Olivier Voinnet, the disgraced former star plant scientist and professor at ETH Zürich, is apparently on extended sick leave, his lab members have been redistributed to other research groups inside the faculty. This I learned from several independent sources, which made the information sufficiently reliable to share here. Previously, Voinnet was investigated by two expert commissions, one very secret by CNRS at his former Institut de Biologie Moleculaire des Plantes (IBMP), and another, more transparent one, at ETH (report here, my overview of the Voinnet scandal here). There, the investigative team comprised of four peers, two of whom were Voinnet’s faculty colleagues, and one was Witold Filipowicz, professor at Friedrich Miescher Institute in Basel. Filipowicz had been evaluating Voinnet’s research as IBMP review board member in 2008, before he nominated him for the 2009 EMBO Gold Medal:
“Olivier Voinnet’s discoveries represent true breakthroughs in his field. He has written several illuminating reviews recently, and participated as a speaker in many prestigious meetings. I consider him to be one of the most talented, original and effective young scientists”.
Update 8.04.2016: The supposedly impartial Voinnet investigator Filipowicz was also a recipient of the 2014 Chaire Gutenberg at Voinnet’s own IBMP as well as neighbouring IBMC in Strassbourg. The Gutenberg Chair is financed by the Alsace Region and the Urban Community of Strasbourg with € 60,000 of which € 10,000 went to Filipowicz personally as ‘Gutenberg Prize’ and € 50,000 were awarded to his host, the teams of LabEx NetRNA of IBMP and IBMC. Coincidently, one of LabEx NetRNA teams is still headed by Voinnet’s IBMP lab keeper and key partner in data manipulation, Patrice Dunoyer.
Under such conditions, it is hardly surprising that the ETH investigative commission concluded that Voinnet’s research was still largely reliable, despite his inexplicably compulsive urge to manipulate his perfectly good experimental data. As ETH press release then announced, Voinnet research was “Conducted properly – published incorrectly”. Well, this depends what ETH leadership understands under proper research.
Below I will show evidence from Voinnet’s peers that the published experimental evidence for his bold discoveries was shaky even before the data manipulations were discovered. Finally, I could not find a single lab which could confirm to me that they reproduced his results.
Instead I found those, who were able to disprove them. What is left of the whole affair, are seven retractions and a large body of corrected, but very questionable literature. These zombie papers continue to pollute the research field of RNA interference (RNAi) and of the wider life sciences. The most notorious paper in this regard is Deleris et al Science, 2006, which even the ETH commission wanted to see retracted, but which the journal chose to correct instead.
An interested reader of my blog, who is also an active scientist, chose to scrutinize this paper and its correction.
The scientist (whose image analysis and summary report are available for download), judges:
“It is a quantitative study (“hierarchical action”) in which the observed ratios of siRNAs within samples and the ratios of the amounts of viral RNA between samples were essential for making the conclusions. Regardless of the picture manipulations, it is already absurd that the experiments for the six figures were seemingly only done once, because this type of quantitative analysis requires repeats and statistical evaluation. The exchange of the correct pictures for the erroneous/manipulated ones led to differences in perceived ratios of amounts of RNA, making the figures agree better with the explanatory models postulated by the authors. At least in several cases this appears to have been done intentionally.
A recurrent reason for picture manipulations seems to have been that the authors did not include internal controls in their gel experiments, despite that such is a prerequisite for allowing quantitative comparisons. Several of the manipulations concern the inclusion of fake controls”.
James Carrington, head of Danforth Plant Science Center in USA and Voinnet’s collaborator on the Deleris et al paper, previously claimed no knowledge of the upcoming correction. He did not reply to my request to comment on the above analysis.
Update 17.04.2016. After I forwarded this article to Science senior editors and AAAS leadership, the journal’s Editor-in-Chief Marcia McNutt has commented over Twitter:
What about Voinnet’s other papers, especially those not even corrected so far?
Prior to the scandal, Voinnet used to be one of the leading scientists in the research field of plant pathogen defense and RNA interference. Voinnet’s PhD advisor, David Baulcombe, was one of the original discoverers of the RNAi mechanisms of plant defence against viruses. The Nobelist Tim Hunt expressed in a Science article his surprise that Baulcombe’s then-PhD student Voinnet did not receive the 2006 Nobel Prize for his role in the discovery of RNAi.
The science behind the discovery is such: the viral mRNA is met inside plant cells by their matching anti-sense RNA, which then together form double-stranded RNA. These RNA molecules are then degraded by DICER proteins into the short interfering RNA (siRNA) duplexes. When loaded into the appropriate RISC protein complex, siRNAs help recognize and destroy the corresponding viral mRNA in a sequence-specific manner. There are different DICER-like (DCL) proteins, and different RISC complexes, which adds a wide range of diversification to the RNAi machinery. In the standard plant model organism Arabidopsis DCL3 generates siRNA fragments which are 24 nucleotides long, these are loaded into a RISC complex containing the AGO4 protein (of the Argonaute family). The 21-nt long siRNA is formed by DCL4 and is loaded onto AGO1-containing RISC complex. All these details become very important when discussing the true impact of Voinnet’s RNAi research.
Voinnet continued working on RNAi in plants. His biggest discovery as young research director at the in Strasbourg IBMP was the nature of the mobile silencing signal in plants. The phenotype, observed by plant scientists from grafting experiments (where a root from the plant of one genotype was grafted to a shoot of a different genotype), was striking: plants were able to initiate gene silencing throughout long distances, across the graft (Palauqui et al, EMBO J, 1997). When a transgene in one part of the plant becomes silenced by RNAi mechanism, this silencing can spread throughout the organism. First the signal spreads locally, from plant cell to plant cell through the plasmodesmata. Eventually, it reaches the phloem and then spreads systemically to the cells at the opposing end of the plant. There is obviously a mysterious mobile signal that spreads the silencing form one end of the plant to another. But what is it?
Until Voinnet postulated his evidence; the issue was an academic debate with many competing hypotheses (and now, it turned back to research and debating). Voinnet’s papers in Science and elsewhere proved that it was the siRNA which was the mobile signal. Now, that Voinnet’s authority is somewhat dented due to his excessive data manipulations, how reliable are his discoveries?
In fact, Voinnet’s own papers, regardless of the data manipulations therein, carry little experimental evidence for siRNA being the mobile signal. This I was told by Dacheng Liang, a former postdoctoral scientist in the lab of Peter Waterhouse at Queensland University of Technology, Australia:
“In all his early papers, e.g. his Nature Genetics papers Dunoyer et al, 2005 and Dunoyer et al, 2007 Voinnet claimed that 21-nt small RNA, rather than 24-nt long RNA, is the mobile silencing signal”.
As Voinnet himself wrote, his model of 2005 “excludes the participation of 24-nt siRNAs in movement and also predicts that, in the absence of active amplification, the extent of short-range cell-to-cell movement should correlate with the levels of primary 21-nt siRNAs accumulating at the site of silencing initiation.” Also in 2007, Voinnet insisted “neither DCL3 nor AGO4 is required for non–cell autonomous RNAi“. Also in his now corrected paper Dunoyer et al Science 2010, Voinnet reiterated the role of the 21-nt siRNA as the mobile silencing signal, yet he cautiously added in the last paragraph: “Of the 21- and 24-bp siRNA species, we studied only the former in this experiment, because AGO1-dependent RNAi generates a measurable silencing movement phenotype […]. Yet, there is no reason to exclude mobility of DCL3-dependent 24-nt siRNAs, which mediate locus-specific chromatin modifications upon loading into AGO4“.
Why did Voinnet add this clause? Because in the same Science issue, his former mentor Baulcombe published a paper presenting the 24nt siRNA is mobile silencing signal (Molnar et al., 2010). In the EMBO J paper of the same year (Dunoyer et al, 2010, now retracted due to heavy image manipulations), Voinnet suggested that both 21-nt and 24-nt siRNAs were relevant, since his “previous study more specifically identified the DCL4-dependent, 21-nt siRNA as being both necessary and sufficient to mediate non-cell autonomous RNAi, leaving open the possibility that 24-nt siRNAs also can move […] at a distance”.
So basically, Voinnet quietly reversed his original claim of the exclusivity of the 21nt siRNA as the sole mobile signal. Yet this is in comparison a minor issue with his papers, and again, even if one for the time being ignores the data manipulations. Liang explains that Voinnet et al actually never really showed that any siRNA, regardless of its size, was the mobile signal:
“All the genetic factors they found in his 2005 and 2007 Nature Genetics papers are responsible for the generation of silencing signals, or small RNAs. But as to whether these are moving or not, the authors did not show any solid evidence regarding the mobile process, movement rate, or the recipient cells responding to any of the classes of small RNAs. And also, the suc2-P19, suc2-P21 and suc2-DCL4 transgenic experiment in his 2010 Science paper is a key experiment to show the generation of 21-nt small RNAs, but unfortunately they did not give any detailed descriptions or statistic data about those transgenic plants”.
The absence of usable statistical data is apparently a recurring feature in Voinnet papers. Yet this apparently never troubled much the editors or peer reviewers at Science, even when Voinnet’s scientific claims were long disproven, by the research community and even Voinnet himself.
Instead, all Voinnet could show, is that siRNA can move between neighbouring cells though his bombardment experiments with siRNA-coated gold particles. This movement through plasmodesmata however is hardly the same as long-distance silencing though the phloem. Yet according to Liang, Voinnet’s papers never demonstrated “that the labelled siRNA can be detected e.g. in the root if it is bombarded into the leaf or vice versa in the perspective of long-distance mobile signal”.
Liang was therefore already sensitized to Voinnet’s peculiar research when he noticed that Voinnet has modified and re-used a published image in his 2010 Science paper. He then reported his criticisms on his Chinese-language blog.

But, as anyone who follows misconduct cases knows, data integrity is often deemed as a minor or even irrelevant issue, when the faked results are being claimed as reproducible otherwise. This was surely the case with ETH, who described Voinnet’s figures as mere “illustrations”, as if a scientific research paper was a children’s book. CNRS’ attitude was not really better.
So, what about Voinnet’s later discoveries on siRNA as the mobile signal in plant immunity? How reproducible are those?
Voinnet’s former PhD advisor Baulcombe is certainly the right authority to ask here. His lab proved to be the only one, aside of Voinnet’s, to have ever published on siRNA as mobile signal, and in Science no less. To me, Baucombe stated in an email in June 2015:
“I think you will find that several labs have published on movement of siRNA including Waterhouse, Lucas, Kehr, Kragler and Vaucheret is, of course, a pioneer of mobile silencing. There is no postulated concept of an exclusive siRNA signal – there may well be many forms of mobile RNA. We have made that point in our discussion sections and reviews”.
I approached all of the plant scientists Baulcombe named, and some more. If any of them ever reproduced in their labs Voinnet’s (and Baulcombe’s) claim of siRNAs as the mobile signal, they decided to keep this vital information secret from the prying public.
Julia Kehr, professor for molecular plant genetics at University of Hamburg, explained:
“We do not work with siRNAs, but with miRNAs, which can be transported over long distances. We could show that numerous miRNAs can be found in the phloem (Buhtz et al 2008, Plant J 53) and at least two nutrient-deficiency associated miRNAs (miR395 und 399) proved movable through phloem in grafting experiments […]. Voinnet always claimed that miRNAs, unlike siRNAs, are only active locally (e.g., Parizotto et al., Genes & Development 2004), yet in 2009 he admitted in regard to published results for miR399 that at least some specific miRNAs can act as systemic signals in exceptional circumstances (Voinnet, Cell, 2009)“.
Hervé Vaucharet, research director at the INRA Centre in Versailles (with whom the young student Voinnet once interviewed to do his PhD thesis, before deciding to work with Baulcombe instead) did not reply to me on this matter. Friedrich Kragler, group leader at the Max-Planck-Institute for Molecular Plant Physiology in Potsdam, ignored every single one of my emails. Same goes for William Lucas, professor for plant biology at UC Davis, USA and finally, Peter Waterhouse.
Instead, Waterhouse’s collaborators offer some insights. His former postdoc Liang stated:
“I don’t think Peter’s lab has shown that siRNA is the long-distance mobile signal. This is really hard stone to crack. And the evidence from Baulcombe’s 2010 Science looks to me weak if you carefully check the data from the whole paper. The problem is, everyone tends to believe that small RNA is the mobile signal because it’s small and also the key player in gene silencing pathway. Either to prove it or disapprove it is very time-consuming, and might not generate any results. Just as Voinnet lab, they used labelled siRNA to show its mobility, but they just showed it can move to neighbor cells”.
Rosemary White, group leader at CSIRO Black Mountain in Canberra, agreed to publicly explain the science of siRNA and the mobile signal.
White used to be a close collaborator of Peter Waterhouse (Liang’s ex-boss), and surely even Baulcombe would agree that her views on this are relevant:
“Much work, both before and after Voinnet’s published work, showed long-distance movement of a range of molecules (and now, even organelles), in the phloem. I suspect that anything that is in the phloem may be able to move long distances through plants. See, for example, a couple of recent grafting papers (Melnyk et al 2015; Lewsey et al 2015) and other recent papers showing that mitochondria and other plastids can also move across graft junctions (Thyssen et al 2012; Gurdon et al 2016). I have also seen many different GFP-tagged proteins move through phloem across graft junctions. This does not seem contentious at all. There are several other papers showing long distance spread of small RNAs.
We and others have also shown that phloem is quite capable of bidirectional transport. This has long been known for plants with both internal and external phloem, such as the various members of the tobacco and cucurbit families, popular subjects for investigation of phloem transport. But this is true even for Arabidopsis phloem, as shown by us (see Fig. 2 Liang et al 2012) and earlier by Kim et al. 2004 (see Fig. 4), and by several others using smaller fluorescent tracers.
We also showed that siRNA could spread long-distance, but only cell-to-cell (http://www.plantphysiol.org/content/159/3/984.long). We added the caveat that this may be a particular feature of siRNAs, generated from artificial hpRNA, which then target non-endogenous mRNA (GFP, in our case). The most interesting (to me) and novel aspect of our paper was that the signal appeared not to spread via the phloem. We discuss implications of this result. Considering that very many RNAs of different sizes are found in phloem exudates, it would be surprising if phloem-mobile siRNAs were not signals for at least some responses. My own suspicion is that this is only the tip of the iceberg as far as signalling molecules in the phloem”.
White concludes:
“I find it very difficult to trust anything in any of his papers. Fortunately, once the field moves on, there may be diminishing need to refer to Voinnet’s publications”.
What about other peers of Voinnet?
In 2010, the American plant scientists Dan Chitwood and Marja Timmermans wrote a review in Nature about Voinnet’s and Baulcombe’s discovery of 21-24-nt siRNAs as mobile signal: “Small RNAs are on the move”. Is the information presented in their Nature article still reliable?
Chitwood, group leader at Carrington’s Donald Danforth Plant Science Center, wrote to me:
“That a mobile silencing signal exists is not in doubt. The precise identity of that signal, and how its mobility changes in different developmental contexts and which small RNA pathways have mobile activity, is more complicated, and remains an area of active research. These two papers [Dunoyer et al, Science 2010 and Molnar et al., Science 2010 , -LS] are critical for the idea that small RNAs themselves (not precursors, for example) are the mobile silencing signal. Like all papers, the persuasiveness of the results stands on performing correct controls and faith by readers that the figures and images in the paper reflect experimental reality and have not been manipulated”.
Timmermans, formerly at CSHL and now professor for developmental genetics at the University of Tübingen in Germany, explained that her lab, just like that of her new German colleague Kehr, only works on miRNAs and not on siRNA. She however withdrew all her previous quotes to me in regard to Voinnet’s research on mobile signal and his papers.
Timmermans also advised that I contact “for additional information” Detlef Weigel, director of the Max-Planck-Institute for Developmental Biology in Tübingen. Weigel however admitted: “My own research group largely gave up the topic of small RNAs”, without further explanations.
But surely Shou-Wei Ding, who personally co-authored several papers with Voinnet, including the seminal Science paper on mammalian RNAi, and a review on mammalian siRNA viral defense, can confirm the discovery of siRNAs as mobile signal in plants? Not really, since Ding could only tell me:
“Unfortunately, my lab does not have funding to work on the silencing signal, and thus won’t be able to help you”.
In summary, I obtained despite my long and intensive search not a single shred of reproducibility evidence for siRNA as mobile signal, from all the plant scientists I approached. Everyone can now make their own conclusions on how reliable Voinnet’s discoveries and what exactly the point of saving his papers was.
What about that of RNAi in mammals, also published in Science (Maillard et al., 2013)? The scientist, who openly criticized this paper and the corresponding Letter in Cell Reports by Ding and Voinnet, was then accused by Voinnet of masterminding the PubPeer smear attack against him. The ETH investigative commission apparently deemed this utterly false and bizarre conspiracy claim credible enough to make it public, uncommented.
Is the nightmare finally over? Many in plant sciences and wider academia prefer to dismiss Voinnet as a case of individual dishonest scientist. To them, science has self-corrected itself, time to forget and move on. Noone involved was sacked (as it seems), yet they all are supposed to have learned their lesson.
Voinnet case is not a stand-alone incident of a dishonest scientist. It is also a failure of the entire plant science community, who even now are largely too scared to air their dirty laundry. Indeed, they have every reason to be: Voinnet and every single one of his main co-authors worldwide are still in their influential faculty positions.
What we are now witnessing is not a self-cleaning, but a chronic failure of science and of academic institutions, like:
- French research center network CNRS, who re-defined what scientific fraud was to ensure Voinnet could not have committed it, kept the entire investigation totally secret, and retained his right hand Patrice Dunoyer in his job as IBMP research group leader.
- Swiss elite university ETH Zürich, who decided to support Voinnet after their own overtly benevolent investigation and failed to even formally interview his ex-team member and presently ETH professor as well, Constance Ciaudo.
- British University of East Anglia, who already trice refused my FOIA request about their investigation into Voinnet’s manipulated PhD thesis. Since I know from reliable sources that this investigation did happen, I must assume the university either buried it, or already acquitted Voinnet against all evidence.
- Elite journals like Science, PNAS, Genes & Development, RNA and others, which unscrupulously allowed Voinnetting to happen. Their editors obviously decided that a research paper can still have its full value, despite deliberate data manipulations with the very likely intention to deceive. Yet by far the worst offender in this regard is Cell Press, which quietly keeps on ignoring all evidence for data manipulation in 3 papers in Cell (Voinnet et al, 1998: Voinnet et al, 2000, Chow et al, 2010, and one paper in Molecular Cell (Garcia et al, 2012). It seems for the Elsevier-owned Cell Press and the EiC Emilie Marcus, the Voinnet scandal either never happened or is long forgotten.
Some publishers, like PLOS and EMBO Press were brave enough to retract Voinnet papers beyond or even against the recommendations of ETH commission. Especially the European research society EMBO, initially reluctant to get involved into the Voinnet affair, acted most exemplary afterwards. Following their own investigation, EMBO withdrew the Gold Medal from this ETH professor and French Academy of Sciences member.
The way how Voinnet misconduct has been dealt with, makes a dangerous precedent. Voinnetting of manipulated papers can soon become a standard rather than exception, and zombie scientists will multiply.


Great job, Leonid.
LikeLiked by 1 person
Yes Leonid, great job, including getting some kind of reply from Marcia McNutt. Maybe you can ask her:
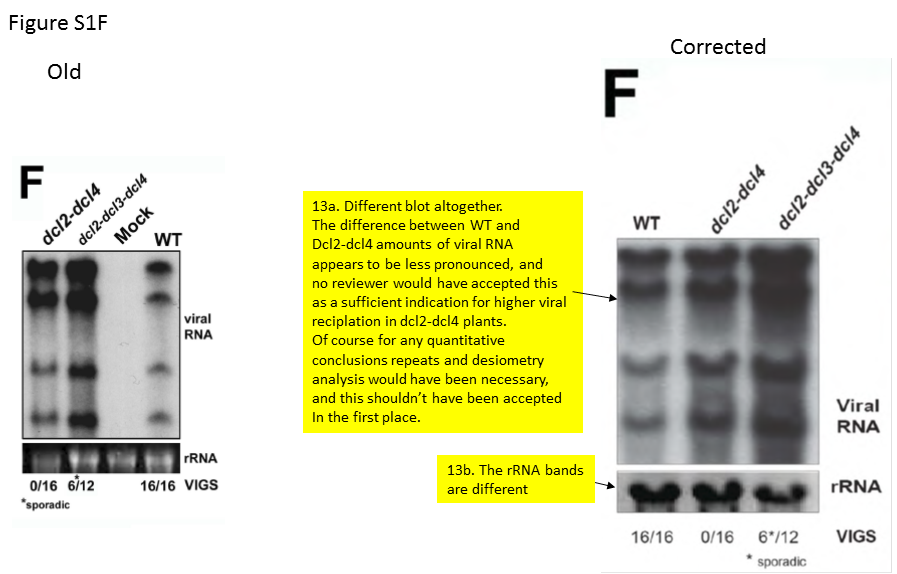
1. How can Fig. S1F be considered a correction, since it only shows that there was no evidence for the conclusion “TRV-PDS accumulates more in the dcl2-dcl4 double mutants”? Or is Science policy that a single experiment showing minimal differences is enough for such conclusion?
2. How can the manipulation of Fig. 1D, which required a fully unnatural action, be an innocent mistake of any kind? Isn’t Science policy that willful manipulation of data should result in retraction? And if it is willful manipulation, wouldn’t it be weird if the conclusions were not affected?
3. How do Science and AAAS feel to stand at the far lenient end in regard to retraction policy, like in comparison with EMBO, in regard to a paper with >6 admitted manipulations, for which the host institute recommended retraction, and in regard to authors with a multitude of proven misconduct? Is such policy ever voted on by AAAS members?
LikeLike
One possible answer may lie in what advice McNutt may have received from Science Editors, specifically those involved with plant science:
http://www.sciencemag.org/about/editors-and-editorial-boards
A glance at the editor board suggests that select individuals (below follows a non-exhaustive list) should be contacted about this issue (perhaps Dr. Schneider could follow up on this).
Jeffery L. Dangl
University of North Carolina
http://bio.unc.edu/people/faculty/dangl/
Ottoline Leyser, University of Cambridge
Director The Sainsbury Laboratory*
http://www.slcu.cam.ac.uk/directory/leyser-ottoline
Jane E. Parker
Max-Planck Institute of Plant Breeding Research
http://www.mpipz.mpg.de/parker
Alison M. Smith
John Innes Centre
https://www.jic.ac.uk/directory/alison-smith/
Note (think of this as a hypothesis): Professor David Baulcombe, formerly of the Sainsbury Laboratory, and a key co-author of several Voinnet papers, has surprisingly been void of any scrutiny. One also sees zero public input from Baulcombe or other high-profile co-authors. Why is that? Therefore, to understand why McNutt would have selected a correction rather than a retraction may require a deeper analysis if there was any bias, influence or pressure by any plant science related editor board member or related individuals (co-authors or individuals in a high profile plant science research institute some how related to or implicated in the Voinnet scandal).
LikeLike
I believe that if you puruse the pages of Science, you will find an article claiming the existence of DNA with an arsenate ester (so-called ‘arsenate DNA’), which is completely discredited. Nevertheless the article still stands (http://science.sciencemag.org/content/early/2010/12/01/science.1197258). So unlike EMBO J and the oft criticised PLOS stable, even in the face of incontrovertible evidence (the simplest and perhaps most damning being the well-known instability of arsenate esters in water!), Science has done nothing. Note it is 6 years since this matter was ‘closed’ from a scientific standpoint.
Also note that this does not prevent this journal (and a good many others) from parading their Imposing Factor and for far too many scientists reacting with shock and awe, usually reserved for religious revelations to the believers, to the presence of papers in such journals on a CV.
LikeLike
Maybe I can add about Marcia McNutt’s comment “Correcting paper was in best interests of advancing science, transparency, and for junior authors”:
-Exposing the pictures that Voinnet did not want to show is not the same as correcting
-The Deleris et al. story is not supported by the data, thus is not transparent
-This is a paper from 2006, who is the junior author that needs protection?
-If Science as a journal wishes to have its own voice in scientific debates, as it does when it comes to climate change, it should try to develop a respectable reputation regarding scientific ability and integrity. The Science effort to save papers that cannot and should not be saved only amounts to the opposite.
The ETH committee went out of its way to have a gentle overall verdict over Voinnet, but nevertheless believed that the Deleris et al. paper could not be salvaged. Doesn’t that ring any bells at the Science office?
LikeLike
The Deleris paper sticks out like a sore thumb drawing fire. Voinnet is going for a second career chance, which is given to him by the ETH (and that’s fine with me). Before that, they intended to clean up the mess, which includes retraction of the Deleris paper which they declared “NON-FACTUAL”.
– From https://forbetterscience.wordpress.com/2016/01/21/olivier-voinnet-case-correcting-the-uncorrectable/ :
The ETH report originally decreed:
“Although it is obviously the journal’s prerogative, the former (category 2) papers, particularly those containing well documented intentional manipulations (PLoS Pathogens 2013 9:e1003435; Plant Cell 2004 16: 1235; Science 2006 313: 68; PNAS 2006 103: 19593 and EMBO J 2010 29: 1699), should be retracted through OV’s requests as being non-factual, irrespectively of whether the reported observations have been reproduced by others.” –
Interesting is that Marcia McNutt, though in a different context, was referred to and quoted http://ngm.nationalgeographic.com/2015/03/science-doubters/achenbach-text :
– Science appeals to our rational brain, but our beliefs are motivated largely by emotion, and the biggest motivation is remaining tight with our peers. “We’re all in high school. We’ve never left high school,” says Marcia McNutt. “People still have a need to fit in, and that need to fit in is so strong that LOCAL VALUES AND LOCAL OPINIONS ARE ALWAYS TRUMPING SCIENCE. And they will continue to trump science, especially when there is no clear downside to ignoring science.” –
If Voinnet has no interest in keeping this paper alive, if junior authors that might have needed protection 10 years ago are not so junior anymore, and if even for Science it seems like a silly reputation risk to not retract a paper despite such recommendation by the author and host institute, who or what then represents the local value that trumps science?
LikeLike
A co-author, Carrington, here http://www.plantcell.org/content/22/2/481/F5.large.jpg shows that he perfectly understands how this type of quantitative analysis should be performed (repeats, quantification). How can he then agree with the Deleris paper being “corrected” while only the lack of data for making conclusions has now been exposed? If they really want to correct it, more experiments are needed.
The same Carrington paper http://www.plantcell.org/content/22/2/481.long suggests that Voinnet was wrong in faking (helping the readers to see) a stronger 24-nt siRNA band in WT background. So how can Carrington, who immediately should spot this, then agree with the statement “conclusions were not changed”?
If people want to correct instead of the retraction recommended by the lead author and host institute, at least they should do it serious.
LikeLike
At http://science.sciencemag.org/content/352/6284/387.full Marcia McNutt states:
Editors are often caught in the crossfire between impatient readers who may reach conclusions about the validity of misconduct allegations on the basis of incomplete information online, and authors who are concerned about damage to their reputation. In the final analysis, the editor’s paramount concern should be for the integrity of the scientific record. –
and
…. the summit attendees agreed that only institutions had the necessary access to conduct investigations, …. –
In case of the Deleris paper, where is the crossfire? The lead-author and the home institute stated that the manuscript was non-factual and recommended retraction. And it is right, only the institute can investigate deeply beyond what is written in the text. So why neglect them?
LikeLike
If it wouldn’t be so sad for the climate debate, one would say that Science (and American science?) deserves someone like Trump. It is quite funny to imagine a Trump rant on the Science-to-Voinnet attitude and credibility issues.
LikeLike
The Voinnet case is only the tip of the ice-berg of the misconduct and problems underlying plant science. Once can begin to grasp a notion of the complexities here that began to be recorded since January 2014:
As for Voinnet, I personally wrote to him and his students indicating that I was displeased with the fact that he had not taken the responsibility of contacting all journals that had cited his retracted and corrected papers to issue a suitable erratum. The literature remains corrupted, and now he is on “sick leave”. Well, when he gets back, I hope he and his collaborators spend a few minutes of every day cleaning up that literature. This includes his co-authors like Baulcombe, Carrington, Deleris and others.
In addition to the spot-on concerns made by Dr. Schneider about wide cronyism and zombieism in the plant sciences, especially among highly ranking elites, one has to ask: how many of Voinnet’s studies still remain problematic? I suspect that a second wave of PPPR is going to be necessary.
LikeLike
I’m guessing you’re the person with all those ‘query’ entries on that thread, huh? I think Pubpeer would be better since it’s not so obvious that it’s the same person all the time and the journals would not be put off as much as they often are when there is a lone avenger who they would like nothing more to ignore as a ‘one-off crank’. Putting everything into an old Ret. Watch thread might be a waste of time. Plus, you need to make cool ‘imgur’ graphics like Claire Francis used to do for all the Frankengels for people like me with short attention spans (but you know, since you’re already sacrificing so much time on this, don’t get mad at me…).
LikeLike
“Olivier Voinnet, the disgraced former star plant scientist and professor at ETH Zürich, is apparently on extended sick leave.”
I’m sorry to hear this. I hope his illness is not stress-related. The situation with Voinnet’s work is very serious, of course, as Leonid has aptly discussed, but there’s more to life than science and it’s never a good thing when someone suffers from depression as a result of work worries.
LikeLiked by 1 person
Me, I suffer from depression because I have been struggling all my life to get a job in science… I kept telling myself I had to be honest, not fake results, not turn a blind eye on any dubious result.
What came out of it? No job, too old to find another job and I see all of Voinnet’s coworkers at the top of the science “food chain”! I am depressed… And I blame it all on Voinnet and all he represents in science!
LikeLike
true or fake?
LikeLike
I strongly agree!!!! Is not only in the plant science field this happens…..but in cancer, neuroscience, cardiology….., any field of science….and if you want to work in science often you will have to work like Voinnet!
But nevertheless hoping in future the situation improves as they are catching these cases!
LikeLike
Omertà in France, Switzerland, UK and other connected labs. A PhD is a publication and can be read in the library or a photocopy obtained by interlibrary loan….
As usual, despite the claims of self-correction, there is little evidence of it, instead we have clear protection of self-interest.
LikeLike
Dear Leonid,
You wrote “Olivier Voinnet (…) is apparently on extended sick leave, his lab members have been redistributed to other research groups inside the faculty. This I learned from several independent sources, which made the information sufficiently reliable to share here.”
I fail to understand why you are using the term “apparently” and why no one seems to be able to get for example an auto-reply from the e-mail account of Olivier Voinnet in which these kind of details are listed. See below for three examples of such auto-replies from researchers of three different universities.
“—– Original Message —– From: @csm.ox.ac.uk; To: Klaas van Dijk; Sent: Friday, April 08, 2016 1:17 PM
Subject: Automatic reply (..); I am away from the office until 11 April with no access to email. I will respond to you as soon as possible after my return. Kind regards (…).”
“—– Original Message —–; From: Simon Broadley; To: Klaas van Dijk; Sent: Saturday, March 12, 2016 10:55 AM; Subject: Out of Office – On Sabbatical Leave until 15 July 2016 (…); Dear Colleagues, I am currently on Sabbatical Leave and will return to the office on Monday 18 July 2016. In the interim, the Acting Head of School will be Professor David Ellwood. During this time, all urgent School matters should be forwarded to Professor David Ellwood. If you are requiring: an appointment with the A/Head of School or assistance with Head of School matters, please contact my PA Teresa Russo. For any Administration matters please contact Assoc Prof Ray Tedman. Regards Simon Broadley. Simon Broadley Professor of Neurology/Dean of Medicine School of Medicine Gold Coast Campus Griffith University QLD 4222 AUSTRALIA”
“—– Original Message —– From: @qut.edu.au; To: Klaas van Dijk; Sent: Friday, February 12, 2016 11:08 PM; Subject: Automatic reply (…); Thank-you for your email. I am on Parental leave from the 22 January until 28th November inclusive. I will only be checking my email sporadically during this period. Cheers (…).”
LikeLike
The Strasbourg address is dead
” olivier.voinnet@ibmp-ulp.u-strasbg.fr: Recipient address rejected: User
unknown in virtual mailbox table”
In contrast the ETH message is accepted, but no reply. Perhaps the therapy for the ‘stress’ that has been prescribed is to count the number of unread e-mails, rather like counting sheep?
It is of course a well established practice, when the unsinkable ship hits an iceberg to:
(1) Reverse, to scrape the ship against the ice and pop the maximum number of rivets.
(2) Re-arrange the deckchairs.
(3) Make sure the band plays on.
(4) On no account request anyone consider heading for the lifeboat, as that is reserved for the Captain.
LikeLike
Hoping in future we can do better and more serious science
LikeLike
HopeinFuture, not sure what Hope you are talking about. As a plant scientist who is both a critic and a defender of the integrity of the plant science literature, and who has been heavily invested, since 2014, in the Voinnet case and literature PPPR, especially at PubPeer, I felt that the demise of Voinnet represented a key event in plant science, and that it merited much greater attention by the peer community. PubPeer changed the title of my topic, and wiped out my introduction. For what reason? Voinnet represents a real risk to the paradigm of ethical safety in plant science. This was an important topic that needed a discussion thread. Yet, PubPeer, playing Big Borther, once again, manipulating the discourse, rather than facilitating it.
HopeinFuture, this is not Hope, this is the sign of the times. Below I add the full text which I take full responsibility for. Despite this, a total abuse by PubPeer of the freedom to be critical.
Olivier Voinnet, the Science Spring, and a call to the (plant) science community for greater PPPR
https://pubpeer.com/topics/1/9EF581519DBFA31BDACE6ACCF1F9EB
I feel highly compelled to issue this statement today, as a topic at PubPeer. I am a middle-class plant scientist of about the same age as Prof. Olivier Voinnet. This case resonates with many and touches all. I wish to share a few of my thoughts and concerns on this platform, PubPeer, which was essential in the downfall of what some are calling the greatest academic disgrace to befall plant science.
And today is an extremely sad day. But also, a day of great initial hope. For many reasons, the first being what the loss of the 2009 EMBO Gold Medal by Olivier Voinnet [1] represents.
The plant science community has put trust in Olivier Voinnet and his colleagues for decades. His work has been lauded, appraised, highly cited, and in most cases, supported or repeated. But 7 retractions and 22 corrections later, all within the space of one short year, and all as a direct result of the time, effort and courage of vigilant sleuths who care deeply about the state of plant science and about its injustices, both in research and in publishing, have indicated that there is hope in post-publication peer review (PPPR) in plant science [2].
In my own dealings, by name and anonymously, over the past few years, I have come to know the fierce opposition that challenging the plant science elite, and even the wider publishing elite, can entail. And who can blame them? Individuals who have invested in a life-time to develop careers will defend them tooth and nail. But what the Voinnet case represents is a new era in plant science. The resounding message that has been sent is that the community will no longer tolerate cheats, nor will it reward them. And EMBO has taken the bold and correct initiative by sending a clear and strong message: if you cheat the system to your advantage, causing irreparable damage of science, then you must suffer the consequences, including being stripped of one’s laurels. Your losses Voinnet, are our losses, too.
Due process is required, but what one short and tumultuous year in the Voinnet case has shown us is that there can be no compromising quality. PPPR is a painful process. It requires time, energy, and most importantly, the deep desire to correct the ills of the literature. Because it is that very same literature that we have used to cite our own work, it is that very same literature that is used to create a support basis for our up-and-coming youth who will hold the reigns of plant science’s literature moving forward.
Before we have the responsibility towards our own selfish careers and legends we have one greater responsibility, as scientists: to defend its integrity. And those who have gone into science for any other reason had better heed the lessons that are being learnt in the Voinnet case.
Ask yourself, do we want our young scientists to embrace distorted values and a corrupted literature? Or do we want to encourage them to speak out, if necessary, against injustice, against a corrupted literature and against excessively powerful individuals – in academia and in publishing – who have gained their power and position from abuse of research and publishing ethics? Plant science is in a revolutionary state, and yet 99% of the community remains either immune to it, or fearful of what this academic revolution represents.
My ideas have been criticized and shot down for years. My challenge to the system and to the powers that control it has led me to lose my career, and be banned from at least two main science publishers who fear my words of truth. Unfortunately, I suspect that this will not be the last of the bans because banning critics of the ills of a system only empowers them more and fortifies their resolve. We have the inherent responsibility, whether undergraduate students or seasoned professors, if we truly love and care for science, to nurture it, and to care for the product of the years of fruitful hard work: the published literature. Without a soundly tested and properly vetted literature, what is science?
The Science Spring is therefore in full swing. But those who concoct images of falling empires and crashing legends had better think again. There is an aspect of “cheating” that is deeply engrained in the publishing system, and that is unable to detect it. The correction of the literature is not a passive “self-correcting” task as some in the mainstream have suggested. No, correction of the literature requires a detailed, paper-by-paper, journal-by-journal and publisher-by-publisher reanalysis.
But who can do this task? Who is a peer? Who is qualified? Who has the right? These are all somewhat irrelevant questions. What PPPR and the correction of the literature requires is:
a) the desire to improve what is faulty;
b) creating a platform that allows for such a correction to take place.
In b), PubPeer provides one such powerful platform that, for now, respects the rights of the anonymous voices. But having this platform is not enough. Reform in how the industry perceives errors, and how it corrects errors, is in need of desperate change. As it stands, the industry is still simply trying to milk profit from scientists, science and the science literature for as long as it can. And I claim, at the expense of quality. Science is being tricked by increasingly creative marketing-driven economics that sugar-coats superficial aspects as being important (such as the impact factor and other useless metrics), allowing them to be gamed, and thus profiting by this gaming. And scientists are constantly being drawn into this trap, which gives them a false sense of reward, one which the humility of science itself cannot give.
So, reform of the plant science literature, or plant science (or any science for that matter), has to begin with a total revolutionary look at the way we do science, the rewards systems in place, the excessive power given to commercial publishers, and increasingly to the parallel corrupted open access movement which insults freedom to knowledge by placing a price tag on it.
So, allow this deeply sad day to also be a day of victories for the injustices that plague the plant science community and literature. Let the fall of one represent the rise of many more voices that live in fear of those in power and of the system. Let the errors and deceit of Prof. Olivier Voinnet inspire us to do more to show the community that not all is well in the plant science literature, even in what was perceived to be the crème-de-la-crème of the plant science literature, and that something – actually many things – needs to be done, urgently.
Let us also today remember the voices of the unknown voices that fought – and continue to fight – battles of injustice and academic corruption. The silent voices of concern, the anonymous voices that diligently examine the literature to discover what years of science prizes and immeasurable funding could never discover, should always be in the back of our minds. It is the courage of the few who feared the grand and powerful majority who are the victors today. They remain the silent minority, but their ranks are increasing, and their voices of discontent are growing larger.
Many questions remain unanswered about Voinnet and the power structure that kept him protected for far too long. There are likely still numerous consequences for Voinnet. But Voinnet did not work alone. He is a team member, and he is collectively responsible for the papers that have been published with his name on it. It would go a long way to restoring public trust if Voinnet’s co-authors would step out publicly, and state clearly that they are against unethical publishing practices, against corrupt powerful protective measures, and that they openly support the corrective measures that are urgently required to the plant science literature, through PPPR, and that the integrity in plant science that has been lost can only benefit, if not now, then in the future.
Greater collective public responsibility is lacking.
Today, I will send an email to Voinnet encouraging him to do the right thing. I will encourage him to get involved in the PPPR movement, and I will request him to please assume the responsibility of restoring trust by actively correcting the literature. This should be done by contacting all of the journals whose papers cited his now corrected and/or retracted papers, requesting them all to issue errata. Voinnet – and possibly the co-authors of those papers – should correct the downstream literature because they have benefitted from those papers, and thus they should also be responsible for it. If Voinnet responds to my call, I will post his response publicly here at PubPeer, because this is an issue of great public importance, at least to the plant science community.
Jaime A. Teixeira da Silva (January 29, 2016)
[1] https://forbetterscience.wordpress.com/2016/01/28/olivier-voinnet-loses-embo-gold-medal-sonia-melo-investigated-by-embo/
[2] Teixeira da Silva, J.A. (2013) The need for post-publication peer review in plant science publishing. Frontiers in Plant Science 4: Article 485, 3 pp.*
http://journal.frontiersin.org/Journal/10.3389/fpls.2013.00485/full
DOI: 10.3389/fpls.2013.00485
Disclaimer: to the critics of Frontiers and who have listed it as a “predatory open access publisher”, I paid no money to have this paper published, and had a tough peer review before it was approved for publication.
I then decided to contact Voinnet myself, in March, also copying ETH and his students Arturo, Florence, Marie-Aude and Gregory, to alert them to the risks of being associated with Voinnet on their careers.
Olivier Voinnet: an open letter
Dear Professor Olivier Voinnet,
We have never met, but I have come to know you through public channels.
At this point, I only care about the damage that you have caused to plant science, and the trust that has been lost as a direct result of your reckless actions.
I therefore recommend, as some ways to restore trust, to:
1) Rescind your PhD.
2) Pay back all wasted funds that were squandered on manipulated science.
3) Pay back all funds that were used on hotels, travel fees, meals and symposium / congress costs associated with falsely advertising your manipulated science.
4) Do community service by helping the post-publication peer review movement clean up the literature of work like yours, that is manipulated and thus misleading. This should be no less than 10 hours of voluntary service a week, in particular, showing young plant scientists the tricks of figure manipulation, and how to prevent and detect them.
5) Face criminal charges.
6) Most importantly, repair the literature. This includes contacting the authors and editors of journals that published papers citing your corrected and retracted literature. You should formally request an erratum to every single paper that has cited one of your flawed papers. As the literature currently stands, several thousand papers are currently misleading the public and the plant science community.
I am a nobody to you, with no glorious science prizes to my name, but a plant scientist nonetheless. Unless you take the initiative to complete tasks 1-6 above, possibly with the exception of 5, I will work tirelessly to convince the plant science community that this complement of measures is the only way of repairing, to a limited degree, the damage that you have caused to decades of trust in plant science publishing. I will be conveying my message to the wider community over time, including to your co-authors.
At first, however, please could you come forward publicly and address my statements made on PubPeer:
https://pubpeer.com/topics/1/9EF581519DBFA31BDACE6ACCF1F9EB#fb44869
I remain, cordially,
Jaime A. Teixeira da Silva
https://www.researchgate.net/profile/Jaime_Teixeira_Da_Silva
Dear Arturo, Florence, Marie-Aude and Gregory,
You are now working with and under Prof. Voinnet. But you need to start rethinking your careers very carefully because the association with Prof. Voinnet may cause irreparable damage to your future careers.
It is therefore fair for me to share my concerns that I am displaying publicly about the damage caused by Voinnet to plant science, and the 6 requests I am expecting of him.
You should feel free to comment publicly at PubPeer.
Regards,
Jaime
And a little figure to support my claims of PubPeer’s topic/comment manipulation, which represents a serious threat to stable PPPR, making PubPeer a potentially dangerous and unreliable tool (but sadly the only one currently available for anonymous PPPR).
LikeLike
Pingback: A personal tale of scientific misconduct – For Better Science
Long and complex story (thank you for that). However, I guess almost all readers got stuck with the very first sentence:
“Olivier Voinnet […] is apparently on extended sick leave”.
I AM SICK AND TIRED TOO!!!
I AM JUST GETTING TIRED OF SEEING SO MANY CHEATERS!!!
After 25 years, I have moved 13 times, I co-authored 250+ papers, I trained 5 PhD, I lectured for 15 years, and I raised funds for 3 laboratories. And here are the results:
Savings: ca. USD 3000
Private goods: two PC, one desk, one bed (single). Period.
Probability to be tenured within 10 years: very close to 0.
Network and contacts in my homeland: lost.
Near future (one week): I have to prepare an audition for a term position of one year.
So, you know what Dr. Voinnet ? [content removed, -LS]. AND PLEASE DON’T COME BACK.
LikeLike
I received this anonymous comment today, which quote here:
The commenter’s email address was fakeDespommier@gmail.com
It’s origin IP address led to an anonymous proxy at tor2e1.privacyfoundation.ch, SOFTplus Entwicklungen GmbH in Switzerland.
I wonder if this comes from someone in Olivier Voinnet circles?
LikeLike
From the acknowledgements of the 271 page PhD thesis.
“This work would not have been possible without support of the Sainsbury Laboratory, funded
by the Gatsby Charitable Foundation. I am also grateful to the European Commission for
supporting my work through a European Union Training and Mobility of researchers cat.20
grant (EBR400IGT974039).
I wish to thank David Baulcombe, my supervisor, for receiving me in his lab during my
National service and for offering me to stay as a PhD student. His constant support and
interest in my work, as well as the freedom he gave me to conduct my experiments were
really essential. I also thank him for the many stimulating discussions we had about the role
and mechanism of silencing, particularly in front of a good pint of Guinness. I am also
grateful to Andy Maule, my advisor. for his help and advice all throughout the thesis.
I am grateful to all members of the lab, past and present, for their support and advice, and for
tolerating my territorial expansions, my messy working habits and my bad tendency to
‘borrow’. Regarding the latter point, I am particularly thankful to Jack Peart, Pere Mestre and
Fabian Vaistij for making clean electroporation cuvettes and buffers fully available… I
would also like to thank Andrew Hamilton, Teresa Ruiz, Frank Ratcliff, Louise Jones, Tamas
Dalmay, and Fabian Vaistij for building such a fantastic ‘silencing team’ spirit over the years,
which made my stay in the lab so exciting and rewarding. Cohesion, that is the key. Many
thanks also to Philippe Vain, Sue Angell, Yvonne Pinto and Carsten Lederer for their help in
various experiments. I also thank Sharyn Carter, Penny Glanfield and Chris Tribley for
making available all I needed to perform my work, Alan Cavill for his constant availability
and Mike Hills and his team for their outstanding job in the greenhouse. I also thank June
Chapman for her help in tissue culture and for transforming plants before I arrived in the lab.
I am also grateful to Shou-Wei Ding, Andrew Jackson, John Stanley and Roger Hull for
providing material and sharing data.
Finally, I would like to thank my parents for their constant support and their faith in me, and
Ruth for her immense patience and kindness, particularly at the end of the thesis, when my
mental health appeared to be more than questionable.”
Telling stuff.
LikeLike
One more correction.
PLoS Pathog. 2016 May 5;12(5):e1005627. eCollection 2016.
Correction: Misregulation of AUXIN RESPONSE FACTOR 8 Underlies the Developmental Abnormalities Caused by Three Distinct Viral Silencing Suppressors in Arabidopsis.
Florence Jay, Yu Wang, Agnès Yu, Ludivine Taconnat, Sandra Pelletier, Vincent Colot, Jean-Pierre Renou, Olivier Voinnet
doi: 10.1371/journal.ppat.1005627
http://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1005627
Original
http://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1002035
LikeLike
A mega correction, but the journal assures us that seeds of “HcPro, P15 and P19′ are available upon reasonable request to the journal. One assumes that a reasonable request would be to repeat this ground breaking work? Perhaps someone in the botany community could request the seeds and then see if they do anything like it is claimed, though this will doubtless waste both time and money, it should produce a PLOSone paper.
LikeLike
However, not all what Olivier did is crap… it is good that Vicky Vance is correcting whatever is obviously wrong, but much of what he did (specially the part concerning the discovery and characterization of viral suppressors of RNAi) is reproducible… I am sure many other things are too… the problem now is how to discriminate the part that is solid and credible, from the part that is not…
LikeLike
Totally agree. Everybody deserves a second chance and we always learnt most from errors and mistakes!
LikeLike
What Voinnet did was cutting corners to increase speed, big time. Like not performing controls and repeats, and fabrication of pictures. That automatically will lead to deviations from the truth, although of course “preliminary data” often have a kernel of truth. I can’t see a genius scientist, as he is portrayed at some places, but apart from the cheating his body of work did require substantial scientific level. Whether he deserves a second chance is discussable, but such decision I ultimately to his employer. Only they can make a balanced decision on his combined values as a scientist, and they are the ones who pay. Personally I don’t care, because if scientists are going to be fired for cutting corners, then probably at least 50% can be fired.
I also can live with the decisions of the ETH concerning his papers. They recommended a substantial number of corrections and retractions. What I can’t stand is their intention to create a myth of the peculiar scientist who has perfectly good data but chooses to ruin them by making falsifications using cutting and pasting of pictures from other data. Are they trying to hypnotize us or so? That is where the science culture is totally wrong, that four established researchers (of the ETH committee) think that they can get away with creating such myths. For a restart it is important that things are called as they are.
The ETH committee may have thought that it was harmless to create such myth, possibly on behalf of Voinnet’s credibility as a teacher of students, but the journal Science took this ETH myth as a chance to not retract the Deleris et al. article despite that the ETH committee recommended its retraction (because the real data were perfect, right?).
Another problem with the ETH report is that it insinuates that it needed malicious energy (a personal vendetta) to make a problem of the figure fabrications by Voinnet. Considering the enormous body of fabrications by Voinnet, this idea is preposterous, shows a very weird concept of science (as if figure fabrications are acceptable gentleman errors), and might be rancorous in itself.
I prefer that scientists themselves guard the integrity of the science body, rather than for example that criminal charges are brought upon those who misbehave. But for such self regulation it is paramount that we call things as they are and take clean and logical decisions. The Voinnet case shows that a number of parties is willing to do so, while some other’s don’t.
LikeLike
Interesting that you said that ~50% of people in academia cut corners, it seems to be a consensus that moral/ethical values are so much needed among researchers… what a pity!
LikeLike
The system selects for speed. The fastest researcher gets the paper, gets the money, gets the position. To find something potentially interesting happens relatively often. Every extra 10% one wants to get closer to being 100% certain probably takes the double amount of time. Many “top-researchers” are selected because of their speed, and they make the rules. These rules of thumb say that it is forgivable to not try to achieve 100% certainty (as it would be “suicidal”). This thinking not only becomes part of addressing the project question, but also in how experiments are performed methodologically. And other researchers than simply repeat those types of methods, based on convenience.
The only way to seriously address the problem is to shift the balance in chances for reward/punishment if cutting corners. But that is very difficult, since there are so many interests at stake.
LikeLiked by 1 person
Perhaps what we need is an urgent renovation of the scientific system: finish with IF, privilege quality instead of speed and number of publications, submit always original data, etc.
LikeLike
The whole research institution needs a complete restructuring (a revolution)… to start with, the standards for becoming a researcher are pretty low (a chicken and egg problem of course); supervisors are afraid of failing candidates because it will look bad in their resumes and may affect their funding opportunities (indeed, the funding agencies have strongly contributed to create this problem). Pretty much anybody that so wishes can become a PhD and, if lucky, get a position and start doing research without having learnt how to write an e-mail properly during his/her time as trainee, not to mention unable to design and conduct sound experiments. That China and India produce PhDs in a massive form, only makes things worse.
On the other hand, research is not regulated. Anybody that have access to reagents (for whatever that means) is entitled to do research. Clinical researchers in many case did not have a proper training on research and therefore conduct research without scientific rigor. In many cases with ghost patients (like Dr. Chandra). Many clinical studies are invented!!!! (not to mention they get money from pharmaceutical companies to make those studies).
Perhaps now that storage is less a problem, Journals could request a copy of all primary data and expose that to the community on-line, that could help to reduce frauds. Strict disciplinary actions from funding agencies would also help, like banning fraudulent researchers for one year for each fraudulent action… Institutions should also assume a more belligerent role otherwise should be also punished by funding agencies. I know all that sounds unreal… too idealistic!
PPPR and whistleblower like Leonid are doing an incredibly important job. In a time when few people dare to speak, there are few brave individuals like Leonid, Paul Brookes and many others, that are not afraid to shout that “scientific misconduct” should not be tolerated… We should definitely support them in this endeavor…
LikeLike
Fully agree!!!!
How many manuscripts are being commented in PubPeer at the present moment?
See the link:
https://pubpeer.com/recent
Isn’t it time to discuss together with journal editors, authors and peer-reviewers, new and fresh policies for paper and research projects submission, peer-review, execution and publication?
LikeLike
Jjovel and Apolo,
Most of what you say is correct, but I don’t think it holds a solution to the problem. We can’t control the supply-line of scientific articles, and we do need some kind of rating system. Despite its problems and limitations, the IFs of both the journal and the article probably form the best possible rating system available in non-intimate contexts.
Your mentioned request for presentation of original data is logical, and some journals do have such policy. The journals which don’t, or do not provide despite claiming otherwise, maybe should be pushed harder to change their policy. But this still won’t solve the problem, because researchers with an intent to cheat will switch to types of experiments which better allow manipulation.
By any logic, the only angle from where the system can be improved is at the discussion/punishment level, thus after publication. Currently this is left to researchers and journals. Most researchers don’t want to be involved in PPPR, although most of them agree with its value. When pressured to take measures for fixing the literature, many journals switch their attitude from “we are a respectable science entity” to “we are merely a vehicle for data presentation by others”, and their efforts to clean the literature are less than meager.
So the big question is how the discussion/punishment level can be so far increased that it stops the bulk of faulty papers. That is either by very hard punishment of a few, like bringing criminal charges upon them, or by soft punishment (public exposure by discussions, errata and retractions) of many. For the criminal charges it would need governments and it will be quite difficult across country borders. For an increase of discussions/errata/retractions to a level which culprits makes consider twice before cheating, scientists will have to become much more active in PPPR. They will never do such because of “the common good”, so their careers should benefit from it. Possibly universities can inquire about PPPR contributions when hiring someone, since after all professors should have an important function in the integrity of their research field. Non-anonymous PPPR should also show one’s intellectual ability, thus be a nice measure for universities when hiring someone.
That’s really the only two things that would have a big impact, either criminal charges or a large increase in PPPR.
LikeLike
Man of the files is absolutely correct, especially the last sentence. And although I am generally against the increased militarization, centralization and control of science (anti Big Brother stance), as it stands, there is little option but to take a more radical approach. However, there are at least two issues that you have not taken into consideration in your equation or future plan, and which could invalidate the whole purpose of PPPR:
a) the choice of “predatory” OA journals, which are expanding. As the rules, and pressure on frauds, in more traditional publishers increase, either by pre-submission checks, or by post-pub punitive measures, this increases the venues for publication of bad or fraudulent science. As we slowly move towards an all-OA publishing platform (spurred by the EU’s decision until 2020), make no doubt that the number of hijacked, false, deceitful and non-academic OA journals is going to expand, to pose a direct business challenge to the half dozen publishers that currently control the vast majority of the science publishing market.
b) I am assuming that you are talking from a Western country, where perhaps criminal charges might be more likely than in dozens of countries where fraud and bribery are likely encouraged as their business model. It is extremely difficult to hold fraudulent scientists, or scientists who engage heavily in misconduct, accountable when the countries they reside and work in do not have the infrastructure to deal with academic fraud.
So, this means that your suggestion, which I also predict will be the way in which science publishing will evolve, will lead to diametrically opposite responses, and thus not resolve the issue, simply transform it into two camps of the publishing world, and a whole bunch of journals in between with something between a legitimate academic model and a predatory one.
The first step, however, is to transform the mental mind-set of researchers to embrace PPPR as part and parcel of research and publishing. Scientists must start to accept that the easy ride is over and that academic scrutiny does not end where acceptance starts. Rather, scientists must be prepared to fight to retain their titles and positions as academic and non-academic critics of their published work. Or face extinction, or a life of a hermit.
As for Voinnet, I have been consistently critical of him in public and have called on criminal charges to be taken. I have also called on community service and a period of a few years in which he is reformed, thus giving him a fair chance to reenter the scientific community as an actively practicing scientist. But several retractions and almost two dozen errata/corrections reflects very serious flaws. Flaws at the level of Voinnet, Voinnet’s teams and collaborators, none of which detected these issues, Voinnet’s institutions, none of which detected any of these issues, yet seemed to only care about their own PR negative fall-out, and by none of the journals, their editors, or publishers.
So, what the Voinnet saga represents is a classical black-swan phenomenon [1] (a shocking revelation of an event that was highly unlikely to occur), and shows that the current research and publishing model is not built to deal with, or embrace, PPPR.
A zero tolerance approach is required, including for self-plagiarism, but this might not be effective if the powers that be (currently Thomson Reuters, Elsevier, SpringerNature, Wiley, Taylor & Francis / Informa, COPE) do not open a publishing model that is based on strict, consistent and transparent rules, which are respected uniformly across all member journals.
As I see it, the Voinnet case is still incomplete, and PPPR is still required. Also, the fall-out of Voinnet et al.’s negative actions will be irreparable for maybe even decades. For this, all co-authors should assume collective responsibility. And, to date, I have seen only silence by 95-99% of co-authors on this issue.
[1] Teixeira da Silva, J.A. (2015) The “black swan” phenomenon in science publishing. Journal of Educational and Social Research 5(3): 11-12.
http://www.mcser.org/journal/index.php/jesr/article/view/7693
DOI: 10.5901/jesr.2015.v5n3p11
LikeLike
Jaime,
The system is deliberately founded on cutthroat self-interest (publish or perish), one of the most powerful human forces. However, currently the chance for wrongdoers to be caught is minimal, and even if caught the punishment is minimal. That leads to massive cutting of corners, as it would in any walk of life.
The defense against misbehavior and the protection of the science body from being clotted with non-reproducible results should be based on a similarly strong primitive drive as is the cheating.
If criminal charges are not possible or desirable, that defense should be based on robust and abundant PPPR (see my previous post). Such PPPR can only be elicited if it is in the self-interest of researchers to critically discuss the papers of others. But who will organize that self-interest?
We have to think of a horse for which it is logical to pull this cart. Journals, COPE, or scientists themselves definitely are not that horse, because at least for the short run they (believe to) have competing interests. But which party is?
Maybe Jjovel or Apolo have some fresh ideas about this, as in this matter I have grown cynical about all parties. Universities? Grant organizations? NIH? Governments? Industry?
LikeLike
Man of the files,
In my opinion, obviously none Universities, grant organizations, NIH, or industry can be the “horse”. I would suggest some sort of international organization such as PubPeer (perhaps a further development of PubPeer) representing different governments, editors, journals, scientists from all levels, ethical interests and where original data can be stored and PPPR can take place and policies are developed to deal with different levels of misconduct.
LikeLike
Apolo,
PubPeer, like PubMed Commons, is about creating the possibilities for PPPR. PubPeer, at least when regarded together with the other possibilities of PPPR, is doing an excellent job in that.
The problem lies within the scientists themselves. In contrast to the public opinion which considers them intellectuals that would enjoy a debate about their work, the large bulk of scientists really hates public debate. Why discuss your data if it is much easier to only convince only one or two reviewers, and then just flaunt with the journal impact factor, right?
All parties probably agree that intensive debate (PPPR) is the cheapest way to make a great improvement in science. Now the problem is that at the individual level that is not true. A scientist has much more to lose from public debate than to gain. So by far most scientists do not publicly criticize the work of others, and do not respond when their work is being criticized. That is what has to change, and that can only be done if scientists are rewarded for public debate. If you apply for a job, you have to fill in things like “Membership of organizations”, “Student education” etc. Why not add “Contribution to the public debate”?
Your suggestion to bring many parties together may be valuable. However, you have to keep in mind that all those organizations have their own agenda, coloring the problem to their benefit. The journals, for example, love to focus on the poor statistics problem rather than acknowledge that poor attitude (or crazy pressure, whatever you like to call it) is at the heart of the problem.
The people with power love their power, so their recommendations will go that direction. People who complain often love complaining, and constructive discussion with them is often not possible. And amidst all those groups you want to come to a decision of how PPPR should be organized? I don’t think that is possible. The end decision of such group will be that “we embrace the enhanced possibilities of PPPR and recommend scientists to participate in it”. So nothing will change.
You state that “policies should be developed to deal with different levels of misconduct”. However, there are such rules already, but people do not stick to them. Just look at the Deleris et al. paper by the Voinnet group and how it is “repaired”. Probably aiming for full exposure rather than to enforce proper measurements is the better and definitely less energy consuming way to go.
I do agree with you that PubPeer is an excellent initiative, and that their team argues in an intelligent, reasonable and eloquent manner. From your suggestions I like the idea of bringing all parties together, though maybe better in a congress/workshop form than in the hope to shape them into an organization. And the PubPeer team should have a prominent presence in that congress/workshop.
Still, we know the problem already, and in my opinion the only question is how scientists can be triggered into more PPPR.
LikeLike
Man of the files, I agree with almost everything you state, but I disagree strongly about your suggestion that PubPeer and a limited number of watchdogs or other parties be central to such meetings and congresses. Your excessive praise of PubPeer in itself suggests bias towards PubPeer, but you fail to appreciate the weakness of handing so much power to an organization led by three relatively unknown individuals. You are willing to hand the fate of science and the destiny of PPPR to three unknown individuals, who have weak research and/or publishing profiles? I don’t think that your suggestion is going to settle nicely with many scientists. As equally as there is a body of critics that believes that just because Beall’s blog raises awareness, that he should be considered an expert on research and publishing ethics, or even on OA. He is just a librarian raising awareness. So, we have to be careful about how we reward those who have strong biases.
Just to give you an example, following my exposure of almost two dozen cases of problematic papers published in 2014-2016 in SpringerNature’s Physiology and Molecular Biology of Plants (PMBP), which I believe will lead (spoiler alert) to an equal number of errata or retractions very soon, I was invited, amid this journal’s crisis, by the EIC, Prof. Rana Pratap Singh, and by the Editor, Prof. N. Raghuram, to become the Ethics Editor of this journal, with immediate effect. As you can imagine, I had to laugh at the suggestion. Can you imagine the uproar among the plant science community if suddenly a critic and whistle-blowing scientist were elected, by a decision made by the top rank of this journal, and not by a democratic vote among all editors and peers? So, you can appreciate that elevating PubPeer (or other similar watchdogs) to the level of leading the PPPR pack to be extremely problematic.
But let me not mince my words here. PubPeer is currently the only integrated platform where signed and anonymous PPPR can take place. So, there is great value as a tool, and the fact that comments link to papers’/publishers’ web-pages is a plus factor, because it alerts readers that their work is being watched, and scrutinized. In that sense, PubPeer deserves unreserved praise.
Asking the plant science community (in this case) to get more involved in PPPR is an uphill battle, maybe even impossible, because you would basically be asking them to support an action that could one day be used against them. Moreover, scientists don’t have time to praise each other, but don’t have the desire to criticize each other, either. So, the battle to make PPPR more acceptable has to hurdle this psychological conundrum first. I suspect that only the hard-core critics and disgruntled few/minority would follow the lead, because only they would appreciate the importance of PPPR. However, I do believe that there are potentially hundreds or thousands of plant scientists that are aware of problems in the literature, or are aware of power abuses by their seniors, PIs, professors, or even research institutes, but who are afraid to report such issues publicly, because it would place their own positions, salaries and grants at risk. So, if we can eliminate the “fear factor”, then we could in essence spur more PPPR. How does one remove “fear” from the publishing equation, especially when publishing is becoming more militarized and/or stringent?
So, to make PPPR work, it has to be moved a notch up from voluntary, to forced. And thus such policies can only take place at the institutional level, because at the individual scientist level, this would never work. Take, for example, the suggestion and institutionalization of “silence” (i.e., the failure to respond to named or anonymous critics) as being misconduct by Kamoun and Zipfel [1], and set into stone by The Sainsbury Laboratory (TSL) [2], most likely as a consequence of this entire Voinnet saga, which embroiled TSL into the entire Voinnet/PPPR/PubPeer battle. When you make lack of accountability equivalent to misconduct at the institutional level, then you automatically encourage PPPR, because then you force your work force of researchers and scientists to be held accountable at the public level, and not simply until the peer reviewer/editor stage. PPPR also needs to be institutionalized at the level of journal and publisher. Publishers, especially COPE members, need to formalize PPPR into their publishing models. Thus, any comment deemed valid by the editor board, and that appears on any public site, like PubPeer or PubMed Commons, must be responded to, or face an expression of concern (at minimum). At the moment, it is neither formal, nor mainstream, so forcing authors to respond remains unacceptable. At some point, this will change as editors and publishers also start to feel shame.
At essence, we have to go back to the vigilante/vigilant mind-set, otherwise nothing of significance takes place. But that may ruffle the feathers of conservatives in the plant science community like Mike Blatt.
[1] Sophien Kamoun, Cyril Zipfel (2016) Scientific record: Class uncorrected errors as misconduct. Nature 531, 173 (10 March 2016). doi:10.1038/531173e
http://www.nature.com/nature/journal/v531/n7593/full/531173e.html
[2] http://www.tsl.ac.uk/about-tsl/scientific-integrity/
LikeLike
Jaime,
I do not want to “hand over” the decisions to the PubPeer team, but just believe that they should take an important part in the discussion. Their words I can understand from beginning to end, while with other parties I feel they are trying to side-direct the real issue to something which is more comfortable for them. Furthermore, the PubPeer formula of anonymity was the one that greatly increased PPPR, so they do seem to have a handle on mass psychology. Of course the formula has its limitations, not only regarding the atmosphere created by (often necessary) anonymity, but also in the quality and quantity of the discussions. Still, the PubPeer team made a big and good impact, and I would like to know their suggestions for further improvements. I can’t say that of many other parties.
In my opinion the focus should neither be on the journals nor on the institutes, but on the scientists. We can force the journals to make a larger set of retractions, and the institutes to deal sharper with misconduct. But that still won’t solve the problem, because the scientists will then soon abandon the silliest types of cheating and therewith stay under the radar for disciplinary measures. Still, they then can produce large nonsense which pollutes the science body. Really the only thing which would work is to increase PPPR, since it can detect all kinds of errors and frauds.
You also focus on that authors should be forced to reply to PPPR, but although I agree with the principle, in my experience that also helps little to fix the problem. Honest answers to severe questions are very rare.
Much more important is that the number of papers that is being discussed in PPPR should increase.
Scientists can’t be forced into PPPR, but can’t we think of a way to trigger them into participating? There must be a way.
LikeLike
Man of the files and Jaime,
I think scientists already engaged in PPPR and it seems the best formula is the anonymous way for now because it seems to me that PubPeer is more often used than PubMed Commons although I also think both anonymous and non-anonymous PPPR should co-exist. Probably, PPPR is the way to go, perhaps other PPPR sites should appear and these sites should not be governed neither by scientists or institutions or universities, but by independent, neutral people with a necessary scientific, ethical and legislation knowledge. Perhaps we can add in these sites access to raw data for PPPR, links to the journals and authors, so that each time it appears a comment both journals and editors will be automatically warned about. The comments and critics done in these PPPR sites do not have to be necessarily negative but in case they are negative, this will prompt authors to respond intuitively as the lack of an answer may mean assuming fault.
Anything will never be perfect but we can improve. For sure, from now on WB manipulation will be harder and harder, although other more sophisticated forms of data manipulation might appear. Also, I agree with that idea of scientists rehab although I also believe scientists must also suffer some sort of penalty for misconduct.
LikeLike
Apolo,
If you believe that scientists are already sufficiently engaged in PPPR, then we fully disagree.
In an ideal world most articles should be discussed on-line in a hard fashion, probably revealing severe flaws in at least 70% of them and highlighting those articles and those parts of articles which are really valuable. Currently, the percentage of articles discussed in PPPR is very, very small (although it may feel like a lot), and most of those PPPR discussions are of low level.
In an ideal world also several scientists should engage in discussion of the same paper, creating a balanced view of the good and bad points of that article. But, currently, in most PPPR cases only “picture duplication seekers” are involved, and real scientific dissections are rare. There are beautiful exceptions, and it is also in those cases that journals are more willing to act.
Most scientists want to stay as far away from PPPR as possible. If I tell them that I do engage in PPPR, the most common phrase is “what is the advantage to you?”. It is this self-advantage thinking which is pumped into us from the beginning of our careers, deciding most steps of most researchers, leading to a great disadvantage for science as a whole. We regard publications as an economic production rather than as something which ought to be beautiful and perfect. I don’t think we can change that culture, so we have to make participation in PPPR beneficial to the economy of the participating scientist.
You focus on improving the level of PPPR. Monitored discussion, which you describe, is already organized by some journals, but for a very selected part of criticisms. It is also possible by writing a correspondence article. Limitations for these options are that the journals are quite political in their choice of which discussions to highlight, that journals are afraid for liabilities thus that correspondence articles often are not allowed to contain insults like “picture duplications”, and that most scientists do not wish to participate as reviewers or otherwise.
Raw data collection as you suggest it may be quite an expensive undertaking. Currently, the system pretends to take care of it by having parties agree to preserve raw data and present them if requested. In many, many cases, however, authors do not provide those data when asked for, without this leading to disciplinary measures by journals or institutes. So maybe your idea should be executed, but I am sure that the idea will always lose in a discussion because parties will choose for the cheaper option and say “OK, if there is a problem, we just implement the current rules stricter”.
The PPPR possibilities are now in place. The journals have to step up in their dealing with flawed articles, but they are moving in the right direction. It really is the scientists which have to change.
(and, as Cato the Elder said:) So we have to think of a way to trigger scientists into PPPR.
LikeLike