The tracheal transplant experiments on three human patients were performed by the German scientists Heike and Throsten Walles without any previous animal testing, as now admitted to me by the Fraunhofer Institute for Interfacial Engineering and Biotechnology (Fraunhofer IGB) in Stuttgart. It also came to light that the pig intestine-based tracheal transplant was eventually deemed as too toxic, and a follow-up animal testing was apparently cancelled. Heike Walles is still associated with Fraunhofer IGB while being together with her husband employed as professors at the University Clinic of Würzburg. A patient of theirs received in 2007 a tracheal transplant of unknown size and shape, prepared from decellurised and in vitro re-populated pig intestine; this unnamed person died only some weeks after, of causes unknown to us. We know however, that two weeks before that operation, a tubular tracheal graft made from pig intestine was test-implanted into the patient’s arm for a week. After a quick analysis, a second in parallel prepared graft was implanted into the patient’s airways. Remarkably, only preliminary first in-arm test, but not the main operation, was ever published (Mertsching et al 2009), around two years after the patient’s death. The next patient, an Indian immigrant Pavninder Singh, received a large tracheal patch, which apparently did not perform as expected: the patient’s tracheostomy had to be re-opened, there was also apparently heavy inflammation. That patient committed suicide 2.5 years after the operation, and became in 2013 a central character of a book chapter which described his and the scientists Walles’ story (Bernhard Albrecht, “Patient of My Life”). Only in 2015 was the case of that patient published as a research paper (Steinke et al, 2015), his tracheostomy went for some reason unmentioned, just like the fact that Singh was no longer alive since 2011. All these tracheal transplants were prepared in the lab of Heike Walles (back then under her former husband’s name Mertsching) at Fraunhofer IGB, the transplantations took place at the neighbouring Robert Bosch Hospital, Clinic Schillerhöhe, where Thorsten Walles used to work as surgeon, before becoming professor in Würzburg. The details can be read in this article.
Humans first
The book author Albrecht wrote, likely after speaking with Walleses, “in animal experiments they already implanted the [tracheal, LS] grafts successfully”. This is what the spokesperson of Fraunhofer IGB, Claudia Vorbeck declared to me in regard to the Fraunhofer tracheal transplants in her email from January 20th, 2017, highlight mine:
“The Fraunhofer IGB itself does not carry out animal tests. If animal experiments are necessary for our work, we will assign these to companies or collaboration partners authorized to perform animal experiments. Whenever possible, however, the Fraunhofer IGB prefers alternative methods for animal experiments. In the case of the project for the production of an autologous trachea graft, no animal experiments were necessary until 2009 due to the preliminary work of Prof. Walles in Hanover. The publications can be found via PubMed”.

The preliminary work in Hannover (exactly, at the Hannover Medical School, MHH) which Vorbeck refers to, was performed by the Walles couple together with their former superior, and now the world-infamous scandal surgeon, Paolo Macchiarini. Not a single Walles publication available on PubMed suggests any previous animal testing of pig intestine-derived tracheal transplants in Hannover (or elsewhere), but one case of an in-human test: that of the patient Ernst Fromhage, who received in 2003 a tiny (“size of a Eurocent-coin”) tracheal patch of decellurised pig intestine. The intervention was published as MacChiarini (sic!) et al 2004 and the method as Walles et al, 2004. Macchiarini is corresponding author on both publications, and was celebrated for this operation in local media, without any mention of Walleses whatsoever. It was also Macchiarini alone (Macchiarini, 2004) who replied to the harsh and most likely very much justified criticism on that operation, which was issued by the late eminent expert of thoracic surgery, Hermes Grillo (Grillo, 2005). The Walleses were left out of the loop here again, on this allegedly exclusively their own discovery. Now however, the two Würzburg professors Walles had a German court sentence me for libel, in absentia, for stating that Macchiarini was in any way involved in that 2003 operation and the associated tracheal transplant technology (unless I can bring up enough money to pay a lawyer to defend myself, the verdict remains guilty, and I also have to pay the huge court costs).
The patient Fromhage died 16 months after the operation, for probably unrelated causes. We have however objectively no clue how the tiny tracheal patch performed at that time, the paper MacChiarini (sic!) et al 2004 declares only:
“Outpatient endoscopies on the 3rd, 6th, and 12th postoperative weeks confirmed the above early postoperative findings and showed that the graft was airtight and surfaced with autologous ciliated respiratory epithelium (Figure 2, C and D), with no evidence of chronic inflammation, granulation tissue, infection, or erosion. Vascular ingrowth was detectable from the patch margins as soon as by the 6th week and completed by the 12th postoperative week. After 12 weeks, the entire patch was functionally and morphologically completely integrated into the adjacent airway”.
Macchiarini and Walles
Let us assume, even if just for the argument’s sake, that this is the only fully trustworthy tracheal transplant paper by Macchiarini (since we already know the accusations, evidence and commission findings about all of his other transplants and publications). Let us for now trust Macchiarini and his partners Walles here that that tiny piece of pig intestine became somehow vascularised and grew a healthy epithelium, and not just overgrown by fibrotic scar tissue. Was it however safe to extrapolate that now large sections of trachea can be replaced with a pig-intestine based grafts, even after the technology was in vitro optimised by Heike Walles at her new Lab at Fraunhofer IGB? Apparently, this federally funded and applied technology oriented research institution decided that the technology was sufficiently proven. Without any animal testing whatsoever, two human patients received Fraunhofer-made large tracheal grafts in a Stuttgart clinic. None of these three is alive today to help assess the actual success of their transplants.
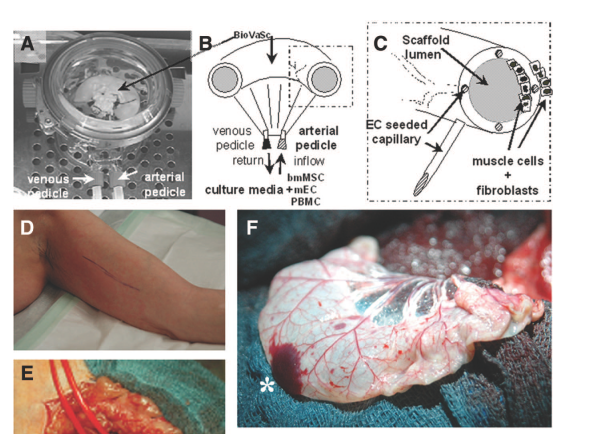
One year after Walleses transplanted in 2007 a patient in Stuttgart with a tracheal graft of unknown size or shape, their former boss Macchiarini performed in Barcelona his famous transplant of a decellurised and bioreactor-“regenerated” small section of bronchus. That patient, Claudia Castillo, is still alive, but she lost her lung (here the full story of Claudia’s Trachea). Noteworthy, Macchiarini is being repeatedly accused of having done this transplant without any previous animal testing. He denies this, but so far refuses to provide evidence. The only testing which Macchiarini did perform before operating the patient Castillo, was an inflammation-assessment of subcutaneously implanted pieces of decellurised trachea carcass in pigs and rats. Heike and Thorsten Walles obviously never even did this, with full support of the former’s academic employer, the engineering-oriented Fraunhofer Society. With Macchiarini, three clinical research institutions were involved internationally, and an ethics approval for compassionate care use was given by the Hospital Clinic Barcelona. With Walles, we don’t know if any ethics approval was sought or granted. As you can read below, the information is secret.
Bo Risberg, emeritus professor of surgery at the University of Gothenburg, famously coined the expression of “Chernobyl of Ethics“ in regard to the Macchiarini scandal. Back in February 2016, he kindly provided me with this Open Letter, where he took issue with the scientific, medical and ethical shortcomings of Macchiarini’s trachea transplants. Now, Risberg declared this in regard to the work of Heike and Thorsten Walles:
“Amazingly the authors introduced xenografts into humans without proper animal testing! That´s Macchiarini style!
These are unethical experiments in the same class as the scandal at Karolinska in Stockholm. There, preclinically untested plastic tracheas were used. Here xenografts, admittedly decellularized and seeded with autologous cells, were implanted, but likewise without any preclinical testing.
Surprisingly none of the journals asked for the written approval of the Ethics committee.”
Secret ethics
Indeed, an interesting question, but very difficult to answer, as I demonstrate below. After the operation on Fromhage, the Walleses left Hannover for Stuttgart and operated the next follow-up patient in 2007, and the last one, Singh, in 2009. We do not know if any ethics approvals were issued or necessary for these transplants, since the German watchdog institution Paul Ehrlich Institute (PEI) keeps avoiding answering my Freedom of Information inquiry on this, placed on December 10th 2016. Which is apparently perfectly OK, since there is really no overarching institution to complain to in Germany. I was only given this general information by Susanne Stöcker, head of the PEI’s Press and Information Office:
“Regarding the treatment of the three patients, I would point out that, in the case of medical emergencies, there is the possibility of a “compassionate use” within the doctor’s therapy freedom, possibly “under justifying emergency”. In such cases, there is no requirement for approval or approval”.
The paper about Singh operation, Steinke et al, 2015, makes no reference to any ethics approvals, only to “patient education and informed consent”. There was however an ethics approval given for the in-arm test of Singh’s unnamed predecessor in 2007. The paper Mertsching et al 2009, which focuses solely on that in-arm test and utterly omits the subsequent airway transplant, and which was published two years after the patient’s death, writes:
“Informed consent of the patient was obtained beforehand and ethical committee approval sought from the Eberhard Karls University Tuebingen. In addition, permission for compassionate use was obtained from the medical association Nordwürttemberg”.
I asked the Ethics Commission of the Medical Faculty and Clinic of the University of Tübingen these two questions:
- was an authorization granted also for the subsequent transplantation of the airways?
- was the transplantation of the patient Singh in 2009 also approved by the same ethics commission?
Right away I received this puzzling reply from the head of the Tübingen ethics commission, Dieter Luft:
“Unfortunately, I cannot yet answer this inquiry because the ethics committee is obliged to maintain confidentiality and discretion in accordance with § 5 para 3 medical profession – Chamber law Baden-Württemberg. Information can only be divulged following a written consent to lift the confidentiality issued by Prof. Walles”.
Update 24.01.2017: Marlies Kepp, head of communications at Robert-Bosch Hospital, where the operations were performed answered in this way when I asked her about who issued the ethics permits:
“…these processes are part of doctor-patient confidentiality”.
That’s probably it, in Germany it’s best to trust your doctor. It doesn’t really matter here that the patient Singh agreed to have this confidentiality fully waived, so the book about his doctor Thorsten Walles and his transplant could be written.
The Albrecht book where the Singh operation is described in detail mentions therefore certain highly relevant clinical aspects aspects which we will not find in any of Walles’ own publications. Like Singh’s re-opened tracheostomy and an “inflammation in his throat”, which was raging for “many months”, following the transplant of the pig-intestine patch. Then there was this incident with Thorsten Walles and German health authorities:
“On a hot Saturday in August 2009 Walles found a letter from authorities in his home mail. […] He broke the Medicinal Produce Law in its most recent form of June 22nd 2009, he read. He was threatened with three years in prison and withdrawal of his licence to practice medicine”.
The issue was resolved a few days after, since the Singh operation took place three months before the new law came into force. Apparently a whistle-blower reported Walles to the authorities: “there were not many suspects”. Nobody likes whistle-blowers. In fact, now it is myself who is threatened with 6 months prison by the German authorities if I dare repeating after the same Albrecht book or mentioning Macchiarini.
Update 24.01. 2017. Today I received two communications. One was a new court letter from Landesgericht Würzburg, giving me less than 2 weeks to reply before a trial against me will be opened on the behest of Walles, primarily for publishing the court documents, tweeting and for allowing to comment and quote from my site, especially to those being abroad and outside of German jurisdiction. Peculiarly, the Walleses now use their home address, which suggest that they wilfully gave false testimony in court before by insinuating to be representing the University Clinic of Würzburg, in order to get that injunction against me passed. The letter is available here.
Too dangerous for animals?
The other communication was an email from Gaston Reeck, of Unit 616 – Health Care, Federal Ministry of Education and Research (BMBF):
“In the following, I would like to answer the questions you have asked about the BMBF-funded project “RegTech: Bioartificial Reconstruction Tissues for Surgery”, as far as it is possible to refer to the TIB (Leibniz-Informationszentrum Technik und Naturwissenschaften Universitätsbibliothek) Final report of the project for free access at the following link. https://www.tib.eu/en/such/download/?tx_tibsearch_search[docid]=TIBKAT%3A872484653&tx_tibsearch_search[searchspace]=tn&cHash=db1348564f8730136468571c4ea2e4e4#download-mark
The clinical trial envisaged in the aforementioned project was not started, as the manufacturing authorization according to § 13 AMG [medicinal produce law, -LS] as basic requirement for a clinical application of the Biological vascularized Scaffolds (BioVaSC) within the project duration was not given.
For the BMBF-funded project, EUR 1,516,300 were earmarked for the planning period from 01.07.2009 to 31.12.2015. The intended purpose of the clinical validation of the bioartificial reconstruction tissue for the tracheal surgery was not achieved. As explained in the final report, the work could not be carried out in full. The amount of funding has therefore not been paid in full”.
In fact, this final project report to BMBF, which was authored by Thorsten Walles and submitted in the 2nd half of 2016, suggests that his tracheal transplants, previously implanted into 3 human patients, proved later on as too toxic (i.e., full of endotoxins) and unsuitable for animal experimenting. The clinical trial (which never was) run from 7.2009 to 9.2012 and was extended twice, until it ended on 31.12.2015. Of the five Milestones, Walles admits only the first one was achieved: Milestone 1, application for the Manufacturing Authorisation, submitted after repeated failure again on 4.08.2015 (and then denied). The Fraunhofer Institute ceased its engagement with the project already on 31.03.2010, the project was managed from 01.10.2009 on at the University of Würzburg, thus basically for the entire 6-year duration of the project. As a reminder, this University denied to me previously any connection to Walles’ tracheal transplants and to any “regulatory or scientific aspects” of those.
Below some quotes, original text of Walles, and his partner Oliver Pullig (then at Fraunhofer IGB, now together with Heike and Thorsten Walles at University of Würzburg). Here for example what they discovered about the tracheal transplants, decellurised obviously by same method as before for two human patients:
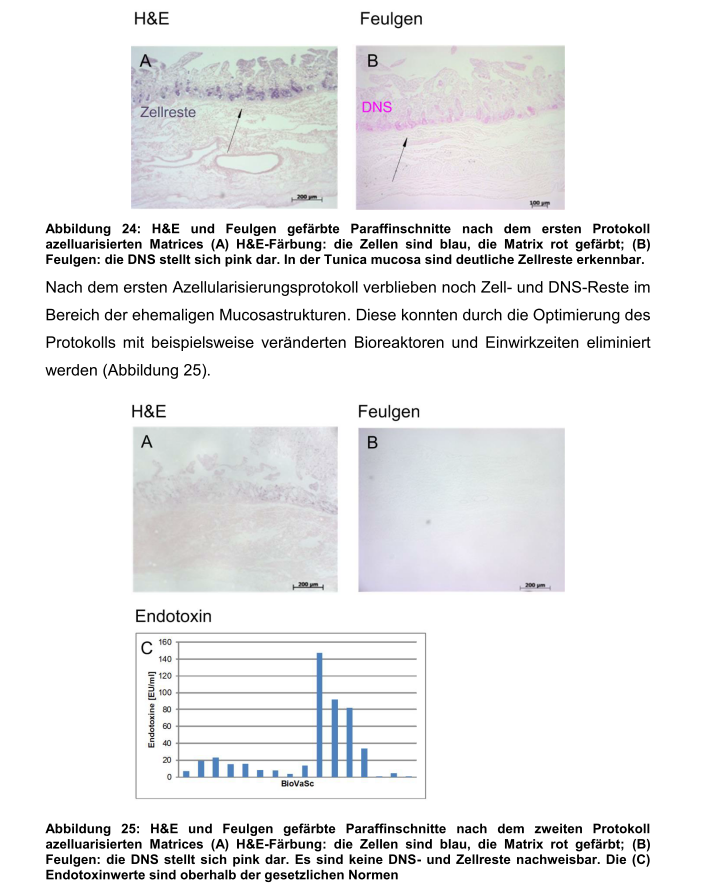
“In the follow-up analysis of the toxin content a too high amount of endotoxins was found. Initially we could achieve though the use of additional cooling steps during decellurisation and use of antibiotics a reduction of endotoxins below the critical level of 0.25 EU/ml.
It proved during the transfer of the R&D technology to BioVaSc manufacturing in GMP conditions that there was no GMP authorisation for some of the previously used substances (e.g., DNAse from Roche [also see reader comment below, -LS] and a change in the process became necessary. Afterwards the pharmacopoeia of BioVaSc showed again critical endotoxin concentrations…”
Update 6.02.2017: In fact, it seems the endotoxins (which a residues of bacteria, capable of causing life-threatening inflammation) were never measured previously when three humans were transplanted. The method was first established and validated by Heike Walles’ PhD student Iris Dally, as evidenced by her dissertation from 2013.
The bioreactor technology, which was previously used on human patients, was found not GMP compliant and was slammed by the state’s authorities during the project evaluation. At the same time, the federal watchdog PEI (the same one which evades answering my FOI inquiry) demanded already in 2010 some animal testing:
“A second consultation took place on 23. 09.2010 at Paul-Ehrlich-Institute in Langen. During the consultation, the necessity and the kind of the animal experiments, relevant for the approval, was discussed. Agreement was the implementation of rodent-implantations to prove and exclude the xenogenic immune reaction to the applied decellurised matrix. The requested experiments on large animals to evaluate the transplants were postponed”.
Thus, Walleses’ tracheal transplants were deemed to dangerous to be implanted into airway of livestock like sheep. They had to be first tested for biocompatibilty as subcutaneous implants in rats, which apparently also never happened (why, actually?). This, mind you, after 3 humans received same technology under compassionate care use.
Walleses now fight in court against me tooth and nail to decry any association with Macchiarini as libel. This is however what Thorsten Walles himself wrote just as the scandal was raging in June 2016, free from every scepticism and in bold:
“In an extensive investigation by the Karolinska Institute, Macchiarini and his coworkers were exonerated of the then raised accusations of scientific misconduct.”
In reference to Macchiarini and his plastic trachea technology, Walles commented that “a scientific assessment of these therapies is currently not possible” and wrote:
“A disadvantage of these two therapy methods [the other one by Emmanual Martinod, using cryo-frozen aortic grafts, -LS], that after the implantation in patients strong inflammation response to the implants occurs, which must be treated extensively and controlled”.
Well, according to the Albrecht book, Walles’ last patient was suffering from very similar symptoms. For “many months”, in fact. According to the final BMBF report, the late patient Singh was examined in the first half of 2011 (shortly before his suicide) and was found fit and active, and able to speak. His re-opened tracheostomy went once again unmentioned.
If you think all this Kafkaesque court attack by Walleses on me makes no sense, tell this to their University Clinic of Würzburg and sign the Open Letter by Prof Rafael Cantera here.
Update 15.03.2017. There were indeed no animal experiments ever performed at all, as confirmed by an investigation by the University of Würzburg (my report here and here, original document here). Nevertheless, the University website still falsely proclaims that “tests in large animals were successfully performed” before Singh was operated (here and here, backup copies here and here).
If you would like to support my court litigation financially, donation amount doesn’t matter, please go to my Patreon site or contact me.

Pingback: Open Letter in support of my investigation of trachea transplants in Germany, by Rafael Cantera – For Better Science
I am not a doctor, but with (artificial) trachea transplants the mechanical stress and infection risks seem particularly high to me. To not test this first in animals seems to amount to what my moral compass would describe as “criminal” attitude. I have to say though that I am surprised that one of the three patients could survive at least a few more years. This patient is the one who the Walleses celebrate as a huge success, despite that also even in this patient complications arose, and despite the fact that according to your descriptions this patient did not perceive the operation as successful himself. Maybe, in the grand scheme of things, the operation in this third patient was a big success indeed from which valuable lessons can be learned. However, that would need a fully detailed and transparent analysis, also of the other two patients, which is not an environment which seems to be fostered here.
The judge was probably wrong. The only relevant difference between the story told to him by the Walleses and the one in your 2nd text version concerns the estimation of success in the third patient. If there were complications, and if the patient himself did not perceive the operation as successful, a judge cannot simply assume that the complainant is the one who is right. He should at least have requested an independent attest for the claim of the transplant being successful. But even if so, as long as there were medical complications, it is hard to see why differences of opinions on the level of success would not be allowed, and why a court action is needed to prohibit the public discussion. After all, this is a new medical approach which came under public scrutiny, and three out of three patients died. A judge shouldn’t be able to stop the normal public debate.
LikeLike
I had a look at the Mertsching et al 2009 publication, and in particular the methods describing “Clinical biopsy procedure to generate the bioartificial transplant”. What strikes me is the fact that most of the culture media used in the procedure are expressively supplied “For Research Use Only”. As a layperson when it comes to law, I would think that use of these media to manufacture “therapeutic goods” (i.e. the transplant) would be illegal, both in a sense of the terms & conditions under which they were supplied, as well as the use of these “therapeutic goods” outside of a research context… Perhaps it would be worth pointing this out to the relevant authorities, and also the suppliers?
LikeLiked by 1 person
“The publications can be found via PubMed”
Biological vascularized matrix for bladder tissue engineering: matrix preparation, reseeding technique and short-term implantation in a porcine model.
J Urol. 2005 Jan;173(1):276-80.
This is the only animal test that I find. It appears that one animal received the implant for 3 hours.
“Acellular matrix was processed from porcine small bowel segments by subsequent mechanical, chemical and enzymatic decellularization, preserving the jejunal arteriovenous pedicles”
I don’t think it is correct to write that no animals tests were done. It would be more correct to ask if one animal with a 3 hour implant is enough to justify a human trial.
LikeLike
This is also an animal study using the acellular matrix. It is in German. Someone should translate it. Interestingly, the author list of this paper and the paper above shows that a D. Schultheiss (corresponding author) was the person that developed the matrix, not the Walleses.
Urologe A. 2004 Oct;43(10):1223-8.
[Biological vascularized matrix (BioVaM): a new method for solving the perfusion problems in tissue engineering].
Schultheiss D, Gabouev AI, Kaufmann PM, Schlote N, Mertsching H, Haverich A, Stief CG, Jonas U.
English Abstract:
A new technique is presented to harvest an acellular matrix from a porcine small bowel segment preserving the mesenteric arterial and venous pedicles. Reseeding of this biological vascularized matrix (BioVaM) with functional cells, i.e. smooth muscle and urothelial cells isolated from the urinary tract, and resurfacing of its vascular structures with endothelial precursor cells results in a vascularized tissue engineered graft for reconstruction and augmentation of the urinary bladder. First promising short term implantation experiments using a porcine model for the evaluation of early graft perfusion after vascular anastomosis are presented.
It is quite possible that the matrix was tested in multiple animals, but as a bladder implant, not trachea.
LikeLike
It was again a very brief test for 1-3 hours, which had nothing to do with trachea, where you need actually airway tests in large animals, not just a brief subcutaneous biocompatibility study. Thank you for pointing out the original source of this “biovascularised matrix”, who seems to be Dirk Schultheiss, then at MHH, now urologist in Giessen.
LikeLike
Pingback: Capes aumenta exigências para bolsas da Universidade Aberta do Brasil | Direto da Ciência
These are not countryside doctors doing everything by the book. These are a doctor and scientist who seeked media attention with a highly controversial procedure, which they established (in whatever part of the trajectory) with someone now condemned for severely breaching ethical and legislation rules. All three of their three patients died, so they are bound to and should get some stick in the media. When they believe that the media coverage is not accurate, they should defend themselves in those media, because the public eye is the platform of their original choosing and that’s what they owe to the public.
Their lawyer probably understands that, and that may be the reason why they only focused on the correctness of a text fragment. But removing of a text fragment only, without specification of what in that text fragment can’t be said, can’t provide the “urgency” in relation to the apparent job search by the Walleses. We are currently seeing the perfect example of that: Text fragment removed, but a lot more negative media attention. Without the urgency, this procedure was not legit. So you may consider not discussing the verdict but to address the procedure (the urgency part). Since your texts were not grossly defamatory beyond common boundaries, and you already showed your willingness to cooperate with them if provided with good arguments (while they were the ones who didn’t want to answer questions), it will be very hard for them to prove that this court procedure had the requested urgency.
LikeLike
I mentioned the possible implication of European law before (which overrules country law), and it does seem hopeful for you:
The European Court agreed to review the injunction, and unanimously held that it violated Brosa’s right to freedom of expression.
https://strasbourgobservers.com/2014/05/21/german-court-injunction-banning-political-leaflet-violated-article-10-brosa-v-germany/
Your case is very fundamental to the freedom of speech, and, especially with the wonderful support given to you by many scientists, you may get a news organization behind you. My sense of humor would be well served if this case, which may have been initiated as financial bullying, would become so big that it would become a financial burden on even the Walleses.
LikeLiked by 1 person
Inspired by Vladimir Teif’s comment in https://forbetterscience.com/2017/01/17/open-letter-in-support-of-my-investigation-of-trachea-transplants-in-germany-by-rafael-cantera/#comment-3705, I was wondering if https://www.crowdjustice.org/ could be a way to help Leonid fight back?
LikeLiked by 1 person
That depends on whether he has any type of legal insurance, and on his strategy. He can choose to pay the 1800 Euro and keep fighting more carefully, he can choose to try to win this case by focusing on the case-specific issues, or he could try to fight the principle of the case for all journalism by focusing on the general issues. Each route has its own length and potential costs.
PubPeer, which must have much deeper pockets than Leonid, got support from Google, Twitter and ACLU in a defamation lawsuit http://retractionwatch.com/2016/12/07/pubpeer-wins-appeal-court-ruling-unmask-commenters/
Leonid is far more outspoken than PubPeer, and I wonder if big brands would associate with him. However, I am happily surprised with the large non-anonymous support he gets from many scientists. Maybe times are changing, and more and more scientists recognize the value and necessity of the aggressive checking of science integrity. Although I don’t agree with everything Leonid writes, I do consider him absolutely integer and in his field he is without competition (especially in bravery!). I am glad that many scientists see it in a similar way.
I think the best what we can do for now is to ask many scientists, but maybe also journalists, to support the letter by Rafael Cantera. If that letter would get 300, or even 500 supporters, that may trigger a larger organization to help Leonid (because then considered mainstream enough). Then it will be possible to go for the long haul and target the legal principle of the case which is that a journalist working for the public good shouldn’t be so easily bullied by injunctions.
If Leonid prefers one of the smaller routes, and if he has no legal insurance for them, then crowdsurfing probably would be an option. But my suggestion is to first try to increase the support for the letter by Prof. Rafael Cantera.
LikeLiked by 1 person
“Urgency” is also an interesting court concept considering their operations. If I am correct, proper testing of new methods in animals etc. can only be bypassed in case of high urgency. But in this case there appear to have been more than 5 years between the operations, and one of the Walleses is a full-time scientist who seems to have spent a big part of her time on this type of research. Shouldn’t the Walleses at least after they started the first experimental procedure in humans have continued with experiments in animals? This “haste” (urgency), necessary for the bypassing of common rules, seems to be self-inflicted. It would be interesting how a judge would look upon that.
LikeLike
Interesting points in the article https://strasbourgobservers.com/2014/05/21/german-court-injunction-banning-political-leaflet-violated-article-10-brosa-v-germany/ which I referred to are:
Imprisonment for defamatory speech on a matter of public interest is absolutely prohibited under Article 10. –
Did “your judge” have the right to threaten you with imprisonment? You could point that out and ask him to revise the verdict, since the judge obviously missed that this concerned a matter of public interest. This actually could be a point interesting for the main media (judge threatens with imprisonment in freedom of speech connected to public interest case).
It must be remembered that (a) the injunction not only banned distribution of the leaflet, but also any “assertions of fact which might depict F.G as a supporter of neo-Nazi organisations,” –
The injunction discussed in that article at least had a logical intent, namely the banning of a leaflet as well as spreading in any form its assertions of fact. Your injunction, only dedicated to a precise text fragment, is absurd.
…. the Court held this was Brosa’s opinion based on his own assessment of the facts, “which might be accurate or not.” The opinion was “not devoid of a factual basis,” … –
It will all come down to the patient No.3 case, the rest seems irrelevant or there is no real conflict in statement. If you had any source (accurate or not) for believing that the operation in this third patient did not work out successfuly, you probably were not legally wrong in writing what you wrote. Because, your “own assessment of the facts” and “not devoid of factual basis”. This is especially so because the Walleses and their institutes refused to answer your questions, so you couldn’t help to have to rely on those other sources.
Do the Walleses really want to go to court with you? The risk for them, except for losing, is that they will have to discuss all the many facts in the respective text fragment in great detail. That may not be such a smart idea, considering that they may have to face more serious court battles (with different strategy points) in which these comments can be used.
LikeLike
Do I misunderstand something? You are supposed to pay the “Kosten des Rechtsstreits” (around 1800 Euro? That’s a real lot if you have no salary), but the article further below seems to explain that this is not allowed in this type of cases where there was no actual “Termin”. They appear not to charge you lawyer-fees (of the opponent) but only administration costs, but still I doubt that the charging is correct. It would mean that every time a journalist has the facts not fully accurate, he can be forced to pay 1800 Euro by a trial in his absence and without any discussion of those facts. From Nr. 3104 VV RVG it probably follows that your case is considered as being under § 105:
§ 105
(1) Das Gericht kann ohne mündliche Verhandlung durch Gerichtsbescheid entscheiden, wenn die Sache keine besonderen Schwierigkeiten tatsächlicher oder rechtlicher Art aufweist und der Sachverhalt geklärt ist. Die Beteiligten sind vorher zu hören. Die Vorschriften über Urteile gelten entsprechend.
(2) Die Beteiligten können innerhalb eines Monats nach Zustellung des Gerichtsbescheids das Rechtsmittel einlegen, das zulässig wäre, wenn das Gericht durch Urteil entschieden hätte. Ist die Berufung nicht gegeben, kann mündliche Verhandlung beantragt werden. Wird sowohl ein Rechtsmittel eingelegt als auch mündliche Verhandlung beantragt, findet mündliche Verhandlung statt.
(3) Der Gerichtsbescheid wirkt als Urteil; wird rechtzeitig mündliche Verhandlung beantragt, gilt er als nicht ergangen.
(4) Wird mündliche Verhandlung beantragt, kann das Gericht in dem Urteil von einer weiteren Darstellung des Tatbestandes und der Entscheidungsgründe absehen, soweit es der Begründung des Gerichtsbescheids folgt und dies in seiner Entscheidung feststellt.
But § 105 (1) says that you should have been at least heard before.
So based on which law do they charge you? In addition, as we have been discussing, your case is very complicated whereas § 105 (1) requires simplicity.
At least you can ask the court based on which law they charged you. You may want a lawyer to write that question.
https://www.haufe.de/recht/deutsches-anwalt-office-premium/ags-012009-keine-terminsgebuehr-im-einstweiligen-verfuegungsverfahren-bei-entscheidung-ohne-muendliche-verhandlung_idesk_PI17574_HI2629201.html
Beitrag aus Deutsches Anwalt Office Premium
AGS 01/2009, Keine Terminsgebühr im einstweiligen Verfügungsverfahren bei Entscheidung ohne mündliche Verhandlung
Entscheidet das Gericht über einen Antrag auf Erlass einer einstweiligen Verfügung gem. § 937 Abs. 2 ZPO ohne mündliche Verhandlung, entsteht keine Terminsgebühr. Anm. Abs. 1 Nr. 1 zu Nr. 3104 VV ist nicht entsprechend anwendbar.
AG Hildesheim, Beschl. v. 18.9.2008–47 C 53/08
Aus den Gründen
Unstreitig hat kein Termin stattgefunden. Deshalb ist auch für eine Terminsgebühr nach Nr. 3104 VV grundsätzlich kein Raum. Die Ausnahmevorschrift der Anm. Abs. 1 Nr. 1 zu Nr. 3104 VV ist – wie alle Ausnahmevorschriften – eng auszulegen. Sie kann nicht durch teleologische Überlegungen überwunden werden. Es ist nicht Aufgabe eines Gerichts, sich an Stelle des Gesetzgebers zu setzen und die Vergütungsregeln des RVG in sich stimmig zu machen – was an so mancher Stelle erforderlich wäre.
Da die Gebühren des RVG ein Entgelt für den Arbeitsaufwand des Rechtsanwalts darstellen sollen und dann, wenn keine mündliche Verhandlung durchgeführt wird, entsprechender Arbeitsaufwand nicht erbracht wird, disponiert der Richter, der im einstweiligen Verfügungsverfahren ohne mündliche Verhandlung entscheidet, auch nicht in unzulässiger Weise über die Vergütung eines Rechtsanwalts. Vielmehr ist die unterschiedliche Vergütung je nach richterlich gewählter Verfahrensweise nur Folge des von der Verfahrensweise abhängigen unterschiedlichen Zeit- und Arbeitsaufwandes des involvierten Rechtsanwalts.
Mitgeteilt von Rechtsanwalt Peter Dreymann, Bockenen
LikeLike
Everything comes down to patient No. 3, so let’s look at that case. There are a few things which strike me in the article https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4333610/
1. Why was that patient not monitored after the immediate weeks following the operation until 2.5 years later? Is that a normal approach of any doctor or scientist doing an experimental procedure? Isn’t the trachea one of the easiest tissues for monitoring?
2. How was the biopsy after those 2.5 years done? And did the physician of the patient, who apparently kept severe problems with his trachea according to the book by Albrecht (which is not mentioned at all in the article by Walles, as Leonid stated!), agree with such biopsy for research purposes?
3. The patient is described as healthy and even playing soccer. That is a very different picture from that given by Albrecht.
4. Why does the article describe absence of immune reactions, whereas Albrecht appears to describe an inflammation lasting for months? Walles repeats the acceptence by the absence of immune rejection under oath in his lawsuit against Leonid.
5. Where is the evidence that the transplant was integrated? It reads as if it may well have worked as an organic band-aid which eventually dissolved. Wouldn’t be bad, but far less spectacular than “an artificial organ”.
6.It probably can’t be decided from this single patient case whether the operation was successful or not, because we can’t know what would have happened without the transplant. If Walles didn’t monitor the patient for 2.5 years, and that patient suffered severe throat problems as described by Albrecht (for whatever reason), Walles can’t know for sure whether those problems had something to do with his operation or not. He only looked after 2.5 years at the patch (transplant) site, but that doesn’t exclude the possibility that inflammations caused by the transplant within those 2.5 years did severe damage.
The crazy part I find the lack of monitoring after the patient left the hospital.
Maybe the physician of this 3rd patient can tell us more?
LikeLike
I should add that this was a biopsy, at the 2.5 years, which included the surface-underlying vascular tissue and kept it intact. I there any reader here who can comment on this type of biopsy for tracheal tissue? Not monitoring for 2.5 years by bronchoscopy and then “suddenly” a rather severe looking biopsy? Is that normal?
LikeLiked by 1 person
In his declaration under oath, Thorsten Walles explains that the suicide of patient No.3 was told to him as well as that it was “aus Eigener Erfahrung bekannt” (known by his experience) whereas Heike Walles only explains that the fact was told to her. Can it be that Thorsten saw the dead body?
LikeLike
As for the case, address the fact that the Walleses under oath declare that there was acceptance by the immune system. They did not present evidence for having found the transplant again, so the immune system may have destroyed it (if so then replacing it by self-tissue, there is something promising in their procedure I have to admit). Plus a book describes a several month inflammation. Plus the Walleses didn’t monitor the patient for 2.5 years, so how should they know. If the declaration under oath is not correct, or can’t be substantiated, presumably the whole case has to be dropped.
Just ask what they mean with their statements. Your requested reaction within two weeks (that’s really fast, normally you can get extensions for that) could consist of questions which you would like to have clarified first.
LikeLike
Critical for their claim that you are wrong is their statement that patient No.3 was healthy. In the Steinke article they explain that “By that time, the patient reported that he could eat and drink normally and was also able to play soccer”. Curiously, they do not repeat the soccer playing in their declaration under oath, although there they do mention that another patient went hunting (for which less “breath” is needed). It is important for you and the judge to be able to assess this situation, so ask them if the Walleses talked directly to the patient 2.5 years after the operation at the time of the biopsy.
It is also important for you to be able to understand what is written in the verdict. Based on “aus eigener Erfahrung bekannt” with the suicide of the patient, written in the declaration under oath by Thorsten Walles, ask them if Thorsten Walles had contact with the corpse (the suicide was roughly 2.5 years after the operation, if I understand correctly), and which of his estimations are based on the possible contact with the corpse. Estimations from a corpse from a train accident should have their limitations in assessing the well-being of a patient, and probably would make them inferior tho the ones by Abrecht (who also seems to be an educated doctor).
So ask.
LikeLike
What also confuses me is that in the court order the Walleses say that patient No.3 probably had a tracheostoma because he could not eat normally, whereas in their article they celebrated his ability to normally eat and drink.
The language in the court order is also puzzling. It says something like “apparently a tracheostoma was created by a third party”. Why did the patient not consult with the Walleses, so that at least they wouldn’t have to use such distant type of languae? And if the patient broke contact with them, why then after 2.5 years he (discussing with the third party doctor?) allowed the Walleses a pretty severelooking biopsy?
Most of all puzzling is that the court order says nothing of the kind that the patient could eat or drink normally, or play soccer. Such would have been quite helpful for the Walleses to counter the descriptions by Albracht who describe a quite miserable state. In the court order the Walleses only say that the patient lived 2.5 years longer, and that the grafted tissue was accepted by the immune system. However, like in the article, no evidence was presented of finding back the transplant, so this acceptance by the immune system can be questioned.
LikeLike
And if the patient broke contact with them, why then after 2.5 years he (discussing with the third party doctor?) allowed the Walleses a pretty severelooking biopsy?
A cynical interpretation is that the biopsy was obtained post-mortem.
LikeLike
But by definition a biopsy is from an alive patient, isn’t it? And the paper describes that by the time of the biopsy the patient was eating, drinking and playing soccer. No, that can’t be, that it was post-mortem. Even though there was little time between November 2011 when Albrecht met the patient, and did not report about the biopsy as far as I know, and December 2011 when the patient committed suicide. Maybe the patient, despite his desolate state described by Albrecht went to visit the doctor that he hadn’t seen for 2.5 years to have part of his tracheal tissue removed for examination, told him he was eating, drinking and playing soccer, and that maybe Walles at the end of the same month had to investigate the dead body.
I think you are too cynical, because how could researchers who would do such thing, namely declare a post-mortem investigation as a biopsy of a soccer playing patient, go to court to accuse another researcher for inaccuracy?
For sure we are missing something, and the responsible university will be able to shine light on these matters and relieve us from our disturbing cynical thoughts.
LikeLike
In the BMBF report, Thorsten Walles writes the biopsy was performed in first half of 2011. The patient was operated in late April 2009, which would make it 2 years. The Steinke et al 2015 paper clearly speaks of 2.5 years. The Clinic Schillerhöhe refuses all information citing doctor-patient confidentiality. The patient Singh expressedly waived this confidentiality, so his medical files could be made available to the journalist Albrecht.
LikeLike
Does Albrecht have any information about the biopsy?
LikeLike
As I understand, Thorsten Walles closed the tracheostoma of patient No.3 during the initial graft operation. This suggests that he considered the operation to take away the need for the tracheostoma. If the tracheostoma had to be introduced again, that probably gave you sufficient reason to assume that the operation did not result in the desired effect (failed). This is especially so because in several reports the Walleses claimed that the patient could eat normally. That now in their lawsuit the Walleses indicate that the tracheostoma probably was introduced because the patient had severe problems with eating, may have to be neglected by the judge.
LikeLike
T. Walles did not re-open the tracheostomy, other doctors did. According to Albrecht book, T. Walles did not see the patient for over 2 years after the operation, which took place on 27th April 2009. I am not sure how it allows Dr. Walles to make in court distance retrospective diagnosis about the medical status of the patient and the graft performance during that time. Neither do we know when exactly the alleged biopsy was performed. T. Walles claims it was done 2 years after in the BMBF report (“first half of 2011”), and 2.5 years after in the Steinke et al paper. The patient committed suicide 2.5 years after the transplant.
LikeLike
Pingback: Evidence is a lie, listen to our authority, say Walles to scientists – For Better Science
Pingback: Image duplication and other irregularities of Heike and Thorsten Walles – For Better Science
Pingback: My Walles trachea transplant reporting fails peer review – For Better Science