Science is amazing. Turns out, the ancient beliefs in vampires who drink blood of young women to retain immortality had solid scientific foundations! It’s just that you must inject the blood, not drink it; and science hasn’t yet determined if the young blood transfusion suffices for immortality or just for life extension, and whether the donor must be a virgin to achieve the maximum rejuvenation effect.
The peer-reviewed anti-aging research, by genius scientists from elite US universities, all with their own biotech startups, proved the blood of young people to be indeed a fountain of youth, rejuvenating the brain, muscles and the rest of the body, while curing all diseases of old age, especially Alzheimer’s. It’s all about parabiosis experiments, where two mice (or sometimes rats) are sutured together, their blood systems connected. The approach seems disgusting and cruel, but it is for the greater scientific good, right?
Well, I am not sure, and neither was Smut Clyde:
“There is some limited support for this from experiments in the 1860s, the golden age of vivisection, where researchers were sewing rats together — creating conjoined twins — so that their blood circulations merged in a single vascular system. Perhaps the idea was that the conjoined entity would become a Rat King, a creature of majesty and arcane powers who would lead the human race into a new era of eternal darkness.
Or else it was just what researchers did back then.
There was a revival of enthusiasm for rodent quilting in the 1950s-70s, with claims that when older rats were stitched up with younger ones they benefited from the propinquity… apart from the 10% or so that died from the operation or the 20%-30% that died of ‘parabiotic disease’. It was one of those avenues of research where results became progressively less positive as the studies became more rigorous.”

I personally wonder what exactly would be the unexplored scientific question such parabiosis experiments could answer? I can only think of some scientist’s desire to prove right a pet theory about some obscure effect of externally supplied blood, but then again, why can’t this same theory be tested by the common practice of blood transfusion? Is it because it wouldn’t work otherwise? But then…
Surely if you seek to market young blood rejuvenation as a therapy to human customers you can only do so via the boring old blood transfusion, and not suturing a hapless young victim onto the back of an ageing millionaire (yes, I see the trash horror film potential here)? So again, what is the scientific point of those bizarre parabiosis experiments if not to squeeze some “positive” results out of a couple of horribly tortured rodents because the same approach of plasma transfusion which you are testing in clinical trials on people, provides exactly zero positive results in mice?
In early 2020, the highest authority on all things science, The Guardian, informed its readers about the 3 leading companies which will soon abolish old age via transfusion of blood from young donors. The article quoted as introduction a certain bearded guru of Anti-Aging:
“There’s still a long way to go – blood is complicated,” says Aubrey de Grey, who leads the nonprofit Sens (strategies for engineered negligible senescence) Research Foundation. “But there are many excellent labs focused on this, so I am optimistic about progress.“
Later it turned out De Grey has always been a sexual predator and abuser, whose own approach to anti-aging was actually much more traditional, namely to have sex with young women, while also coercing these women to have sex with the wealthy ageing donors of his SENS foundation.
That was reported by STAT News, not by Guardian because the Grauniad never writes bad things about scientists, and instead rather writes good things about bad scientists, on principle and for reasons I personally cannot fathom.
So I will take example of the Guardian professionals and introduce you to the same 3 blood-letting anti-aging companies. In my own, evil anti-science troll ways.

Ambrosia
Ambrosia is the young blood company of the most notorious young blood quack out there, Jesse Karmazin, who threatened HuffPost over their lengthy investigation into his scam with a defamation lawsuit, claiming that a deceased Ambrosia patient had “faked his own death.”
The Guardian introduces:
“In 2016, Jesse Karmazin began offering infusions of young whole-plasma as a way to help beat ageing, without published evidence that the treatment worked. By the end of 2018 Ambrosia, the US startup he founded, which charges $8,000 for one litre or a discounted $12,000 for two, had opened clinics in five US cities.”
By the way, the prices now dropped, you can buy 2 litres of blood for just $8k at Ambrosia, shippable to all of the USA, just write to ambrosiaplasma@gmail.com!
Smut Clyde is better informed about Ambrosia’s history. The scam started already years earlier. The precursor was Karmazin’s xVitality Sciences, which used to peddle the mysterious “all-natural” product named Prophylaxia before the company disappeared at the end of 2015:

Unsurprisingly, Prophylaxia was actually human blood, to be applied in Peru and Ukraine.

Somehow, xVitality never took off. Smut Clyde continues in his old post from 2016:
“Dr Karmazin has scrubbed off all trace of xVitality from his LinkedIn entry and FB account, and picked Ambrosia as the name for his new company. It has received a fair bit of attention around the Intertubes, what with its “clinical trial” that lacks control cases but charges a $8000 fee for “subjects”. Tech billionaires, concerned that youth is wasted on the young and that the poors are consuming valuable resources —“It’s this extremely abundant therapeutic that’s just sitting in blood banks”— are recruiting Dr Kamarzin to fill the role of Renfield. The prospect of Peter Thiel sinking millions into this cargo-cult (blithely sanguine that having won the golden ticket in the dot-com lottery, and being a Nietzschean Übermensch, he is as much an expert on biology as he was on nautical engineering), it is enough for one to wish Karmazin the best of luck.“
In February 2019, because of people like Karmazin the American young blood market became such a criminal free-for-all that FDA issued a warning, to Karmazin personally, and to the public in general:
“The FDA has recently become aware of reports of establishments in several states that are offering infusions of plasma from young donors to purportedly treat the effects of a variety of conditions. The conditions range from normal aging and memory loss to serious diseases like dementia, Parkinson’s disease, multiple sclerosis, Alzheimer’s disease, heart disease or post-traumatic stress disorder. We have significant public health concerns about the promotion and use of plasma for these purposes. There is no proven clinical benefit of infusion of plasma from young donors to cure, mitigate, treat, or prevent these conditions, and there are risks associated with the use of any plasma product.
[…] We strongly discourage consumers from pursing this therapy outside of clinical trials under appropriate institutional review board and regulatory oversight.”
The year before, Ambrosia’s much touted clinical trial NCT02803554 with 200 old gits who handsomely paid Karmazin for the young blood therapy ($8000 each) completed, its results were of course never released.
The Guardian however still believes in Karmazin, because Karmazin still believes in himself:
“The company wasn’t warned first by the FDA. “It came out of the blue,” says Karmazin, who ceased operations out of caution and to understand the FDA’s position. Last October, after an eight-month hiatus, Ambrosia reopened as a scaled-back operation with a clinic in San Francisco. As Karmazin sees it, the law is on his side.“
And so is surely science, right? Because Karmazin is far from alone.
Elevian
The biotech start-up Elevian was co-founded by Amy Wagers and two other Harvard professors in 2018. The Guardian informs that Elevian
“is working on therapies to elevate the activity of Growth Differentiation Factor 11 (GDF11) – a protein in blood plasma present in very small amounts that it believes decreases with age in mice.
Studies published in 2013 and 2014 by Wagers’s laboratory and those of the company’s other scientific co-founders, showed that old mice injected with GDF11 reproduced several of the parabiosis findings – with regeneration seen in the heart, skeletal muscle and brain. It is the only factor that has been demonstrated in old mice to have beneficial effects across a number of major organs that decline with age, notes Wagers. Elevian is working on scaling up GDF11 production before launching its first human trial. […]
GDF11 is controversial. It has proved hard to replicate some of the regeneration findings, calling into question its use as a potential therapy.”
What Guardian didn’t tell you because it’s apparently irrelevant, is that the parabiosis rejuvenation researcher Wagers previously retracted two papers for fraud (Mayack et al Nature 2010 and Mayack & Wagers Blood 2012), while her postdoc Shane Mayack took the full blame.
Wager’s GDF11 discovery in Science (Sinha et al 2014) was soon debunked by Novartis scientists in Egermann et al Cell Metabolism 2015:
“Recent studies showed an age-related decrease in GDF11 and that GDF11 treatment improves muscle regeneration, which were contrary to prior studies. We now show that these recent claims are not reproducible and the reagents previously used to detect GDF11 are not GDF11 specific.“
They also found out that Wager’s antibody GDF11 was not specific:
“…we next tried the antibody that was used to demonstrate that GDF11 declines with age (Loffredo et al., 2013, Sinha et al., 2014). This antibody was also first tested for its specificity. By western blot analysis, the antibody was found to recognize both recombinant myostatin and GDF11 to a similar degree, indicating cross-reactivity and a lack of preferential binding to GDF11 (Figure 1B).“
The other quoted paper (Loffredo et al 2013) was from the lab of another Elevian co-founder, the Harvard professor Rich Lee. So Lee and Wagers protested:
“Now, the Wagers and Lee group says the assay Novartis used to detect GDF11 and GDF8 was itself flawed. They found that the main protein detected by the antibody test is immunoglobulin, another protein that rises in blood level with age. Mice lacking the gene for immunoglobulin tested negative for the active form of GDF11/8 that the Novartis assay was thought to reveal, they report today online in Circulation Research.“
The extra irony is that Wagers’ and Lee’s research was also disputed (Smith et al Circulation Research 2015) by two Temple University professors whose own scientific achievements are tainted by massive Photoshop fraud: Steven Houser and Abdelkarim Sabri.
Whom to believe?
Alkahest
There is not just Harvard’s but also Stanford’s authority. In July 2021, Newsweek covered the young blood market situation, informing:
“Earlier this year, Grifols closed on a $146 million-deal to buy Alkahest, a company founded by Stanford University neuroscientist Tony Wyss-Coray, who, along with Saul Villeda, revealed in scientific papers published in 2011 and 2014 that the blood from young mice had seemingly miraculous restorative effects on the brains of elderly mice. […] In the last six years, Alkahest has identified more than 8,000 proteins in the blood that show potential promise as therapies. Its efforts and those of Grifols have resulted in at least six phase 2 trials completed or underway to treat a wide range of age-related diseases, including Alzheimer’s and Parkinson’s.”
Let me show you what kind of research earned Wyss-Coray almost 150 million dollars.
Jian Luo, Amy H Lin , Eliezer Masliah , Tony Wyss-Coray Bioluminescence imaging of Smad signaling in living mice shows correlation with excitotoxic neurodegeneration Proceedings of the National Academy of Sciences (2006) doi: 10.1073/pnas.0605077103


The first author Jian Luo assured on PubPeer:
“These images were not used for statistical analysis. Therefore, the mistake in Fig. 3A does not undermine our work’s conclusion. Nevertheless, I apologize for the mistake. I will try to find the raw data and fix it if possible.
Again I really appreciate the efforts you and your colleagues at Pubpeer put in post-publication review. I will definitely be more careful in the future when making figures.”
It wasn’t his only misstep.
Jian Luo, Fiona Elwood , Markus Britschgi , Saul Villeda , Hui Zhang , Zhaoqing Ding , Liyin Zhu , Haitham Alabsi , Ruth Getachew , Ramya Narasimhan , Rafael Wabl , Nina Fainberg , Michelle L. James , Gordon Wong , Jane Relton , Sanjiv S. Gambhir , Jeffrey W. Pollard , Tony Wyss-Coray Colony-stimulating factor 1 receptor (CSF1R) signaling in injured neurons facilitates protection and survival The Journal of experimental medicine (2013) doi: 10.1084/jem.20120412

Luo explained:
“It is an image duplication mistake for Figure 3I. We apologize for the mistake and will try to find the original data and fix it.“
There was also another image duplication in Figure 6, but that was apparently intentionally reused image of a control brain staining, because apparently the Wyss-Coray lab financially couldn’t afford to stain more than one section of a mouse brain.
Jian Luo, Peggy Ho , Lawrence Steinman , Tony Wyss-Coray Bioluminescence in vivo imaging of autoimmune encephalomyelitis predicts disease Journal of Neuroinflammation (2008) doi: 10.1186/1742-2094-5-6

On PubPeer, Luo assured:
“Figure 1C of GFAP staining indeed has a repeated image. The representative image of day 35 was a mistake. Attached corrected Figure 1 here. We’re sorry for the mistake and will work with the editorial office to fix it if possible.“
Jian Luo, Andy Nguyen , Saul Villeda , Hui Zhang , Zhaoqing Ding , Derek Lindsey , Gregor Bieri , Joseph M. Castellano , Gary S. Beaupre , Tony Wyss-Coray Long-term cognitive impairments and pathological alterations in a mouse model of repetitive mild traumatic brain injury Frontiers in Neurology (2014) doi: 10.3389/fneur.2014.00012

Again, Luo was grateful to his post-publications reviewers:
“We checked carefully and found that the highlighted left side of Figure 8 was a mistake introduced during figure making. This part of the Figure was intended to show little p-Tau immunoreactivity in the CA3 of sham animals. While this mistake does not affect the conclusion of the paper, we sincerely apologize for it. We are working with the editorial office to correct it.
Thank you also for identifying potential mistakes in other papers. We will check each of them and respond accordingly. To avoid similar mistakes in the future, we revised our workflow of figure making and added additional steps of quality check.“
Those papers still wait to be corrected. It wasn’t just Luo who made mistakes:
Julia Marschallinger , Tal Iram , Macy Zardeneta , Song E. Lee , Benoit Lehallier , Michael S. Haney , John V. Pluvinage , Vidhu Mathur , Oliver Hahn , David W. Morgens , Justin Kim , Julia Tevini , Thomas K. Felder , Heimo Wolinski, Carolyn R. Bertozzi, Michael C. Bassik, Ludwig Aigner, Tony Wyss-Coray Lipid-droplet-accumulating microglia represent a dysfunctional and proinflammatory state in the aging brain Nature Neuroscience (2020) doi: 10.1038/s41593-019-0566-1



Wyss-Coray then issued two corrections in January 2020 and in July 2020:
“In the version of this article initially published, errors occurred in Figs. 1k and 3b. In Fig. 1k, the percentages for the Ceramids (CE) groups were mistakenly included into the Other group; in Fig. 3b, the graph was inadvertently duplicated from Fig. 5h.“
Well, those papers were nto about young blood rejuvenation, so what do they matter, right? However, also the decisive young blood paper was criticised, for itsunconvincing claims:
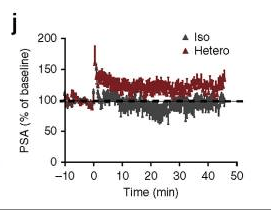
Saul A Villeda, Kristopher E Plambeck , Jinte Middeldorp , Joseph M Castellano , Kira I Mosher , Jian Luo , Lucas K Smith , Gregor Bieri , Karin Lin , Daniela Berdnik , Rafael Wabl , Joe Udeochu , Elizabeth G Wheatley , Bende Zou , Danielle A Simmons , Xinmin S Xie , Frank M Longo , Tony Wyss-Coray Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice Nature Medicine (2014) doi: 10.1038/nm.3569
A PubPeer critic pointed out:
“—All these ‘rejuvenation’ could be ‘undone’ with the a single panel of Figure 1 and namely 1 J
—There are no differences and the purpose of SEM bars are diminished by using one and opposite polarities and it is still an accepted practice
—And words of authors :
‘Although long-term potentiation (LTP) in the DG of isochronic parabionts quickly reached baseline levels, LTP in heterochronic parabionts was maintained above baseline throughout the recording period (Fig. 1j). We detected no changes in synaptic strength (Supplementary Fig. 4e). These functional data indicate that synaptic plasticity in aged mice is enhanced by exposure to young blood ’“
Well, it passed peer review in Nature Medicine, a journal which will publish anything provided the authors are eminent enough and affiliated with eminent US institutions. Hoya Camphorifolia commented:
“Meanwhile the research team have moved on to promising benefits from umbilical-cord blood instead, as if mere young blood has been abandoned.“
That was the study, its main advantage is that umbilical cord blood can be bought relatively cheap from certain vendors, and there is no need to specifically recruit and bleed young people, what with the costs and the ethics hassle:
Joseph M. Castellano , Kira I. Mosher , Rachelle J. Abbey , Alisha A. McBride , Michelle L. James , Daniela Berdnik , Jadon C. Shen , Bende Zou , Xinmin S. Xie , Martha Tingle , Izumi V. Hinkson , Martin S. Angst , Tony Wyss-Coray Human umbilical cord plasma proteins revitalize hippocampal function in aged mice Nature (2017) doi: 10.1038/nature22067
Nick Brown took the liberty to recalculate the p-values of the decisive experiments of that study:
“Does anyone else see a problem here:
“(Z-score = 2.12; P = 2.68 x 10^-2) or young (Z-score = 0.96; P = 2.59 x 10^-3) treatment, but not elderly (P > 0.05) plasma treatment. An additional predicted disease and functional pathway was memory (Z-score = 1.38; P = 2.61 x 10^-2)…”
Those are regular Z-scores, right? The kind where 1.96 gives you a two-tailed p value of 0.05? So why do we have (after cleaning up the unnecessary exponential notation):
Z = 2.12 p = 0.0268 (actual two-tailed p value: 0.034)
Z = 0.96 p = 0.00256 (actual two-tailed p value: 0.337)
Z = 1.38 p = 0.0261 (actual two-tailed p value: 0.168)“
And now the groundbreaking results of rejuvenation with umbilical cord plasma went from extremely significant to not significant at all. But it passed peer review in Nature, so there.
Hoya camphorifolia found something else in that Nature paper:
“An interesting but underpublicised aspect of this Letter was the failure to replicate earlier reports that injection of plasma from young (teenage) donors would rejuvenate mice. E.g., from Wyss-Coray’s Alkahest team“
Indeed, just the year before in 2016, Wyss-Corray’s company Akahest was celebrated for its breakthrough achievement:
“Blood plasma from young people has been found to rejuvenate old mice, improving their memory, cognition, and physical activity. The method has the potential to be developed into a treatment for people, says Sakura Minami of Alkahest, the company behind the work. […]
They found that human plasma does have the power to rejuvenate. Treated mice ran around an open space like young mice. Their memories also seemed to improve, and they were much better at remembering their way around a maze than untreated mice.
“Young human plasma improves cognition,” says Minami, who presented her findings at the Society for Neuroscience annual meeting in San Diego, California, on Monday. “Their memory was preserved.” […]
The team then examined the brains of the treated and untreated mice. They looked for clues on the birth of new neurons in the hippocampus – a process called neurogenesis, which is thought to be important for memory and learning. Sure enough, the treated mice appeared to have created more new cells in their brain. “Young human plasma treatment can increase neurogenesis,” says Minami.”
Yet when those conference data was published next year in the Castellano et al Nature 2017 paper, all these amazing results with young adult blood turned out to have been wrong. As if someone made them up out of wishful thinking, to get a clinical trial set up and to enrich themselves, evil tongues might say. But as we know, data fudgery does not happen in the Wyss-Coray’s lab.

“Notably, young adult human plasma treatment increased expression in a smaller subset of these genes, while elderly plasma did not lead to significant changes (Extended Data Fig. 3a, b). At the protein level, only cord plasma treatment significantly increased the number of c-Fos-expressing cells within aged dentate gyrus (Fig. 1e and Extended Data Fig. 3c). The activation seemed to depend on context, since the pattern was not observed in other regions, including amygdala and motor cortex (Extended Data Fig. 3d, e), nor was it observed in young hippocampi (Fig. 1f). Human plasma treatments did not alter phosphorylated cyclic AMP responsive element-binding protein (CREB)16 or hippocampal neurogenesis…“
Already in early 2017 Wyss-Coray and his company Alkahest knew the therapy does not work. Their open-label clinical trial NCT02256306 with 18 participants was named “The PLasma for Alzheimer SymptoM Amelioration (PLASMA) Study (PLASMA)“, its results were published two years after completion, as Sha et al JAMA Neurology 2019:
“The efficacy of yFFP [young fresh frozen plasma, -LS] in patients with mild to moderate AD could not be determined because of the small sample size, a change in the design of the study, and the short duration of treatment. Therefore, assessments of cognition, mood, functional ability, and default mode network changes were exploratory. Analyses of these measures did not find that infusions of yFFP altered mood, global cognition, or functional connectivity. However, improvements in functional abilities were reported by caregivers.“
Basically, they found no measurable effect whatsoever, and others noticed it also. But this is exactly why Alkahest started a phase 2 clinical trial NCT03765762 with more Alzheimer’s patients, as their CEO Karoly Nikolich announced back then:
“Based on the promising data from our PLasma for Alzheimer SymptoM Amelioration (PLASMA) study last year, we have progressed the development of GRF6019, a proprietary plasma fraction developed and provided by Grifols, our development and clinical partner.“
The results from the clinical trials NCT03765762 (Alzheimer’s) and NCT03713957 (Parkinson’s) haven’t been published in a peer-reviewed journal or as a preprint yet. They don’t have to be, Alkahest was just sold for 150 million, whether the young blood therapy works or not, is someone else’s problem now.
Dr Wyss-Coray didn’t reply to my email asking to comment on PubPeer issues and his company’s clinical trials.
PS: one doesn’t need to bleed any teenagers anymore. A stool transplant will do, as the Irish professor John F Cryan discovered in Boehme et al 2021!
PPS: More anti-aging cures (I expect you to pay me a kickback fee!):

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Or send blood (young readers only)!
€5.00



A small comment regarding the percentages in Figure 1k of Marschallinger et al. (2020), doi:10.1038/s41593-019-0566-1: it is in principle possible that such a sum of percentages is a little greater (or smaller) than 100% due to rounding of the individual values. Not sure if that’s the case here, but it is in itself not so strange.
LikeLike
That Fig 1k issue was corrected. Wyss-Coray explained on Pubpeer:
“In the pie charts we had the “Ceramids” (Ce) included into the group “other” at the beginning. Then decided to visualize the ceramids separately. So the % of the others is the value minus the ceramids (8.8% for Hippocampus; 7.4% for Microglia).”
https://pubpeer.com/publications/169E529839E6DE61FFCBB320A7C68B#5
LikeLike
The rounding up or down could only be in the last digit, which means that even in the very worst possible case, the discrepancy could only be 6 times 0.1 in each sum (as there are 6 summands in each sum). So the total could not possibly be more than 100.6, and in each case it is more. So rounding up does not explain the errors here.
(Of course, it is not possible to have all summands increasing when rounding them, so the maximum possible legitimate discrepancy is even smaller than 0.6.)
LikeLike
Why did we stop using monkey testicles?
A reader sent me this account of Dr Serge Voronoff, the Russian-French surgeon who lived a century ago and who invented the term “rejuvenation” and the therapy to go with it, by suturing pieces of monkey testicles into his ageing patients’ scrotums.
https://www.atlasobscura.com/articles/the-true-story-of-dr-voronoffs-plan-to-use-monkey-testicles-to-make-us-immortal
“By 1923, Voronoff was the director of the experimental laboratory at the Collège de France, and his testicular grafts had gained such popularity and acclaim that a special reserve was being set-up in Africa specifically to capture and maintain monkeys for gland transfer. He presented his procedure to the thousands-strong International Congress of Surgeons in London, wowing the assembly with his seemingly ground-breaking solution to aging (and, ideally, impotence). At the conference he also touted that his procedures would soon be available to women as well, and promised to turn “grandmothers into debutantes.” Voronoff’s enthusiasm for shoving monkey testicles into the human body was both ambitious and infectious. By the mid-1920s, at least 300 people had undergone his procedure, including at least one woman receiving a xenograft of monkey ovary.
The demand for Voronoff’s procedure continued to increase, as did his ambition. Following in the mold of any classic mad scientist, Voronoff bought himself a castle in Grimaldi, Italy in 1925. Citing the prohibitive costs and hassle of transporting monkey parts for his procedures, Voronoff built a primate enclosure in the garden and hired a former circus trainer to manage his new farm. Castle Voronoff was also outfitted with a small hospital where the doctor could perform the grafts.
In a 1927 article printed in the Delaware Star, Voronoff claimed that his procedure had been performed over 1,000 times throughout the world, restoring septuagenarians to the strength of their youth. Even more startlingly, Voronoff said that he had begun administering the rejuvenating testicle grafts to young sheep, essentially creating a race of super-sheep. “
LikeLike
William Empson’s 1937 villanelle Missing Dates seems to refer to an earlier version of this scam, but I’ve never bothered to track it down. No doubt Smut Clyde has the matter in his copious files.
========
Slowly the poison the whole blood stream fills.
It is not the effort nor the failure tires.
The waste remains, the waste remains and kills.
It is not your system or clear sight that mills
Down small to the consequence a life requires;
Slowly the poison the whole blood stream fills.
They bled an old dog dry yet the exchange rills
Of young dog blood gave but a month’s desires.
The waste remains, the waste remains and kills.
It is the Chinese tombs and the slag hills
Usurp the soil, and not the soil retires.
Slowly the poison the whole blood stream fills.
Not to have fire is to be a skin that shrills.
The complete fire is death. From partial fires
The waste remains, the waste remains and kills.
It is the poems you have lost, the ills
From missing dates, at which the heart expires.
Slowly the poison the whole blood stream fills.
The waste remains, the waste remains and kills.
LikeLike
Pingback: Paul Craddock’s “Spare Parts” – Book Review – For Better Science
Pingback: Toppling Giants in Stanford – For Better Science