddled by Ajan’s incessant stream of lies and flatteries and clueless of what those magic iMP cells are and what plans his Celixir has with them. Stephen Westaby, retired celebrity heart surgeon and Celixir shareholder, was the man behind Celixir’s experiments in Greece, all done without any control arm. Westaby explained:
“The work with advanced heart failure in Greece was undertaken by some very bright surgical trainees of mine who had returned home. We began by using left ventricular assist devices as an alternative to heart transplantation. They then developed an interest in the use of autologous bone marrow derived stem cells intending to generate new microvasculature in areas of ischemia not amenable to satisfactory coronary bypass. We waited hours for the autologous cells to be processed with the patients under anaesthetic which was not ideal. The results as for most stem cell trials were less than impressive. In the meantime the LVAD patients did very well.
I then met Prof Sir Martin Evans in the UK who was working with modified allogenic mesenchymal stem cells that now have a specific categorisation I believe – immunomodulatory progenitor cells that we non immunogenic . It meant that we could use them without the inordinate delay and my Greek colleagues were keen to have that opportunity. This must be the trial in coronary bypass patients that you refer to. I don’t have any direct knowledge about the negotiations with the regulatory authorities in Greece. However what we found was unexpected and quite remarkable. The patients seemed to have significant reduction in volume of scar within the myocardium on thallium scanning that needs to be confirmed in a larger trial using MRI in the UK. The prognostic importance of fibrosis and scar removal in heart failure prevention cannot be underestimated.“
Westaby added:
“Martin Evans would be the person to ask about the cells and the Greek authors about regulatory issues.“
Later on, Westaby was less sure about the iMP cell magic he witnessed:
“As a busy cardiac surgeon I was not party to any of the scientific or regulatory background . Obviously I was interested in the opportunity to work with a Nobel Prize winning scientist and my colleagues in Greece were already using autologous BM cells . I introduced the two in good faith.
A number of people have reviewed the scans in Europe and the US and believe the scar removal effect to be real . Certainly the patients who could not be completely revascularised are remarkably well several years later (I went back to interview them for the company) and I was impressed by their exercise tolerance. They clearly suffered no adverse effects but it was clear to me that a prospective randomised trial should be performed using MRI imaging. My colleagues in Greece are in a similar position- motivated to improve the outcome for heart failure patients with new approaches- but we have to accept what we are told about the science to a certain extent.“
Westaby is still Celixir’s board member, as Chief Medical officer. While Evans, Ajan and the allegedly unbiased and patient-oriented UK authority MHRA can’t wait to see 50 British patients experimented upon. With sod knows which cells.
Update on the UK company Celixir and the MHRA’s response to my concerns
By Patricia Murray
I have already reported some of Celixir’s dishonest and fraudulent activities to the Medicines and Healthcare products Regulatory Agency (MHRA) and Health Research Authority (HRA), and have made my concerns publicly available (here and here). Through FOI requests, I have now obtained more evidence of inappropriate behaviour, which I have included in the current report. Most notably, it appears that Celixir was involved in a clinical trial in Greece that was conducted without regulatory approval having first been obtained. Further problems are highlighted below.
Background
Celixir are planning to undertake a Phase IIb clinical trial at the Royal Brompton Hospital, UK. The trial will recruit 50 patients, 30 of whom will have Celixir’s allogeneic bone marrow-derived ‘immunomodulatory progenitor cells’ (‘iMPs’, aka ‘HeartCel’) injected into their heart muscle during coronary artery by‐pass grafting (CABG). This Phase IIb trial is following on from an 11 patient Phase IIa safety trial that took place in the AHEPA Hospital in Greece in 2012‐2013, the results of which were published in Anastasiadis et al 2016.

I became concerned about Celixir after discovering that the CEO, Ajan Reginald, had a track record of fraudulent and dishonest behaviour. As indicated by excerpts from the following news article, Reginald is a struck-off dentist whose previous activities should have raised concerns about his suitability for developing experimental therapies:
- “…the GDC [General Dental Council -PM] said the dentist had to be struck off to protect the public”.
- “He also used 20 of his patients to fleece the Dental Practice Board out of cash for work that was not done.”
- “There was substantial evidence of a continuing pattern of neglect which covered a wide range of failings resulting in a real risk to patients, including children”.
The above article was published in 2005, but it appears that Reginald did not turn over a new leaf after this; since this date he has engaged in a catalogue of questionable activities, which include falsely claiming to be a medical doctor, falsely claiming to have a PhD, plagiarising and misrepresenting research data(here and here).
Celixir’s 2012-2013 Greek trial was allegedly a success, and in 2016, the company set up a GMP (Good Manufacturing Practice) facility in Greece near to the AHEPA Hospital. It was therefore surprising that instead of undertaking their Phase IIb trial at the AHEPA Hospital with surgeons experienced in the use of ‘iMPs’, Celixir instead opted to undertake the Phase IIb trial in the UK. One of the more serious issues I have now discovered is that the trial in Greece was undertaken without the necessary regulatory approvals having first been obtained.
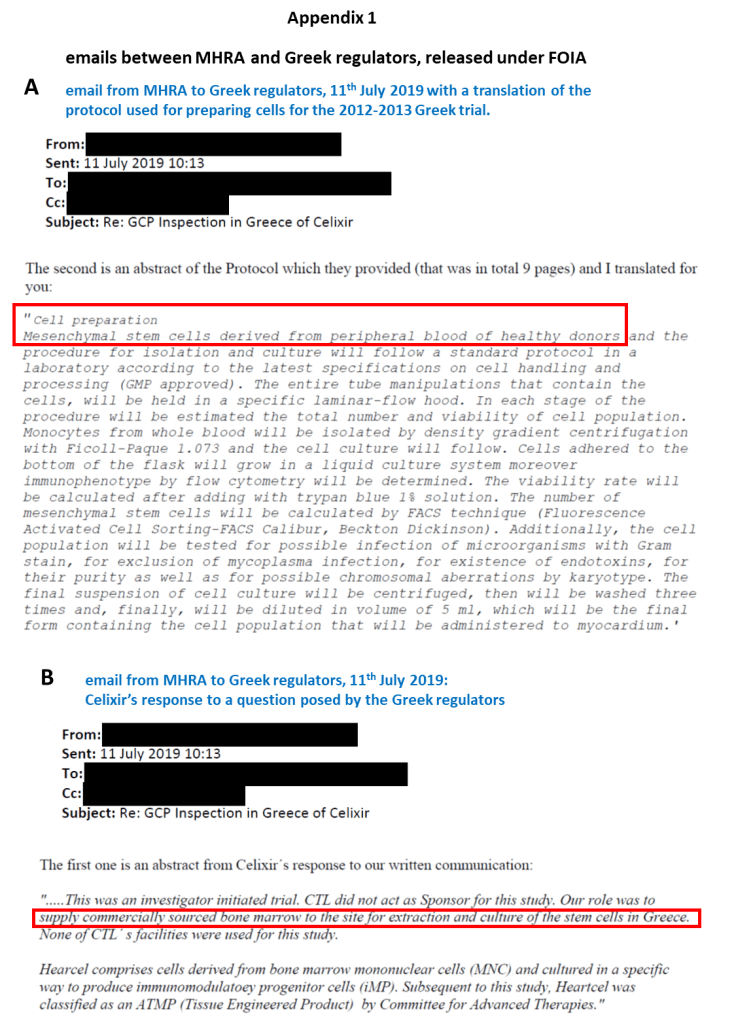
Following a complaint to the European Medicine’s Agency (EMA) in 2017 (by an individual unknown to me), an investigation revealed that Celixir had falsely claimed to have used bone marrow-derived ‘iMPs’ in the 2012-2013 Greek trial.
The cells used were in fact donor-derived peripheral blood-mesenchymal stromal cells (PB-MSCs) (Appendix 1). At the time the trial took place, the only PB-MSCs that Celixir had access to were cells that had been procured from the peripheral blood of patients in the Morriston Hospital in Wales[Pieper et al 2017].
Celixir did not have approval to use these cells as therapies in other patients. The Greek regulators imposed penalties on Celixir, the AHEPA hospital, and the trial’s principal investigator. The fact that penalties were imposed raises the possibility that Celixir may have decided to undertake their Phase IIb trial in the UK because they are not welcome to do so in Greece.
Despite these issues, the MHRA informed me in Dec 2019 that they were happy for the Brompton trial to proceed. To understand why, I submitted an FOI request to the MHRA for internal emails concerning my complaints about Celixir. These emails showed that initially, some MHRA staff appeared to take my concerns seriously (see timeline below). Surprisingly, however, after MHRA discovered what had happened in Greece, instead of withdrawing approval for the Brompton trial, this information appeared to have the opposite effect: i.e., following these revelations, MHRA decided that Celixir had no case to answer and the Brompton trial should proceed. This was despite there being irrefutable evidence that Celixir had provided false information to regulators, the Health Research Authority, and in their Anastasiadis et al 2016 publication.
The MHRA were even happy to condone Celixir retrospectively changing the description of the cell type used in the Greek trial on the Clinical Trials.gov registry, despite this being an attempt to conceal fraudulent behaviour. MHRA’s actions in the Celixir case appear to be inconsistent with their mission to “enhance and safeguard the health of the public by ensuring that medicines and medical devices work, and are acceptably safe”.

Timeline showing my interactions with the MHRA and how they have responded to my concerns
1. 21st March 2019: My initial complaint about Celixir to MHRA
I contacted MHRA to express concerns that Celixir (aka Cell Therapy Ltd) had obtained MHRA approval to conduct a clinical trial at the Royal Brompton Hospital. In brief, Celixir’s trial will recruit 50 patients, 30 of whom will have allogeneic (donor-derived) ‘immunomodulatory progenitor cells’ (‘iMPs’, aka ‘HeartCel’) injected into their heart muscle during coronary artery by‐pass grafting (CABG). This Phase IIb trial is following on from an 11 patient Phase IIa safety trial that took place in Greece in 2012‐13, the results of which were published in 2016 in the ‘Journal of Cardiovascular and Translational Research’ (a ‘Springer’ journal).
My main concerns were as follows:
- The CEO of the company, Ajan Reginald, had a track record of fraudulent behaviour.
- The entry on Clinical Trials.gov (NCT01753440) for the 2012-13 Greek trial indicated that mesenchymal stromal cells were used, whereas the 2016 publication indicated that bone marrow-derived ‘iMPs’ were used3.
It thus appeared that Celixir’s ‘iMPs’ had not been used in the previous trial and that the 2016 publication contained false information.

2. 1st April 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
The Acting GCP Operations Manager & Lead Senior GCP Inspector of MHRA’s Inspections, Enforcement and Standards writes [highlights here and elsewhere by PM, -LS]:
“We received an email from HRA [Health Research Authority, –PM] with concerns regarding a trial that has received approval in the UK and Celixir. We previous (sic) received a complaint about this and will look for the details, but we couldn’t take any action previously as even although I believe that although the sponsor was based in the UK the trial was only being conducted in Greece. It now appears they are about to start a trial here, and the concerns seem to indicated (sic) this is worth a further review before we put any UK patients at risk.”
This email indicates that MHRA had previously received a complaint about Celixir’s conduct in the Greek trial but could not take action because the trial was being undertaken in Greece. The individual who made the complaint to the MHRA is unknown to me.

3. 2nd April 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
An MHRA employee writes:
“To me, the issue appears to be regarding the potential for fraudulent activity. Providing sufficient hard evidence is available, this would warrant an investigation and the suspension/withdrawal of the trial whilst the investigation is ongoing.”
The MHRA’s Medical Assessor writes:
“If the information we were provided [by Celixir, -PM] is incorrect then an investigation is warranted.”
The view of these MHRA employees was that if Celixir had provided false information, the trial should be suspended or withdrawn pending an investigation.
4. 7th April 2019: My second complaint about Celixir to MHRA
I contacted MHRA to inform them that Celixir had plagiarised and misrepresented research data in a patent they had filed in 2011 for a cell type they called ‘progenitor cells of mesodermal lineage’ (‘PMLs’). In brief, Celixir had inserted data in their patent from an unrelated US group(Granero-Molto et al 2009) showing that MSCs could home to injured bone in mice. In addition to plagiarising the data, Celixir renamed the MSCs as PMLs(patent PCT/GB20 12/05 1600). This was significant because the Investigational Medicinal Product Dossier (IMPD) that Celixir had submitted to MHRA to obtain Clinical Trial Authorisation (CTA) indicated that the ‘iMPs’ were generated from ‘PMLs’.
The fact that Celixir had deliberately misrepresented MSCs as ‘iMPs’ in their 2016 publication, and as ‘PMLs’ in their patent, indicated a pattern of fraudulent behaviour.

5. 8th April 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
The Deputy Unit Manager / Senior Medical Assessor of the Clinical Trials Unit reviewed my concerns and drafted a report that included the following statements:
“However, the authors do include a comprehensive results table of the cell surface markers, showing a comparison between iMPs and MSCs. It is clear from the paper [the 2016 publication –PM] that their intention was to culture iMPs rather than MSCs, so we’ll assume that the profile of surface markers provided for the iMPs is relevant to the product used.”
Here, the Deputy Unit Manager (DUM) is saying that s/he assessed the 2016 publication and noted a table containing flow cytometry data allegedly generated from ‘iMPs’. However, these data were not generated from bone marrow-derived ‘iMPs’. The data were generated from peripheral blood-derived MSCs (PB-MSCs, see Celixir letter) and deliberately misrepresented as bone marrow-derived iMPs in the 2016 publication.
The DUM made the following conclusions:
“It appears it is possible that the cells may have changed to those used in the phase 2 trial in Greece, and that Heartcel product in the current trial is not the same as that used previously. However, this is only based on the entry for the phase 2 trial in Greece on Clinicaltrials.gov, as stated in the trial paper from 2016. This may warrant further investigation for clarification, although if the Clinicaltrials.gov is a more accurate reflection of the trial then this implies the paper is significantly flawed and fraudulent.”
The DUM acknowledges in this initial review that if the 2016 publication did not accurately reflect the 2012-2013 trial it would be significantly flawed and fraudulent. Although the DUM might not have known it at this time, further evidence would confirm that the paper was indeed significantly flawed and fraudulent; i.e., (i) data presented as being generated from bone marrow-derived ‘iMPs’, were actually from PB-MSCs; (ii) the study protocol for the 2012-2013 Greek trial that was provided to the MHRA by the Greek regulators, indicated that the cells used were PB-MSCs (Appendix 1).

6. 25th April: My third complaint about Celixir to MHRA
I provided MHRA with a report I had compiled on Celixir. Amongst other things, the report mentioned that although Celixir claimed in their 2016 paper that the cells used in the 2012-2013 Greek trial were procured under Good Manufacturing Practice (GMP) conditions, Celixir did not have access to any GMP facility in the UK. When challenged, Celixir stated that the cells were prepared in an “accredited Greek laboratory”. However, there is no evidence for this. If a Greek GMP unit had been used, information regarding its name and location should have been included, along with the name of the authorised person in charge of the facility who oversaw the preparation of the cells.
I also highlighted in my cover email that the Greek surgeons who led the 2012-2013 trial had undertaken a very similar trial in 2009-20119, the only difference being that the cells used in the earlier trial were autologous bone marrow-derived cells. I thought it odd that the paper Antonitsis et al 2012 reporting on the 2009-2011 trial was not referred to in the 2016 publication, and that it had not been registered on Clinical Trials.gov. I expressed my concern to the MHRA that patient data from the earlier trial might have been re-used in the 2016 publication.
It was noteworthy that cardiac images (thallium scans) of only one patient had been included in the 2016 publication. I later contacted the radiologist, Prof Doumas, a co-author on the 2016 publication, to request the images from all 11 patients. He told me that he had been retired for 2 years and that the data had been “transferred to the trash” (Appendix 2). I found it very worrying that these crucial data had apparently been destroyed. Had the raw date-stamped images been available, it would have been possible to confirm that 11 patients had been recruited to the second trial.

7. 9th May 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
The Deputy Unit Manager / Senior Medical Assessor of the Clinical Trials Unit writes that she has received the following information from the Greek regulators:
“Additionally, kindly be informed that following a complaint submitted to the EMA [European Medicines Agency –PM], our Inspectorate investigated this issue in 2017. As a result of this investigation, our Management Board imposed penalties to the manufacturer [Celixir], the Institution/site and Principal Investigator, as foreseen by national law.”
The Deputy Unit Manager (DUM) comments:
“I don’t think this changes the CTU decision though, that even if the previous data was with a different product, we would still have approved the current UK trial. This is certainly a case for doing an inspection though, and sooner rather than later if possible”.
The Deputy Unit Manager (DUM) now says that even if different cells were used in Greece, MHRA would still have approved the UK trial. This seems to contradict the conclusions of his/her 7th April review, because if the cells used in Greece were different, this would mean that Celixir had presented false information in their 2016 publication, which according to the DUM, would make it “significantly flawed and fraudulent” (see section 5). It is noteworthy that the DUM displays no curiosity about the nature of the penalties that the Greek regulators imposed.
8. 11th July 2019: From emails between MHRA and Greek regulators obtained on Dec 9th 2019 under FOI
The Greek regulators informed MHRA that during their investigation, Celixir were asked some questions. Celixir’s response to one of these questions was picked out by the Greek regulators, presumably because they felt it should be of interest to the MHRA:
“CTL [Cell Therapy Ltd, aka Celixir -PM] did not act as Sponsor for this study. Our role [Celixir’s role –PM] was to supply commercially sourced bone marrow to the site for extraction and culture of the stem cells in Greece. None of CTL΄s facilities were used for this study.”
Celixir’s response contradicts the information provided in the 2016 publication; i.e., the paper stated that the cells had been isolated in the UK and were derived from the bone marrow of a healthy donor. The 2016 publication even contains a detailed description of how the bone marrow was aspirated from the donor, and how the cells were then harvested from the bone marrow. Yet here, Celixir is now claiming that the bone marrow was sourced from a commercial supplier. It is noteworthy that they do not name the supplier.
The Greek regulators also informed the MHRA that they had obtained a copy of the informed consent form (ICF) used in the 2012-2013 Greek trial along with the protocol describing the cells and how they were prepared. These documents were written in Greek and had presumably been provided to the Greek regulators by the AHEPA hospital during the investigation. The Greek regulators kindly translated the part of the protocol that they felt should be of interest to the MHRA (Appendix 1). This included the following sentences:
“Cell preparation: Mesenchymal stem cells derived from peripheral blood of healthy donors and the procedure for isolation and culture will follow a standard protocol in a laboratory according to the latest specifications on cell handling and processing (GMP approved).”
The protocol makes it clear that the cells used in the trial were PB-MSCs, not bone marrow-derived ‘iMPs’ as falsely claimed in Celixir’s 2016 publication. This information confirms that the 2016 publication did not accurately reflect the 2012-13 Greek trial, which according to MHRA’s Deputy Unit Manager, meant that the publication was “significantly flawed and fraudulent”(see section 5).

9. 16th July 2019: My fourth complaint about Celixir to MHRA
I highlighted my concern that the cells used in the 2012-2013 Greek trial may have been obtained from the peripheral blood of patients in the Morriston Hospital in Swansea. At this time, I was not aware of the information from the Greek regulators confirming that peripheral blood-derived cells had indeed been used, but my suspicion was aroused by documents I had obtained under FOI from the Research Ethics Committee (REC) in Wales. These documents revealed that Celixir were requesting permission to use the cells they obtained from the Morriston patients as therapies in other patients. The REC did not give approval for this.
10. 29th July 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
The Deputy Unit Manager / Senior Medical Assessor of the Clinical Trials Unit writes:
“A further review is top of my list this week, and this additional info could be helpful, although a lot seems out our remit (the accuracy of the paper for example). We have managed to get some data from Greece, but its minimal as it didn’t go to the Greek regulator for approval, so I can’t get an IMPD.”
I found it difficult to understand why the Deputy Unit Manager (DUM) now says the accuracy of the 2016 publication is outside MHRA’s remit, when the publication was providing supporting data for the 2016 trial. Moreover, the DUM had analysed the paper in his/her initial review, so this statement appeared contradictory. Even more difficult to understand was that the DUM mentions that the data from Greece was minimal. The information from Greece was highly significant because it confirmed that Celixir’s 2016 publication was significantly flawed and fraudulent, and that they had provided false information in the CTA application submitted to MHRA.
For instance, Celixir’s Investigator’s Brochure that was submitted to the MHRA stated that ‘iMPs’ are prepared from bone marrow, and that they had previously been used in the 2012-2013 Greek trial; this was untrue because PB-MSCs were used in the 2012-2013 trial.
The DUM does not seem troubled by the fact that the Greek trial had been undertaken without the necessary approvals having first been obtained.

11. 5th August 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
The Deputy Unit Manager / Senior Medical Assessor of the Clinical Trials Unit writes:
“I have done a full review of all the information available to MHRA (including information from regulator and GCP in Greece) and concluded that no regulatory action is required.”
The Deputy Unit Manager (DUM) goes on to say:
“The Royal Brompton trial is not on hold from MHRA perspective and the halt has been lifted by HRA for Ethics.”
It is not clear why the DUM informed his/her colleagues that the HRA had lifted their halt on the trial because this information was not correct. HRA had not lifted their halt.
The DUM’s review dated 1st August (referred to in the above email), which was made available under FOI, lists the following conclusions:
“• The 2009 trial (2012 Antonitsis paper) used autologous BMSCs [bone marrow-derived stem cells –PM] and there is no evidence the data was carried forward and mixed with new data to fraudulently present new data in the 2016 paper”
Unfortunately, this assessment is based on the assumption that the information presented in the 2016 paper is true and accurate. Given that key information in the paper is now known to be false, a question is raised as to whether any of the information in the paper is trustworthy. An experienced cardiologist who has reviewed the data has highlighted the following problematic issues concerning the functional heart data.

Opinion of an experienced cardiologist on Celixir’s 2016 publication
“The left ventricular ejection fraction (LVEF) is important for assessing cardiac function. It can be calculated using the following calculation: LVEF (%) = left ventricular end diastolic volume (LVEDV) – left ventricular end systolic volume (LVESV) x 100/ LVEDV. If one calculates the baseline LVEF from the LVEDV (208.9) and the LVESV (106) given in Table 2 of the 2016 publication, we get an LVEF of 49.3% [i.e., (208.9-106) x100/208.9]. It is therefore difficult to understand why the value for LVEF in Table 2 is given as 37.2%, as such a disparity could not be explained by rounding errors.
Turning to Figure 2, we see that baseline LVEF is presented in what appears to be half a box-and-whisker plot, showing a median LVEF of 40% for the trial participants. If the median LVEF was 40%, this would mean that a proportion (i.e., half) of the participants must have had an LVEF that was over 40%, which would be more consistent with the calculated average above of 49.3%. This is curious, because to be included in the trial, participants should have had an LVEF that was below or equal to 40%.
Figure 2 suggests that LVEF improves from baseline to 1 month post-operatively, and then stays relatively stable. However in Figure 3, one sees that the LVEDV was decreased compared with baseline at 1, 3, 6 and 12 months and LVESV changed little. So from these data, one would expect that LVEF would have decreased compared with baseline at each time point, and not increased as claimed. One would expect that LVEF would improve following coronary artery bypass grafting, so if LVEF decreased, this might suggest that the cell injection had a detrimental effect. The authors gave 3 doses of iMPs, but made no comment about a dose-response effect.“
The only way to confirm there was no patient overlap between the 2009-11 and 2012-13 trials would be to inspect the raw cardiac images (thallium scans), as these would be date-stamped. However, the radiologist has indicated that these are now “in the trash” so it would not be possible to do that (Appendix 2). The fact that these crucial data were destroyed raises further question about the conduct of the 2012-2013 trial.
The DUM further stated:
“The 2012 trial (2016 Anastasiadis paper) has some detail lacking to allow a full comparison, but there is no evidence the cells used in the trial are different to those being used in the current Royal Brompton trial”
It is difficult to understand how the DUM came to this conclusion. The authors themselves now admit that the cytometry data presented in the 2016 paper were derived from PB-MSCs, rather than from bone marrow-derived ‘iMPs’. Moreover, the information from the Greek regulators indicates that the cells used in the 2012-13 trial were PB-MSCs, not bone marrow-derived ‘iMPs’ (Appendix 1) as falsely claimed in the 2016 paper. The cells that Celixir plan to use in the Brompton trial will be derived from bone marrow. Hence, there is clear evidence that the cells used in the Greek 2012-13 trial are different from those in the Brompton trial.
“Whether iMPs and MSCs are truly unique cannot be clarified….”
This is a valid point because the protocol for generating ‘iMPs’ and indeed ‘PMLs’ in Celixir’s patents is just a standard protocol for generating MSCs. This raises the question of why Celixir insist on calling the cells ‘iMPs’ and/or ‘PMLs’? Could it be that by positioning them as novel cell types they hope to make substantial profits, which would not be possible if they were simply called MSCs? A presentation by Celixir reveals that they plan to charge >£72K for each patient treated with ‘iMPs’.

However, even if ‘iMPs’, ‘PMLs, and MSCs can be regarded as one and the same, this only applies if they are derived from the same source. Of note, the cells used in the 2012-13 Greek trial were generated from peripheral blood-derived mononuclear cells, whereas those in the Brompton trial will be from bone marrow-derived mononuclear cells. Mononuclear cells from blood and bone marrow are not the same(Capiod et al 2009).
“The MHRA has no reason to suspect fraudulent activity related to the submitted data to support approval of the CTA”
Here, the DUM appears to be contradicting his/her previous review from April, where she stated: “if the Clinicaltrials.gov is a more accurate reflection of the trial then this implies the paper is significantly flawed and fraudulent”(section 5). The paper was meant to be providing evidence of safety for the Brompton trial. As indicated above, there is clear evidence that the original entry on Clinical Trials.gov was a more accurate reflection of the trial and that the paper is indeed significantly flawed and fraudulent. It is therefore difficult to understand why the DUM states that the MHRA now has no reason to suspect fraudulent activity.
“The clinical trials.gov entry for the second Greek trial has been updated and now reflects the cells in a more consistent manner.”
What the DUM refers to as ‘updating’ the Clinical Trials.gov entry for the 2012-2013 trial, actually involved retrospectively changing the description of the cell type that was used. It is difficult to understand why MHRA decided to condone Celixir’s actions here, rather than viewing this unethical and dishonest behaviour as unacceptable.
12. 16th Sept 2019: My fifth complaint about Celixir to MHRA
I provided MHRA with Celixir’s rebuttal to my initial report and my response to their rebuttal. I highlighted that Celixir’s claim that they had (i) developed a protocol for isolating ‘iMPs’ from bone marrow, (i) characterised the cells, (iii) established GMP conditions, and (iv) performed preclinical tests within 12 months was implausible. At this time, I had not yet obtained the emails between the MHRA and the Greek regulators, so was unaware that this claim of Celixir’s would later be proven to be false (i.e., it later being confirmed that the cells used in the 2012-2013 Greek trial were derived from peripheral blood, not from bone marrow).
Celixir also indicated in their rebuttal that despite them having openly engaged in serious research misconduct (including plagiarism and data misrepresentation), the MHRA were happy for them to proceed with the Brompton trial. I asked MHRA if this was correct.
13. 18th Sept 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
It appears here that the Deputy Unit Manager has prepared a draft response to send to me and is asking his/her MHRA colleagues for comments. One colleague states the following:
“Is there a danger that the statement ‐ We are unable to comment on accuracy of data in publications or patents, nor the nature of the cells used in prior clinical trials, including the timeline for the products development -undermines any of the data supporting the application ?”
The statement is indeed problematic as it suggests that the MHRA now have no issue with the fact that (i) Celixir’s claims regarding their ‘PMLs’ and ‘iMP’ are based on plagiarised and misrepresented data; (ii) Celixir knowingly misrepresented the cells used in the 2012-2013 Greek trial as being derived from bone marrow when they were in fact derived from peripheral blood; (iii) Celixir could not possibly have undertaken the necessary preclinical tests before administering the cells to participants in the 2012-2013 Greek trial; (iv) Celixir appear to have sourced their peripheral blood samples from patients in the Morriston Hospital without approval from the Research Ethics Committee; (v) Celixir do not appear to have prepared the cells for the Greek trial in a GMP facility, thereby exposing patients to serious risk.
This colleague goes on to ask a very pertinent question:
“…is it possible to state why the allegation that the data that [Patricia Murray -PM] alleges to be falsified would not raise a significant concern to the trial approval?”
The Deputy Unit Manager does not provide an answer to this very important question.
14. 12th Nov 2019: My sixth complaint about Celixir to MHRA
I drew MHRA’s attention to the fact that Celixir had provided false information to the European Medicine’s Agency (EMA). Specifically, Celixir had informed the EMA that all 11 patients in the 2012-2013 Greek trial were New York Heart Association (NYHA) Class IV, when only 4 were NYHA Class IV; 2 were NYHA Class III; and 5 were NYHA Class II. This was important because by indicating the patients were all NYHA Class IV, Celixir were falsely claiming that they were more seriously ill than they actually were. In the same document submitted to the EMA, Celixir had also falsely presented surface marker analysis of PB-MSCs obtained from patients in the Morriston Hospital as bone marrow‐derived ‘iMPs’. I asked MHRA if they had a view on whether there should be an absolute bar on Sponsors that provide false information to regulators. I also requested the correspondence between the MHRA and Greek regulators concerning Celixir under FOI.
15. 12th Nov 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
MHRA’s Director of Licensing Division states:
“I wonder given the number of repeated requests from this enquirer, on the same issues, whether it is time for us to decline answering anymore (unless there are new issues raised) and treat this as a nuisance request?”
It was disappointing to see that MHRA’s Director of Licensing Division viewed my valid concerns as a nuisance and suggested that MHRA should decline responding to me.
16. 13th Nov 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
MHRA colleagues discuss a report that has been drafted by the HRA concerning their investigation into Celixir. The HRA report contains the following statement by the Greek regulators:
“Additionally, kindly be informed that following a complaint submitted to the EMA, our Inspectorate investigated this issue in 2017. As a result of this investigation, our Management Board imposed penalties to the manufacturer, the Institution/site and Principal Investigator, as foreseen by national law.”
The MHRA colleagues find this statement problematic and indicate that they will ask the HRA not to publish their report.
The Deputy Unit Manager (DUM) goes on to ask his/her colleague the following:
“Do you have details of what these penalties were?”
I found it quite remarkable that despite being informed 6 months previously that penalties had been imposed on Celixir by the Greek regulators (see section 7), MHRA did not think it important to ask what these penalties were.

In my first report on Celixir, I posed the following question “why did Celixir set up a GMP facility in Greece in 2017 to prepare cells for a UK trial?” Given that the first trial at the AHEPA Hospital had allegedly gone so well and the GMP facility was close by, it would have seemed sensible to conduct the subsequent trial in Greece. The reason for Celixir wanting to undertake the trial in the UK might have been because of the penalties imposed on them in Greece. Are the MHRA comfortable with the fact that companies who may not meet the standards of honesty and integrity required to undertake clinical trials in Greece are welcome to do so in the UK?
It appears that the MHRA is not acting in the best interests of UK patients, but rather prefers to facilitate the activities of a dishonest, unethical and fraudulent company.
17. 10th Dec 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
I had contacted the MHRA on 9th Dec to ask if they could let me know the date that the protocol describing the PB-MSCs used in the 2012-2013 Greek trial had been submitted. This information was important as it would show if the date of the protocol corresponded with Celixir’s request to the Research Ethics Committee in Wales for permission to use cells derived from the peripheral blood of the Morriston patients as therapies in other patients.
An email between two MHRA employees on 10th Dec stated the following:
“I think if she wants that she [Patricia Murray –PM] should go directly to the Greek authority rather than us now doing the work for her.”
Investigating Celixir is not really my work as such because I am not a regulator. However, it is my view that scientists have a duty to challenge and expose pseudoscience and research misconduct, especially when patients are being put at risk. In the case of Celixir, I am very concerned that a company with such an extensive track record of dishonest, unethical and fraudulent behaviour is being allowed to test experimental therapies on UK patients. I am also concerned that companies such as Celixir are bringing the field of regenerative medicine into disrepute. I had rather hoped that MHRA might have seen it as their work to properly investigate this company.
18. 13th Dec 2019: My seventh complaint about Celixir to MHRA
After reading the emails between the MHRA and the Greek regulators that had been released to me under FOI, I alerted MHRA to a number of disturbing issues regarding Celixir, including the following:
- The 2012-2013 Greek trial had been undertaken without the necessary approvals having first been obtained.
- The protocol confirmed that PB-MSCs had been used in the 2012-2013 Greek trial, rather than bone marrow-derived ‘iMPs’ as falsely claimed by Celixir.
- Celixir had provided false information to the HRA to obtain approval from the Research Ethics Committee (i.e., they had falsely declared that the ‘iMPs’ had been authorised for use in a clinical trial in Greece conducted by themselves as Sponsor).
- Celixir had stated in their rebuttal letter to my initial report that manufacture of their ‘iMP’ cells takes a minimum of 15 days, but preferably about 22 days. Despite this, information from the Greek regulators indicated that the cells that had been sent to the UK for use in the Brompton trial had been manufactured in just 10 days. This suggested that Celixir were not even adhering to their own Standard Operating Procedures for ‘iMP’ manufacture.
- Celixir’s CTA application had indicated that the ‘iMPs’ to be used in the Brompton trial would be generated in Greece from human bone marrow from the US company ‘AllCells’. The CTA application indicated that the bone marrow would be sent directly to Greece. However, the emails revealed that the bone marrow had first been sent to the UK. This was troubling because Celixir do not have a GMP facility in the UK, nor do they have an HTA licence for storing human samples.

19. 17th Dec 2019: From internal MHRA emails obtained on Jan 21st 2020 under FOI
The Deputy Unit Manager drafted a response to my concerns of 13th Dec that would later be sent to me. The draft included the following statement:
“The dates for the manufacture of the batch [of Celixir’s ‘iMPs –PM] to be used in the current trial [the Brompton trial –PM] have been reviewed and are consistent with the current IMPD. Therefore our position remains that there are no grounds to take regulatory action to halt the trial.”
Of note, the above draft was prepared without MHRA first verifying that its content was correct. The Standard Operating Procedure (SOP) for ‘iMP’ manufacture states a minimum of 15 days, but the cells for the Brompton trial were manufactured in just 10 days. It is therefore irrefutable that Celixir were not adhering to their own SOPs. Nevertheless, it appears that this was of no concern to the MHRA.
20. 13th Jan 2020: Response from Dr June Raine, Interim Chief Executive of MHRA
Dr Raine assures me in her letter that MHRA are “satisfied that there are no discrepancies” regarding Celixir’s CTA application for the Brompton trial.
This is despite the fact that Celixir had falsely claimed to have used bone marrow-derived ‘iMPs’ in the 2012-2013 Greek trial, when in fact peripheral blood-derived MSCs had been used.
Dr Raine states:
“Publications are not a requirement [i.e. the 2016 publication that reported on the 2012-2013 Greek trial -PM], and it is not a routine aspect of MHRA review to check all references in full for an IB (Investigator’s Brochure).
Dr Raine appears to have overlooked the fact that on 2nd April, her own MHRA staff had advised that if there was hard evidence that the cell type described in the 2016 publication had been falsely described “this would warrant an investigation and the suspension/withdrawal of the trial whilst the investigation is ongoing” (see section 3). Even the MHRA’s Deputy Unit Manager had previously stated that “if the Clinicaltrials.gov is a more accurate reflection of the trial [which it was –PM] then this implies the paper is significantly flawed and fraudulent” (see section 5).
Dr Raine states:
“…for the current trial to be undertaken at the Royal Brompton Hospital we have no evidence that data provided is fraudulent.”
On the contrary, there is irrefutable evidence that Celixir provided fraudulent data. They falsely claimed to have used bone marrow-derived ‘iMPs’ in a previous trial when they in fact used a different cell type altogether (peripheral blood-derived MSCs). This means that their bone marrow-derived ‘iMPs’ have not previously been tested in patients. Moreover, the cells that were used in the Greek trial appear to have been derived from the peripheral blood of patients in the Morriston Hospital without REC approval, and without having been prepared in the necessary GMP facilities. Celixir also conducted the 2012-2013 Greek trial in the absence of regulatory approval. Dr Raine’s confidence in this company therefore appears to be misplaced.
Dr Raine states:
“Please also be aware that consideration for company staff is not part of the trial review by either MHRA or HRA. The company Director [Ajan Reginald –PM] is not named in any trial documentation and is not the “researcher” named for the trial, rather the trial investigators are from the Royal Brompton and their suitability has been reviewed by HRA.”
Dr Raine appears to be saying that it is of no concern to the MHRA if Celixir’s CEO has previously engaged in a catalogue of dishonest, unethical and fraudulent activities because he is not named on the trial documentation. It is difficult of follow the logic here. Applying the same reasoning, this would mean that if Andrew Wakefield or Paolo Macchiarini (individuals who are known to have published fraudulent data with devastating consequences) set up a UK company and wished to test an experimental therapy in a clinical trial, the MHRA would see no problem with this as long as they were not named on the trial documentation.
Notes:
Pieper IL, Smith R, Bishop JC, Aldalati O, Chase AJ, Morgan G, Thornton CA (2017). Isolation of Mesenchymal Stromal Cells From Peripheral Blood of ST Elevation Myocardial Infarction Patients. Artificial Organs doi: 10.1111/aor.12829
This paper by Ina Laura Pieper describes the isolation of mesenchymal stromal cells (MSCs) from the peripheral blood of patients admitted to the Morriston Hospital in Swansea following a myocardial infarction. Pieper undertook her PhD studies with Celixir during 2010-2013, which encompassed the period that patients were recruited to the 2012-2013 Greek trial. The paper states: “This pilot study demonstrates for the first time that MSCs as defined by the ISCT [International Society for Cell and Gene Therapy –PM] minimal criteria can be isolated and cultured from the peripheral venous blood of STEMI [ST elevation myocardial infarction –PM] patients, but not from healthy volunteers.”
Update 20.10.2020
Yesterdays, the publisher Springer Nature issued an expression of concern for Anastasiadis et al 2016. The note goes:
Following an investigation into a number of concerns raised in relation to this article after publication, the journal has found that the study did not receive all necessary approvals from regulatory and ethical bodies. While it has been confirmed that the study received approval from both the AHEPA University Hospital IRB and the IRB of the School of Medicine, Aristotle University of Thessaloniki, our investigation has found that advance approvals were not obtained from the National Ethics Committee nor the National Organizations for Medicines, as required for clinical trials conducted in Greece. This Expression of Concern does not comment on whether the research followed applicable ethical standards.
Additionally, during the investigation, the authors clarified the following about the methods: left ventricular ejection fraction (LVEF) at baseline was manually corrected by the independent referring cardiologists before inclusion in the study. After reviewing the raw data, discrepancies can be found between the estimated and calculated LVEF data. As this raises questions about the accuracy of the method to determine whether patients met the inclusion criterion of an LVEF of less than 40%, the readers are advised to interpret the findings related to the baseline LVEF data with caution.
The authors would also like to correct the following details:
Methods – culture of iMP
The manuscript states that “the iMP cells used in this study were prepared under GMP/ISO 9001 conditions.”
To avoid confusion, the authors would like to correct this to “the iMP cells used in the study were prepared under ISO 9001 and ISO 15189 conditions.”
Authors’ Affiliations
Dr Sabena Sultan, Ajan Reginald and Prof. Sir Martin Evans correct affiliation were with Cell Therapy Ltd., Celixir House, Innovation Way, Stratford upon Avon
Conflicts of Interest
– Stephen Westaby wishes to declare he was issued options in Cell Therapy Ltd. in 2012, which were later converted into shares.
– While the authors Ajan Reginald, Sir Martin John Evans and Sabena Sultan declared that they held shares in Cell Therapy Ltd., they would also like to declare their affiliation with Cell Therapy Ltd. at the time of submission.
Authors Polychronis Antonitsis, Kyriakos Anastasiadis, Argirios Doumas and Stephen Westaby agree to this Editorial Expression of Concern. Authors Ajan Reginald, Martin John Evans and Sabena Sultan disagree with this Editorial Expression of Concern. Author George Efthimiadis has not responded to the editor about this Editorial Expression of Concern.

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
5.00 €


Absolutely shocking. I thought that Brexit is a horrible thing but didn’t suspect it would be THAT horrible SO soon.
Prof Murray and Leonid should continue creating waves. Maybe contacting UK national newspapers, BBC, tabloids, anyone! As to the DUM and MHRA, their bosses should publicly declare their personal financial independence from the Celixir’s highly dubious crew. I am sure there is some bribery or other “contribution in kind” involved. There is no other explanation to their conniving of Celixir’s crimes.
LikeLike
I would like to thank Leonid for making this publicly available. My opinion is that the patients who will be recruited to the Brompton trial will not be signing an informed consent, as they will not be told anything about the history of the company or the so-called ‘therapy’ that will be administered to them. At least patients searching to understand more about the trial now have a chance of finding the information themselves.
The behaviour of the MHRA is baffling, which is why I requested the internal emails. From my reading of those, I think the main problem stemmed from the fact that MHRA were already aware that there were problems with this company before they received the CTA application to undertake the Brompton trial. However, this information seemed to have been overlooked. Had the MHRA performed due diligence at that point, they would have discovered that there had been serious Good Clinical Practice (GCP) violations in relation to the Greek trial, and that penalties had been imposed on the company by the Greek regulators. The improprieties included, but were not restricted to, undertaking the trial without first obtaining approval from the appropriate Greek authorities (i.e., regulatory approval from the National Competent Authority (EOF) and ethics approval from the Independent Ethics Committee).
The Deputy Unit Manager (DUM) dealing with the case for MHRA did not become aware of this until after I informed them of my concerns in in 2019. I would have thought that the correct response at this point would have been to suspend the trial and find out more about the nature of the improprieties in Greece and what penalties had been imposed. But instead, the DUM appeared to immediately adopt a defensive position, stating: “I don’t think this changes the CTU [Clinical Trials Unit –PM] decision though, that even if the previous data was with a different product, we would still have approved the current UK trial.”
From my point of view, this was a much bigger problem than not having done the due diligence in the first place. In a big organisation, it is understandable that things can sometimes get missed, but it is not acceptable to then try and defend that by whitewashing serious breaches and improprieties. By doing so, the DUM converted a relatively minor error into a much more serious one. This potentially speaks of an unhealthy defensive culture at the MHRA.
However, on a brighter note, some MHRA staff did appear to act appropriately, and although the Director of Licensing was keen to refuse my FOI requests, the DUM made the right decision to respond positively. The transparency here was commendable.
I hope that the MHRA will now do the right thing and withdraw their approval for this trial. I would also like to see the MHRA being more welcoming in future of individuals who inform them of potential serious improprieties rather than viewing them as a nuisance. The goal of both parties should be the same –protecting patients from harm.
LikeLike
Even if the heart trial doesn’t go ahead, fear not, because Ajan has plenty more things up his sleeve, like making gene therapies for cancer: see Pubpeer discussion of their PLoS One paper:
https://pubpeer.com/publications/F0ED142380122E04FEAAFEA10CC606#
Check out what he has to say in this Spotify interview from July 2019:
Interesting bits below. Does anyone know that it means to ‘breglish’ a patent? Does it mean you have stolen it from someone else?
8:36: “Most recently we had a hypothesis that siRNA in particular would be very helpful in oncology in solid tumours that are resistant, and that seems to have panned out. We’ve got a couple of papers in oncology recently where our siRNAs are focussed on STAT6. STAT6 is highly expressed in the tumours of patients with very poor survival.
9:27: “But then as a CEO we had this good hypothesis that siRNA would work so we went out and we bought that siRNA. So we bought it, we did the acquisition, we integrated, we’ve now developed it, we’ve got it BREGLISHED etc etc. “
Some info below on Dr Lee Chapman, senior author of Celiixr’s PLoS One siRNA paper who declares on PubPeer that his multiple roles as Celixir/Cell Therapy Ltd pose “no conflict of interest”. We learn from Ajan that as well as working at the cutting edge of siRNA biology, Lee is also creating new cells and patenting them. He is a co-inventor on the infamous ‘iMP’ patent. What versatility we have here!
38:39: “So Lee was a scientist. He did a DPhil at Oxford and then became a patent attorney. And he was our patent attorney from day 1, so from 2010 he was our patent attorney. And then we persuaded him to leave the world of patents and go back to being a scientist. So now he’s our Head of Discovery Sciences. Because ultimately Lee realised that he loves solving problems. And his expression of how he solved the problem used to be writing a patent. Now his expression is to design an experiment, create a new cell and patent it if that makes sense. So again it’s that theme of become a world expert in something and then apply it to something else.“
“None of our lab scientists have PhDs”.
LikeLike
The Daily Telegraph reporting.
https://www.telegraph.co.uk/news/2020/03/13/exclusive-nobel-laureate-embroiled-research-scandal/
“A Nobel laureate has become embroiled in a major research scandal after it emerged that a stem cell company he co-founded conducted unlawful medical trials.
Professor Sir Martin Evans, one of the country’s leading geneticists and the former Chancellor of Cardiff University, was involved in trials in Greece which took place without authorisation, and were described by the country’s regulators as a “violation” of national legislation.
He is accused of going on to mislead the British medical watchdog when seeking approval for a new round of trials at one of the UK’s leading hospitals.”
LikeLike
Pingback: Stem cells to cure COVID-19 because why not – For Better Science
Story in the new issue of Private Eye: “Bones of Contention”
MHRA has stopped the trial, but unfortunately, only after 1 patient was ‘treated’. The patient was treated on 16th March. Trial was stopped on 24th March. No more patients will be treated and trial will end when this single patient has completed follow-up.
Serious failings here by MHRA, Health Research Authority and the Brompton Hospital.
Serious failings by heart surgeon Prof Stephen Westaby, who despite being fully aware of Celixir’s improprieties, continues to work for Celixir and promote them.
Serious failings by SpringerNature who failed to retract the article about Celixir’s Greek trial that was published in the Journal of Cardiovasc Trans Res. Publishers knew that along with other serious problems, the trial had no ethical or regulatory approval, and that the Greek authorities imposed penalties on Celixir. Had they acted promptly, it is likely that the trial could have been stopped before a patient was ‘treated’.
Disappointing behaviour by Brompton staff Profs Kim Fox, John Pepper and Dr Ali Vazir; Dr Darrin Disley, CEO of Mogrify; and Prof Fiona Murray from MIT. All are fully in the picture about Celixir’s previous behaviour in Greece, but appear comfortable about continuing to work with them.
LikeLike
Update in this issue of Private Eye: ‘Trial and Error’
The article indicates that the MHRA is now formally investigating Celixir. This is great news and hopefully it will lead to action being taken against the company.
A flaw in the system for trial approvals in the UK though is that no matter how badly a company has behaved, and no matter what penalties are imposed, it seems there is no mechanism to prevent them from seeking permission to conduct further trials. I asked both the MHRA and HRA if a tipping point could ever be reached where the extent of fraudulent, dishonest and unethical behaviour that a company has engaged in could render them unsuitable for sponsoring a clinical trial. Neither authority said ‘yes’.
LikeLike
Update in this issue of Private Eye : ‘Paper thin evidence’
The article reports that Celixir’s 2016 article on their ‘iMP’ cells (aka ‘Heartcel’) now has an Expression of Concern. Readers are advised to interpret data “with caution” and there is an admission that the cells injected in patients’ hearts were not prepared under GMP.
We also learn that Celixir now plans to target cancer patients with a new invention: the ‘mesodermal killer cell’ . Mesodermal killer cells are made by simply growing Celixir’s ‘iMPs’ in low oxygen conditions and adding a few supplements.
Patent here: https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2020148520&tab=PCTCLAIMS&_cid=P11-KH9MRJ-91893-1
The following excerpt from the patent suggests all types of cancer may be treated with the magic MKs:
LikeLike
Pingback: Colchicine or Liquorice? – For Better Science
Pingback: The Cardiff Coronatest – For Better Science