The misconduct-tainted University of Gothenburg professor of transplantation biology Suchitra Sumitran-Holgersson (see my detailed report here), has now lost a critical publication due to data manipulation, her funding by the charity Hjärt-Lungfondens (Heart-Lung-Foundation) has been frozen. This is what the General Secretary of Hjärt-Lungfondens, Kristina Sparreljung told me:
“Suchitra Sumitran-Holgersson has an ongoing support from us during 2016 and 2017 with 500,000 Swedish crowns per year. She has received 500,000 Swedish crowns so far. As soon as the cheating issue came to our attention, we have made every effort to assess the situation and act properly based on the information we have. As a first step, we decided to freeze the payments. We have also asked the institution [University of Gothenburg, -LS] to freeze its payments, but they have decided to let Suchitra Sumitran-Holgersson research further until the investigation is complete. She will not receive any more money from us until the investigation is complete”.
Also Björn Aschan, Chairman of the Gothenburg-based charity IngaBritt and Arne Lundbergs Research Foundation informs that their 2.5 Mio SEK funding to Sumitran Holgersson has been suspended:
“The funding of equipment to the Gothenburg University for Professor Sumatran-Holgerssons research has temporarily been suspended awaiting the outcome of the investigation re. Professor Sumitran-Holgersson’s research”.
The retracted paper by Sumitran-Holgersson (Berg et al, Tissue Eng Part A. 2014) concerns her experiments with Macchiarini-style “regenerated” trachea. There, an alleged cure of a 76-year old chronic asthma patient is described, who suffered death from infection shortly after receiving the trachea transplant. The operation was performed by Sumitran-Holgersson’s close collaborator, Michael Olausson, director of the Transplant Center at the University of Gothenburg.
I informed on March 3rd the journals Editors-in-Chief Antonios Mikos and Peter Johnson about the suspicions raised on PubPeer, yet received no reply. Now the retraction came:
“The Editors of Tissue Engineering are officially retracting the published article entitled, ‘‘Replacement of a Tracheal Stenosis with a Tissue-Engineered Human Trachea Using Autologous Stem Cells: A Case Report,’’ by Berg M, et al., Tissue Eng Part A; 2014;20 (1/2):389–397; DOI: 10.1089/ten.tea.2012.0514.
The corresponding author, Suchitra Sumitran-Holgersson, of the above-named article contacted the Editors via email to ask for a figure in the article to be replaced nearly three years after publication because it was duplicated from a previously published article1 from her group. As requests of this kind are highly irregular and are often cause for concern, the editors determined that a retraction was in order.
The editorial leadership of Tissue Engineering is committed to the highest standards of scientific content and integrity, and does not tolerate any improprieties”
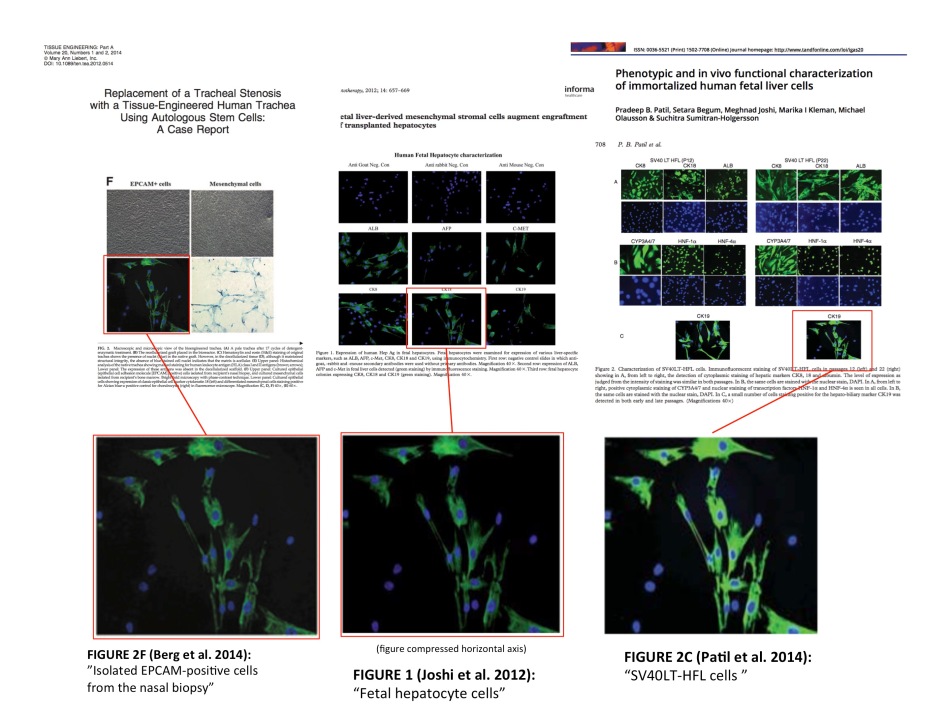
This is Sumitran-Holgersson’s second retraction for data manipulation, the first one for Elsheikh et al, Blood 2005 happened after an external investigation at her former place of work, the Karolinska Institutet (KI) in Stockholm. Other journals promised to investigate, when I contacted them in March 2016. There may be more retractions, or maybe nothing at all will happen, as one journal boldly announced. Though the same image was also reused in another paper of Sumitran-Holgersson’s in the Scandinavian Journal of Gastroenterology (Patil et al, 2014), its Editor-in-Chief Helge Waldum wrote this to me in his email from March 16th, 2016:
“Thank you for contacting us about double publishing of figures by Suchitra Sumitran-Holgersson. We agree with you that some parts of figures first published in Scandinavian Journal of Gastroenterology later were used in another publication. We have written to the author about this, but will not do anything more about it. After all, the pictures used twice only made up a minuscule part of the figures, and we do not find this error of such a degree to do anything more”.
Waldum likely oriented himself on the decision by the Swedish Research Council (Vetenskapsradet, VR) from 2011 to acquit Sumitran-Holgerssson of all proven accusations of research misconduct and data manipulation and to restore all her funding. The story of this investigation by the biomedicine professors from Uppsala University, Olle Kämpe and Lena Claesson-Welsh, is so bizarre that it could make an entertaining film. Kämpe and Welsh were able to prove beyond doubt that Sumitran-Holgersson has been manipulating data (their report is available exclusively from my site), because of this, her above mentioned paper in Blood had to be retracted. Yet since the main evidence concerned a manuscript which was never published, VR decreed that this was not actual research misconduct as such, since there was no intention to deceit in a scientific publication. Moreover, VR used the rather unambiguous evidence of deliberate research misconduct as pretext to dismiss the entire investigation as biased and to claim that Kämpe and Welsh overstepped their mandate by finding it. The decision by the then-VR president Mille Millnert is available exclusively here, where he declared:
“The expert group was not supposed to judge whether such a lack of adherence to good scientific practice had been done in good or ill faith, or whether scientific fraud had been committed“.
However, neither Kämpe nor Welsh were part of this expert group. They were specifically invited as external investigators, while the VR expert group had separate meetings and finalized their report on their own, as I was informed by a reliable source.
Even more puzzling, a detailed report on Sumitran-Holgersson’s misconduct, which Kämpe purportedly personally delivered to KI, disappeared into thin air. This file was allegedly hand-delivered to the KI Registrar and addressed to KI professor Martin Ingvar, but now it could not be found anywhere at KI. I however provide a (incomplete) copy exclusively here. Finally, because of their investigations, Kämpe and Welsh were threatened with a ruinous lawsuit by the company Adsorber, owned by Sumitran-Holgersson’s husband and Gothenburg professor, Jan Holgersson. VR apparently chose to stay out of this. Despite our extensive communications, the Research Council chose not to explain to me on why they allowed this blackmail to happen, through their explicit refusal to provide legal protection to their own investigators.
What next for this controversial Gothenburg professor of regenerative medicine? I will provide updates below, whenever new information or developments on the Sumitran-Holgersson case arrive.
Update 11.05.2016. Sumitran-Holgersson featured prominently in the 2015 magazine and a now deleted 2016 interview of the Swedish Njurfonden (Kidney Fund), with the suggestion that she was funded by this charity. I introduced myself and my relevant articles to the president of Njurfonden, Håkan Hedman, who then declared to me:
“On behalf of Njurfonden (National Kidney Foundation) I confirm that that Njurfonden has not given Professor Holgersson any funding during the passed 10 years. However, she is until March a famous and a well known scientist and her research is exiting. This is the reason why we presented her on our web among many other scientists in order to show all interesting renal research going on in Sweden. Professor Holgersson is still under investigation and not judged“.
However, a related charity, “Stiftelsen Professor Lars-Erik Gelins Minnesfond” funds kidney research and its president is the same Hedman. The Gelin Foundation supported Sumitran-Holgersson regularly until 2012, in 2014 and 2015 her collaborator Olausson was financed for “reconditioning of kidneys from heart-dead donors”. I asked Hedman to comment on the status of this funding to the Gothenburg duo, and was brusquely told:
“I do not know who you are and your purposes. We have been informed about the investigation. Until now I will not answer any further questions about this matter“.
Update 13.05.2016, updated again 20.05.2016. Sumitran-Holgersson and Olausson still enjoy generous funding from other public sources, at least until the investigations are concluded.
The 2.1 Mio SEK from the Swedish Research Council (VR), for the purpose of pancreas regeneration are being duly paid out, as VR Chefjurist Anna Hörnlund notified me:
“Research Council grants are ongoing and we are following this case“
The LUA-ALF grant funding from Swedish government, which is distributed locally through universities. Lars Grip, research director of Sahlgrenska Hospital, who is also responsible for the LUA-ALF funding management there, stated to me in an email:
“There is currently an investigation ongoing regarding suspected misconduct at the University of Gothenburg. The LUA-ALF grants are handled in close collaboration between the university and the Sahlgrenska University hospital. The disposition of the actual grants are not yet decided on but will be handled in connection to this investigation”.
I asked Grip whether this means LUA-ALF funding to Sumitran-Holgersson was suspended, yet he did not reply anymore. One week later however, he notified me, together with Christina Jern, vice dean for ALF at Sahlgrenska:
“The funding for 2016 is paid out at the beginning of the year. The funding for 2017 will depend on the outcome of the investigation that is ongoing at the university“
Incidentally, the now retracted paper about the lethal trachea transplant declared to had its research funded by LUA-ALF. However, Grip explained to SVT that this operation “was health care. It was not a research project in the usual sense“. Yet the Swedish Medical Products Agency, which usually grants such hospital exceptions for already approved drugs, is not so sure. Also, the information Sumitran-Holgersson and Olausson originally provided in that retracted paper, that “the Swedish Medical Products Agency was informed and approved this surgery according to ‘‘hospital exception’’ was apparently untrue. Ann Marie Janson Lang, MPA expert in neuropharmacology and advanced therapy medicinal products, informed me:
“The Medical Products Agency has not given any approval (see legal requirement in Medicinal Product Act 2015:315), i.e. no hospital exemption has been authorized for tissue engineered human trachea using autologous stem cells replacing tracheal stenosis“.
Update 18.05.2016. It seems Olausson and Sumitran-Holgersson were repeatedly giving untrue statements about ethics approval on their human experimental transplants, both for trachea as well as regenerated veins. These approvals were never granted, unlike the two scientists assured in their publications.
This well-promoted Olausson et al 2014 paper in Cell Press/Lancet-supported Elsevier journal EBioMedicine reported regenerated vein transplants into two children, 2 and 4 years old., and contained a statement:
“As in the previous published case (Poole-Warren et al., 1996, – an obviously unrelated Australian study made in sheep, -LS) in the present two cases, a pediatric team performed the evaluation in a multidisciplinary fashion, which also includes abiding to the UNICEF Convention on the Rights of the Child. […] The parents also provided informed consent, which was given to the pediatric team in the absence of the operating surgeon to avoid pressure. Moreover, permissions from the two regulatory bodies in Sweden—The National Medical Board and The Medical Products Agency were granted.”
The MPA expert Janson Lang inform me however that, once again:
“No hospital exemption for the tissue-engineered veins using autologous peripheral whole blood has been granted by the Medical Products Agency. The classification of the product as an advanced therapy medicinal product and the requirement for GMP was communicated in the beginning of 2012, after receiving a question on this matter from the corresponding author of the paper you refer to”.
Duc H Le, Lancet-associated EiC of EBioMedicine, wrote to me:
“we will carry out our own independent investigation into this issue, starting with the authors’ institutes and the authors themselves. Depending on the outcomes of the investigation we will consider appropriate editorial actions“.
What about other Olausson/Sumitran-Holgersson papers on vein transplants into juvenile patients? This impactful Olausson et al 2012 Lancet publication was the first to report stem-cell bioengineered vein transplant into a 10-year-old patient and issued this ethics statement
“We obtained consent from all the relevant ethical organisations, including the parents, local ethical commission, and two regulatory bodies in Sweden (National Medical Board and Medical Products Agency; appendix -see relevant information in comment below, -LS). Consent for use of donated organs follows the rules of organ donation for organ transplantation at the University Hospital”.
To this, Janson Lang declared on behalf of MPA:
“I’m afraid the answer is the same – i.e. no hospital exemption or manufacturing authorization/GMP certificate for bioengineered vein transplantation with autologous stem cells has been given by the Medical Products Agency”.
Absence and especially deliberate misinformation about the ethical approval in clinical research papers usually leads to their immediate retraction. It is possible that this was the true reason for the retraction of Berg et al, Tissue Eng Part A. 2014 paper on the lethal trachea transplant. We will now have to see how journals respond to this new information.

How can different journals have such different reactions to the same piece of misconduct? Aren’t they all (don’t laugh) members of COPE?
LikeLike
I feel challenged to reply here. The cool thing about COPE guidelines is their “small print”. Most academics just read the bold bits, and then wave those about to demand retractions or correction. Nonsense, read on people. COPE guidelines are formulated in a way that they can mean whatever you want them to mean. Please read page 3 of this document:
Who decides if the part is big enough or the paper is so seriously flawed that a retraction is in order? Journal editors, that’s who. Same for self-plagiarism. COPE will generally side with them, since COPE’s COI is that they are exclusively funded and controlled by the very publishers they are advising.
Indeed, the EiC of Scand J of Gastroenterology decreed that only an insignificantly minuscule part is affected and did nothing. EiCs of Tissue Engineering saw that the paper is unreliable and retracted it. Both acted in agreement with COPE guidelines.
Hope I could answer your question.
LikeLike
Given this massive range of do-nothing to retract, it makes one wonder precisely why COPE is receiving so much money from the publishers to become “members”. If ethics are so flexible, and their implementation (or reading) is equally flexible, as Dr. Schneider correctly points out, then this makes COPE a very rich farcical organization, or at best a toothless one, that was established by the commercial publishers, to give a mask of ethical integrity to its rich backers.
One could argue that having COPE is better than not having COPE at all, but one has to question the legitimacy of this powerful organization, and its funding, face such radically different outcomes given the same “ethical” issue, in this case, figure duplication. It is precisely the inconsistency about the implementation of COPE rules, and the fact that so many COPE member editors are failing to act in accordance with COPE guidelines (without impunity), that the readership must hold COPE, and its members (editors, journals and publishers) more accountable. Because a seriously corrupting factor is involved in this transaction of trust with the academic community: money.
As I see it, Helge Waldum is not following COPE guidelines, and given the other concerns with papers by Sumitran-Holgersson stemming from her misconduct, surely Dr. Waldum has the responsibility of reassessing her position and that of her journal? At minimum, one would expect an expression of concern.
COPE-related queries here:
https://www.pubpeer.com/topics/1/2E91E8916236A6EE03F372E64FBBAF
LikeLike
COPE could do something to remedy this situation by issuing case-by-case guidance (e.g. in this case, they could say: “We endorse Tissue Engineering’s interpretation” or not as the case may be).
LikeLike
Unlike some people I believe that COPE is a net force for good because it embodies valuable ideals in a publishing sector which often seems to be free of them. Do we really think that the world would be better with no COPE? However COPE could do more to make these ideals a reality.
LikeLike
COPE provides good guidelines for honest and decent editors, which is surely a valuable contribution. It however has zero sanctioning power, in fact also little inclination to act upon those who place politics and simple personal convenience above research integrity.
As an example I can only mention the policies of one of the COPE’s own founders, Richard Horton, EiC of The Lancet: https://forbetterscience.wordpress.com/2016/04/05/does-the-lancet-care-about-patients/
Another example is COPE’s condoning of excessive self-plagiarism:
https://forbetterscience.wordpress.com/2016/04/11/academic-self-plagiarism-misconduct-or-a-literary-art-form/
LikeLike
Neuroskeptic’s naive ideals could be true if COPE removed a core corrupting factor from this entire equation: “membership fees”. As is stands, monetary contributions by some members theoretically amount to tens or even hundreds of thousands of British pounds a year, which sounds far from being a “charity”. It is a powerful financial entity, disguised as a charity (even though it is formally registered as one in the UK). The ethical ideals and guidelines espoused by COPE would be no less noble if there was no money involved. But the fact that such a tremendous amount of money continues to be involved, indicates that ethics is currently being bought.
How would COPE and its members act if their ethical advice were truly free, and thus free of financial conflicts of interest?
Your question is as rhetorical as asking is it better to have a corrupted editor than no editor at all (if you are in that camp of scientists that automatically associates the term “editor” with “academic quality”?
One has to ask why COPE has zero sanctioning power if it receives so much money? This is precisely what makes it farcical.
LikeLike
Another mess, at Linkoping University this time.
Linkoping is shy about identifying professor, or perhaps policy prevents identification?:
http://liu.se/liu-nytt/LiU-nytt?l=en&newsitem=674390
http://headlinesnewstoday.blogspot.com/2016/05/corneas-intended-for-pigs-underwent.html
This seems to say funding withdrawn/frozen(?) but since I don’t use FB or Twitter I can’t read it properly
http://www.techsite.io/p/327504
Is this the researcher at issue? (PLEASE correct me ASAP if I have this wrong) : https://liu.se/medfak/igen/griffith-may?l=en
publication, NB first author at Filatov Institute
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4676913/
Take home point: keep control of your investigational products
LikeLike
Changing of subject:
Why scientists knowingly publish fraudulent data?
Simple answer: because of grant money!
LikeLike
Another problem is when the authors know very well the editors, when the authors take coffee with the editors, when they discuss what experiments to include in the papers even before the experiments are done.
LikeLike
https://pubpeer.com/publications/AD930970194DC9714AC9E5F5090DF3#fb51582 new finding of inappropriate image reuse by SSH
LikeLike
Re: 18.05.2016 Update:
I am a Swedish philosopher and medical ethicist, living in Gothenburg.
The various quoted passages certifying ethical approval look extremely strange, as they mostly refer to agencies/authorities whose role are not to approve human trials or research.
The only agency/authority empowered by law to issue permission for human trials/research is the Ethical Review Board (Etikprövningsnämnden). This is regulated by the Ethical Review Act, see here: http://www.epn.se/en/start/the-organisation/ .
If there is a valid ethics permission, the proof would be a registered approval with a sequence number, attached to the application made by the team. A copy of this should be held by the person responsible (in this case the treating physician, presumably). It should also be on file with the Ethical Review Board, as a publicly available document that can be requested by any citizen. Its existence should be possible to verify or falsify through a simple phone call to the office of the regional Ethical Review Board of Gothenburg. One could then also verify if the produres described in the articles match the ones permitted. If a human trial lacks thgis kind of permission, or is conducted in another way than what has been approved, it is illegal and punishable with financial penalty or jail. However, as the statute of limitation is very short, it seems none of the cases here would be possible to prosecute.
Medical Product Agency: Swedish FDA, may approve the production of an unlicenced medical product on a case by case basis. However that approval does not mean an approval of the use of that product (only the production). The responsibility for use rests entirely with the treating physician within standard conditions (that is, this person is not exempted from standard requirements of good care, etc.). This also means that if a procedure is a piece of research (a trial), standard legal requirements say that it needs approval of the Ethical Review Board.
National Medical Board: There is no authority/agency by that name, but presumably the authors mean the National Board of Health and Welfare, alternatively the Health Care Inspectorate. More likely the former. NBHW can, however, not authorise human trials or experiments; its role is to issue guidelines describing routine procedures and standards of care in different areas and to issue clarification of applicable law regarding health care. The Health Care Inspectorate is empowered to issue a number of special permissions in the health care area, but none of these regards experimental treatments or human trials.
I have no idea what “local ethical commission” may indicate. It may be that the authors do mean the Ethical Review Board, and in that case they would be able to prove this easily by making public their registered and numbered permission. However, from the local reporting over here it appears more likely that what is referred to is an informal body for ethical consultation, discussion and education at the Sahlgrenska Hospital called “Etiskt Forum”, which has no legal empowerment to authorise any kind of research or procedure.
From a legal research ethical and ethical approval standpoint, the statements on how the informed consent has been collected has no valence whatsoever. If the research has been undertaken without permission of the Ethical Review Board, there has been no prior description or valid assessment or vetting of these procedures.
LikeLike
I now obtained the Appendix of the Lancet publication by Olausson et al 2012, which the authors have indicated that it would contain consent documents “from all the relevant ethical organisations, including the parents, local ethical commission, and two regulatory bodies in Sweden National Medical Board and Medical Products Agency)“.
The Lancet Appendix contains no documentary evidence of institutional ethics approval.
The approvals by the MPA and the national Board did not exist, unlike authors pretended to. The approval granted by “Etiskt Forum” carries, as explained above by Christian Munthe “no legal empowerment to authorise any kind of research or procedure“. Olausson and Sumitran-Holgersson therefore performed an experimental transplant surgery on a child patient who was absolutely not in any directly life-threatening or even critical condition (as authors themselves admit below), and in absence of any ethics vote, to which they pretended nevertheless in this publication.
Previously, both The Lancet and Elsevier refused to share this Appendix with me, by ignoring all my requests. I am posting it here, and welcome the publisher to contact me to claim the copyright.
What the authors declare about the “Ethics Process” is only this:
LikeLike
Pingback: “Notification of Suspected Research Misconduct”, by 4 Macchiarini-whistleblowers – For Better Science
Pingback: Brexiting out of EU research and patient rights – For Better Science
Pingback: Pontus Boström: cheater carousel in Sweden – For Better Science
It is really gratifying seeing that some justice still exists… It makes me think that the debacle of Science is still salvageable and perhaps reversible…
LikeLike
Pingback: Professors: innocent victims of snakes at their bosoms? A guest post from Sweden – For Better Science
Pingback: Sumitran-Holgersson and Olausson guilty of misconduct and unethical experiments on children – For Better Science
Pingback: Swedish police investigation acquitted Macchiarini on advice of misconduct-tainted trachea transplanters – For Better Science
Pingback: Sumitran-Holgersson and Olausson to retract 8 papers for research misconduct – For Better Science
Pingback: Gothenburg to sack Sumitran-Holgersson, requests 7 retractions – For Better Science