It is the Nobel Prize week, and yet you, dear reader, came to this website where Nobelists are constantly being pooh-poohed upon. So you shall not be disappointed, for our hero here is the winner of the 2019 Nobel Prize for Physiology or Medicine, Gregg Semenza, professor at the Johns Hopkins University in USA.
Semenza shared his 2019 Nobel Prize with two other researchers, but only his papers keep popping up on PubPeer bearing telltale signs of data fakery. There are some recurrent author names suggesting naughty mentees or collaborators, but still, in many cases Semenza is the last and corresponding author, so the final responsibility is his. After all, also Nobel Prize recognition comes from that same last authorship.
First, some scientific background. The Nobel Prize Committee explains the 2019 award scientific rationale:
“Animals need oxygen for the conversion of food into useful energy. The importance of oxygen has been understood for centuries, but how cells adapt to changes in levels of oxygen has long been unknown. William Kaelin, Peter Ratcliffe, and Gregg Semenza discovered how cells can sense and adapt to changing oxygen availability. During the 1990s they identified a molecular machinery that regulates the activity of genes in response to varying levels of oxygen. The discoveries may lead to new treatments of anemia, cancer and many other diseases.”
Like everything else in cancer research, the hypoxia field is full of fraud, just search “HIF” (hypoxia-inducible factor which Semenza discovered) alone on PubPeer. One should hope the Nobel Prize (and Lasker, and other award-winning) labs might provide a pleasant exception, but alas…
In an interview with the Journal of Clinical Investigation (JCI) on the occasion of his Nobel Prize, Semenza was quoted:
“The one thing I pretended when I was younger was that I was an FBI agent. Investigating crimes, being a detective, it’s like being a scientist, right?“
Well, now science detective Clare Francis had a look into Semenza’s papers. And there was a lot, over 20 flagged papers on PubPeer before Clare Francis gave up and moved on to another case. The findings in Semenza’s research are rivalling those of another winner of the Nobel Prize in Physiology or Medicine, Louis Ignarro, so this is rather serious.
Have a look at this 8-year old paper on HIF-1 from the Semenza lab:
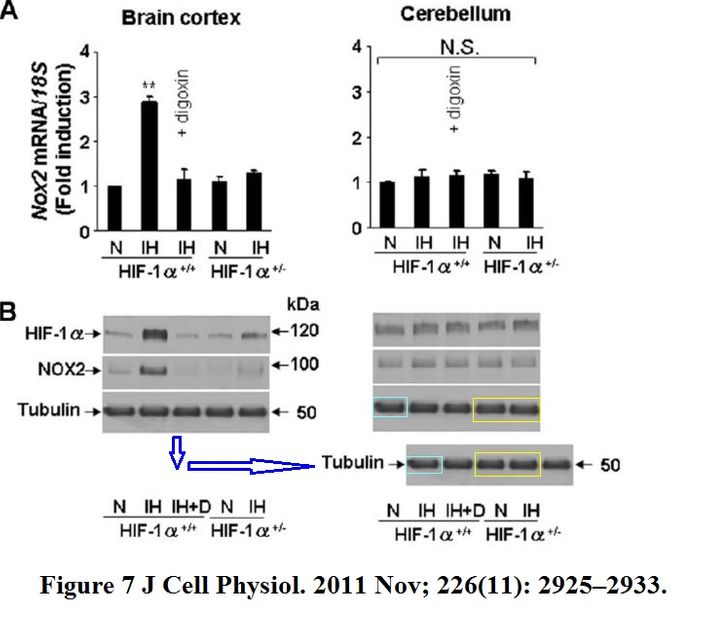
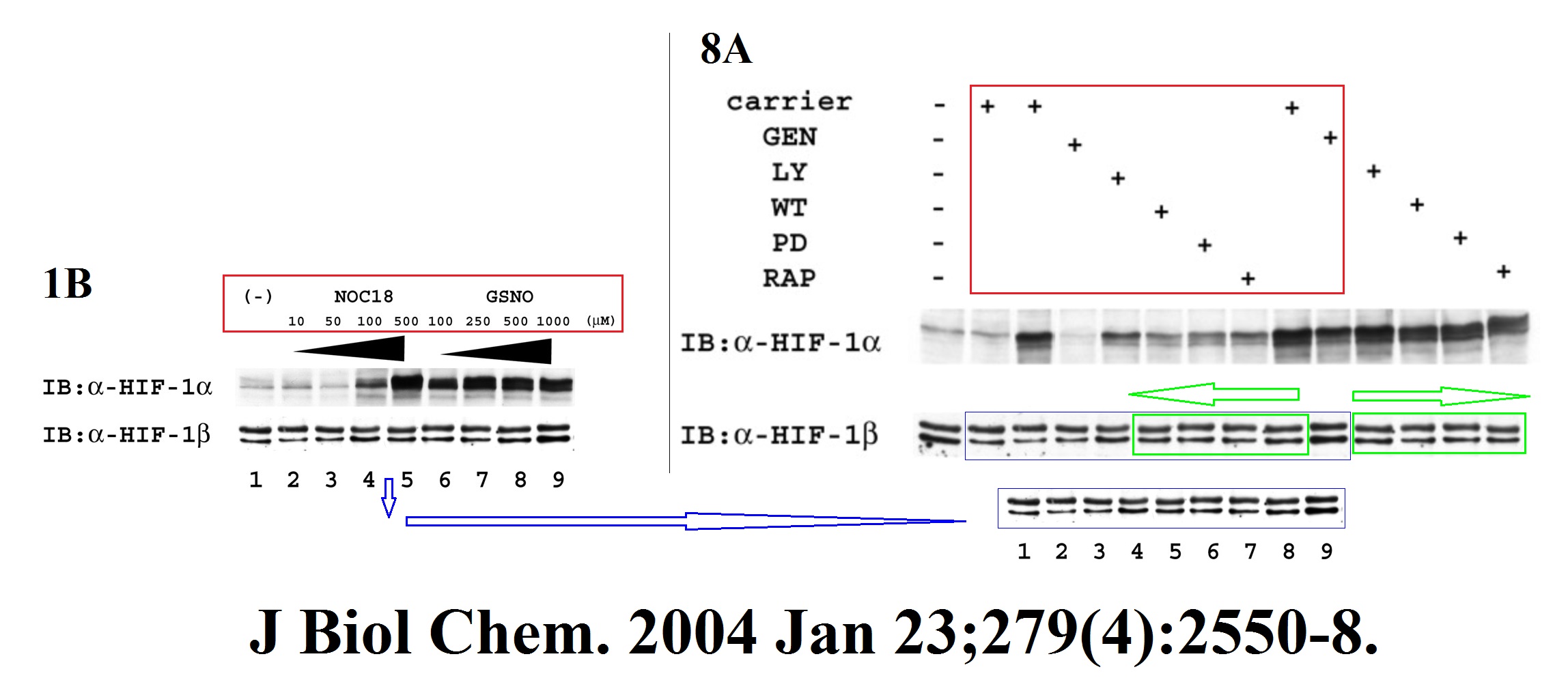
H Zhang, C C L Wong, H Wei, D M Gilkes, P Korangath, P Chaturvedi, L Schito, J Chen, B Krishnamachary, P T Winnard, V Raman, L Zhen, W A Mitzner, S Sukumar, G L Semenza HIF-1-dependent expression of angiopoietin-like 4 and L1CAM mediates vascular metastasis of hypoxic breast cancer cells to the lungs Oncogene (2012) doi: 10.1038/onc.2011.365
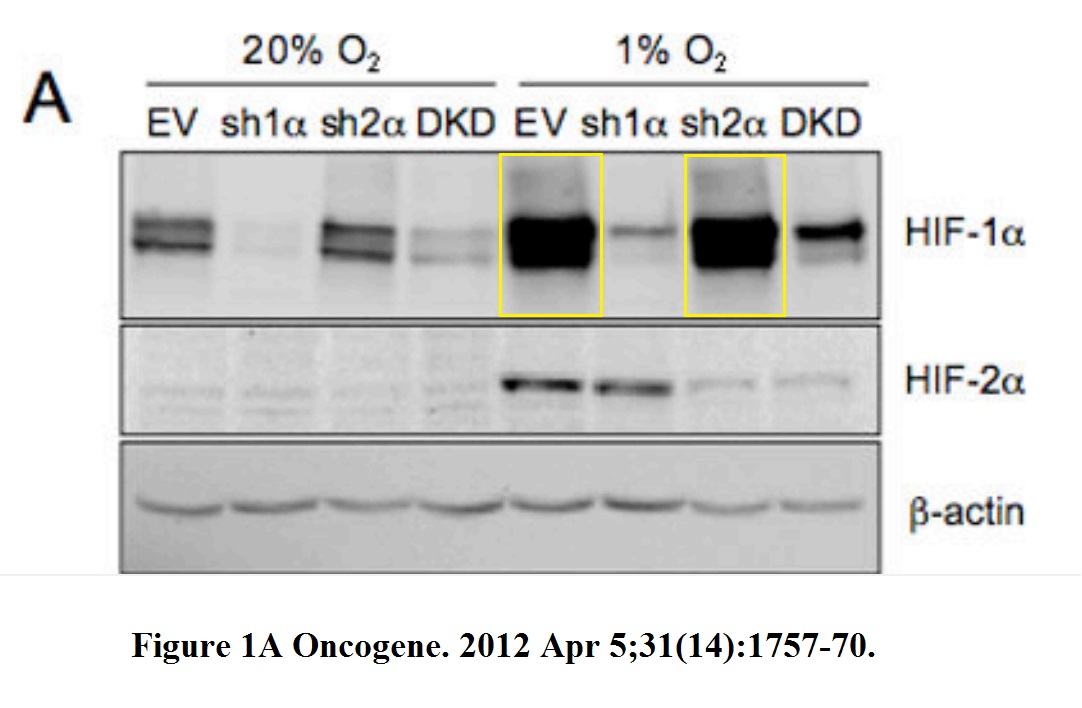
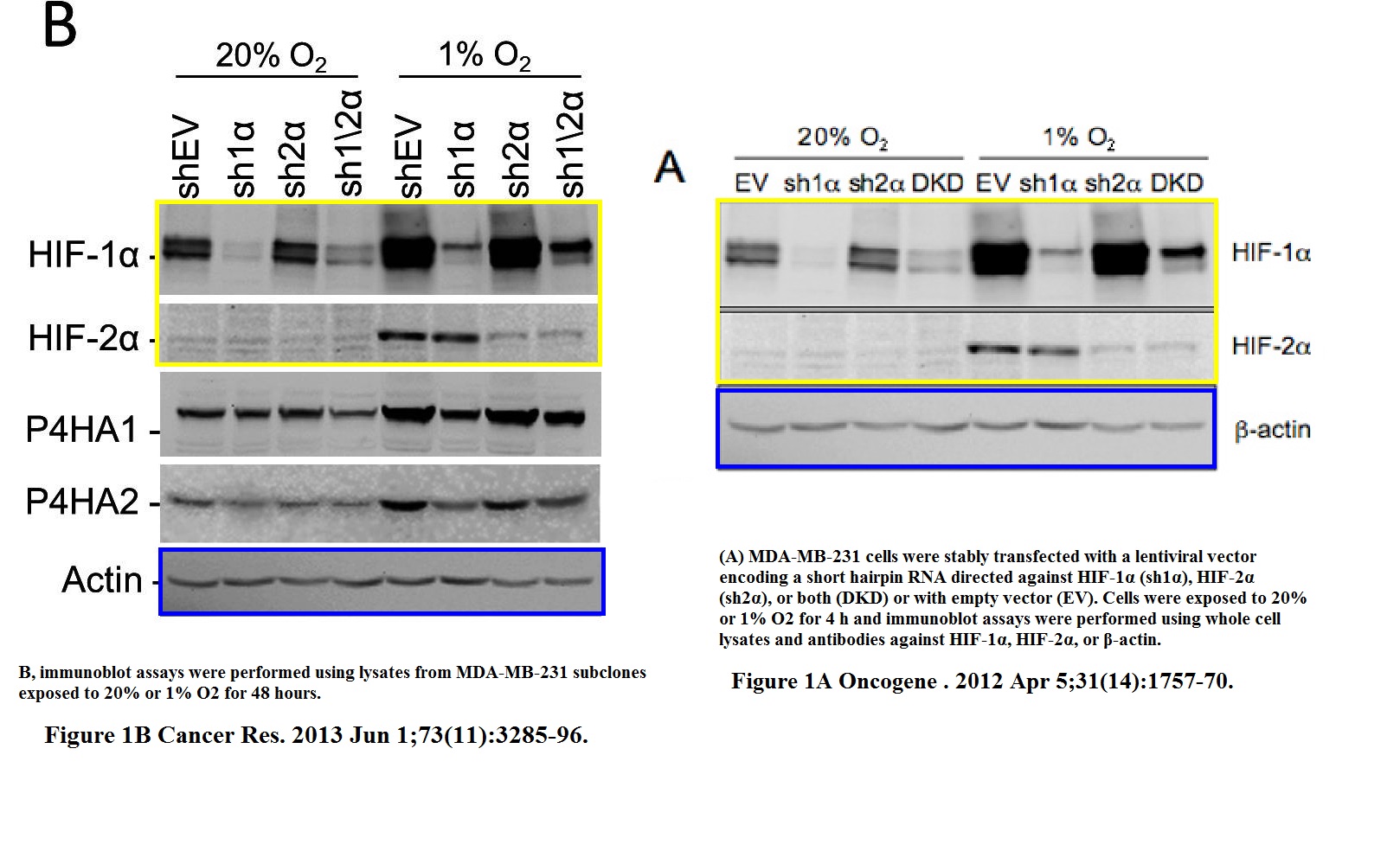
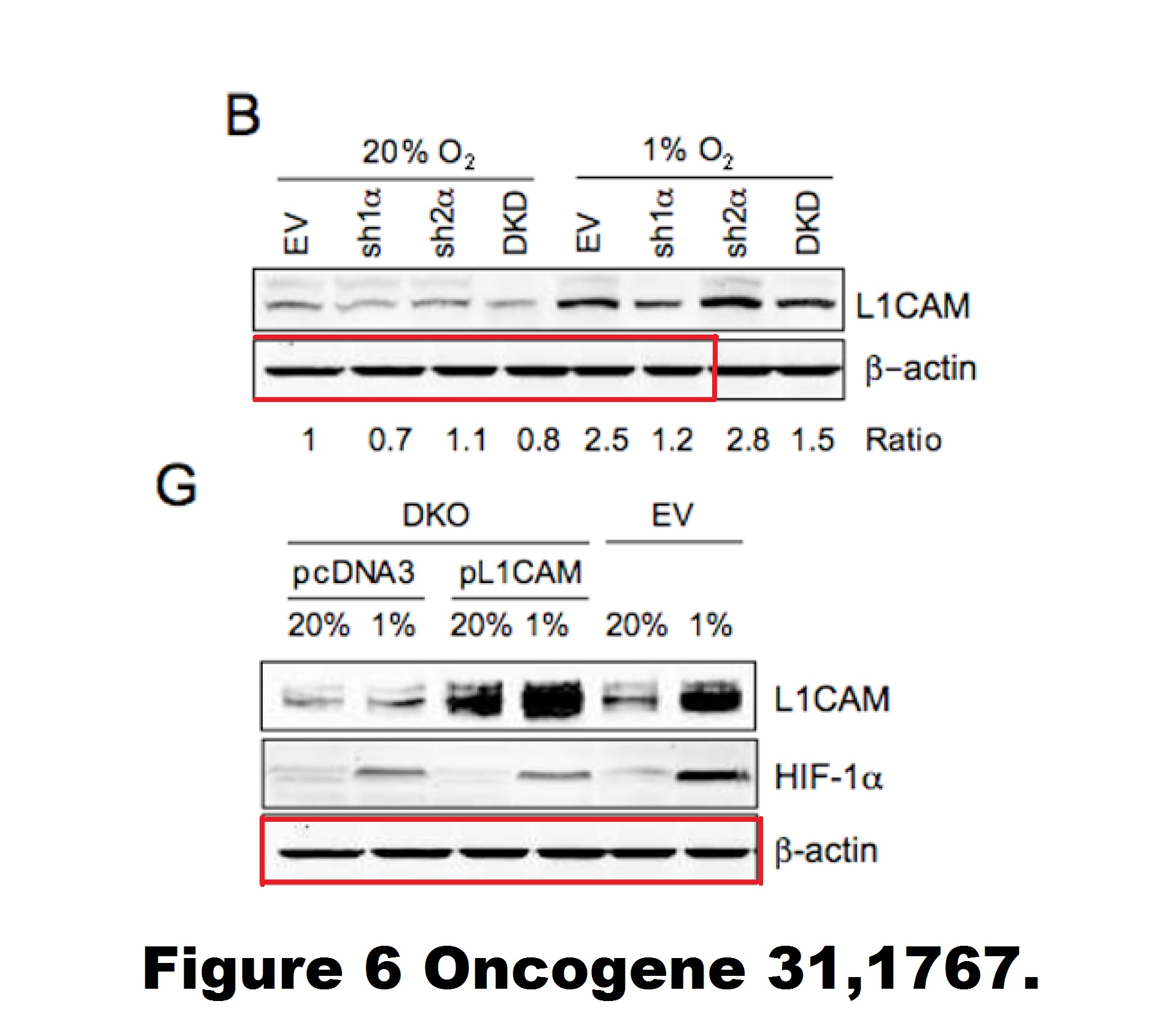
A gel in Figure 1A contains two bands which are likely copy-pasted. Figure 6 reuses a loading control for utterly unrelated experiments and samples. Judging from the band shapes and spacing, the beta-actin blot does not match either the gel shown in panel B nor G.
The issue with Figure 1A is minor compared to that: the research results were recycled in a different paper, Gilkes et al Cancer Research 2013. Luckily for all involved, Oncogene‘s Editor-in-Chief Justin Stebbing practices research integrity only in theory.
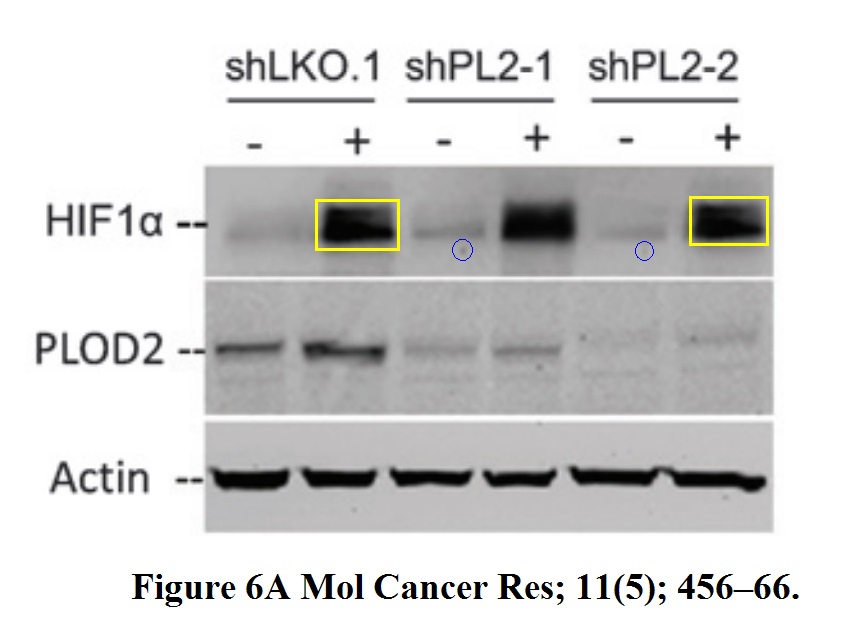
Or how about this 7 year old paper, again with Semenza as last author, where the Nobel-Prize-winning HIF1a data suffered some copy-pasting.
Daniele M. Gilkes, Saumendra Bajpai, Carmen C. Wong, Pallavi Chaturvedi, Maimon E. Hubbi, Denis Wirtz, Gregg L. Semenza Procollagen lysyl hydroxylase 2 is essential for hypoxia-induced breast cancer metastasis Molecular cancer research : MCR (2013) doi: 10.1158/1541-7786.mcr-12-0629
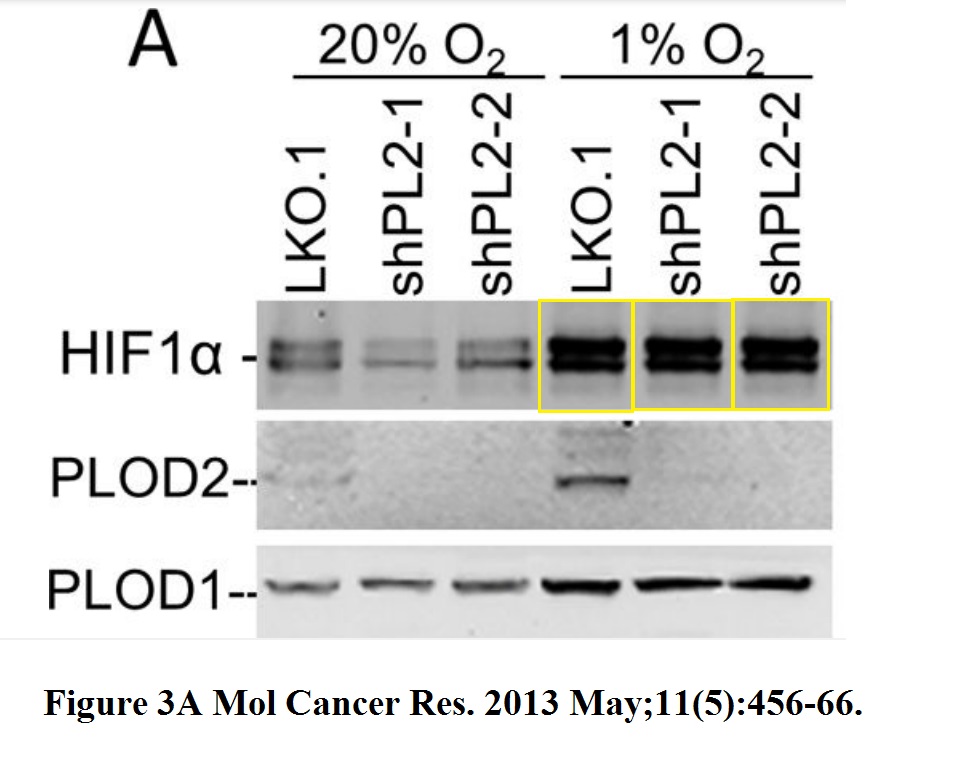
The first author of the above paper, Daniele Gilkes, remained close to her mentor and is now assistant professor at Johns Hopkins. In case you might think Semenza had no clue what his postdoc was doing, here their own words:
“Having a mentor that was both a clinician and a scientist was extremely important and impactful for my training,” Gilkes said. “Gregg helped me to focus on research that could make a true impact on improving outcomes.” […]
She noted that even with as many as 15 people in the lab, Semenza always responded quickly to her questions.
“I try to do that for my students, because that was really important to me,” she said. “It made me feel like he was very interested in the work that I was doing and that it was important to him. But it’s difficult to emulate.”
Is Gilke teaching her student the Art of the Photoshop now? Maybe. Here another first-author paper of hers from Semenza lab, where Johns Hopkins University’s Vice Provost for Research, Denis Wirtz, is again coauthor. In this capacity, Wirtz would be in charge of investigating the Nobelist’s papers (including those he himself coauthored) for suspected research misconduct.
Daniele M. Gilkes, Saumendra Bajpai, Pallavi Chaturvedi, Denis Wirtz, Gregg L. Semenza Hypoxia-inducible factor 1 (HIF-1) promotes extracellular matrix remodeling under hypoxic conditions by inducing P4HA1, P4HA2, and PLOD2 expression in fibroblasts The Journal of biological chemistry (2013) doi: 10.1074/jbc.m112.442939
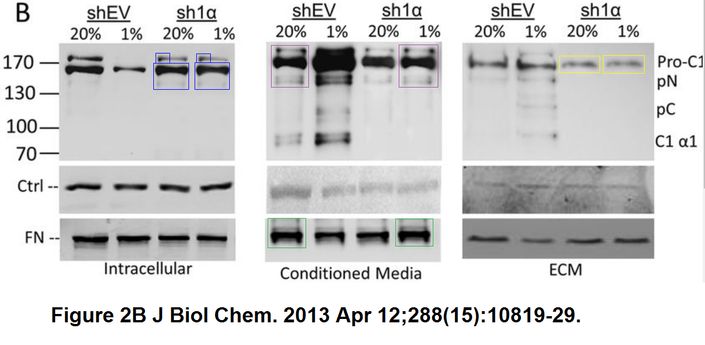

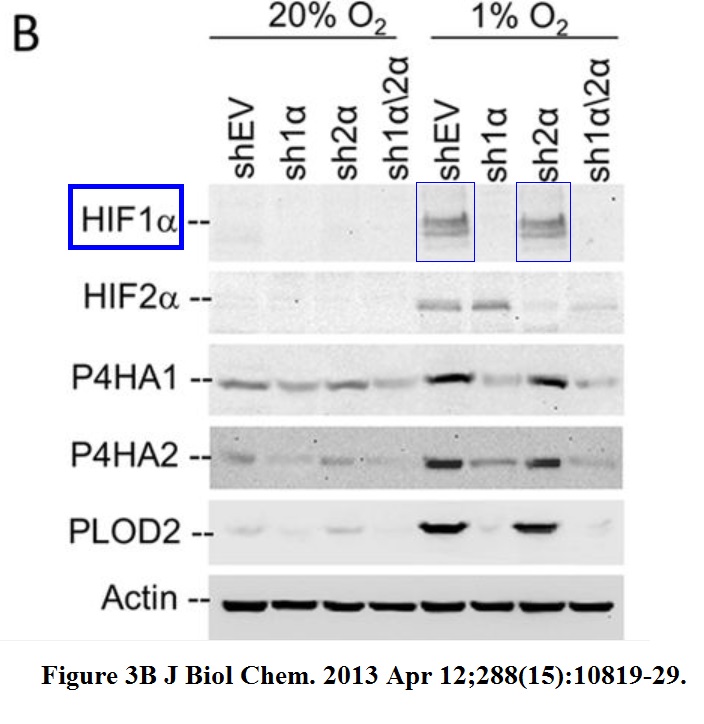
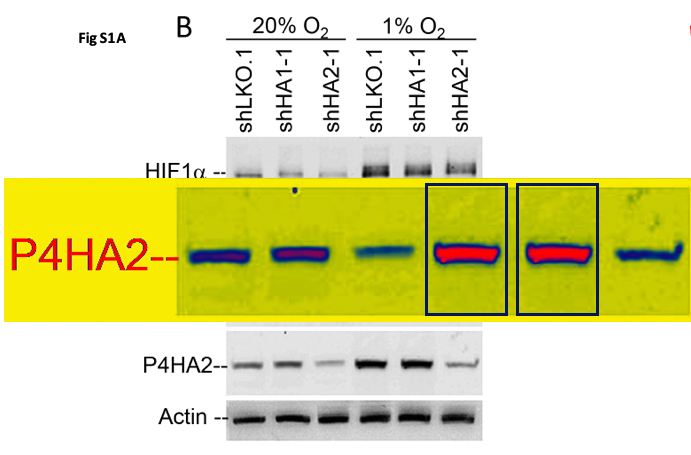
An utter disaster. Someone should be worried now, because the journal JBC and its society publisher ASBMB are quite tough on data fakery.
The next Gilkes et al paper from Semenza’s lab, again with Wirtz as coauthor, is also an utter train wreck, and is only safe from any danger of retraction because PNAS never cares about such things, certainly not with contributed (i.e., not really peer reviewed) papers. And this one was eminently contributed by the National Academy of Sciences member Semenza himself.
Daniele M Gilkes, Lisha Xiang, Sun Joo Lee, Pallavi Chaturvedi, Maimon E Hubbi, Denis Wirtz, Gregg L Semenza Hypoxia-inducible factors mediate coordinated RhoA-ROCK1 expression and signaling in breast cancer cells Proceedings of the National Academy of Sciences of the United States of America (2014) doi: 10.1073/pnas.1321510111

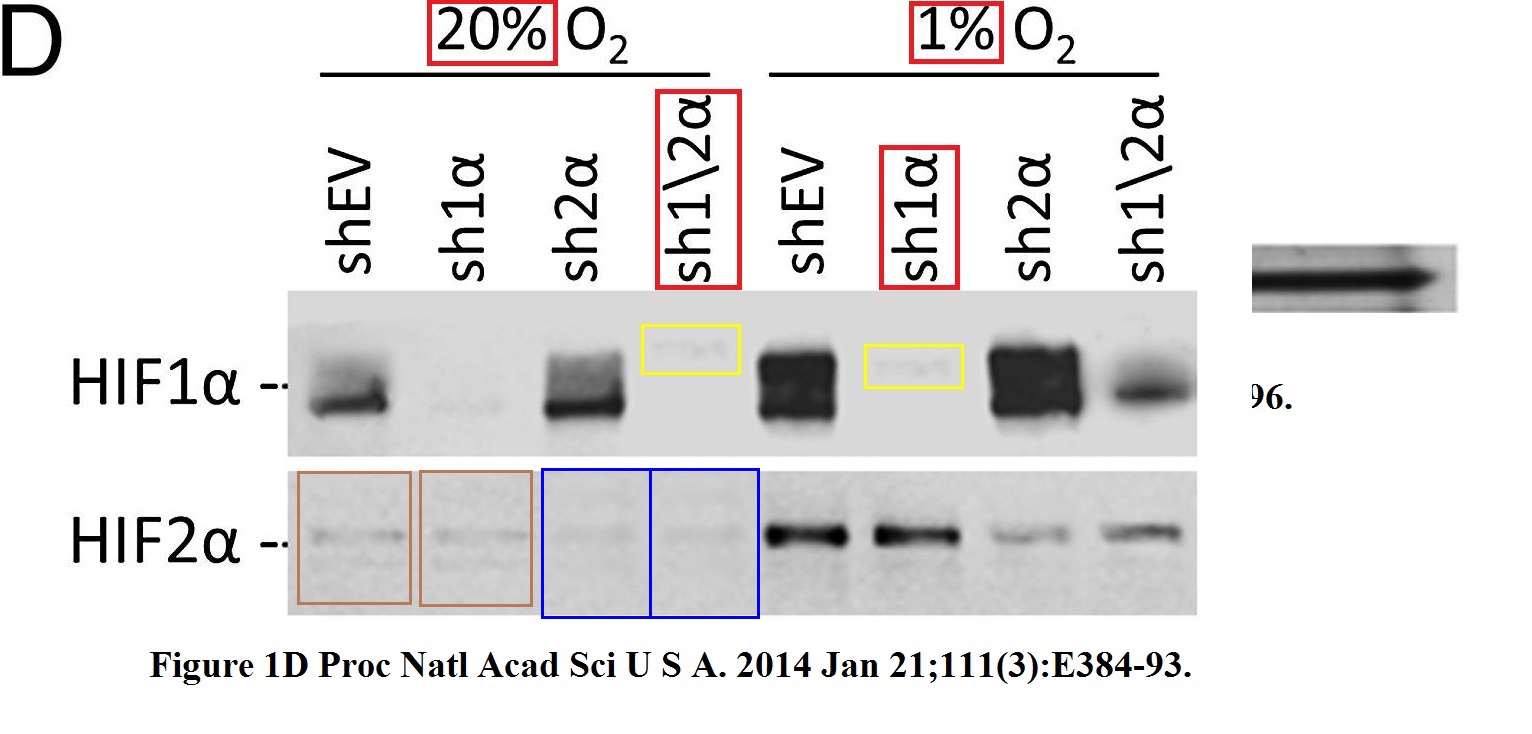
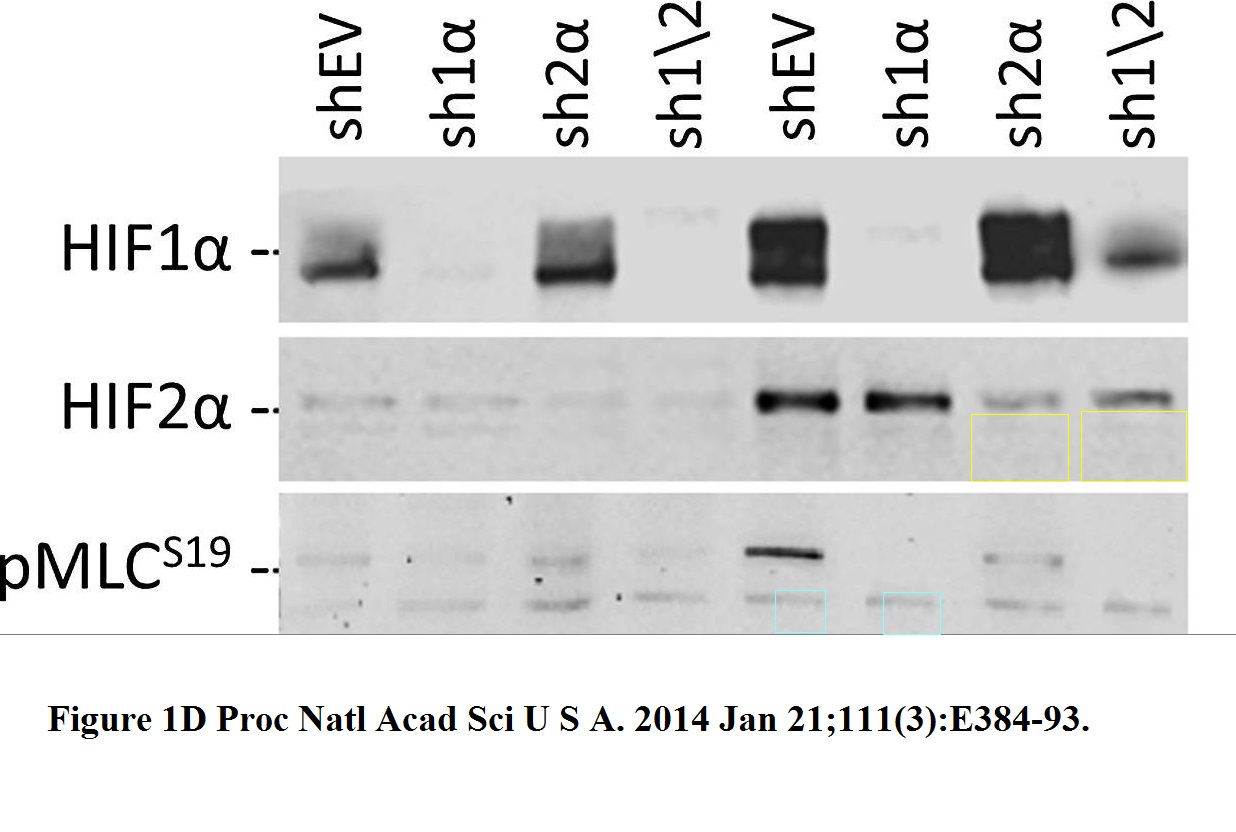

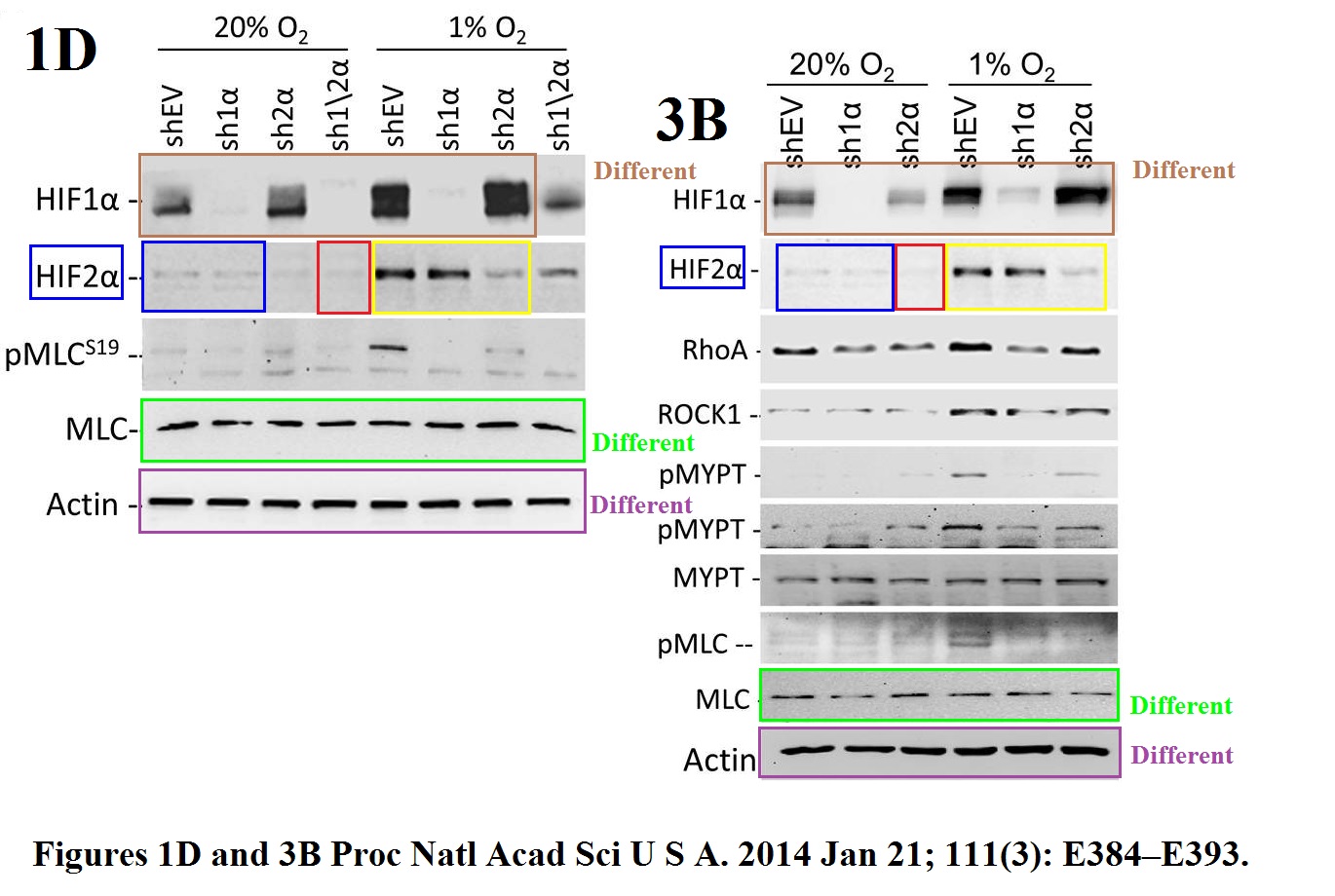
The HIF2a gel was reused, after some splicing, the sample labels remained same, except the red-boxed one. But why did the authors recycle only that gel, and not the loading control Actin, or HIF1a or pMLC? Maybe becasue the two figures show slightly different results for these?
The Figure 5C of the next Gilkes et al paper from Semenza lab was fixed in 2013, because it was “incorrect”. No details were provided by the journal, of which Semenza is deputy Editor-in-Chief, so here Clare Francis:
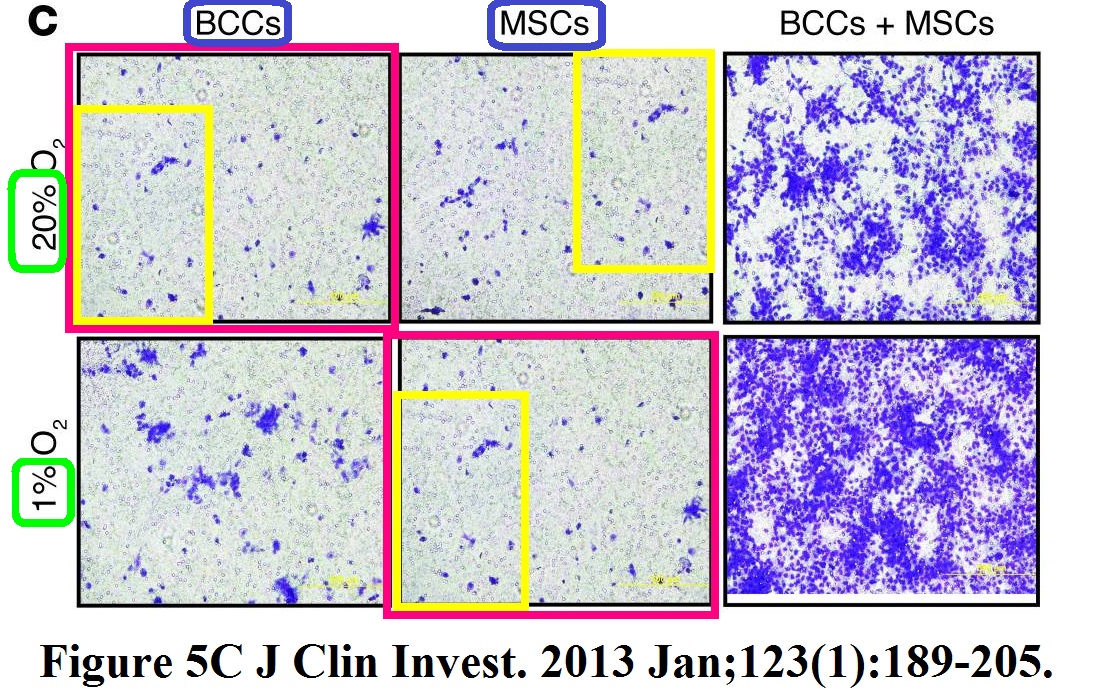
Pallavi Chaturvedi, Daniele M. Gilkes, Carmen Chak Lui Wong, Kshitiz, Weibo Luo, Huafeng Zhang, Hong Wei, Naoharu Takano, Luana Schito, Andre Levchenko, Gregg L. Semenza Hypoxia-inducible factor–dependent breast cancer–mesenchymal stem cell bidirectional signaling promotes metastasis The Journal of clinical investigation (2012) doi: 10.1172/jci64993
Semenza’s deputy Chief Editor colleague at JCI, Arturo Casadevall, is an internationally celebrated champion of research ethics, but only in theory, e.g. it does not seem to apply to his own papers.
When the Nobelist was interviewed in January 2020 about his work as JCI Deputy chief editor, by his editorial board colleague Elyse Dankoski, she somehow mentioned that “JCI’s current editorial board seems committed to tackling problems in scientific publishing, such as bias in first authorship as well as data manipulation issues“. I wonder if Semenza winced, or chuckled darkly. He was also asked: “Can you share any insights into what you’re looking for when you evaluate a submitted article?“.
The reply was hilarious:
“Pretty simple, just answer two questions:
Is this work novel and impactful?
Do the results adequately support the conclusions?“
Do you, dear reader, think those cloned gel bands were sufficiently novel and impactful and do they affect the conclusions of Nobel Prize winning papers from the Johns Hopkins lab of Dr Semenza?
Let me show you a bit more, now some collaborative research.
For example, there was a collaboration of Semenza with Nanduri Prabhakar of University of Chicago. Prabhakar even wrote an editorial on the occasion of Semenza’s Nobel Prize in the society journal Physiology. All three papers below have the same first author Guoxiang Yuan from Chicago, but would they have been published in these journals if not for Semenza’s significant coauthorship? Not in PNAS for sure, where Semenza “contributed” the paper to bypass peer review.
All these copy-pasted gel bands are depressing. But even more depressing is the knowledge that none of these three journals is likely to retract them, or to do anything in the first place. Because Semenza won the Nobel Prize.
There were certainly strange things going on in Semenza’s lab. The following might look like a misunderstanding, or a mistake, but seems to be more sinister:
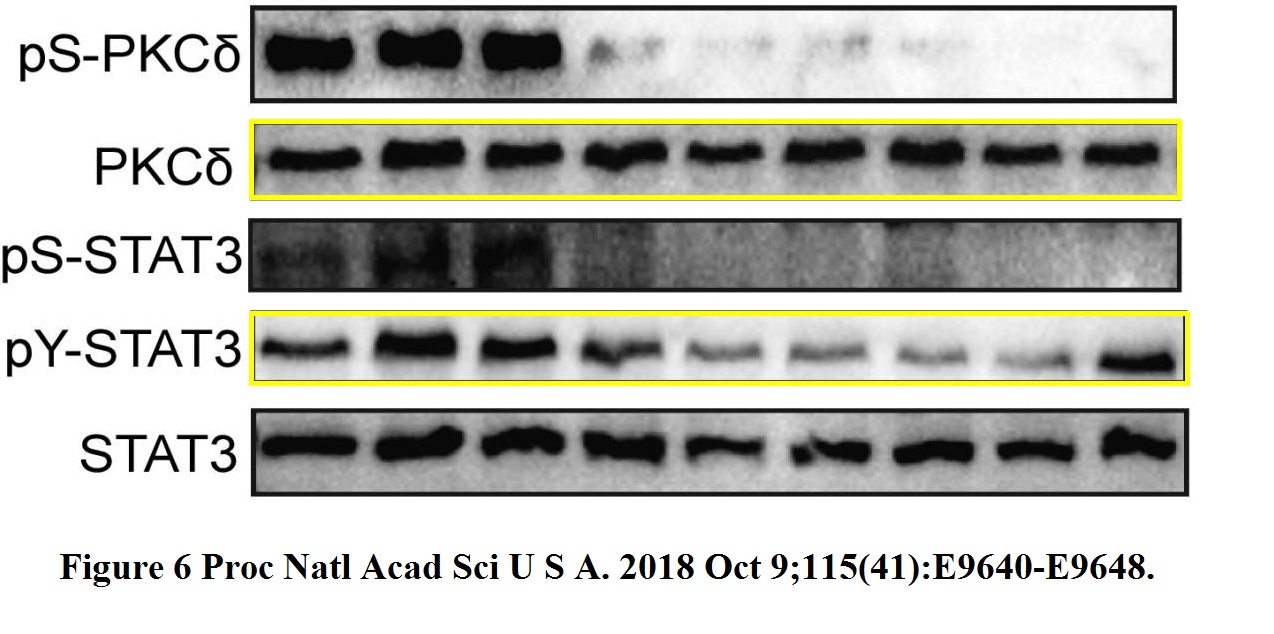
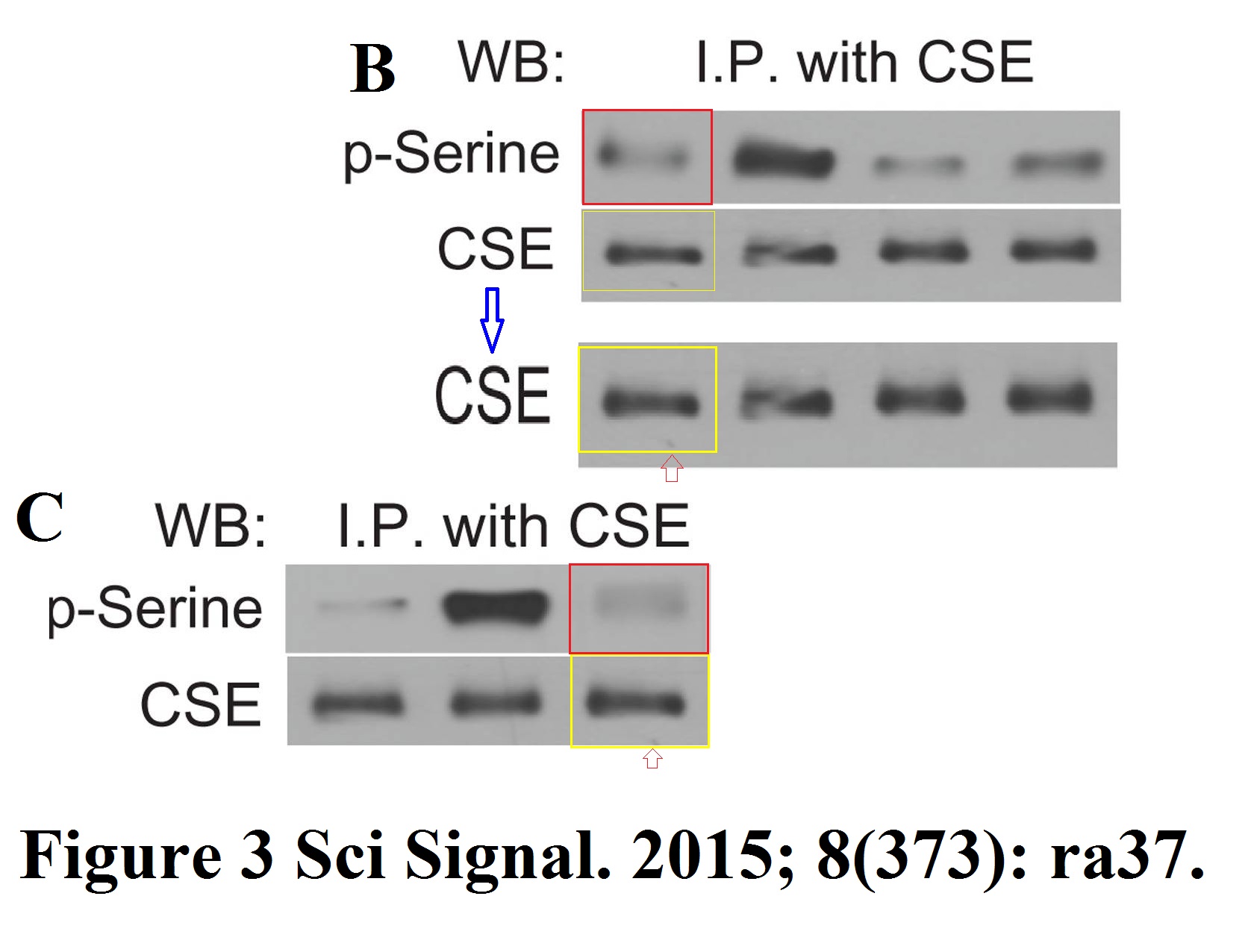
Jie Lan, Haiquan Lu, Debangshu Samanta, Shaima Salman, You Lu, Gregg L. Semenza Hypoxia-inducible factor 1-dependent expression of adenosine receptor 2B promotes breast cancer stem cell enrichment Proceedings of the National Academy of Sciences of the United States of America (2018) doi: 10.1073/pnas.1809695115
There should be two gels, one probed for PKCdelta (total and phosphorylated) and another probed for STAT3 (total and phosphorylated). But the PKCdelta and pY-STAT3 bands are so similar they must have come from same gel. Is the declared pY-STAT3 signal really the pS-PKCdelta signal? In this case, what happened to the correct pY-STAT3 gel, and what are the other gels really showing then? This cannot happen by mistake. Luckily, also this PNAS paper was contributed by Semenza, so there is no case, as far as the journal is concerned.
When the discoverer of the HIF proteins received his Nobel Prize phone call, he said:
“Yeah, well certainly nobody expects that, that’s for sure. Even after people have been telling you for, you know, 20 years or more that it’s going to happen, no one expects it.“
He might have expected the Nobel, but did Semenza expect Clare Francis?
Another collaborative paper with a similar problem prompted one Cell Press editor to admit he either has no clue of molecular biology, or does not care a bit about basic data integrity.
De Huang, Tingting Li, Xinghua Li, Long Zhang, Linchong Sun, Xiaoping He, Xiuying Zhong, Dongya Jia, Libing Song, Gregg L. Semenza, Ping Gao, Huafeng Zhang HIF-1-mediated suppression of acyl-CoA dehydrogenases and fatty acid oxidation is critical for cancer progression Cell Reports (2014) doi: 10.1016/j.celrep.2014.08.028
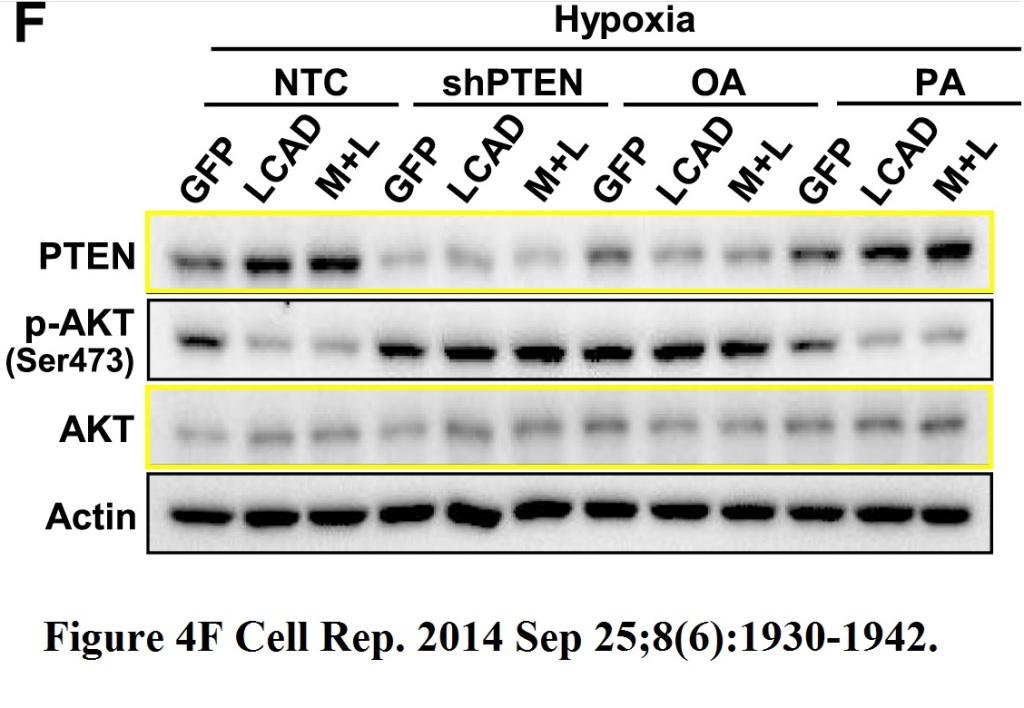
Basically, the phospho-AKP blot comes from a different gel for which no loading control was done. Stephen Matheson, Editor-in-Chief of Cell Reports, wrote to Clare Francis:
“I agree, but I don’t think those at all need to be from the same blot. This is not problematic.“
That is only partially correct, Steve. There can be indeed more than one gel, but each of those must be supplied with its own loading control. How do you know that for the p-AKT gel the same amount of protein lysate was loaded in, say, lane 1 as opposed to lane 2 and 3, or lane 4? Answer: blind trust because one of the authors went on to win a Nobel Prize. Semenza is by the way the only non-China-based author on that paper, you can guess what his actual contribution was. Yes, I am aware that gift authorships are a form of scientific misconduct. But see, it helped not only to get that paper published in a Cell family journal, but also to get it to remain published there.
As it will have helped another Cell Press paper, this time from South Korea, where Semenza’s name again stands out like a sore thumb, as the only contributor from Johns Hopkins.
Jason S. Lee, Yunho Kim, Ik Soo Kim, Bogyou Kim, Hee June Choi, Ji Min Lee, Hi-Jai R. Shin, Jung Hwa Kim, Ji-Young Kim, Sang-Beom Seo, Ho Lee, Olivier Binda, Or Gozani, Gregg L. Semenza, Minhyung Kim, Keun Il Kim, Daehee Hwang, Sung Hee Baek Negative regulation of hypoxic responses via induced Reptin methylation Molecular cell (2010) doi: 10.1016/j.molcel.2010.06.008
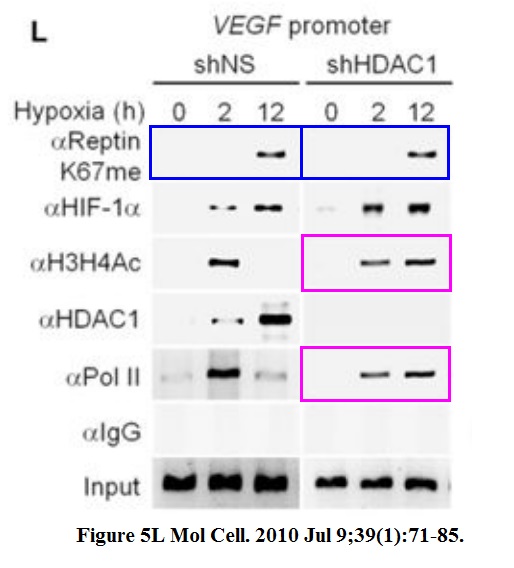
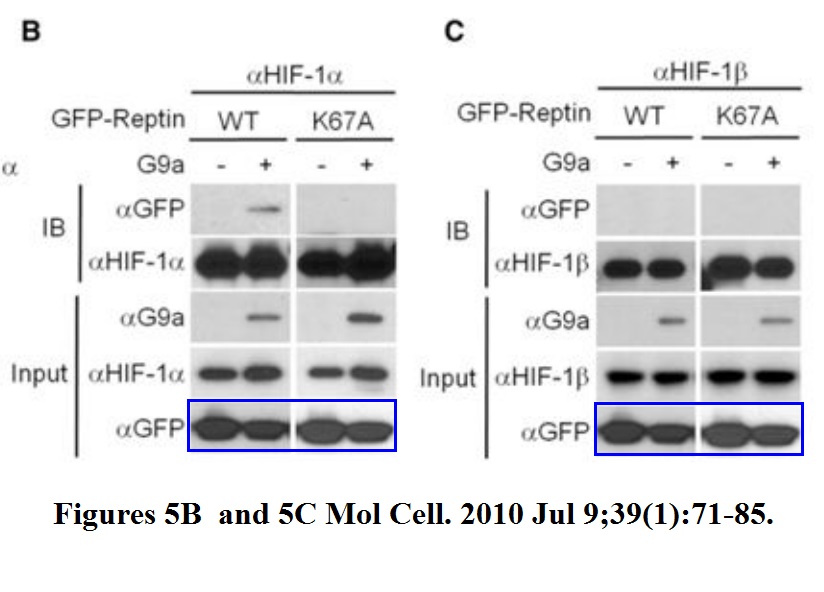
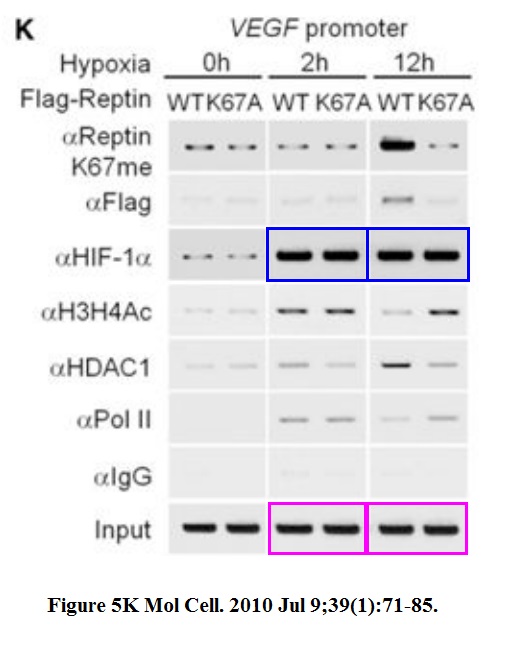
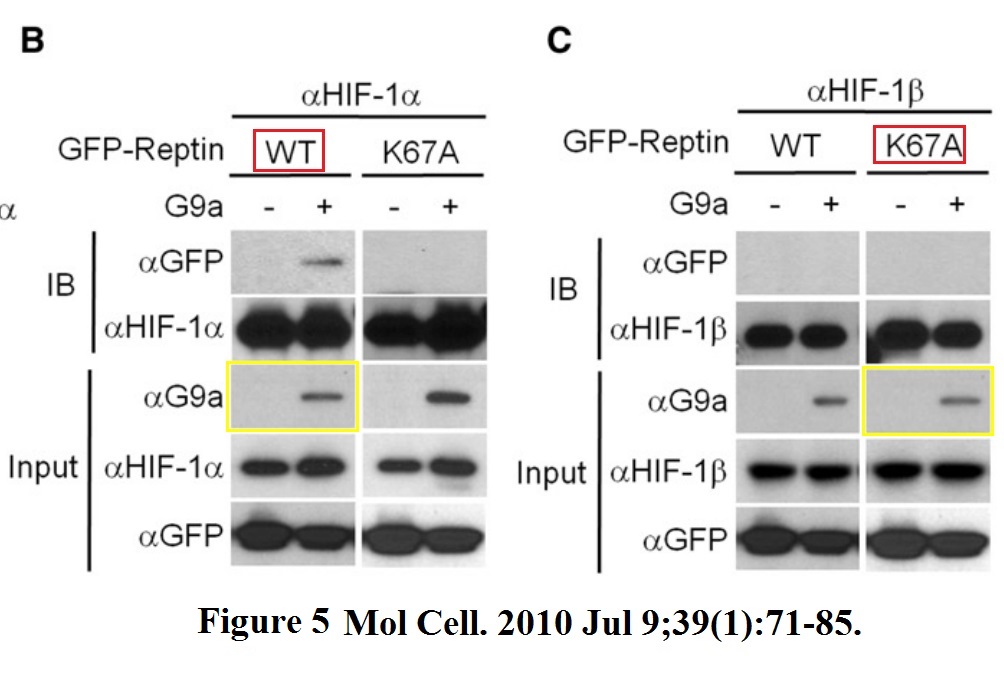
Nobody, absolutely nobody at Molecular Cell will have any problem with this. This is a journal which rejects retraction requests from universities. Maybe this is a good opportunity to wonder, sure, Cell Press and Elsevier in general does not give a flying toss about falsified data, but how about torturing small animals? Is it their thing also? Does it get them off or will they finally get off their bums and do something about scientific malpractice? Don’t hold your breath. Have a look at what was done to these mice:
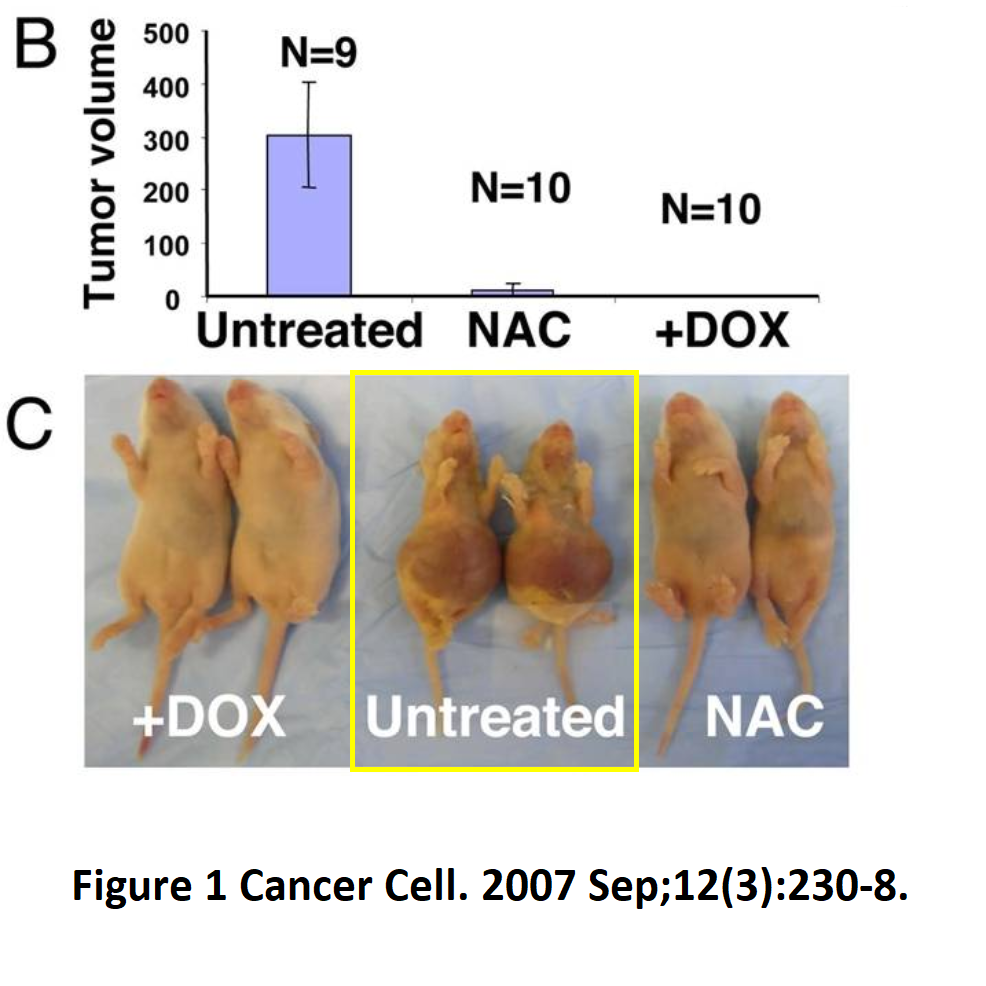
Ping Gao, Huafeng Zhang, Ramani Dinavahi, Feng Li, Yan Xiang, Venu Raman, Zaver M. Bhujwalla, Dean W. Felsher, Linzhao Cheng, Jonathan Pevsner, Linda A. Lee, Gregg L. Semenza, Chi V. Dang HIF-dependent antitumorigenic effect of antioxidants in vivo Cancer Cell (2007) doi: 10.1016/j.ccr.2007.08.004
This animal research work was done at Johns Hopkins. Did nobody really notice that the xenograft tumours grew so huge that the animals must have horribly suffered? That the nude mice should have been euthanised long before the tumours became this big, as per standard animal experimenting guidelines in USA? The authors failed there, and so did the editors and peer reviewers. Will something happen to this Cancer Cell paper now? Don’t be silly, Semenza won the Nobel Prize.
Better let’s look at some cloned western blot bands again, those are at least funny. Here some older stuff Semenza published with Kiichi Hirota, now professor at Kansai Medical University in Japan.
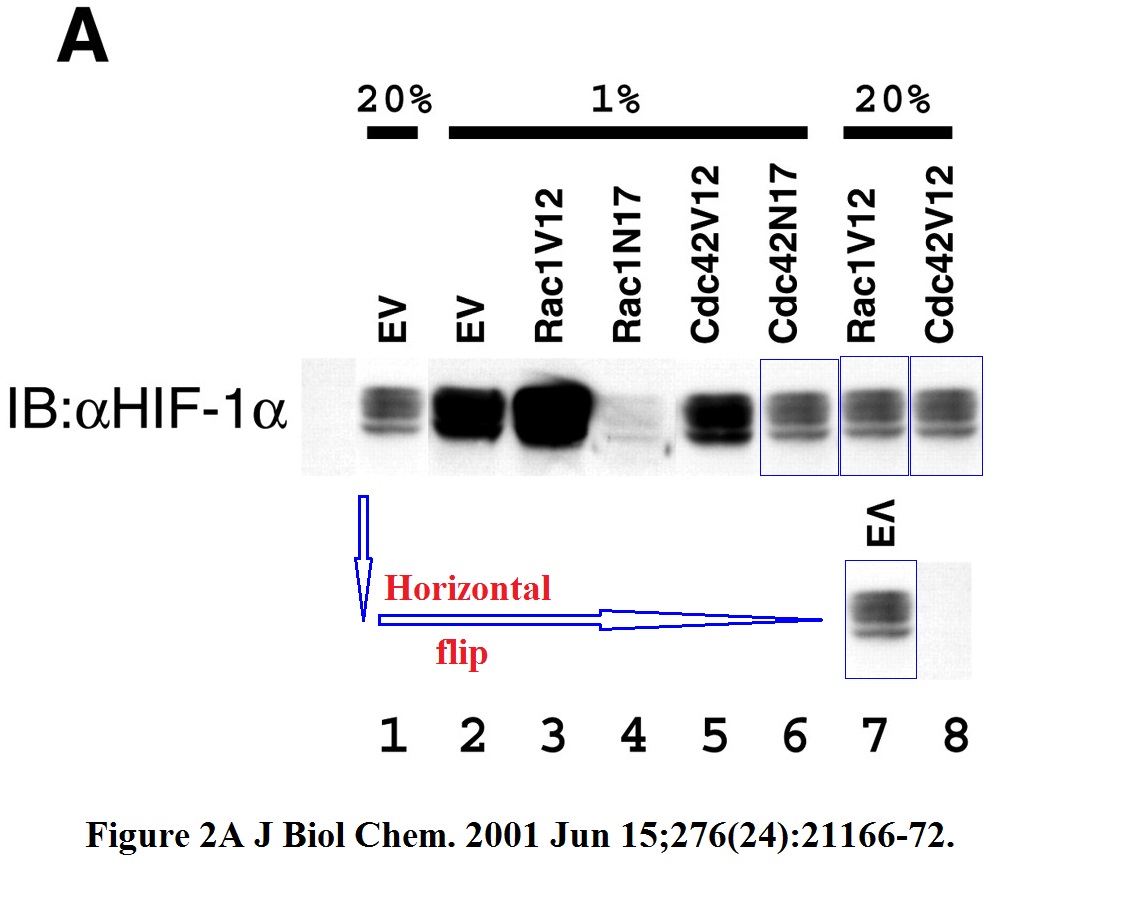
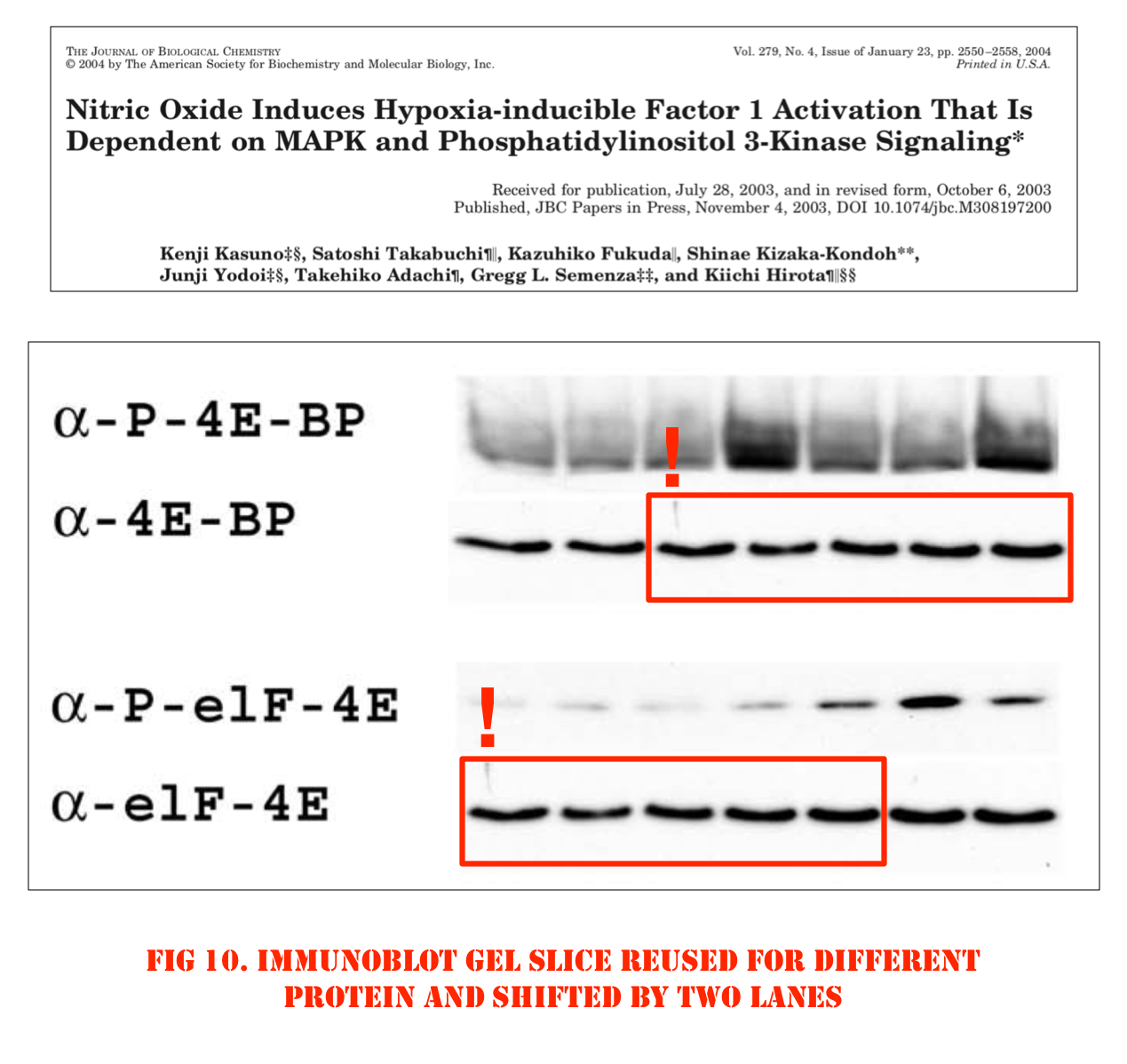
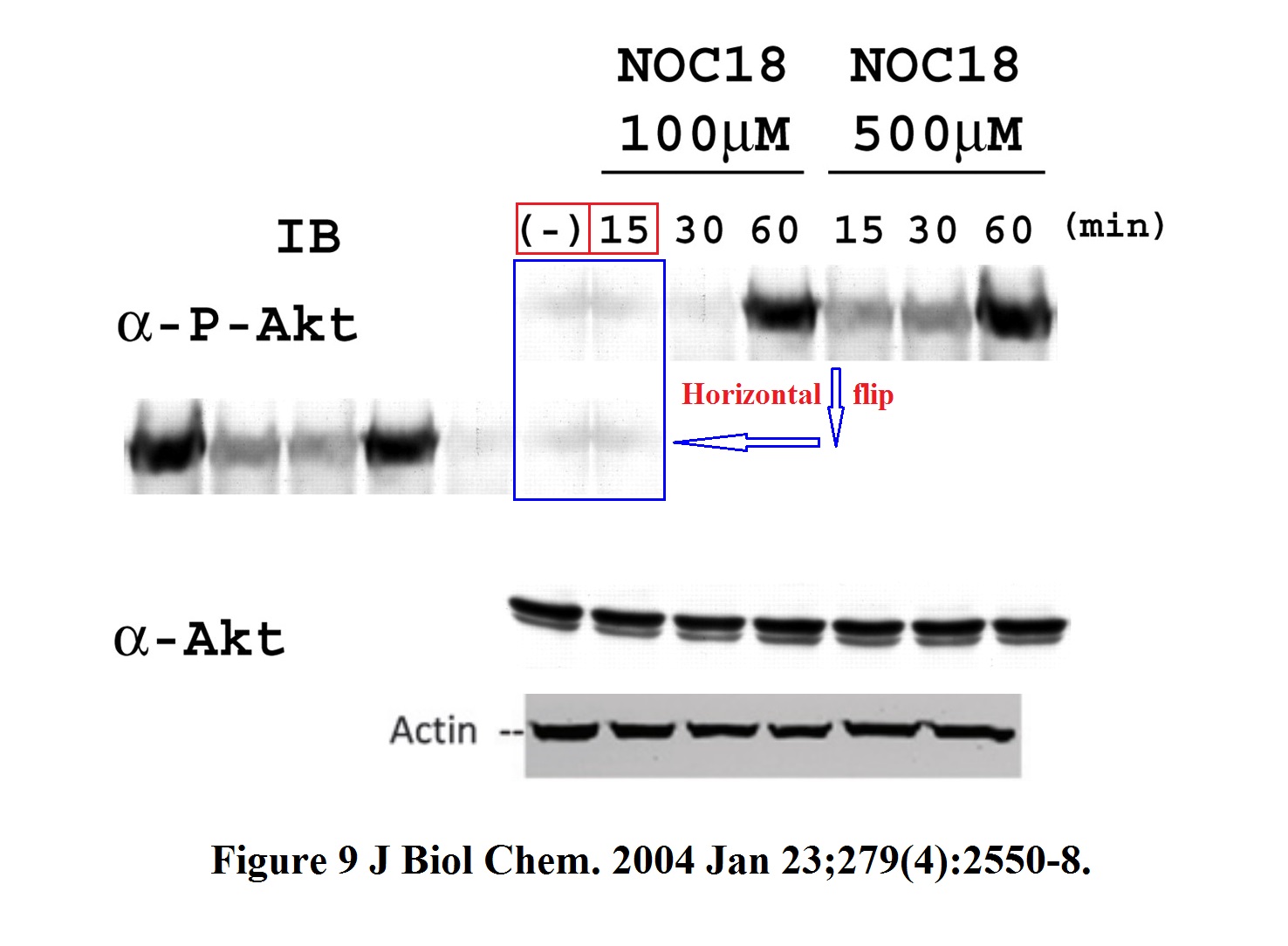
Hirota (“my h-index is “49“) became Semenza’s collaborator when the former joined Johns Hopkins School of Medicine as visiting professor in 1999-2002. In this regard, also this study in Nature Genetics, where a gel was falsified:
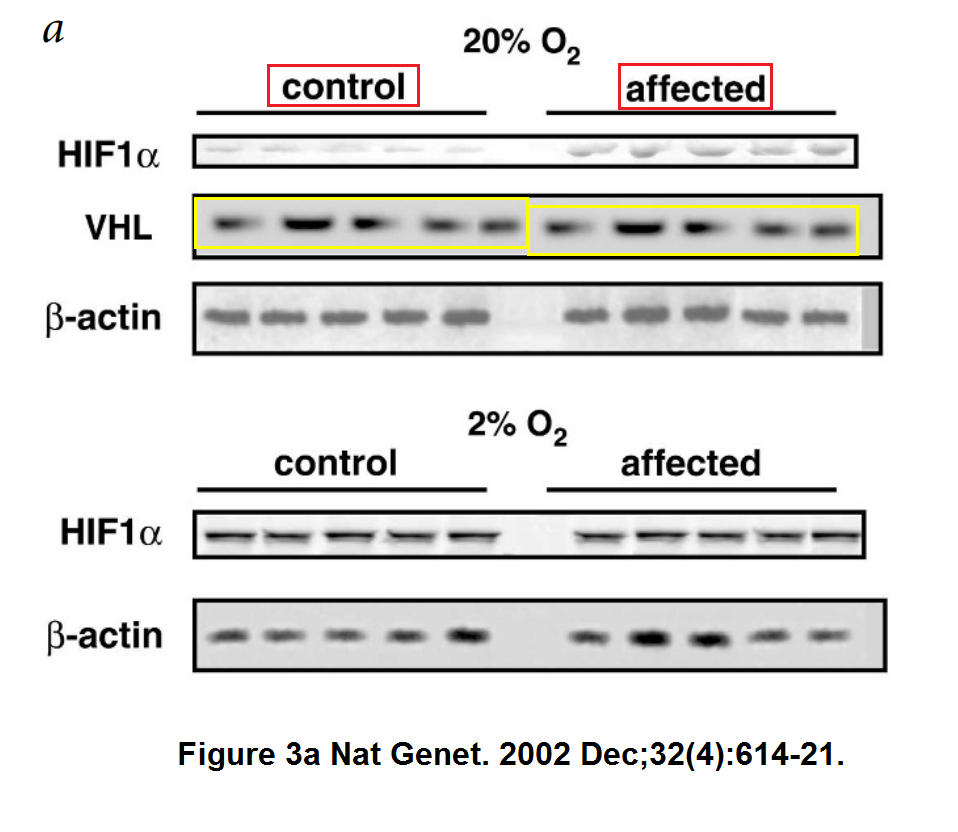

So far, Semenza had one retraction, in 2011 and with some other Japanese colleagues, for Tomita et al Biochemical Journal 2007. Back then the notice said “The last author, Naoki Mori, takes full responsibility for the misrepresentation of data in this paper“.

When he won the Nobel Prize, Semenza said in an interview with ACS magazine c&en:
“All science is a collaborative effort. I have all these hardworking postdocs and graduate and undergraduate students in the lab. Everybody works together as a team. I have a really fine group of people who work in the lab. My major criterion for picking people is that they’re really nice to work with because we spend a lot of time together.“
You took the responsibility for their work when accepting your Nobel Prize and other awards, Professor Semenza. Time to take responsibility for the fabricated figures your team published.
Update 8.10.2020
Johns Hopkins University confirmed to me to be aware of the issues. Meanwhile, Clare Francis resumed checking Semenza’s publications (see comment section). One is worth posting here, because it is another PNAS contributed paper, where one of the friendly reviewers Semenza hand-picked was…. Maria Alfonsina Desiderio, from University of Milan, Italy. Remember her? Desiderio has 5 retractions now, for data fakery.
This is the paper:
Debangshu Samanta, Daniele M. Gilkes, Pallavi Chaturvedi, Lisha Xiang, and Gregg L. Semenza Hypoxia-inducible factors are required for chemotherapy resistance of breast cancer stem cells PNAS (2014) doi: 10.1073/pnas.1421438111

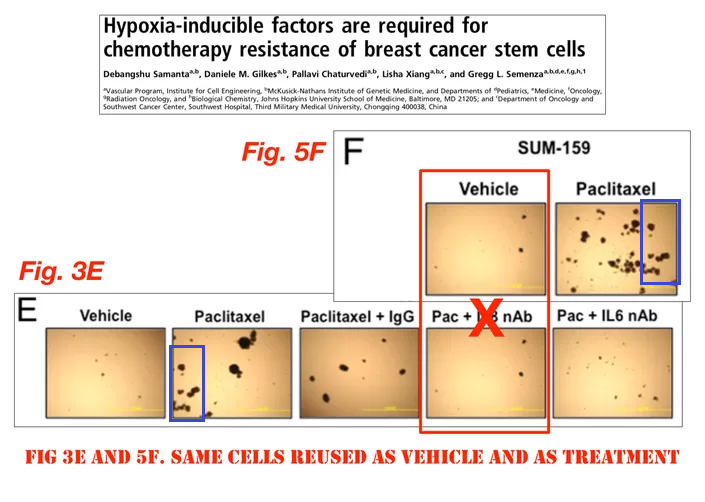
Update 10.10.2020
This gets worse and worse. Another one contributed by Semenza to PNAS:
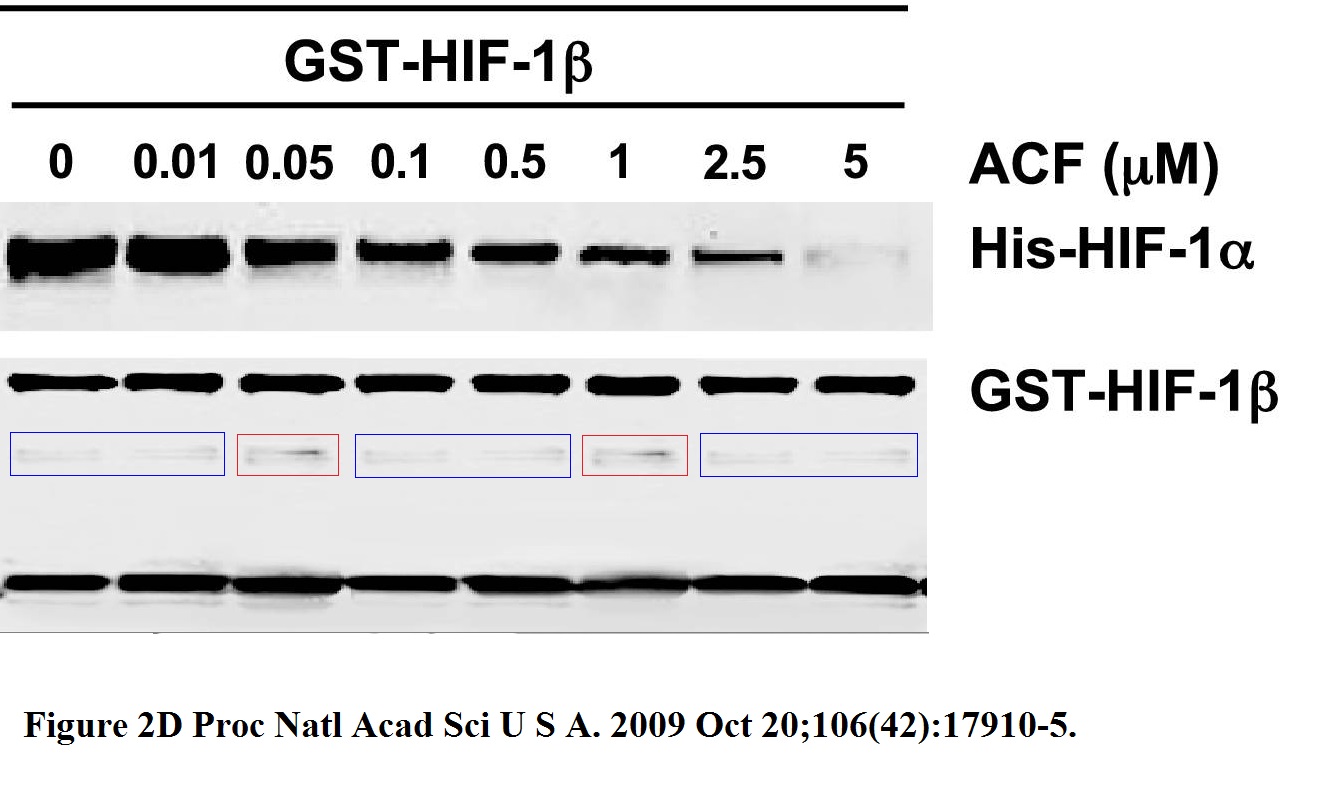
KangAe Lee, Huafeng Zhang, David Z Qian, Sergio Rey, Jun O Liu, Gregg L Semenza Acriflavine inhibits HIF-1 dimerization, tumor growth, and vascularization Proceedings of the National Academy of Sciences (2009) doi: 10.1073/pnas.0909353106
Someone erased all the lower gel bands, as evidenced by the cloned fragments. Does this affect the conclusions? It sure does, very much. And how about this, by similar author collective (yes, also contributed by Semenza):
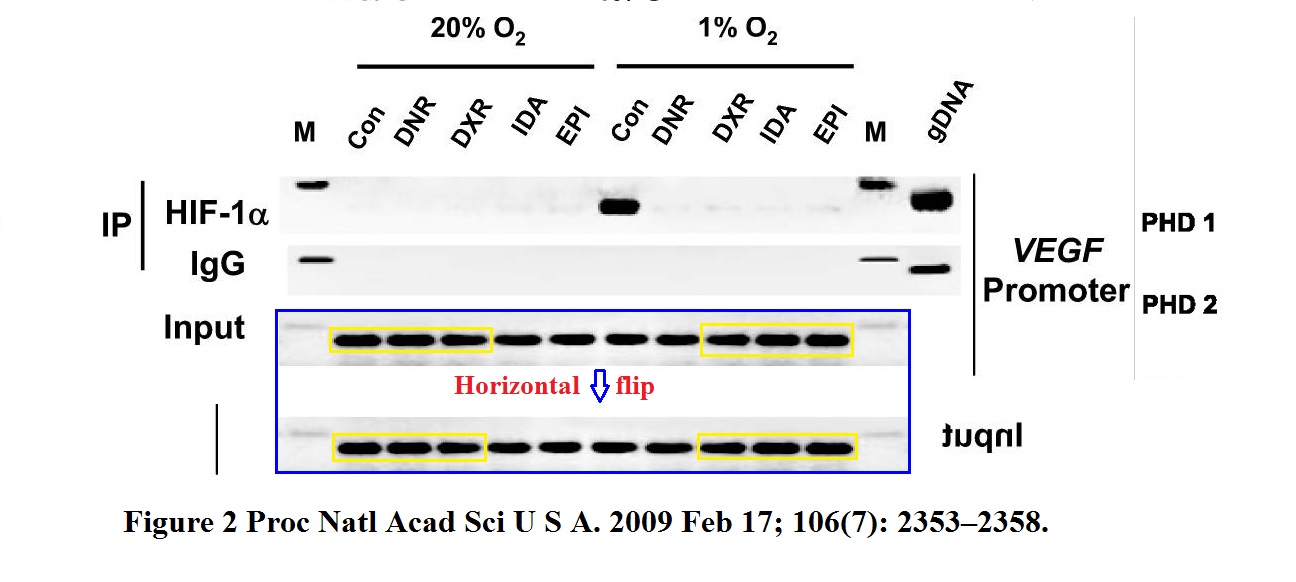
KangAe Lee, David Z Qian, Sergio Rey, Hong Wei, Jun O Liu, Gregg L Semenza Anthracycline chemotherapy inhibits HIF-1 transcriptional activity and tumor-induced mobilization of circulating angiogenic cells Proceedings of the National Academy of Sciences (2009) doi: 10.1073/pnas.0812801106

Someone commented on PubPeer: “I think the whole row is a mirror“. It does look like a falsified loading control. In the same paper, mouse images are sometimes strangely similar, as if that mouse was imaged just seconds and not days apart as the study description claims. The first author, KangAe Lee is now at Princeton University, here is another classic of hers flagged by Clare Francis.
Did you perchance also have trouble reproducing Semenza’s research results, as some of my readers already complained?
Update 14.10.2020
The treasure hunt continues! This article became popular with Japanese readers, so here a collaborative paper of Hirota and others, where Semenza will probably now regret to have agreed to signing as coauthor:
Tomoyuki Oda, Kiichi Hirota, Kenichiro Nishi, Satoshi Takabuchi, Seiko Oda, Hiroko Yamada, Toshiyuki Arai, Kazuhiko Fukuda, Toru Kita, Takehiko Adachi, Gregg L. Semenza, Ryuji Nohara Activation of hypoxia-inducible factor 1 during macrophage differentiation AJP Cell Physiology (2006) doi: 10.1152/ajpcell.00614.2005
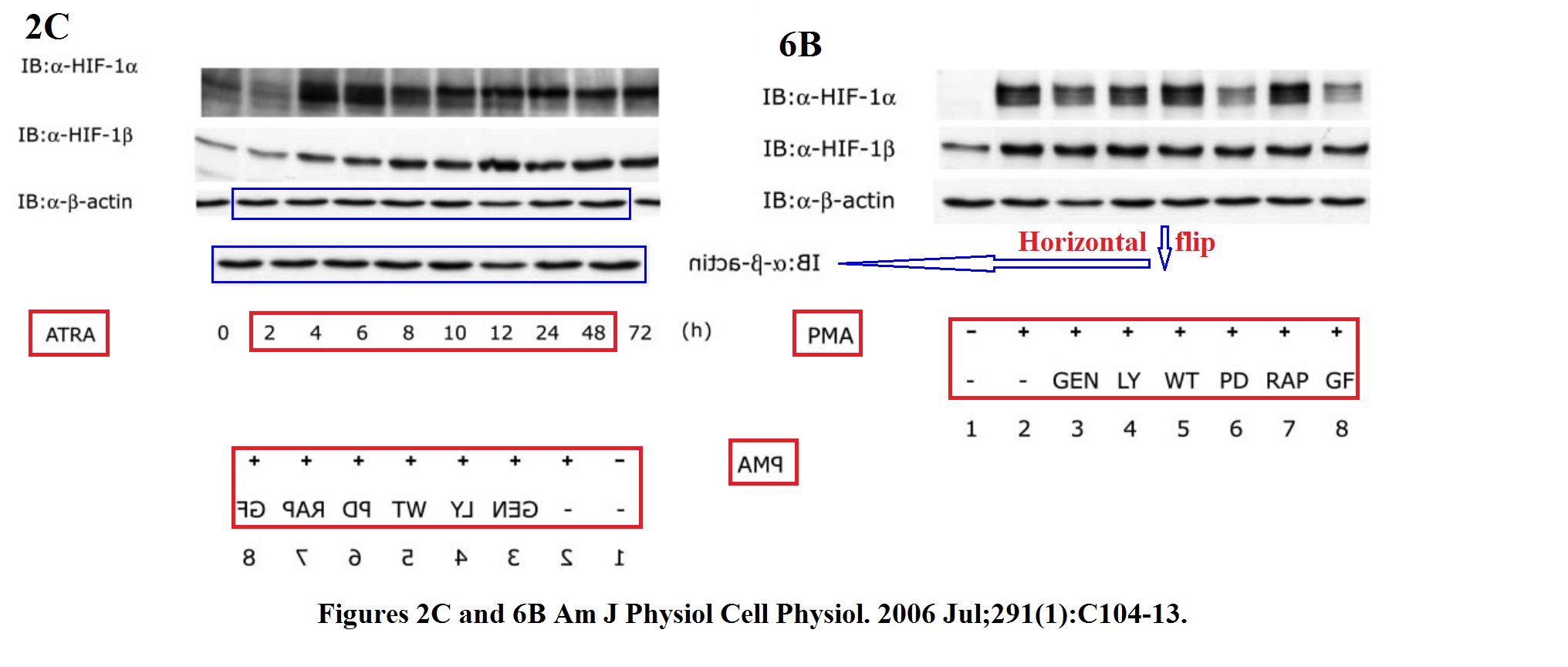
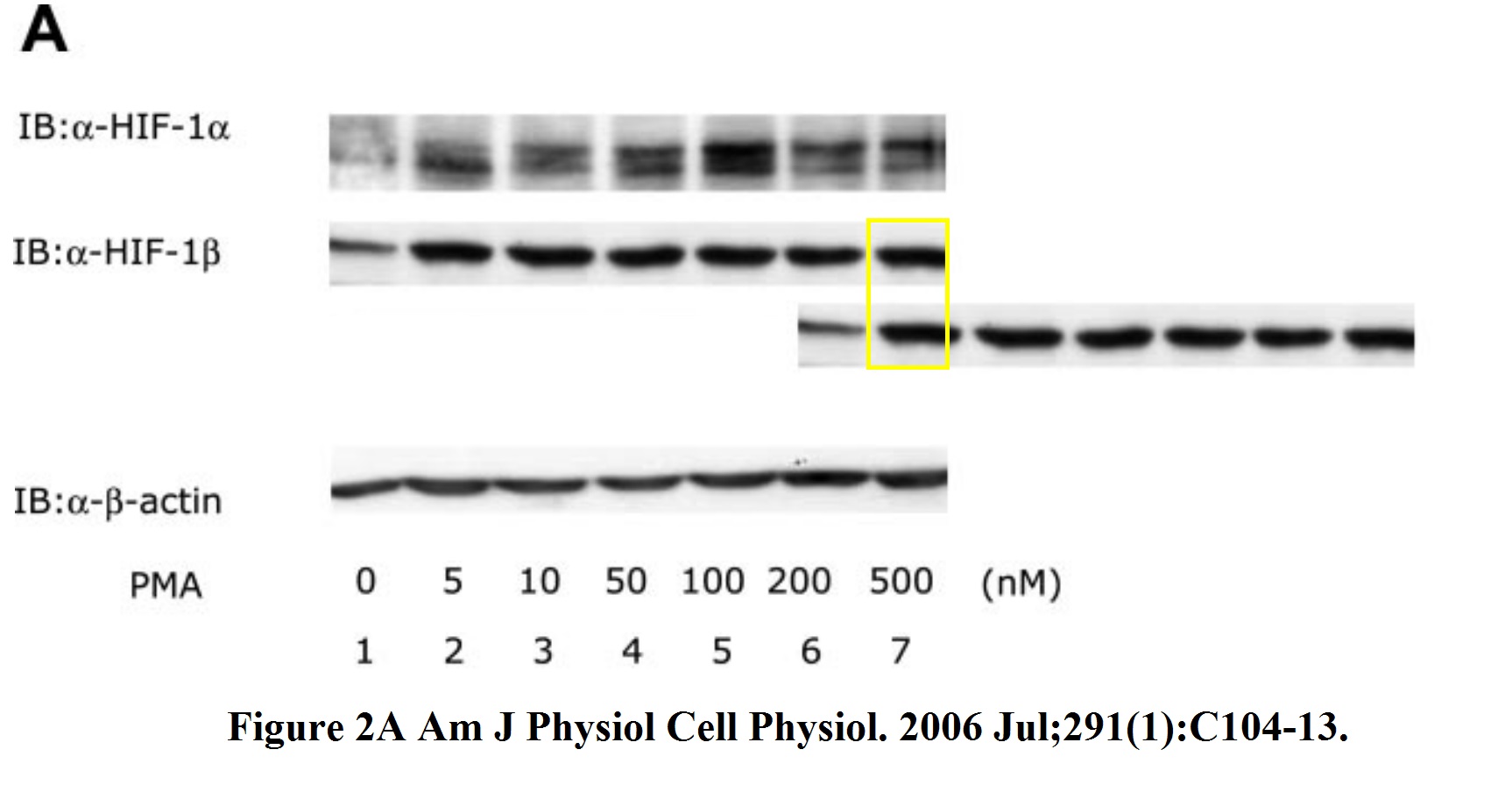
From Japan to China. Remember Huafeng Zhang, coauthor on several problematic papers from Semenza lab, and now professor at the University of Science and Technology of China in Hefei? Here another one:
Huafeng Zhang, Marta Bosch-Marce, Larissa A. Shimoda, Yee Sun Tan, Jin Hyen Baek, Jacob B. Wesley, Frank J. Gonzalez, Gregg L. Semenza Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia Journal of Biological Chemistry (2008) doi: 10.1074/jbc.m800102200
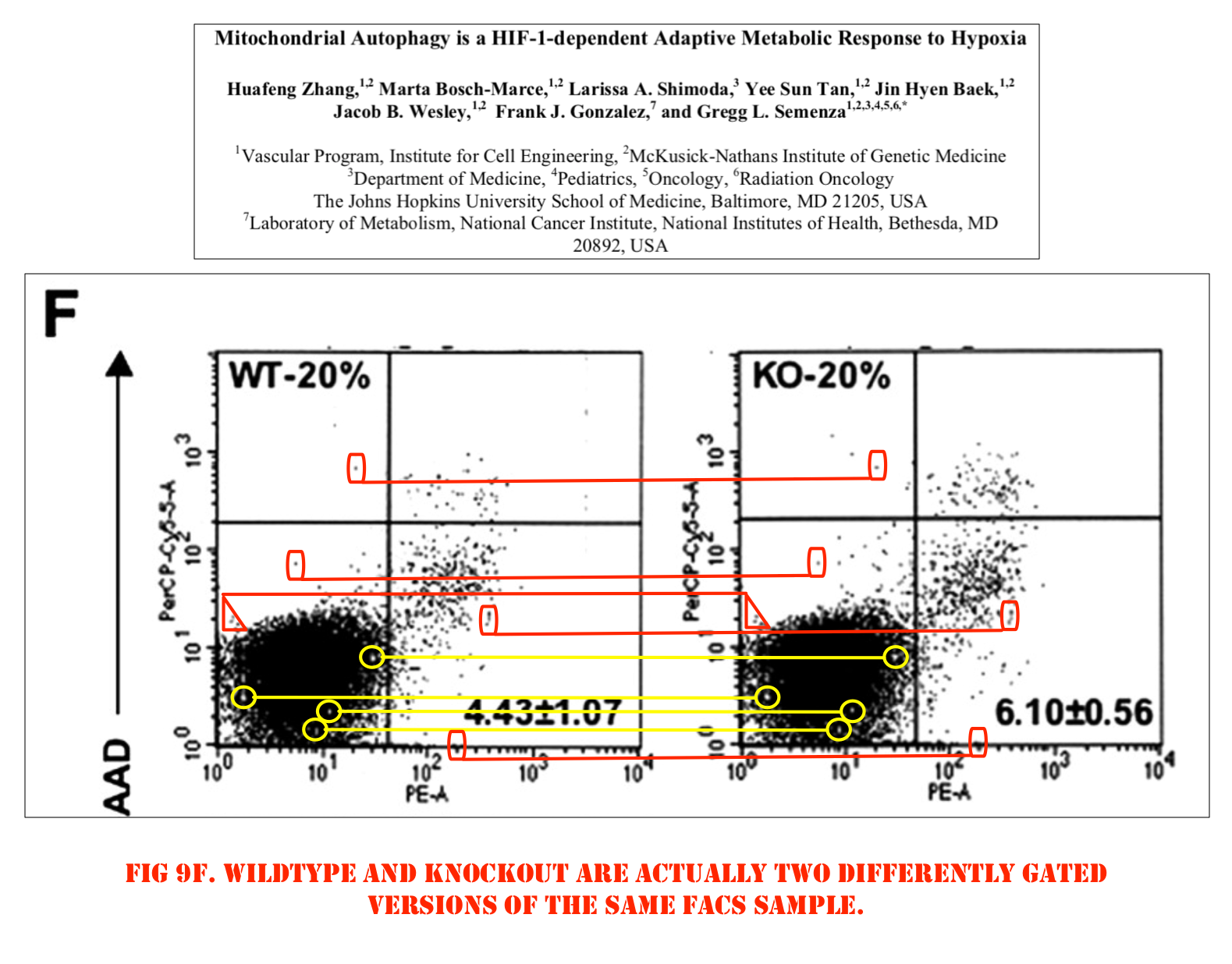
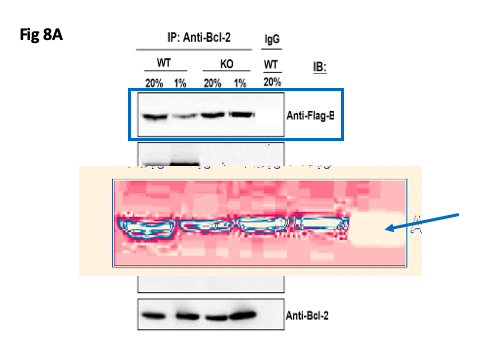
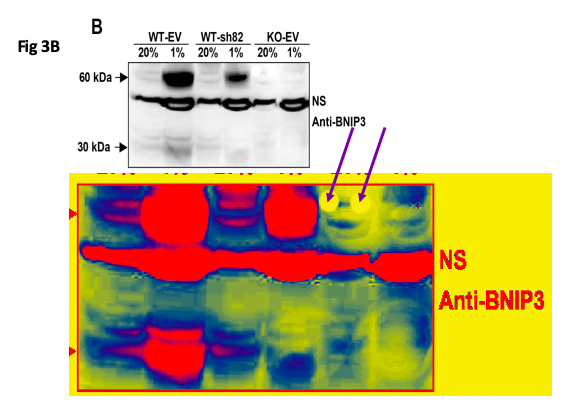
Update 17.10.2020
Welcome to the new episode of Gregg’s Photoshop Adventures. By now, first media reported, NRC (in Dutch). Recall the Gilkes et al Cancer Research 2013 paper briefly flagged at the very beginning above, for data reuse? It’s worse, than originally thought; contributed by Elisabeth Bik:

Daniele M Gilkes, Pallavi Chaturvedi, Saumendra Bajpai, Carmen C Wong, Hong Wei, Stephen Pitcairn, Maimon E Hubbi, Denis Wirtz, Gregg L Semenza Collagen prolyl hydroxylases are essential for breast cancer metastasis Cancer Research (2013) doi: 10.1158/0008-5472.can-12-3963
Update 19.10.2020
There are presently over 40 Semenza papers on PubPeer with serious concerns raised. Here a somewhat fresh one, for Cell Press to do diddly-squat about, as part of their advertisement programme for the ethically challenged among scientists:
Haiquan Lu, Ivan Chen, Larissa A. Shimoda, Youngrok Park, Chuanzhao Zhang, Linh Tran, Huimin Zhang, Gregg L. Semenza Chemotherapy-Induced Ca 2+ Release Stimulates Breast Cancer Stem Cell Enrichment Cell Reports (2017) doi: 10.1016/j.celrep.2017.02.001
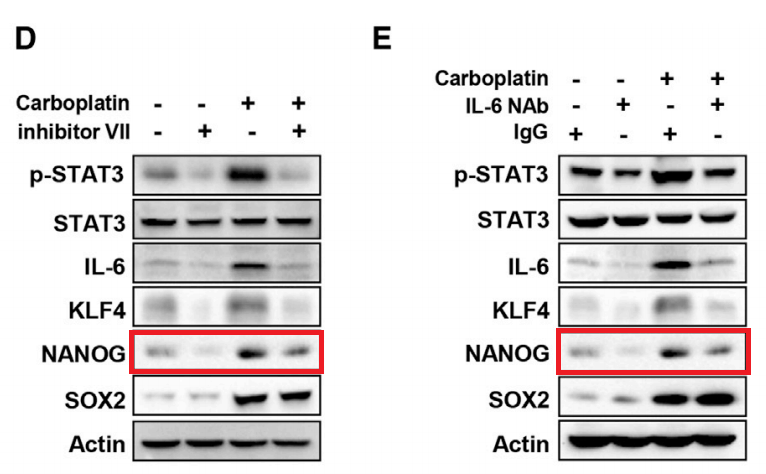
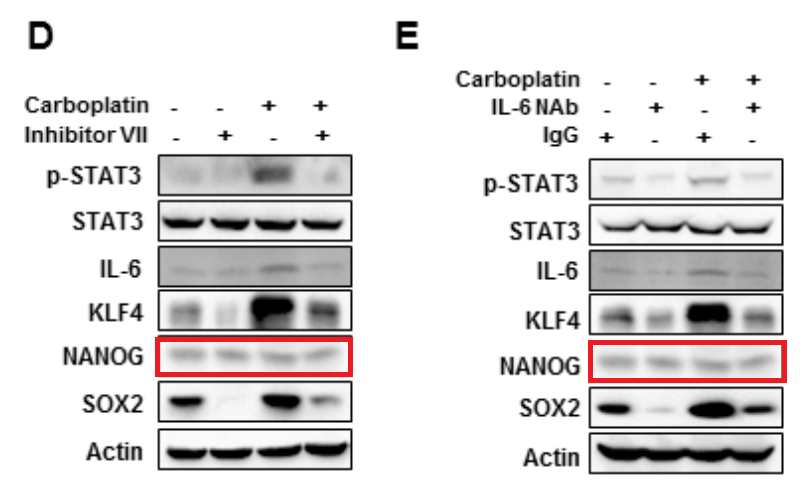
Very lazy approach, also the loading controls (STAT3, Actin) were from a “library”, and not from the same gel.
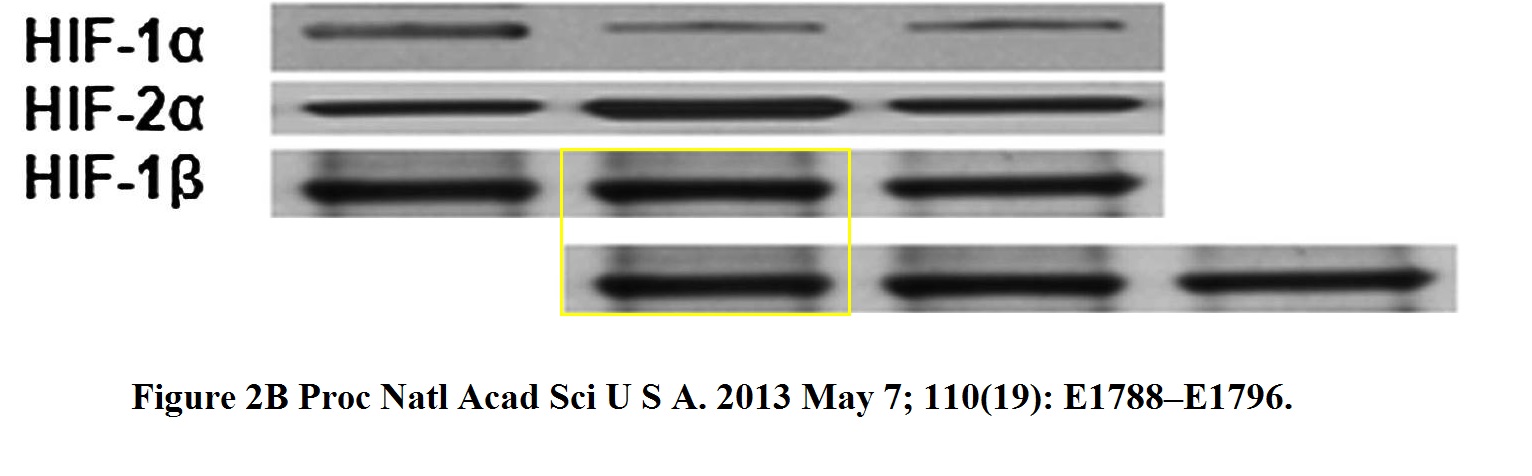
This is the oldest Semenza paper flagged so far, it is likely to be part of the Nobel Prize qualification:
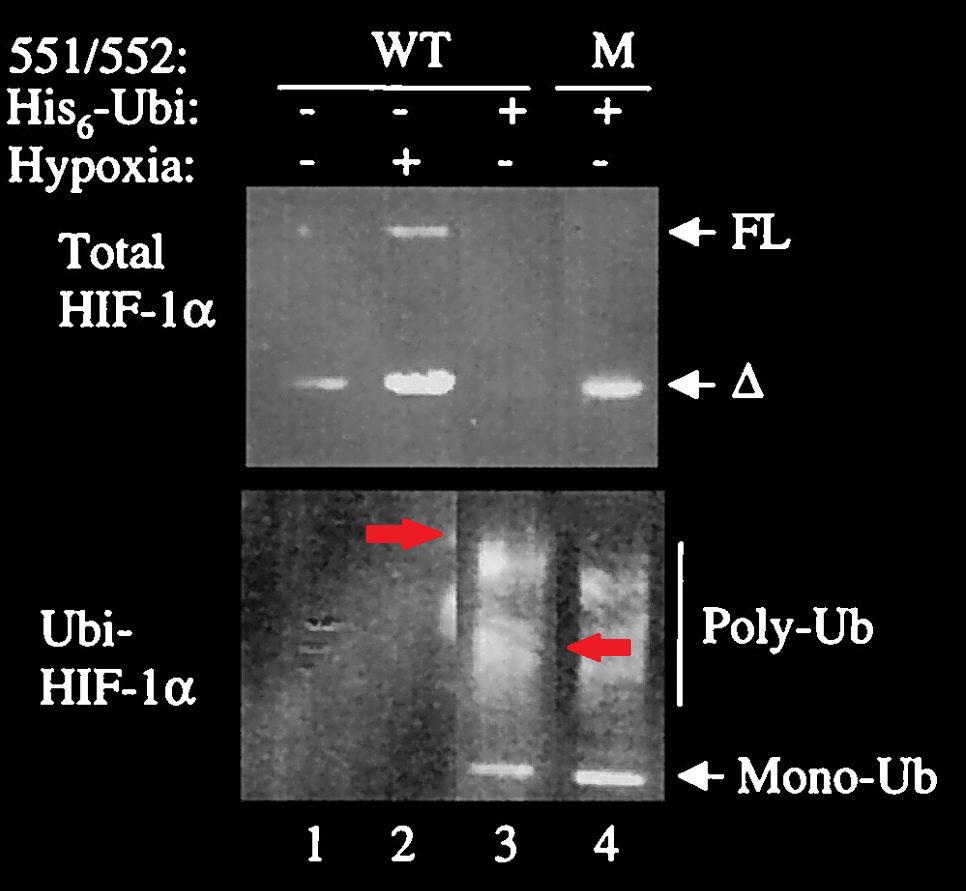
C. H. Sutter , E. Laughner , G. L. Semenza Hypoxia-inducible factor 1alpha protein expression is controlled by oxygen-regulated ubiquitination that is disrupted by deletions and missense mutations Proceedings of the National Academy of Sciences of the United States of America (2000) doi: 10.1073/pnas.080072497
Apparently, someone disagreed with the signal in the third lane, so it was overlaid with “better” results.
Jonathan Links, Vice Provost and Research Integrity Officer at Johns Hopkins University, replied to me again:
“I am acknowledging receipt of this additional information. I am reiterating what I told you previously: Please refer to the Johns Hopkins University Research Integrity policy—which I provided to you previously. Johns Hopkins will adhere to the federal research misconduct regulations and the provisions of that policy, which specifically address confidentiality.“
The policy is here, its procedure stipulates the appointment of an internal inquiry by the Hopkings School of Medicine before deciding on a formal investigation. I however do not know how the faculty can find anyone with “unresolved personal, professional or financial conflicts of interest” in such a high profile case.

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your contribution to getting the Nobel prize abolished.
5.00 €


“The 2019 #NobelPrize in Physiology or Medicine has been awarded jointly to William G. Kaelin Jr, Sir Peter J. Ratcliffe and Gregg L. Semenza “for their discoveries of how cells sense and adapt to oxygen availability.””
Hum Mol Genet. 2001 May 1;10(10):1029-38. doi: 10.1093/hmg/10.10.1029.
Contrasting effects on HIF-1alpha regulation by disease-causing pVHL mutations correlate with patterns of tumourigenesis in von Hippel-Lindau disease
S C Clifford 1, M E Cockman, A C Smallwood, D R Mole, E R Woodward, P H Maxwell, P J Ratcliffe, E R Maher
Affiliations collapse
Affiliation
1Section of Medical and Molecular Genetics, Department of Paediatrics and Child Health, University of Birmingham, The Medical School, Edgbaston, Birmingham B15 2TT, UK.
PMID: 11331613
Figure 2B. Much more similar than expected.
LikeLike
“Affiliations collapse”
If only…
LikeLike
Personally, I’m not very concerned about the impact of the Semenza scandal on his many co-authors. He himself probably didn’t fake data, but he is ultimately responsible for the lab practices and culture that led to this mess. His co-authors and labmates were happy to associate themselves with the “gifted scientist” and collectively they aided and abetted the publishing of some very questionable work. Anyone involved could have put a stop to it by seeing the evidence and speaking up; the fact that no one took that risk may impact them all.
Sadly, Semenza himself will likely escape scot-free; too much is at stake for the institutions involved. If there is evidence of recently faked data, the responsible individuals may be identified and called out. Early career researchers from the lab may have an asterisk on their CV. Semenza will probably apologize for permitting some bad practices to creep into the lab and say he’s putting this behind him so he can go do more #science… move along people, nothing to see here.
~ Cheshire
LikeLiked by 1 person
I wholeheartedly agree with this. I expect exactly this to play out. At one point you’re bringing in so much for the institution and it takes a lot more than a couple of photoshopped images to cause repercussions.
LikeLiked by 1 person
” a couple of photoshopped images to cause repercussions.”
Many more than that.
LikeLike
I think the culture is….gee, if I work for a NAS member (Semenza is one), all I need to do is get one first author paper in high impact journal (PNAS or above), and I get a great job and career. So lets massage the data a little to make sure it gets into PNAS or above, and not PLOS one and below….
I think I earlier called this the Croce effect. Notice that Semenza is not even really involved, and maybe his major fault here is that he is just too trusting.
In a way, Semenza has been hurt by his own success. This is how the system works. Play it right, and/or massage it (wrong), or your out.
LikeLiked by 1 person
The public reaction is… Wang Chung, Dance Hall Days (1981): “Take your baby by the hand (mentoring someone with a paid fellowship)….make her do a high hand stand …(get her a first author papers in PNAS).”
LikeLiked by 2 people
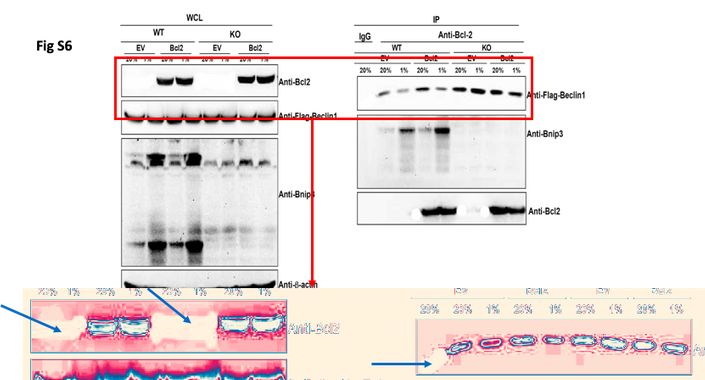
I was just casually looking at the publications of other associated people. Found a quick addition from Denis Wirtz lab: EB1 and cytoplasmic dynein mediate protrusiondynamics for efficient 3-dimensional cell migration Hasini Jayatilaka,,†Anjil Giri,,†Michelle Karl,,†Ivie Aifuwa,,†Nicholaus J. Trenton,‡Jude M. Phillip,§Shyam Khatau,* and Denis Wirtz,†,{,k,#,1Department of Chemical and Biomolecular Engineering, and†Johns Hopkins Physical Sciences–Oncology Center, The Johns HopkinsUniversity, Baltimore, Maryland;‡Department of Bioengineering, Rice University, Houston, Texas, USA;{Department of Pathology,kDepartment of Oncology, and#Sydney Kimmel Comprehensive Cancer Center, The Johns Hopkins School of Medicine, Baltimore, Maryland,USA; and§Division of Hematology and Oncology, Department of Medicine, Weill Cornell Medicine, New York, New York, USA
In supplementary figure 1A and 1C, loading controls were probably recycled from some completely unrelated experiments.
LikeLiked by 1 person
Can you please provide a visualisation and post it on pubpeer?
LikeLiked by 1 person
“Daniele M Gilkes, Lisha Xiang, Sun Joo Lee, Pallavi Chaturvedi, Maimon E Hubbi, Denis Wirtz, Gregg L Semenza Hypoxia-inducible factors mediate coordinated RhoA-ROCK1 expression and signaling in breast cancer cells Proceedings of the National Academy of Sciences of the United States of America (2014) doi: 10.1073/pnas.1321510111”
Figure 2B continued. Much more similar than expected.
LikeLiked by 1 person
Pingback: Il prezzo della selettività - Ocasapiens - Blog - Repubblica.it
Pingback: mTOR: conclusions not affected? – For Better Science
Pingback: 原创 去年诺奖得主涉嫌学术造假!30多篇论文被曝P图、复制粘贴 - 网铮网
Pingback: Hyperbaric Oxygen Therapy extends life, the telomeres, and everything – For Better Science
Pingback: 原创 去年诺奖得主涉嫌学术造假!30多篇论文被曝P图、复制粘贴 -搜狐资讯
Pingback: 2019諾貝爾獎得主塞門紮翻車!論文涉嫌造假,網友:P圖才是科研利器? | hny健康
Pingback: Leonid Schneider & ‘For Better Science’ – Crank or Cure? | This Ȼ Life
Pingback: Episode 22: When Science Goes Sideways - Nobel Prizes and Opioids - Breaking Bad Science
“H Zhang, C C L Wong, H Wei, D M Gilkes, P Korangath, P Chaturvedi, L Schito, J Chen, B Krishnamachary, P T Winnard, V Raman, L Zhen, W A Mitzner, S Sukumar, G L Semenza HIF-1-dependent expression of angiopoietin-like 4 and L1CAM mediates vascular metastasis of hypoxic breast cancer cells to the lungs Oncogene (2012) doi: 10.1038/onc.2011.365”
2021 correction. https://www.nature.com/articles/s41388-020-01618-z
“Since publication of the article the authors noticed the following errors:
(1) The actin immunoblot (loading control) from Fig. 6B was mistakenly included in Fig. 6G. The correct actin immunoblot is attached. It demonstrates that there was equal loading in all of the lanes.
(2) The three panels in Fig. 1A (HIF-1alpha, HIF-2alpha, and actin immunoblots) are identical to three panels in Fig. 1B of Gilkes et al. (Cancer Res. 73:3285, 2013). These are validation immunoblots for knockdown subclones, which were generated through the combined effort of the first authors of the Oncogene and Cancer Research papers.
We regret the two errors that were made. They do not alter any of the paper’s conclusions.”
I think Pubpeer caused the correction.
https://pubpeer.com/publications/0694D956FA93FB972962C5C5C150D7
LikeLike
Cardiovasc Res. 2012 Jan 1;93(1):162-9. doi: 10.1093/cvr/cvr282. Epub 2011 Oct 25.
Tie2-dependent knockout of HIF-1 impairs burn wound vascularization and homing of bone marrow-derived angiogenic cells
Kakali Sarkar 1, Sergio Rey, Xianjie Zhang, Raul Sebastian, Guy P Marti, Karen Fox-Talbot, Amanda V Cardona, Junkai Du, Yee Sun Tan, Lixin Liu, Frank Lay, Frank J Gonzalez, John W Harmon, Gregg L Semenza
Affiliations collapse
Affiliation
1Vascular Program, Institute for Cell Engineering, The Johns Hopkins University School of Medicine, Baltimore, MD 21205, USA.
PMID: 22028336 PMCID: PMC3243042 DOI: 10.1093/cvr/cvr282
2021 correction.
https://academic.oup.com/cardiovascres/advance-article/doi/10.1093/cvr/cvab020/6124096
Corrigendum to: Tie2-dependent knockout of HIF-1 impairs burn wound vascularization and homing of bone marrow-derived angiogenic cells [Cardiovasc Res. 2012;93(1):162-9; doi: 10.1093/cvr/cvr282]
The authors have contacted the Editorial Office to request a correction to Figure 3 of this manuscript.
The two insets, which are located on the right side of Fig. 3A, were not derived from the larger photomicrographs that they overlay, but rather are magnified images of representative vascular structures in the dermis that stained with the CD31 and αSMA antibodies, respectively.
Although there is no error involved, the authors would like to clarify and have provided a new figure in which the images are presented in a separate column.
This change does not affect the overall scientific message of the paper.
The figure legend for Figure 3 has also been updated:
This matter was investigated by the ESC Journals Family Ethics Committee, which recommended this resolution.
LikeLike
Am J Physiol Cell Physiol. 2020 Nov 1;319(5):C922-C932. doi: 10.1152/ajpcell.00309.2020. Epub 2020 Sep 16.
Hypoxia-inducible factor-1 mediates pancreatic β-cell dysfunction by intermittent hypoxia
Ning Wang 1, Xue-Feng Shi 1, Shakil A Khan 1, Benjamin Wang 1, Gregg L Semenza 2, Nanduri R Prabhakar 1, Jayasri Nanduri 1
Affiliations
1Biological Sciences Division, Institute for Integrative Physiology and Center for Systems Biology of O2 Sensing, University of Chicago, Chicago, Illinois.
2Johns Hopkins University School of Medicine, Baltimore, Maryland.
PMID: 32936698 PMCID: PMC7789966 (available on 2021-11-01) DOI: 10.1152/ajpcell.00309.2020
Problematic data figure 2C. Much more similar than expected after vertical flip and horizontal resizing.
LikeLike
J Physiol. 2018 Aug;596(15):3087-3100. doi: 10.1113/JP274833. Epub 2018 Jan 4.
DNA methylation in the central and efferent limbs of the chemoreflex requires carotid body neural activity
Jayasri Nanduri 1, Ying-Jie Peng 1, Ning Wang 1, Shakil A Khan 1, Gregg L Semenza 2, Nanduri R Prabhakar 1
Affiliations
1Institute for Integrative Physiology and Centre for Systems Biology of O2 Sensing, Biological Science Division, The University of Chicago, Chicago, IL, USA.
2Vascular Program, Institute for Cell Engineering; Departments of Pediatrics, Medicine, Oncology, Radiation Oncology, and Biological Chemistry; and McKusick-Nathans Institute of Genetic Medicine, The Johns Hopkins University School of Medicine, Baltimore, MD, USA.
PMID: 29148180 PMCID: PMC6068255 DOI: 10.1113/JP274833
Problematic data.
Figure 8A. Dmnt1 panel. Vertical, straight changes in signal either side middle lane, i.e. middle lane “slotted in”.
Problematic data.
Figure 6B. Dmnt1 panel. Vertical, straight change in signal right end band in middle lane.
LikeLike
You all must have heard of “product placement”, for example when someone in a film for no apparent reason at all starts extolling branded fast food.
Well, The Scientist introduces the new concept: Tainted Scientist Placement. In an article published on 13 May 2021, the magazine covered some cancer research paper in Frontiers by two Minnesota researchers, where cells deficient in DNA repair genes were analysed. Now, the strange thing was that out of nowhere, this bit came in the text:
Daniele Gilkes works on hypoxia, not DNA repair, and she is not at University of Minnesota but at Johns Hopkins University. Moreover, Gilkes is not a coauthor of that paper, and neither the editor or the reviewer (Frontiers names those).
LikeLike
There 2 things you should know about people who make it up:-
They don’t give a shit,
They don’t give a shit.
Works for them!
LikeLike
Well, some fall into this category. Some are born in the dark side. That being said:
I just left a lab where a post-doc was, IMO, was mis-representing data. I think he knew it was wrong, so he felt guilt, indicating he gave a shit. The problem here was that the post-doc was intent on being a faculty member and felt he had to do it. Even worse, the environment, set by the “advisor”, was perfectly conducive for him doing it, as the “advisor” was a prima-donna assistant professor MD where not getting tenure was not an option. I really felt if the advisor had been less of an ambitious narcissist and more of a normal scientist humbled by the vagaries of nature the post-doc would have not flipped to the dark side. But I fear the post-doc is, forever, lost……
LikeLike
“The problem here was that the post-doc was intent on being a faculty member and felt he had to do it”
Yes.
This is not new.
The outcome for those who misrepresent data AND get caught, is the same for those who do NOT mis-represent data: no career.
LikeLike
Beg your pardon?
https://forbetterscience.com/2018/05/03/zombie-scientist-sonia-melo-awarded-by-astrazeneca/
LikeLike
The floors of the modern academic laboratory are the new battleground of good versus evil, where souls are either saved or lost forever! And the devil is winning…..
LikeLike
….which makes Shneider, Smutley, and the Bikster the light of god…..
LikeLike