Corrado Viotti is one of the most prolific internet sleuths who exposed many major research fraudsters in Europe and the Americas. You probably loved his previous work under an assumed name, but this is his first “outing”, so please give Corrado a warm welcome.
This time, Corrado leads us to another major university in USA, specifically to the Warren Alpert Medical School of Brown University, whose probably most famous researcher is the antivaxxer, internet troll and the most shameless science cheater, Wafik El-Deiry. Corrado will introduce you to his faculty colleagues, cardiovascular researchers like Frank Sellke, but also some nephrologists and orthopaedicians.
I contacted the main characters in this story, but none of them replied. Well, their research papers speak for themselves.

Mad School of Brown University
by Corrado Viotti
The Warren Alpert Medical School of Brown University is one of those places where students drop a 350 grand on an education they could have got for $3.50 in late charges at the public library11). While those hefty fees cover the running costs and staff salaries, the research is funded by multi-million dollar government grants, aka public money. I have filtered out several hundreds biomedical research articles from Brown containing the words “western blot” in the text, and found out the usual, unavoidable ocean of manipulated data. After major cases of data manipulation were found at Stanford, Harvard, Duke, MIT, Yale, etc., yet another top-ranked US institution and Ivy League member got caught with hands in the cookie jar.
Cancer at Duke? Better call Sal!
“I have NEVER faked data. If you wish to carry on what appears to be a vendetta please supply me the name of your lawyer and I will have my lawyer contact him.” – Sal Pizzo, Duke University
Frank Sellke
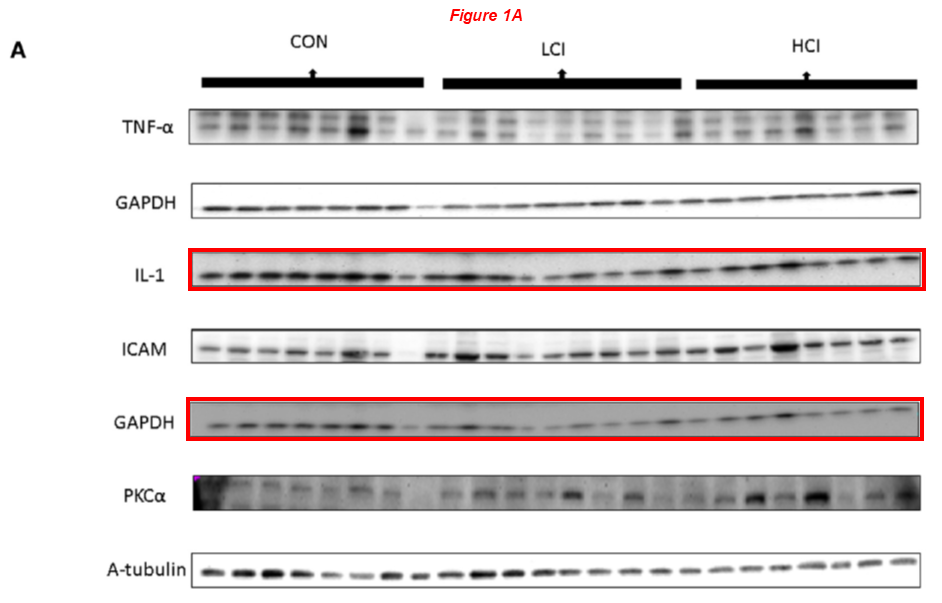
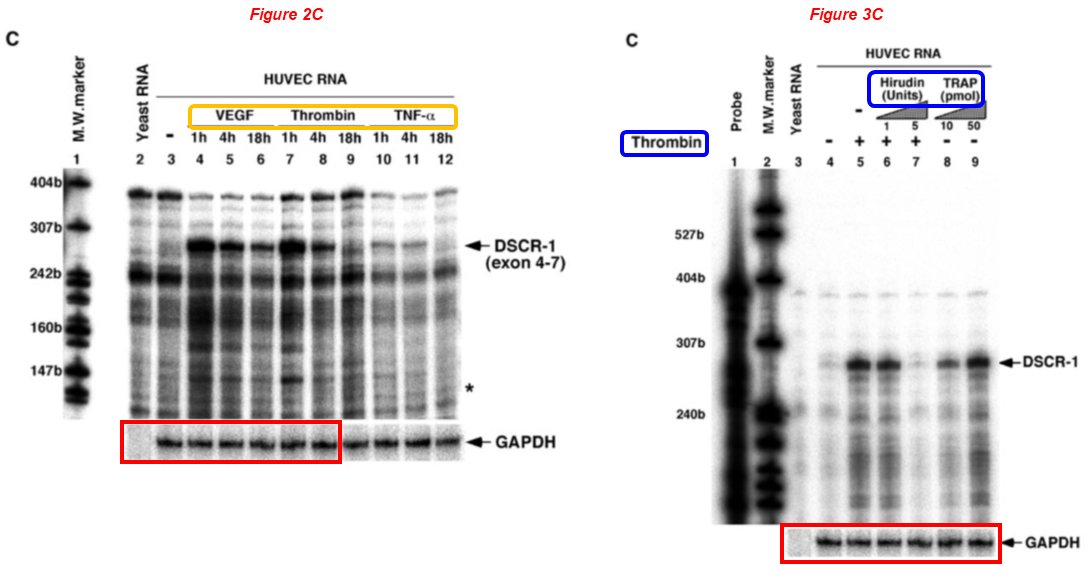
Please meet Frank Sellke, professor and chief of cardiothoracic surgery at the Warren Alpert Medical School (WAMS) and Lifespan Hospitals, and director of the Lifespan Cardiovascular Institute. Dr Sellke received NIH grants worth $9.5 million over the last decade, and sports over 40 papers on PubPeer with a troubling pattern of troubling data spanning 20+ years. Let’s start with a very recent one, where the same blots describe both ischemic and non-ischemic myocardium conditions:
Brittany A. Potz, Sharif A. Sabe, Laura A. Scrimgeour, Ashraf A. Sabe, Dwight D. Harris, M. Ruhul Abid, Richard T. Clements, Frank W. Sellke* Calpain inhibition decreases oxidative stress via mitochondrial regulation in a swine model of chronic myocardial ischemia Free Radical Biology and Medicine (2023) doi: 10.1016/j.freeradbiomed.2023.09.028
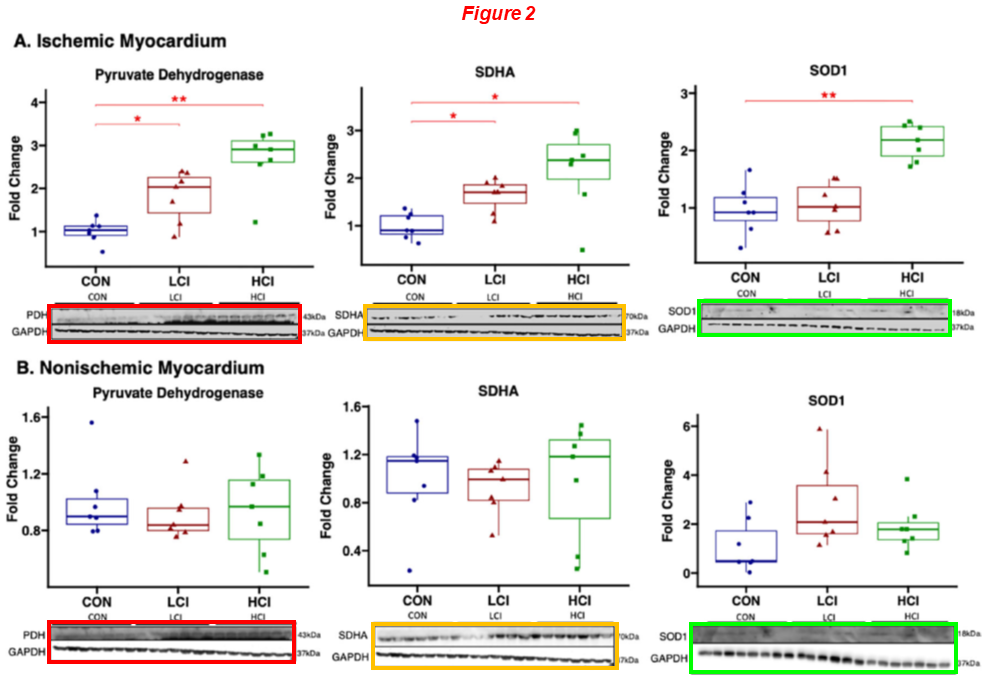

The six duplicated blots (PDH, SDHA, SOD1 and their controls) display different exposure times and were rescaled vertically. In the next one, a similar constellation of authors reused the same western blot for different proteins and conditions:
Laura A Scrimgeour, Brittany A Potz, Ahmad Aboul Gheit, Yuhong Liu, Guangbin Shi, Melissa Pfeiffer, Bonnie J Colantuono, Neel R Sodha, M Ruhul Abid, Frank W Sellke* Intravenous injection of extracellular vesicles to treat chronic myocardial ischemia PLOS One (2020) doi: 10.1371/journal.pone.0238879
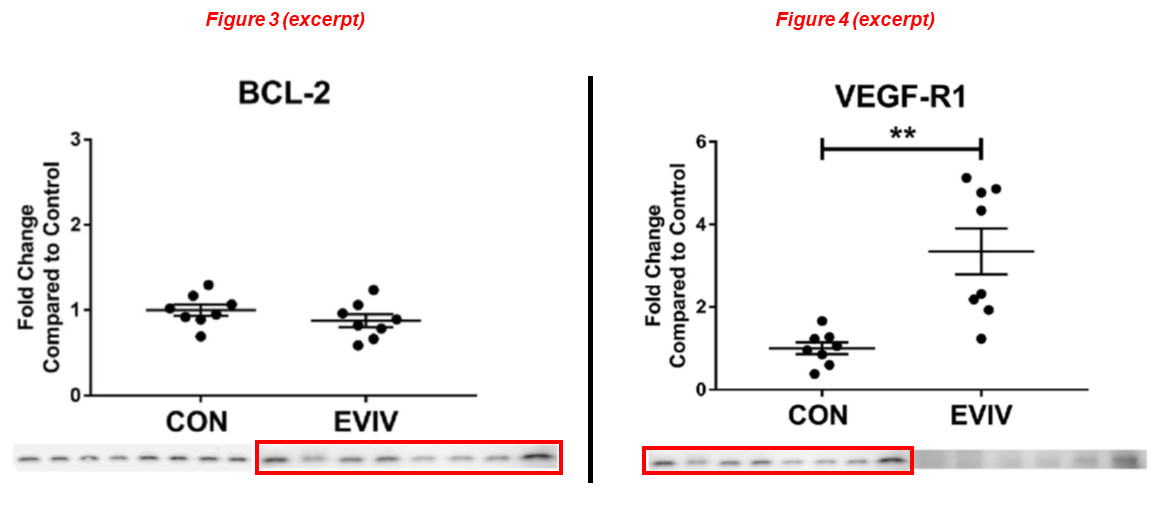
Another one below with Dr Brittany Potz in first position, she is now assistant professor at Penn State. Again the same blot, acquired at different exposure times, describes different things:
Brittany A. Potz, Ashraf A. Sabe, Nassrene Y. Elmadhun, Sharif A. Sabe, Benedikt J.V. Braun, Richard T. Clements, Anny Usheva, Frank W. Sellke* Calpain inhibition decreases inflammatory protein expression in vessel walls in a model of chronic myocardial ischemia Surgery (2017) doi: 10.1016/j.surg.2016.11.009
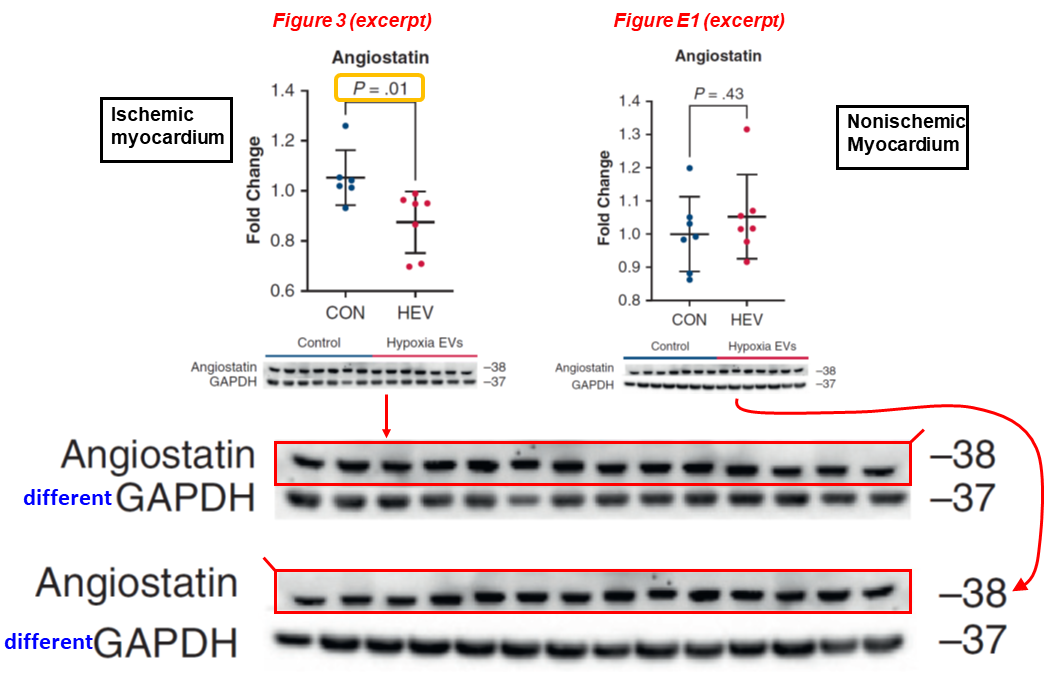
The young authors may change, but the same kind of problems keep cropping up. In the next study, once more the same blot describes both the ischemic and non-ischemic myocardium. Yet, the effect of HEV injection (the key topic of the paper) on angiostatin expression is reported to be significant (P = 0.01) only for the ischemic condition. The blots are also mirrored horizontally:
Dwight D. Harris, Sharif A. Sabe, Mark Broadwin, Christopher Stone, Cynthia Xu, Meghamsh Kanuparthy, Akshay Malhotra, M. Ruhul Abid, Frank W. Sellke* Intramyocardial injection of hypoxia-conditioned extracellular vesicles increases myocardial perfusion in a swine model of chronic coronary disease JTCVS Open (2024) doi: 10.1016/j.xjon.2024.06.003
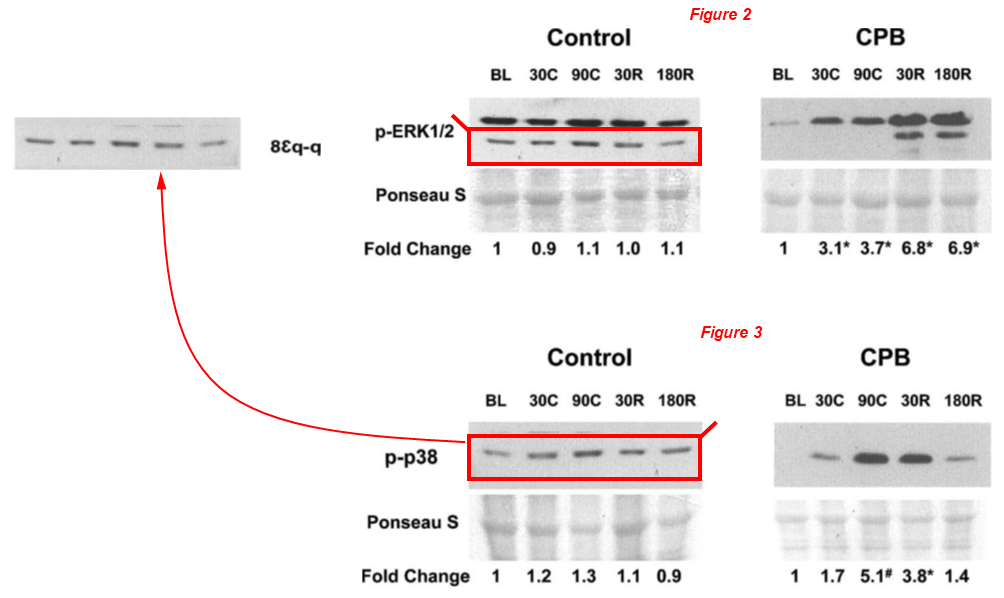
Now let’s go back almost quarter of a century! In this study from 2003, the lower band of p-ERK1/2 was cropped out, mirrored horizontally and presented as p-p38:
Tanveer A Khan, Cesario Bianchi, Eugenio G Araujo, Marc Ruel, Pierre Voisine, Frank W Sellke* Activation of pulmonary mitogen-activated protein kinases during cardiopulmonary bypass Journal of Surgical Research (2003) doi: 10.1016/s0022-4804(03)00236-1
Some of you may be wondering whether Sellke is maybe the victim here, betrayed by young associates seeking fortune and fame, and unaware of all that went on behind his back. The next case may give us some insights. Here we see a rescaled blot representing Tubulin and GAPDH, across papers published three years apart, a kind of forgery one would expect from anywhere but Ivy League universities:
- Yuhong Liu, Victoria Cole, Isabella Lawandy, Afshin Ehsan, Frank W. Sellke, Jun Feng* Decreased coronary arteriolar response to KCa channel opener after cardioplegic arrest in diabetic patients Molecular and Cellular Biochemistry (2018) doi: 10.1007/s11010-017-3264-x
- Yuhong Liu, An Xie, Arun K. Singh, Afshin Ehsan, Gaurav Choudhary, Samuel Dudley, Frank W. Sellke, Jun Feng* Inactivation of Endothelial Small/Intermediate Conductance of Calcium‐Activated Potassium Channels Contributes to Coronary Arteriolar Dysfunction in Diabetic Patients Journal of the American Heart Association (2015) doi: 10.1161/jaha.115.002062
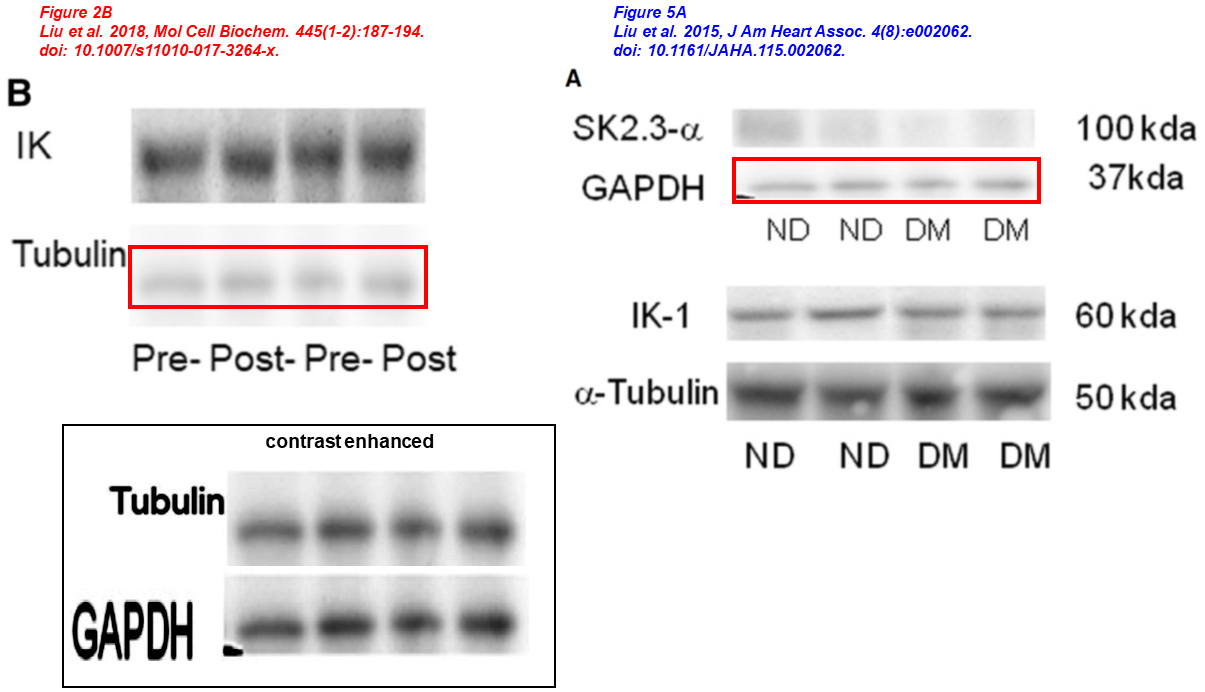
This is where it gets interesting. On the issues shown above, Sellke commented at PubPeer with a laconic:
“They are not same bands“
How it is possible that a thoracic surgeon – who should have excellent eyesight – could fail to recognize the identity of the bands shown above is a mystery I fear could be easily solved.
Jun Feng
The two papers listed immediately above have the same corresponding author. Please meet Jun Feng, long-time Sellke’s associate, former managing director of the department of surgery at the Warren Alpert Med School, who left the harsh climate of New England and moved south to the University of South Florida in 2025. Dr Feng received NIH grants worth $5 million in the last ten years, and sports 23 papers on PubPeer, all with Frank Sellke. Here we have two articles published simultaneously, in the same issue of Circulation, where Sellke is the lead author. At the time twenty years ago, Feng was an eager young mind in Sellke’s group at Harvard University.
- Jun Feng, Cesario Bianchi, Jennifer L. Sandmeyer, Frank W. Sellke* Bradykinin Preconditioning Improves the Profile of Cell Survival Proteins and Limits Apoptosis After Cardioplegic Arrest Circulation (2005) doi: 10.1161/circulationaha.104.524454
- Jun Feng, Cesario Bianchi, Jennifer L. Sandmeyer, Jianyi Li, Frank W. Sellke* Molecular Indices of Apoptosis After Intermittent Blood and Crystalloid Cardioplegia Circulation (2005) doi: 10.1161/circulationaha.104.526160
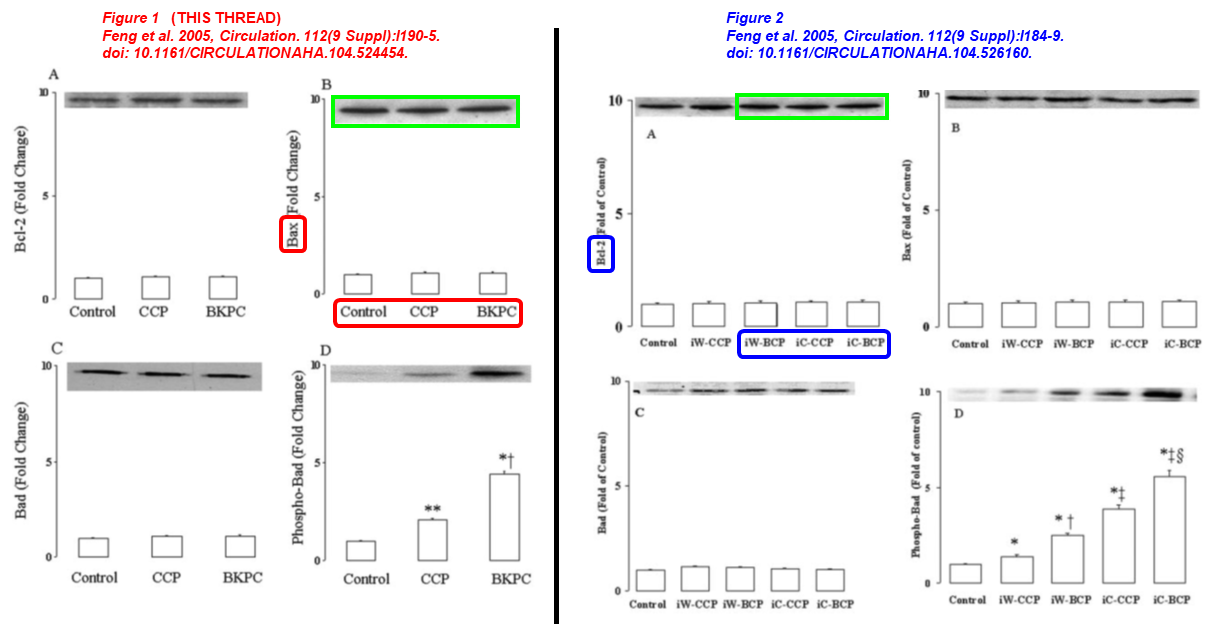
The same blot describes Bax and Bcl-2 expression under completely different treatments.
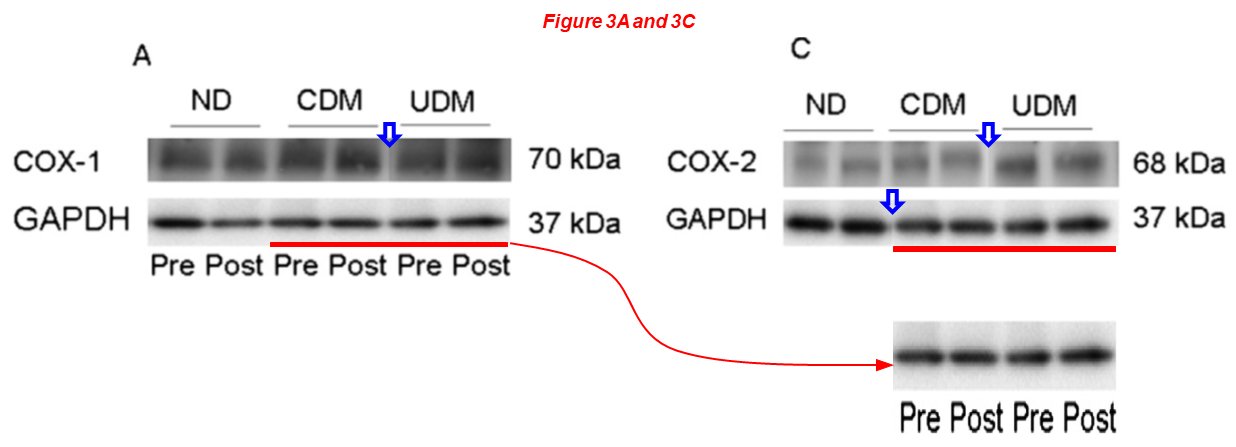
In the next one, Feng is again first author and Sellke last. They inform us that “upregulation of COX-2 expression may contribute to bradykinin-induced coronary arteriolar relaxation in diabetic patients undergoing cardiac surgery“. Look at the COX2 blot and at other things:
Jun Feng, Kelsey Anderson, Arun K. Singh, Afshin Ehsan, Hunter Mitchell, Yuhong Liu, Frank W. Sellke* Diabetes Upregulation of Cyclooxygenase 2 Contributes to Altered Coronary Reactivity After Cardiac Surgery The Annals of Thoracic Surgery (2017) doi: 10.1016/j.athoracsur.2016.11.025
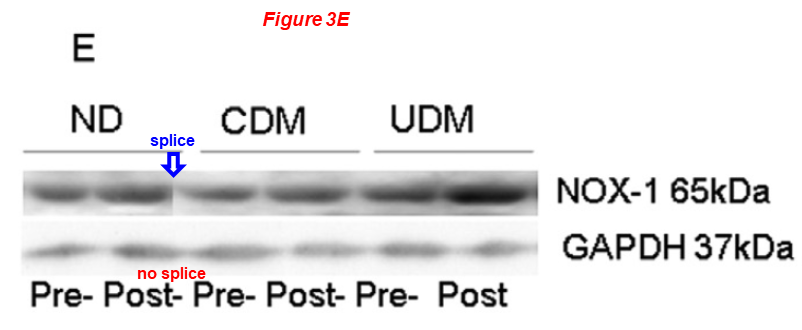
The next two papers have on board the pharmacology professor at the University of Fribourg, Switzerland, Csaba Szabo, an activist against scientific fraud and contributor at For Better Science.
“Unreliable” by Csaba Szabo – book review and excerpt
Csaba Szabo’s book “Unreliable: Bias, Fraud, and the Reproducibility Crisis in Biomedical Research” – review by Zoltan Ungvari and excerpt.
Dr Szabo found himself entangled in this affair due to an ill-fated collaboration with Sellke that took place 17 years ago. All the experimental work was done in Sellke’s lab. Csaba urged Sellke to take action2, and it actually had an effect, as Sellke began to acknowledge some of the issues on PubPeer.
The first author on both papers is a former collaborator of Sellke, Robert Osipov, now working as surgeon and vein specialist in Arizona. Jun Feng is again on board:
Robert M. Osipov, Michael P. Robich, Jun Feng, Vincent Chan, Richard T. Clements, Ralph J. Deyo, Csaba Szabo, Frank W. Sellke* Effect of hydrogen sulfide on myocardial protection in the setting of cardioplegia and cardiopulmonary bypass☆ Interactive CardioVascular and Thoracic Surgery (2010) doi: 10.1510/icvts.2009.219535
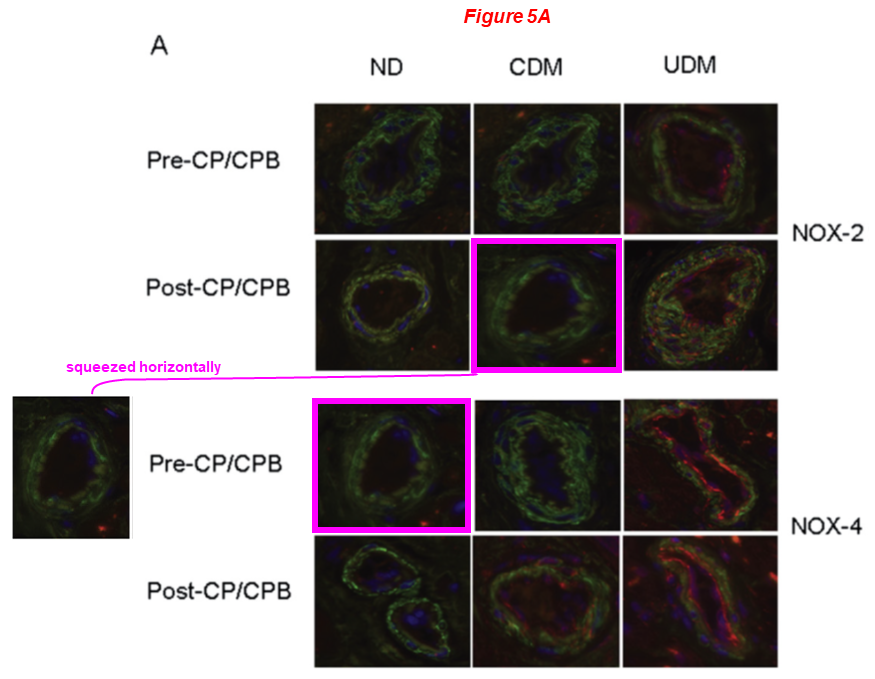
In the next one, four sets of identical blots describe different proteins and/or conditions in three main figures, some are rescaled:
Robert M Osipov, Michael P Robich, Jun Feng, Yuhong Liu, Richard T Clements, Hilary P Glazer, Neel R Sodha, Csaba Szabo, Cesario Bianchi, Frank W Sellke* Effect of Hydrogen Sulfide in a Porcine Model of Myocardial Ischemia-Reperfusion: Comparison of Different Administration Regimens and Characterization of the Cellular Mechanisms of Protection Journal of Cardiovascular Pharmacology (2009) doi: 10.1097/fjc.0b013e3181b2b72b
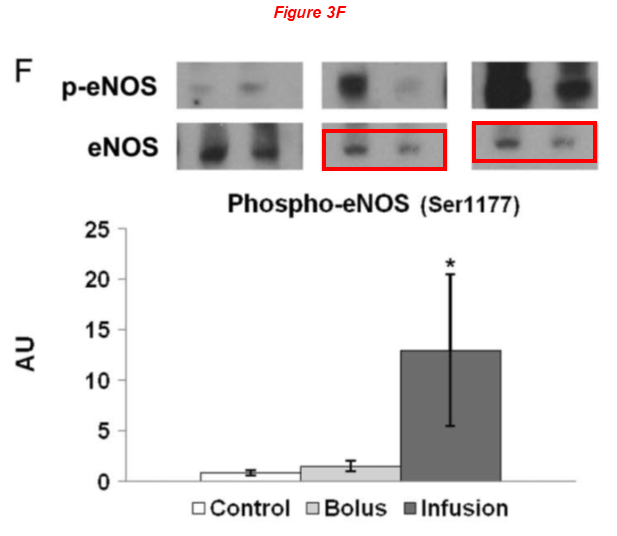
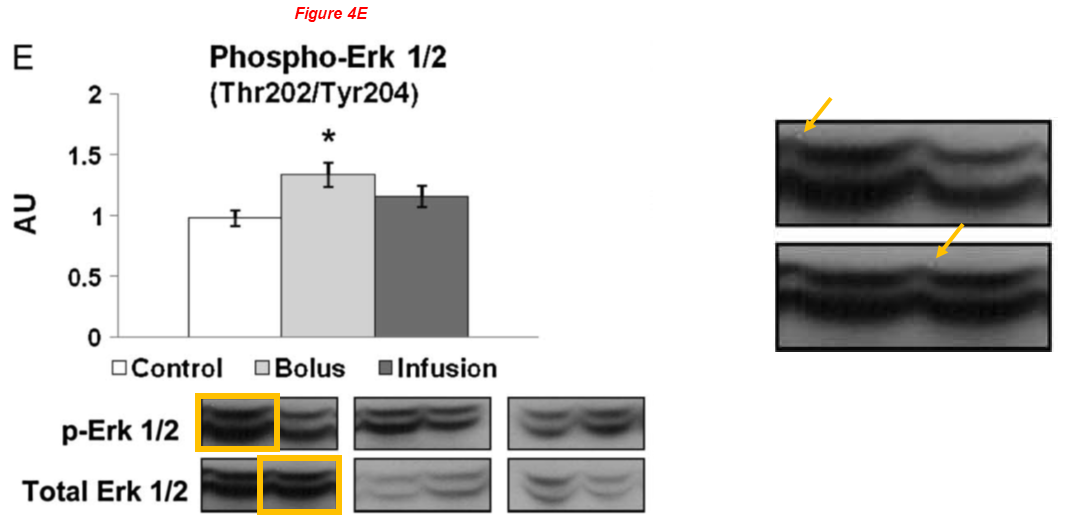
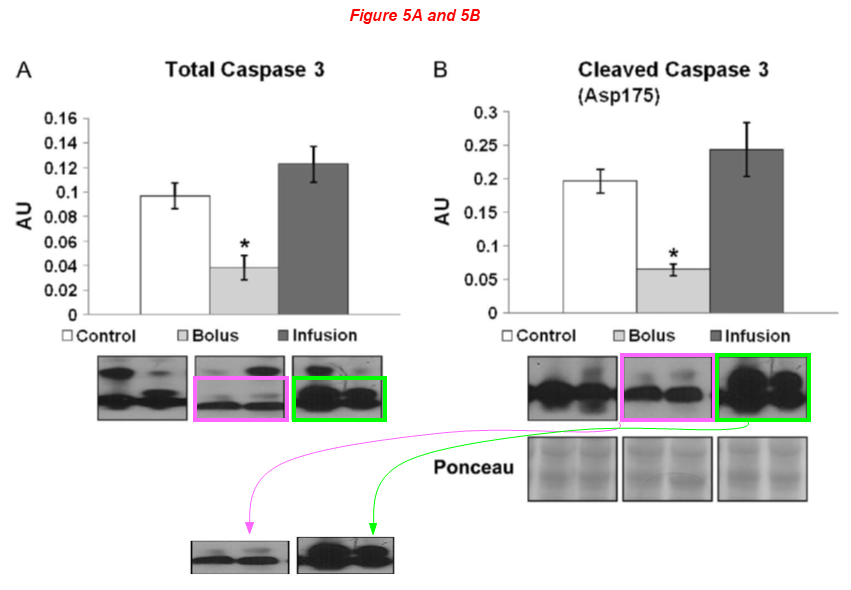
There are several regular Sellke’s collaborators on board the article above, namely Brown professors Cesario Bianchi and Neel Sodha, the assistant professor at University of Rhode Island Richard Clements, and finally Michael Robich, now employed at Banner Health in Colorado. Here are two works with Robich as first author:
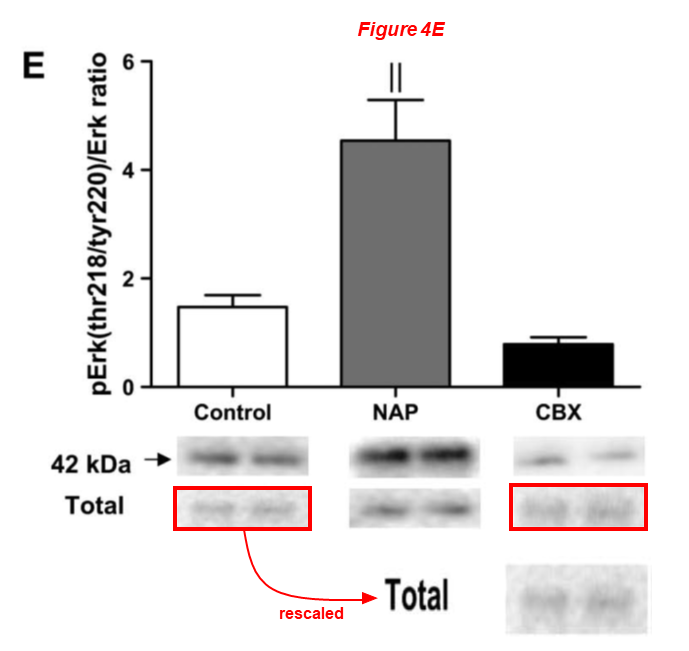
Fig 4E
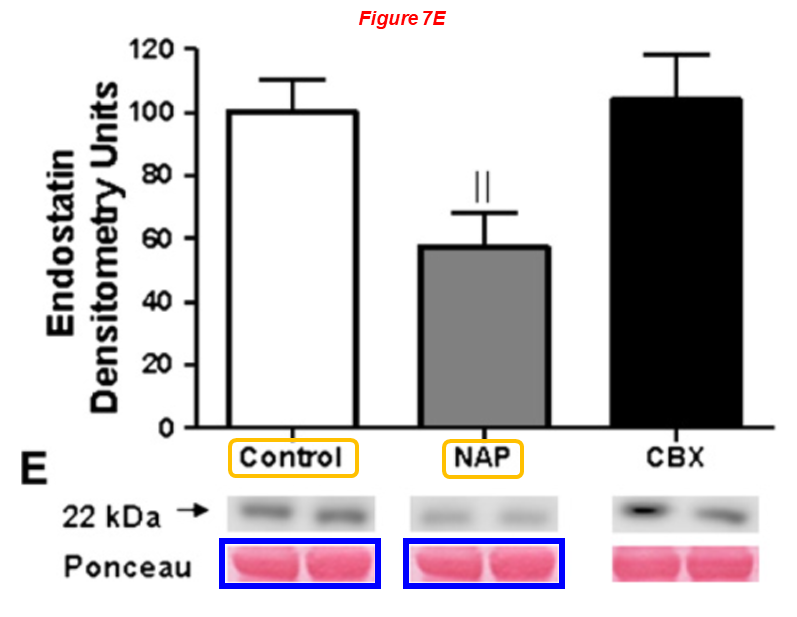
Fig 7E
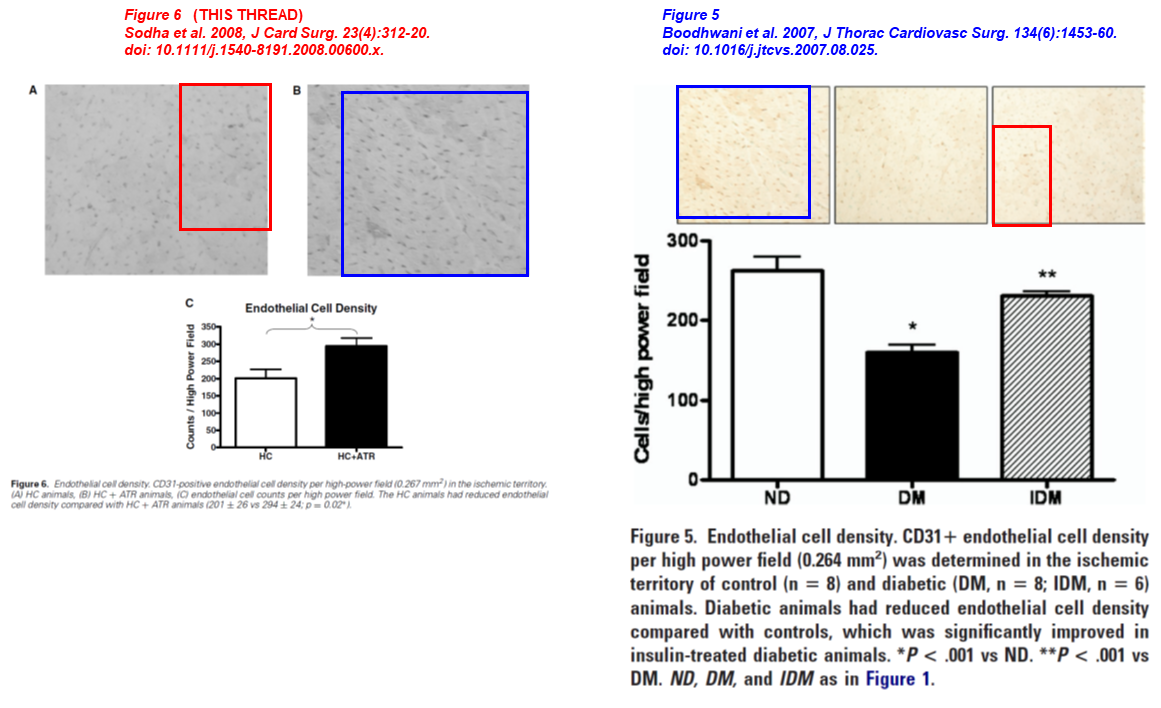
Here are two old works with Professor Sodha as lead author. Two micrographs overlap, in both papers the images describe endothelial cell density, but the animals are either treated/not treated with atorvastatin (left) or represent animals diabetic/non-diabetic (right):
- Neel R. Sodha, Munir Boodhwani, Basel Ramlawi, Richard T. Clements, Shigetoshi Mieno, Jun Feng, Shu-Hua Xu, Cesario Bianchi, Frank W. Sellke* Atorvastatin Increases Myocardial Indices of Oxidative Stress in a Porcine Model of Hypercholesterolemia and Chronic Ischemia Journal of Cardiac Surgery (2008) doi: 10.1111/j.1540-8191.2008.00600.x
- Munir Boodhwani, Neel R. Sodha, Shigetoshi Mieno , Basel Ramlawi , Shu-Hua Xu , Jun Feng, Richard T. Clements , Marc Ruel , Frank W. Sellke Insulin treatment enhances the myocardial angiogenic response in diabetes The Journal of Thoracic and Cardiovascular Surgery (2007) doi: 10.1016/j.jtcvs.2007.08.025
Ruhul Abid
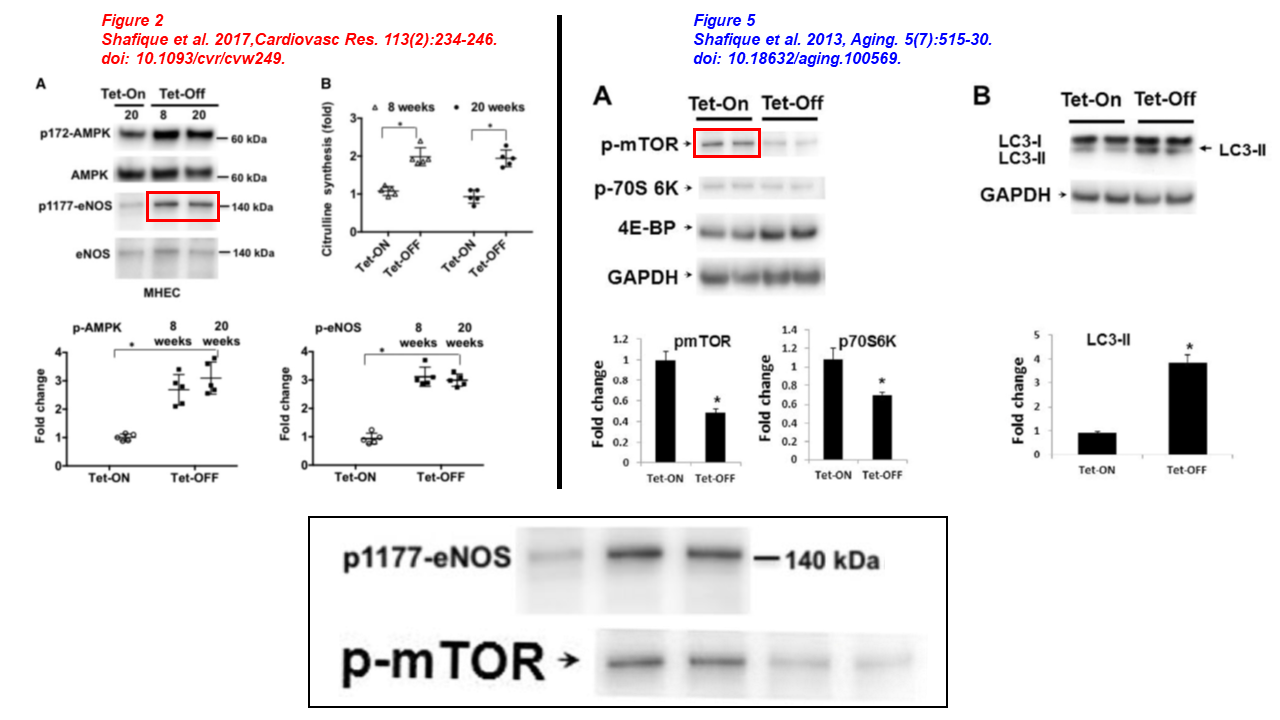
Please meet Ruhul Abid, yet another regular collaborator of Sellke’s, co-author of several papers described above, professor at WAMS and principal investigator at the Cardiovascular Research Center, recipient of $3.3 milion in NIH grants in the last decade. Abid sports 13 papers on PubPeer. In the article below one eNOS blot used to be p-mTOR four years before, Sellke and Feng are on board too:
- Ehtesham Shafique, Anali Torina, Karla Reichert, Bonnie Colantuono, Nasifa Nur, Khawaja Zeeshan, Vani Ravichandran, Yuhong Liu, Jun Feng, Khawaja Zeeshan, Laura E. Benjamin, Kaikobad Irani, Elizabeth O. Harrington, Frank W. Sellke, Md. Ruhul Abid* Mitochondrial redox plays a critical role in the paradoxical effects of NAPDH oxidase-derived ROS on coronary endothelium Cardiovascular Research (2017) doi: 10.1093/cvr/cvw249
- Ehtesham Shafique , Wing C. Choy , Yuhong Liu , Jun Feng , Brenda Cordeiro , Arthur Lyra , Mohammed Arafah , Abdulmounem Yassin-Kassab , Arthus V.D. Zanetti , Richard T. Clements , Cesario Bianchi , Laura E. Benjamin , Frank W. Sellke, Md. Ruhul Abid Oxidative stress improves coronary endothelial function through activation of the pro-survival kinase AMPK Aging (2013) doi: 10.18632/aging.100569
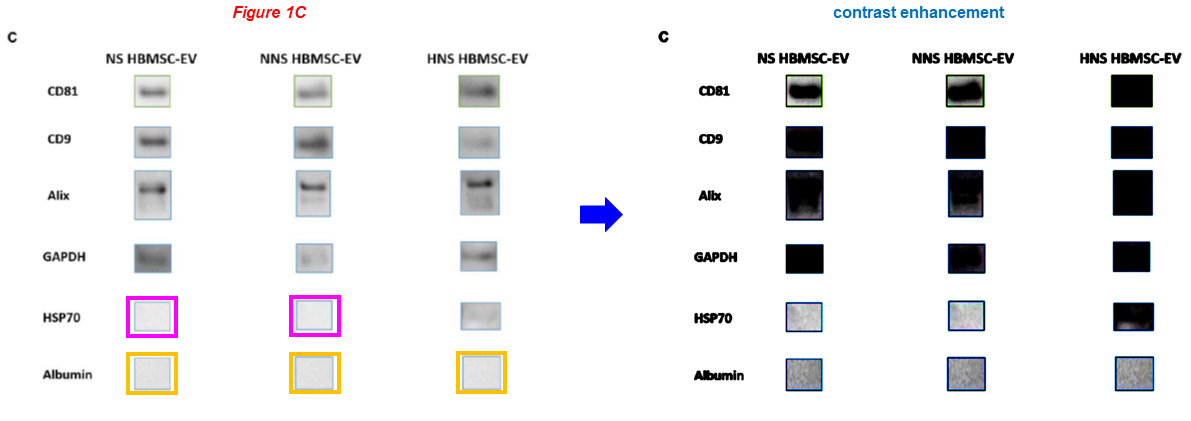
Now two very recent studies by Abid cosigned by Sellke, where blots were reused for different experiments across papers, while others are duplicated. Please also note the individual cropped bands, something that is no longer standard practice for thirty years already:
Cynthia M. Xu, Catherine Karbasiafshar, Rayane Brinck Teixeira, Nagib Ahsan, Giana Blume Corssac, Frank W. Sellke, M. Ruhul Abid* Proteomic Assessment of Hypoxia-Pre-Conditioned Human Bone Marrow Mesenchymal Stem Cell-Derived Extracellular Vesicles Demonstrates Promise in the Treatment of Cardiovascular Disease International Journal of Molecular Sciences (2023) doi: 10.3390/ijms24021674
Here the other postage stamp album, reusing same data:
Cynthia M. Xu, Sharif A. Sabe, Rayane Brinck‐Teixeira, Mohamed Sabra, Frank W. Sellke, M. Ruhul Abid* Visualization of cardiac uptake of bone marrow mesenchymal stem cell‐derived extracellular vesicles after intramyocardial or intravenous injection in murine myocardial infarction Physiological Reports (2023) doi: 10.14814/phy2.15568
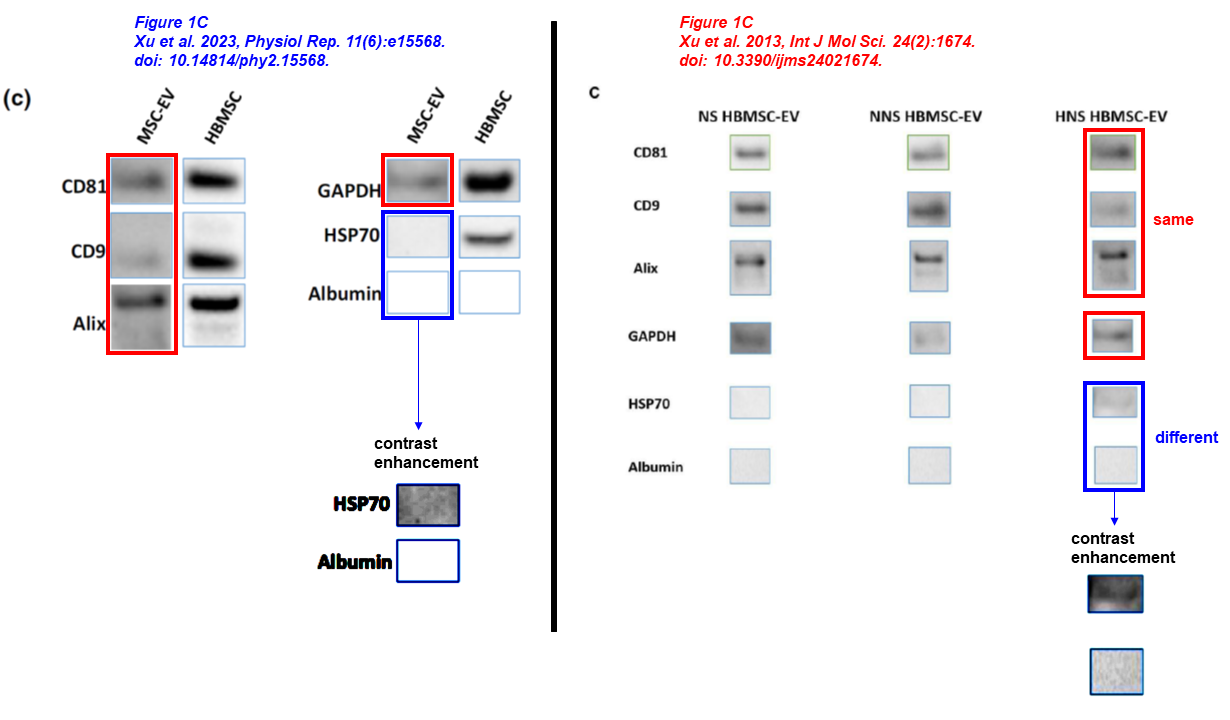
Abid was an early adopter. The following one is 22 years old, and Abid is first- and corresponding author. At the time, he was working at the Beth Israel Deaconess Medical Center of Harvard University. Two sets of identical blots describe different proteins, four sets of mRNA controls describe different conditions within this paper and across papers:
Md. Ruhul Abid*, Ivo G. Schoots, Katherine C. Spokes, Sheng-Qian Wu, Christina Mawhinney, William C. Aird Vascular endothelial growth factor-mediated induction of manganese superoxide dismutase occurs through redox-dependent regulation of forkhead and IkappaB/NF-kappaB Journal of Biological Chemistry (2004) doi: 10.1074/jbc.m408285200
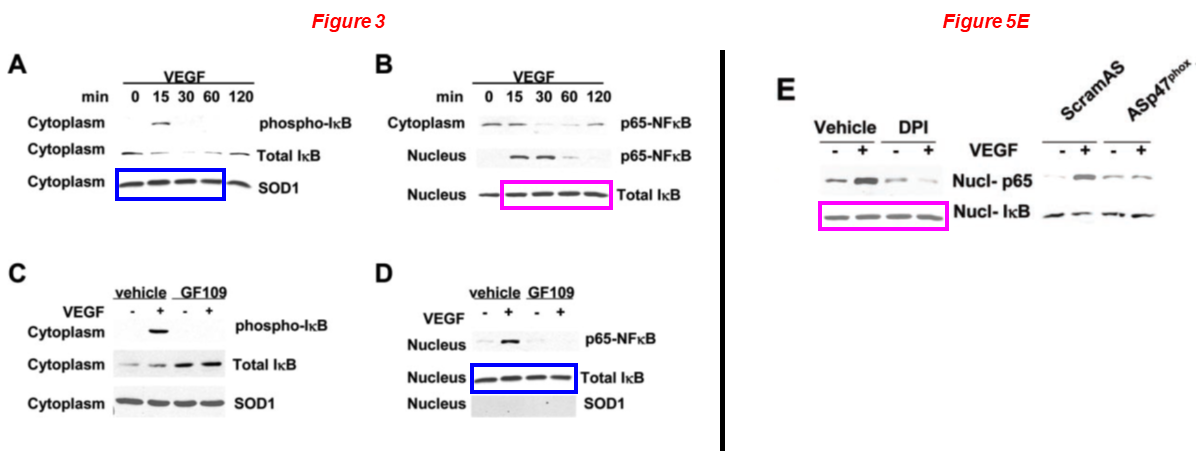
The same blot has been reused to describe Total IκB in Figure 3B and Nucl-IκB in Figure 5E (magenta boxes).
The same The 28S rRNA control has been used in Figure 1D and 1E, but conditions are different (green boxes).
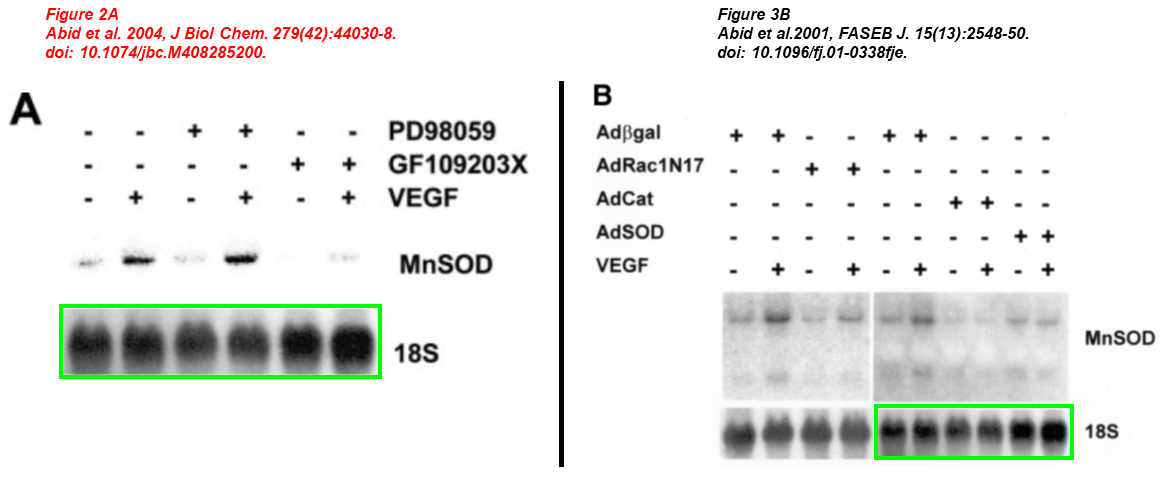
Md. Ruhul Abid , Jo C. Tsai , Katherine C. Spokes , Shailesh S. Deshpande , Kaikobad Irani , William C. Aird Vascular endothelial growth factor induces manganese‐superoxide dismutase expression in endothelial cells by a Racl‐regulated NADPH oxidase‐dependent mechanism The FASEB Journal (2001) doi: 10.1096/fj.01-0338fje
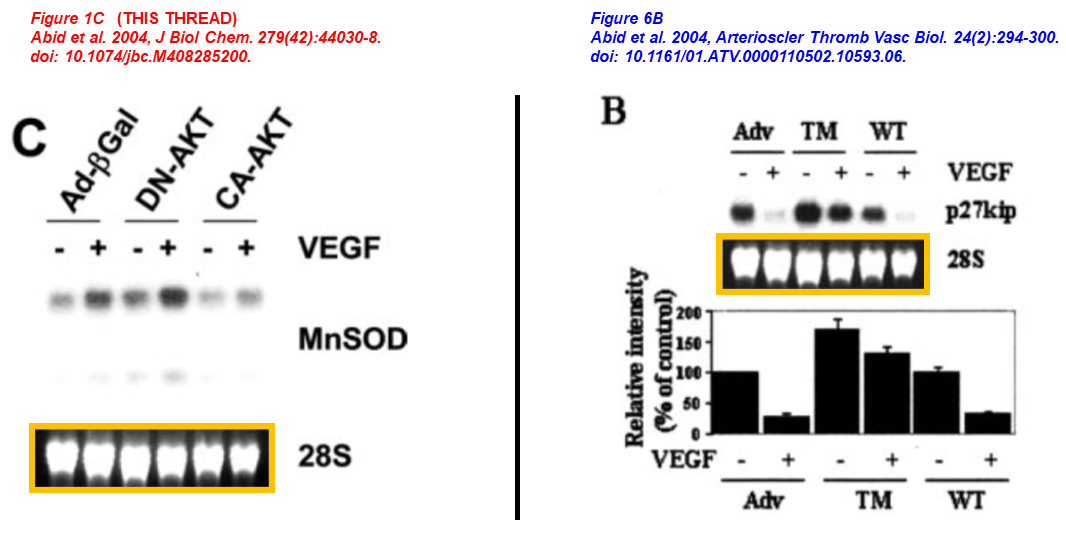
Md. Ruhul Abid, Shaodong Guo , Takashi Minami , Katherine C. Spokes , Kohjiro Ueki , Carsten Skurk , Kenneth Walsh , William C. Aird Vascular Endothelial Growth Factor Activates PI3K/Akt/Forkhead Signaling in Endothelial Cells Arteriosclerosis, Thrombosis, and Vascular Biology (2004) doi: 10.1161/01.atv.0000110502.10593.06
Yet another vintage classic for Abid, a quarter of a century old, this time the corresponding author is William Aird, professor of medicine at Harvard Medical School and a practising hematologist at the Beth Israel Deaconess Medical Center. Among other things, please appreciate the all identical flow cytometry plots:
Md. Ruhul Abid, Jo C. Tsai, Katherine C. Spokes, Shailesh S. Deshpande, Kaikobad Irani, William C. Aird* Vascular endothelial growth factor induces manganese‐superoxide dismutase expression in endothelial cells by a Racl‐regulated NADPH oxidase‐dependent mechanism FASEB Journal (2001) doi: 10.1096/fj.01-0338fje
Another one from William Aird’s lab, Abid is again on board:
Takashi Minami, Keiko Horiuchi, Mai Miura, Md. Ruhul Abid, Wakako Takabe, Noriko Noguchi, Takahide Kohro, Xijin Ge, Hiroyuki Aburatani, Takao Hamakubo, Tatsuhiko Kodama, William C. Aird* Vascular Endothelial Growth Factor- and Thrombin-induced Termination Factor, Down Syndrome Critical Region-1, Attenuates Endothelial Cell Proliferation and Angiogenesis Journal of Biological Chemistry (2004) doi: 10.1074/jbc.m406454200
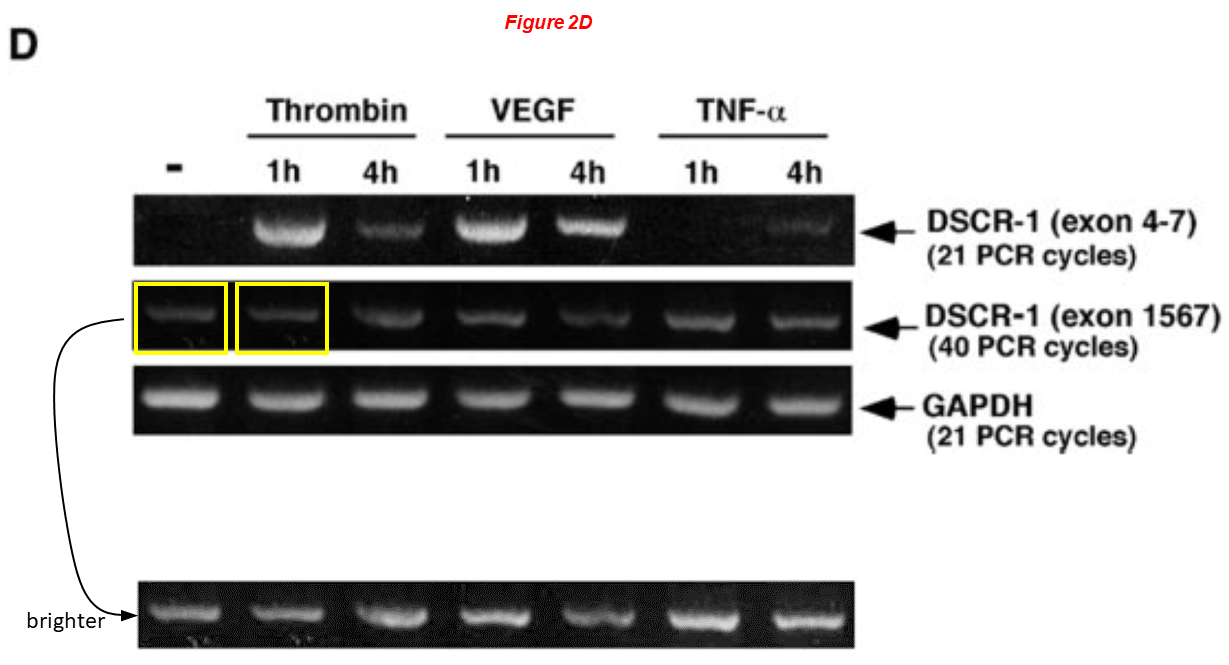
Anny Usheva
Anny Usheva, professor of surgery at WAMS, is the last member of Sellke’s cluster of people that I present you, we have already encountered her in some papers shown above. Dr Usheva is co-recipient with Sellke of an NIH grant worth $5.7 million. She has 8 papers on PubPeer. Here the common problem, a rescaled and mirrored blot represents different experimental conditions:
Martin Lange, Tatsuya Fujikawa, Anna Koulova, Sona Kang, Michael Griffin, Antonio Lassaletta, Anna Erat, Edda Tobiash, Cesario Bianchi, Nassrene Elmadhun, Frank Sellke, Anny Usheva* Arterial territory-specific phosphorylated retinoblastoma protein species and CDK2 promote differences in the vascular smooth muscle cell response to mitogens Cell Cycle (2014) doi: 10.4161/cc.27056

Like the paper above, the next one is from the time when Dr Usheva was at Beth Israel Deaconess Medical Center of Harvard, which is the place where also Adid and Sellke used to work before landing at Brown. There seems to be a very special relationship between these institutions.
Boian S. Alexandrov, Vladimir Gelev, Sang Wook Yoo, Ludmil B. Alexandrov, Yayoi Fukuyo, Alan R. Bishop, Kim Ø. Rasmussen, Anny Usheva* DNA dynamics play a role as a basal transcription factor in the positioning and regulation of gene transcription initiation Nucleic Acids Research (2010) doi: 10.1093/nar/gkp1084

Three sets of lanes are badly duplicated, presumably the reviewers were reviewing while in the shower.
Let’s move away from Sellke & Co and discuss other people at WAMS. Also in their research, you’ll see the issues are just as baffling.
Shougang Zhuang
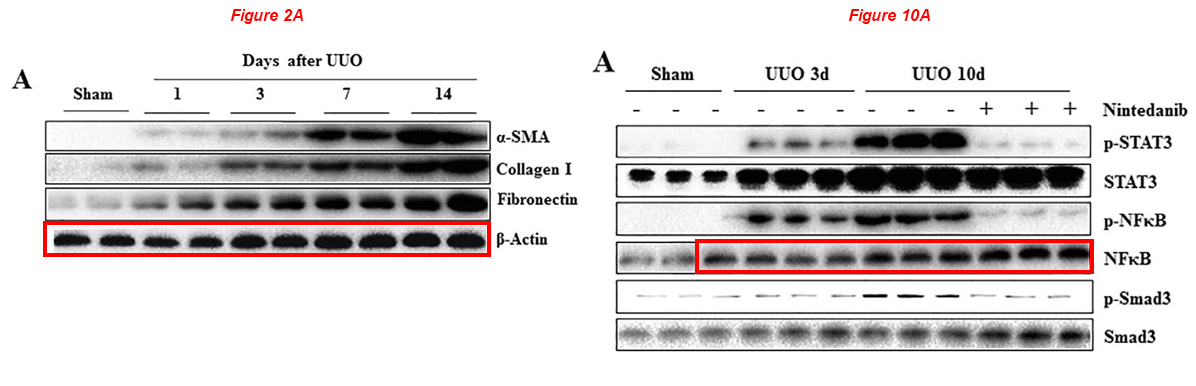
Please meet Shougang Zhuang, professor of medicine at WAMS and director of Kidney Research at Rhode Island Hospital. Dr Zhuang received $2.6 million of NIH grants in the last ten years, and boasts 40 papers on PubPeer. Here is an article about Nintedanib, sold under the brand names Ofev and Vargatef, an oral medication used for the treatment of idiopathic pulmonary fibrosis, which the authors claim has beneficial effects on kidney fibrosis too. Blots describe different proteins across figures, and some others are patchworks of copied & pasted bands. Look at this joke:
Feng Liu, Li Wang, Hualin Qi, Jun Wang, Yi Wang, Wei Jiang, Liuqing Xu, Na Liu, Shougang Zhuang* Nintedanib, a triple tyrosine kinase inhibitor, attenuates renal fibrosis in chronic kidney disease Clinical Science (2017) doi: 10.1042/cs20170134
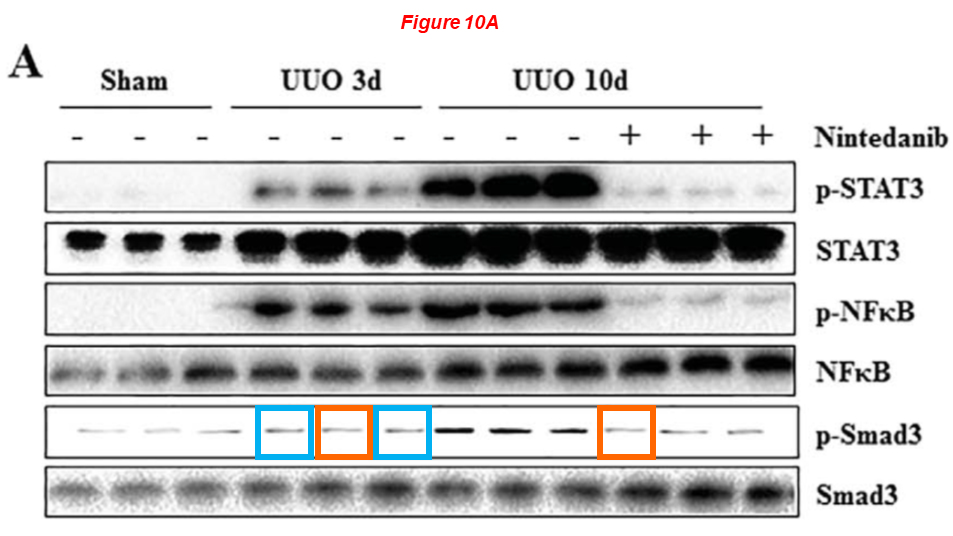
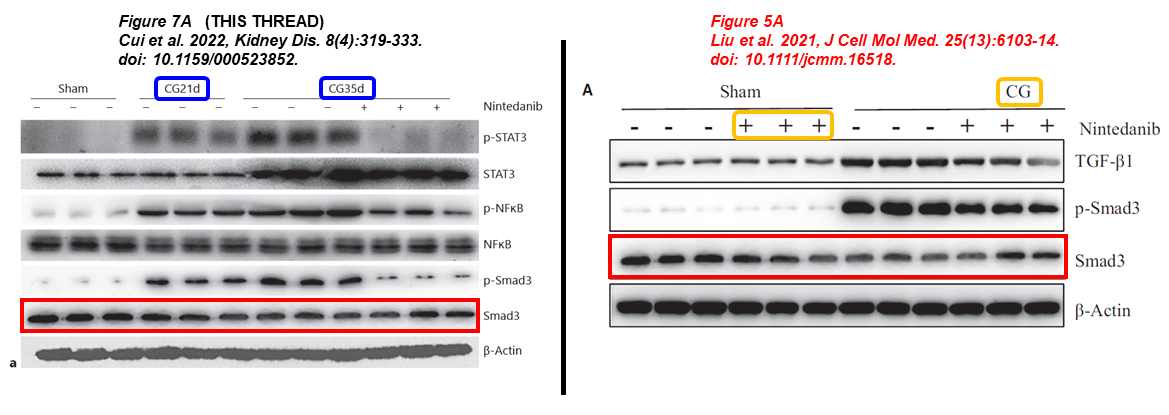
Again a paper about Nintedanib for treating kidney fibrosis, reusing same gel bands:
Binbin Cui, Chao Yu, Shenglei Zhang, Xiying Hou, Yi Wang, Jun Wang, Shougang Zhuang, Feng Liu* Delayed Administration of Nintedanib Ameliorates Fibrosis Progression in CG-Induced Peritoneal Fibrosis Mouse Model Kidney Diseases (2022) doi: 10.1159/000523852
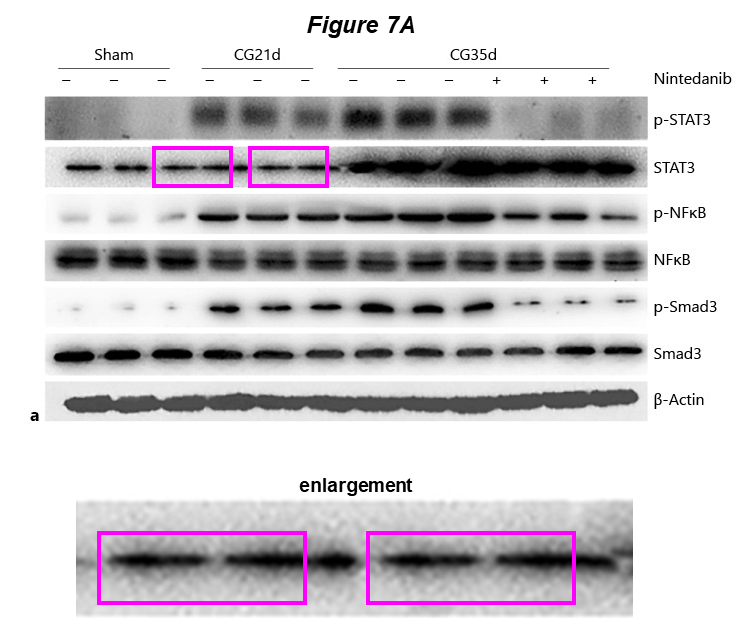
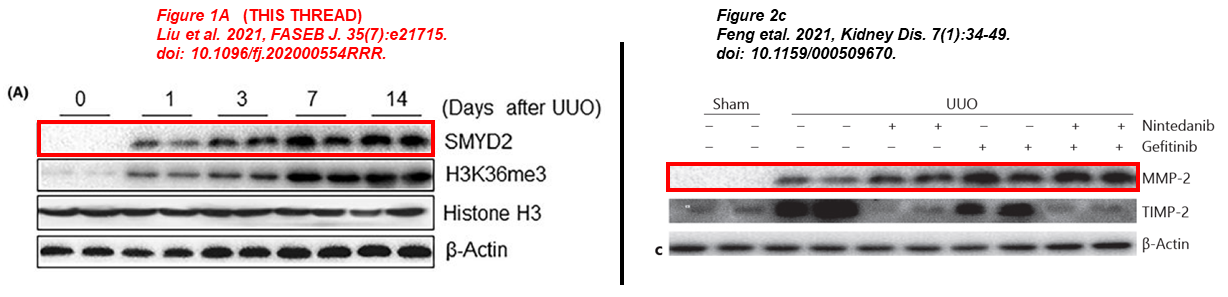
Here two papers published months apart, where the same blot describes SMYD2 and MMP-2. One of the papers focuses on the role of SMYD2 in kidney fibrosis, the other one describes the benefits of simultaneous treatment with Nintedanib and Gefitinib in a murine model:
- Lirong Liu, Feng Liu, Yingjie Guan, Jianan Zou, Chunyun Zhang, Chongxiang Xiong, Ting C. Zhao, George Bayliss, Xiaogang Li, Shougang Zhuang* Critical roles of SMYD2 lysine methyltransferase in mediating renal fibroblast activation and kidney fibrosis The FASEB Journal (2021) doi: 10.1096/fj.202000554rrr
- Liu Feng, Wang Li, Yu Chao, Qin Huan, Fang Lu, Wang Yi, Wang Jun, Cui Binbin, Liu Na, Zhuang Shougang* Synergistic Inhibition of Renal Fibrosis by Nintedanib and Gefitinib in a Murine Model of Obstructive Nephropathy Kidney Diseases (2020) doi: 10.1159/000509670
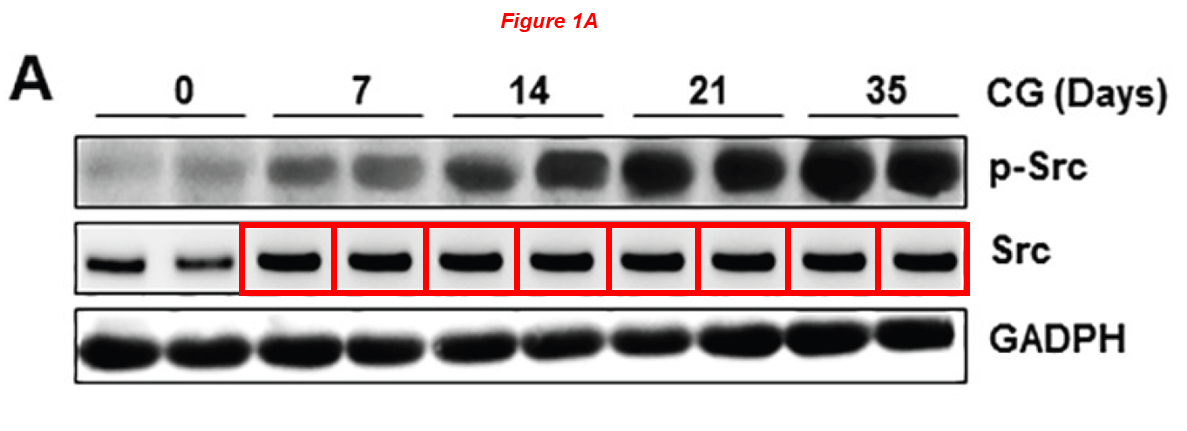
I’ll present you just one more paper by Shougang Zhuang, a scientific atrocity published in Oncotarget, a journal that will allow me to introduce another incredible character from WAMS in the next chapter. Look at these forgeries from an Ivy League university, and follow the PubPeer link to see much, much more:
Jun Wang, Li Wang, Liuqing Xu, Yingfeng Shi, Feng Liu, Hualin Qi, Na Liu, Shougang Zhuang* Targeting Src attenuates peritoneal fibrosis and inhibits the epithelial to mesenchymal transition Oncotarget (2017) doi: 10.18632/oncotarget.20040
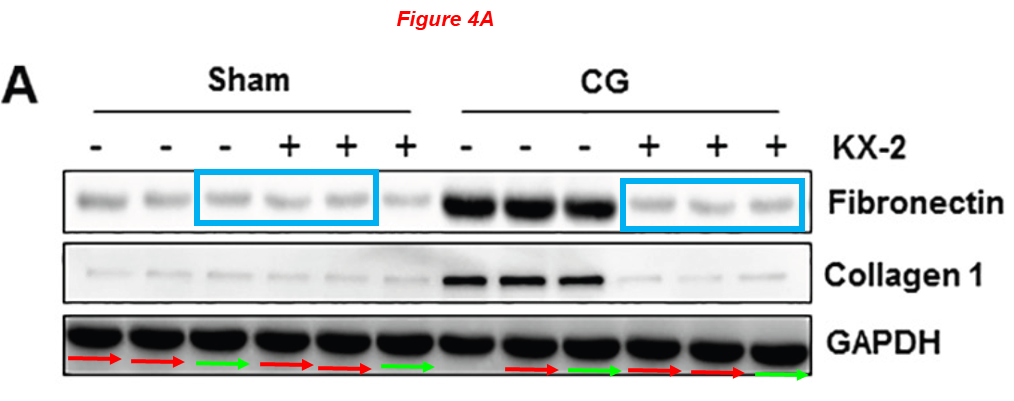
As I mentioned, there are 40 such papers on PubPeer for Shougang Zhuang, and I can’t show them all here. So, let’s continue with the Editor-in-Chief of Oncotarget, who incidentally also works at WAMS.
Wafik El-Deiry
Here we finally reach with the legendary Wafik El-Deiry, a man whom regular For Better Science readers know for the truckload of questionable data he published across 30 years, and notorious for his support of the worst of antivaxxers and his wild and bizarre outbursts on social media. El-Deiry sports 94 papers tagged on PubPeer and 1 retraction as of today, yet he received NIH grants worth $4 million in the last decade.
El-Deiry even used to be Trump’s candidate to lead the National Cancer Institute (NCI), because he claimed COVID-19 vaccines would cause cancer. Luckily for American people, his candidacy for that position never materialized, at least so far. More insights about El-Deiry can be gathered from the article below, and also in June 2025 Shorts and January 2026 Shorts.
Wafik El Deiry, the anti-qualified Science Guardian
Both Harvey Risch and Wafik El Deiry are perfectly anti-qualified candidates to lead the National Cancer Institute.
El-Deiry is most certainly one of the people behind the notorious Science Guardians website, run by a gang of individuals who call the sleuths “The PubPeer Mob” and whine about the “weaponization of post peer-review”, whatever that means. Here are some examples of El-Deiry’s science, in a predatory journal from e-Century:
Cassandra S Parker, Lanlan Zhou, Varun V Prabhu, Seulki Lee, Thomas J Miner, Eric A Ross, Wafik S El-Deiry* ONC201/TIC10 plus TLY012 anti-cancer effects via apoptosis inhibitor downregulation, stimulation of integrated stress response and death receptor DR5 in gastric adenocarcinoma American Journal of Cancer Research (2023) pubmed: 38187068


In the last weeks the American Association for Cancer Research (AACR) have issued four Expressions of Concern for El-Deiry‘s fake papers (read March 2026 Shorts), which triggered Wafik’s rage on social media. He asked the NIH, the Department of Health & Human Services and even the White House to investigate AACR and the “weaponization of post peer-review platforms such as PubPeer“.
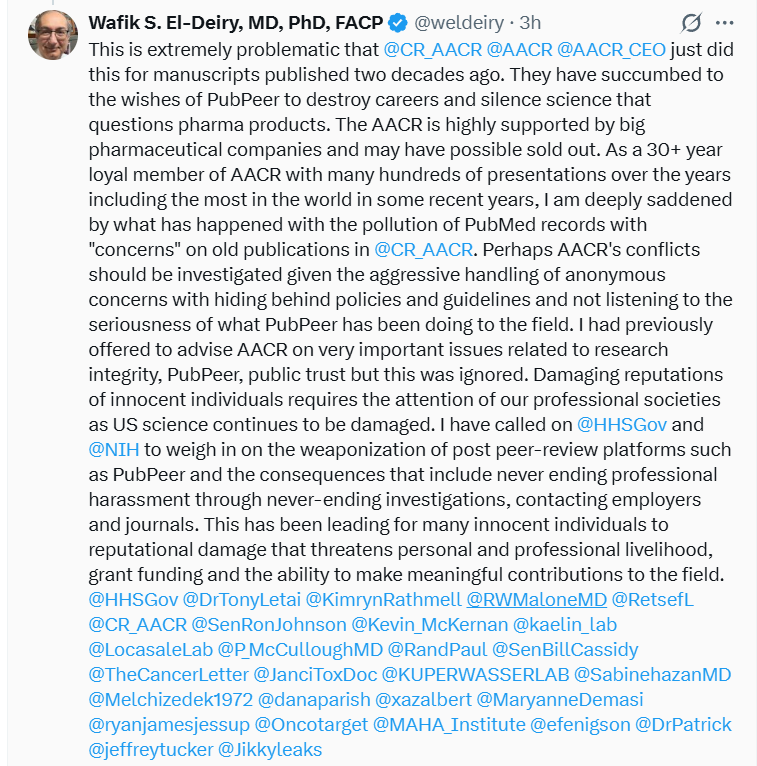
Incredible. Truth is, the only person who should be investigated is El-Dery himself. Look at other two examples of Dr El-Deiry’s genius, flagged by Elisabeth Bik:
Amriti R. Lulla, Michael J. Slifker, Yan Zhou, Avital Lev, Margret B. Einarson, David T. Dicker, Wafik S. El-Deiry* miR-6883 Family miRNAs Target CDK4/6 to Induce G Phase Cell-Cycle Arrest in Colon Cancer Cells Cancer Research (2017) doi: 10.1158/0008-5472.can-17-1767
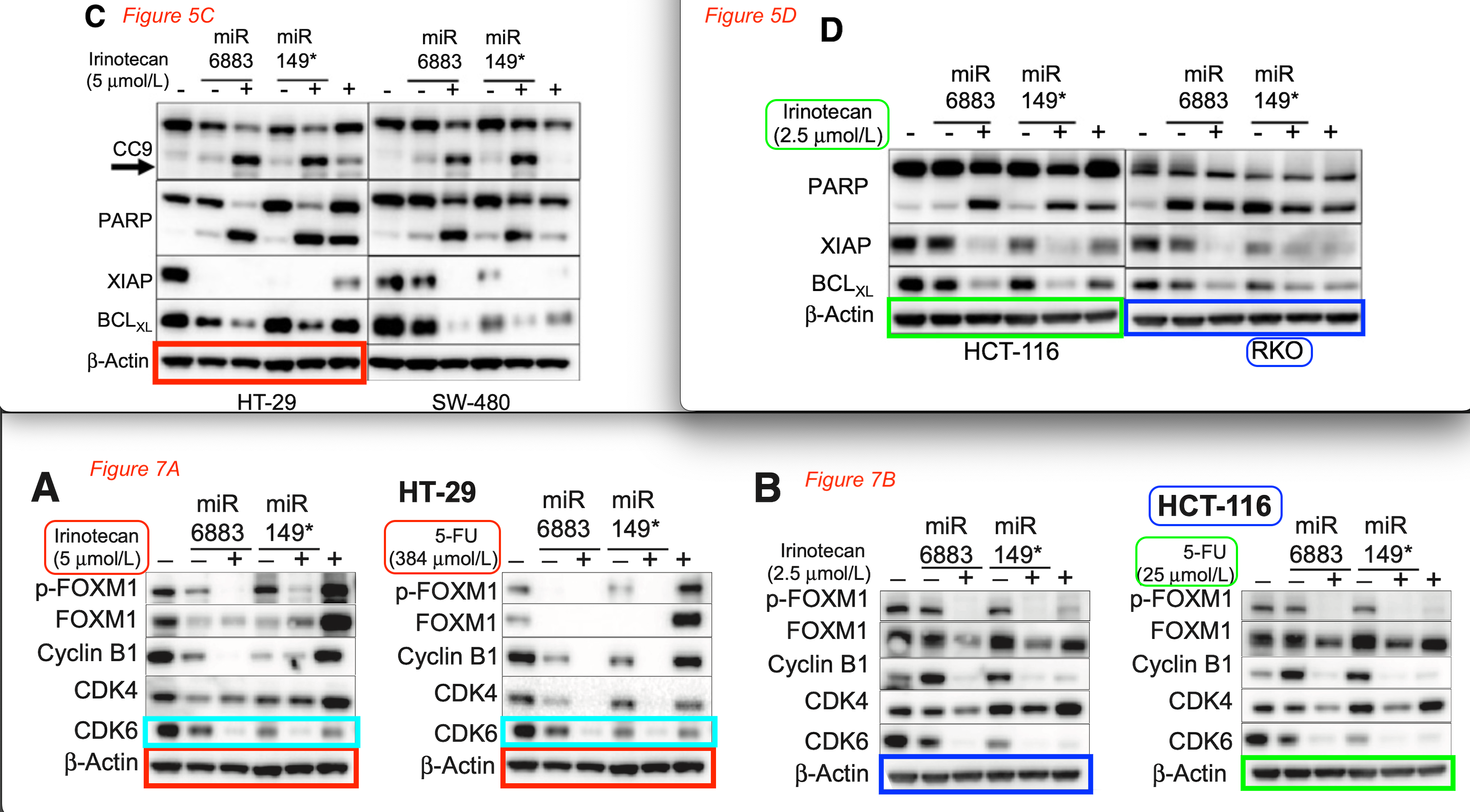
Just two authors on this paper:
Zhaoyu Jin , Wafik S. El-Deiry* Distinct signaling pathways in TRAIL- versus tumor necrosis factor-induced apoptosis Molecular and Cellular Biology (2006) doi: 10.1128/mcb.00257-06

There are 90 such papers on PubPeer for El-Deiry, you decide who should be investigated.
Ting C. Zhao
Please meet surgery professor at WAMS Ting C. Zhao, recipient of NIH grants worth $3 million in the last ten years, who sports 20 papers flagged on PubPeer, the vast majority in collaboration with Shougang Zhuang, encountered above. Dr Zhao’s medical specialty is cardiology, his lab focuses on the molecular mechanisms and translational implications of cardiovascular pathology, metabolic disorders, and tissue repair.
In the paper below from Zhao’s lab, overlapping CLSM micrographs describe different samples, and echocardiographic pictures were reused from Zhao’s earlier works where they describe completely different conditions. Shougang Zhuang is also on board:
Jianfeng Du, Ling Zhang, Zhengke Wang, Naohiro Yano, Yu Tina Zhao, Lei Wei, Patrycja Dubielecka-Szczerba, Paul Y. Liu, Shougang Zhuang, Gangjian Qin, Ting C. Zhao* Exendin-4 induces myocardial protection through MKK3 and Akt-1 in infarcted hearts AJP Cell Physiology (2016) doi: 10.1152/ajpcell.00194.2015
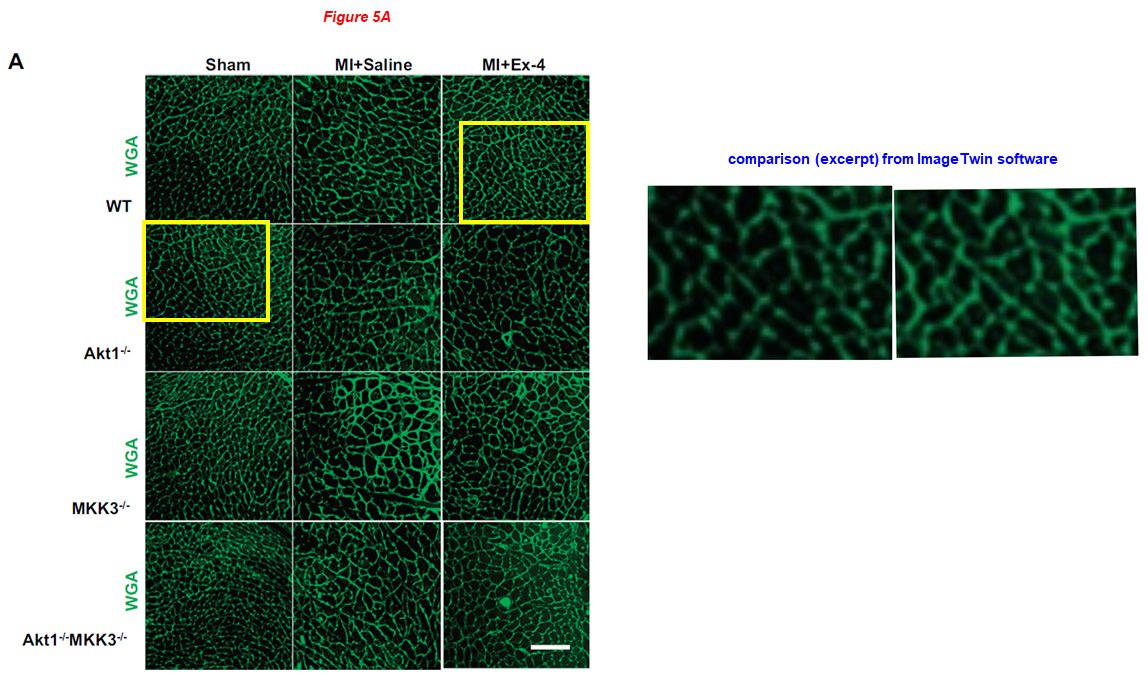
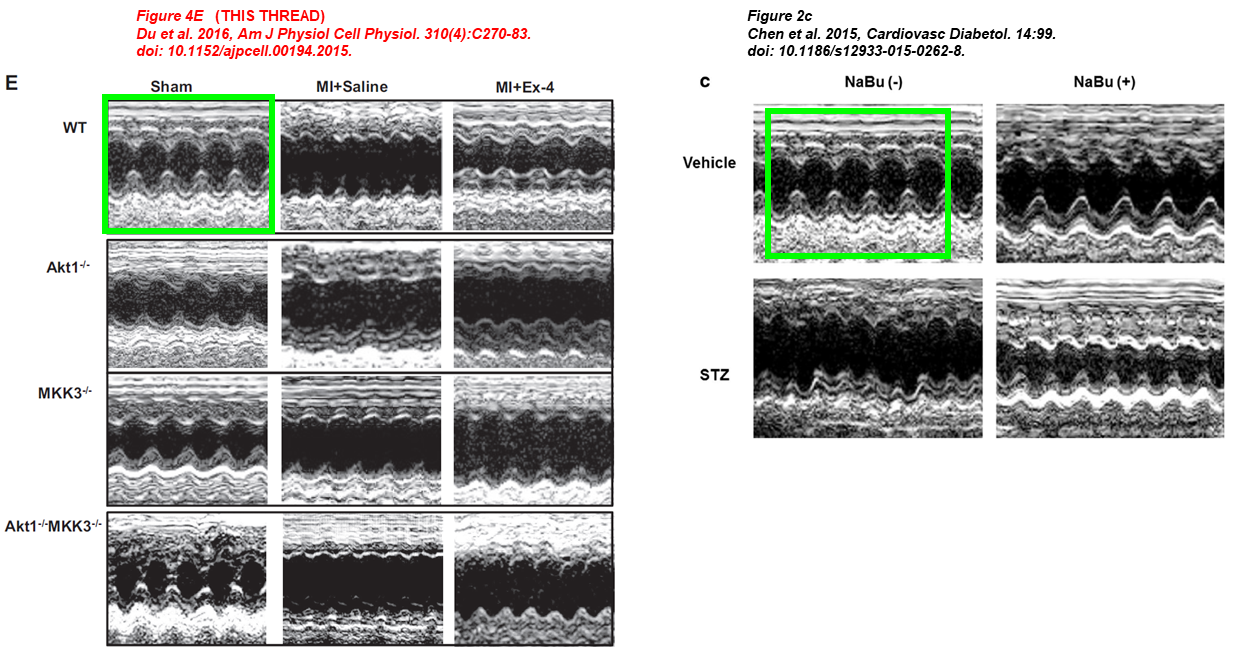
Youfang Chen, Jianfeng Du, Yu Tina Zhao, Ling Zhang, Guorong Lv, Shougang Zhuang, Gangjian Qin, Ting C Zhao Histone deacetylase (HDAC) inhibition improves myocardial function and prevents cardiac remodeling in diabetic mice Cardiovascular Diabetology (2015) doi: 10.1186/s12933-015-0262-8
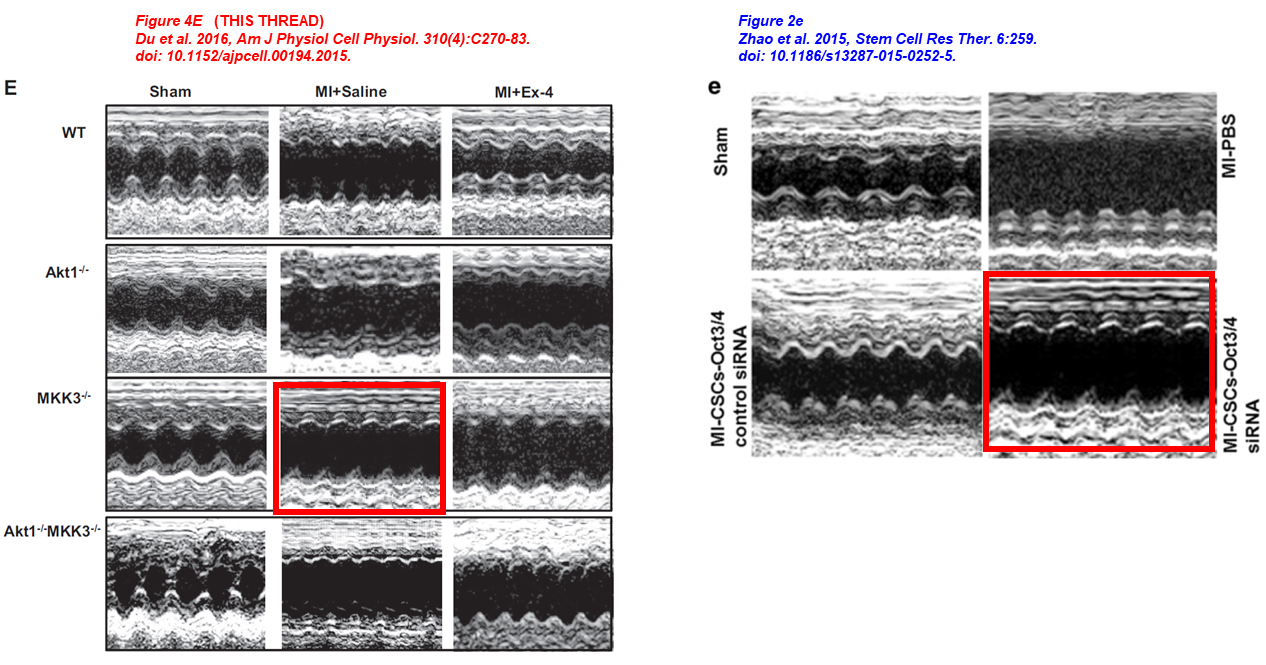
Yu Tina Zhao , Jianfeng Du , Youfang Chen , Yaoliang Tang , Gangjian Qin , Guorong Lv , Shougang Zhuang , Ting C. Zhao Inhibition of Oct 3/4 mitigates the cardiac progenitor-derived myocardial repair in infarcted myocardium Stem Cell Research & Therapy (2015) doi: 10.1186/s13287-015-0252-5
In the next article, Zhao is listed last among the authors, but the lead is Naohiro Yano, a guy currently employed as research associate at the Providence VA Medical Center. This paper has on board also Y. Eugene Chin, who worked for twelve years at Brown University but is now back in China. Eugene Chin has 30 papers on PubPeer and 2 retractions as of today, the latest materialized a few weeks ago. Look at this Yano-Chin-Zhao production:
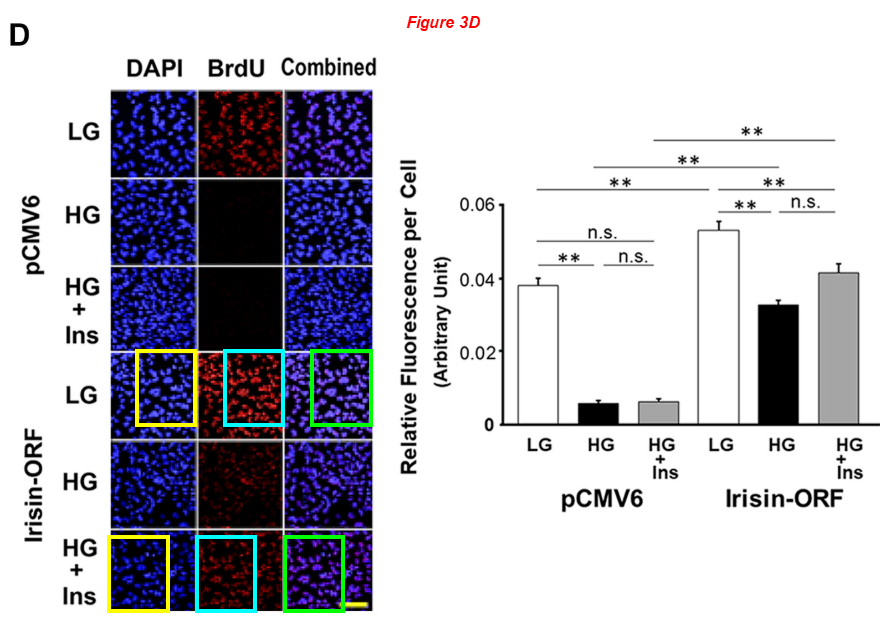
Naohiro Yano*, Ling Zhang, Dennis Wei, Patrycja M. Dubielecka, Lei Wei, Shougang Zhuang, Ping Zhu, Gangjian Qin, Paul Y. Liu, Y. Eugene Chin, Ting C. Zhao Irisin counteracts high glucose and fatty acid-induced cytotoxicity by preserving the AMPK-insulin receptor signaling axis in C2C12 myoblasts American journal of physiology. Endocrinology and metabolism (2020) doi: 10.1152/ajpendo.00219.2019

Here are two in-house collaborations between Ting Zhao and Patrycja Dubielecka-Szczerba, associate professor of medicine at WAMS and director of translational hematology at the Rhode Island Hospital, who in turn is on board in the paper above:
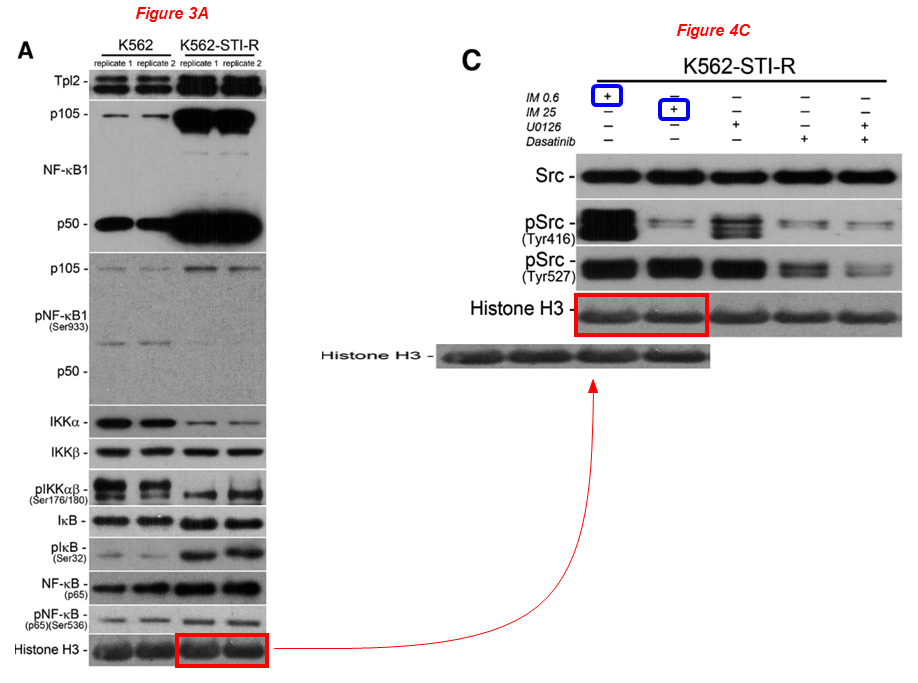
Figure 3A and Figure 4C

Figure 3D and Figure 7C
Now a splendid collaboration between Zhao and Gangjian Qin, medicine professor now back in China, at SUSTech in Shenzhen, who worked for 25 years at many US universities like Illinois, Tufts, Northwestern and Alabama. This is a production carrying the Northwestern and Alabama affiliations for Qin. The same rescaled/mirrored blots describe IKKβ and actin, or actin and LaminA:
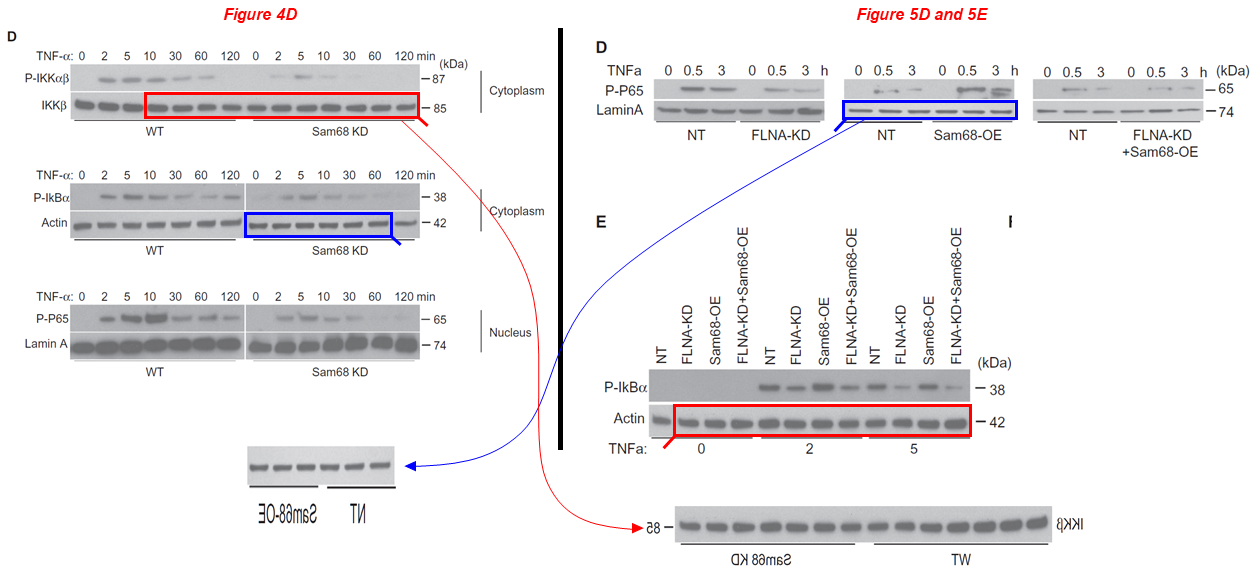
Shuling Han, Shiyue Xu, Junlan Zhou, Aijun Qiao, Chan Boriboun, Wenxia Ma, Huadong Li , Dauren Biyashev, Liu Yang , Eric Zhang, Qinghua Liu, Shayi Jiang, Ting C. Zhao, Prasanna Krishnamurthy, Chunxiang Zhang, Stéphane Richard, Hongyu Qiu, Jianyi Zhang, Gangjian Qin* Sam68 impedes the recovery of arterial injury by augmenting inflammatory response Journal of Molecular and Cellular Cardiology (2019) doi: 10.1016/j.yjmcc.2019.10.003
I’ll show you just one more paper by Ting Zhao, an almost 20-year-old work with Naohiro Yano again as first author, with former Brown pediadrics professor James F. Padbury (now at UCSF), and a certain Yi-Tang Tseng, whom I wasn’t able to locate. Tseng is the corresponding author of this nonsense:
Naohiro Yano, Daisuke Suzuki, Masayuki Endoh, Ting C. Zhao, James F. Padbury, Yi-Tang Tseng* A novel phosphoinositide 3-kinase-dependent pathway for angiotensin II/AT-1 receptor-mediated induction of collagen synthesis in MES-13 mesangial cells Journal of Biological Chemistry (2007) doi: 10.1074/jbc.m610537200
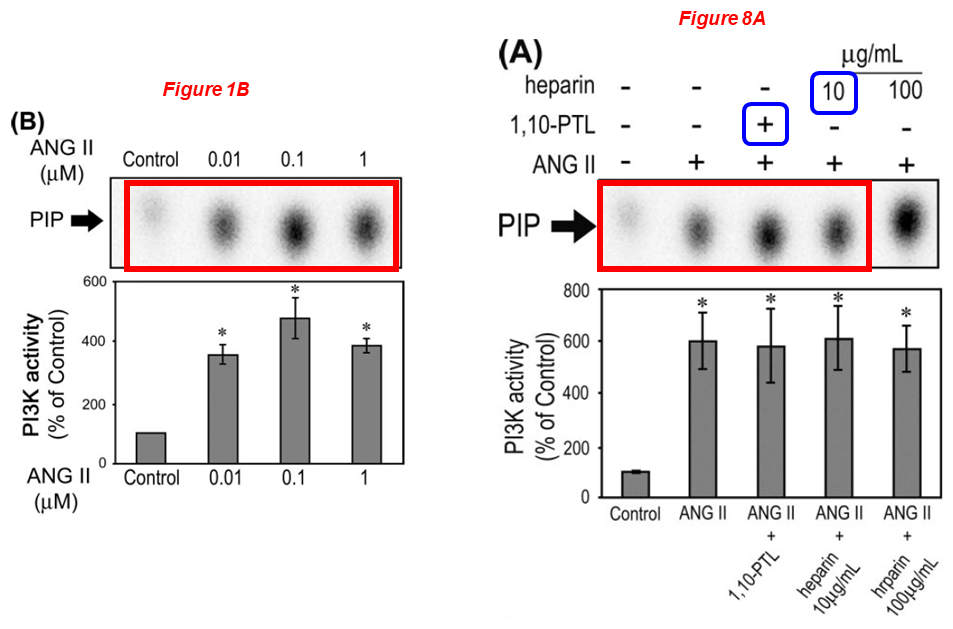
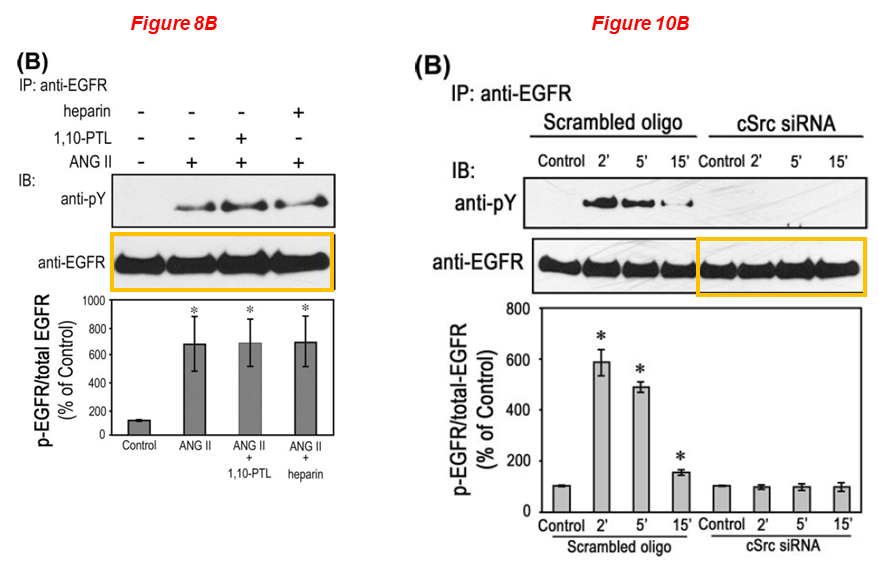
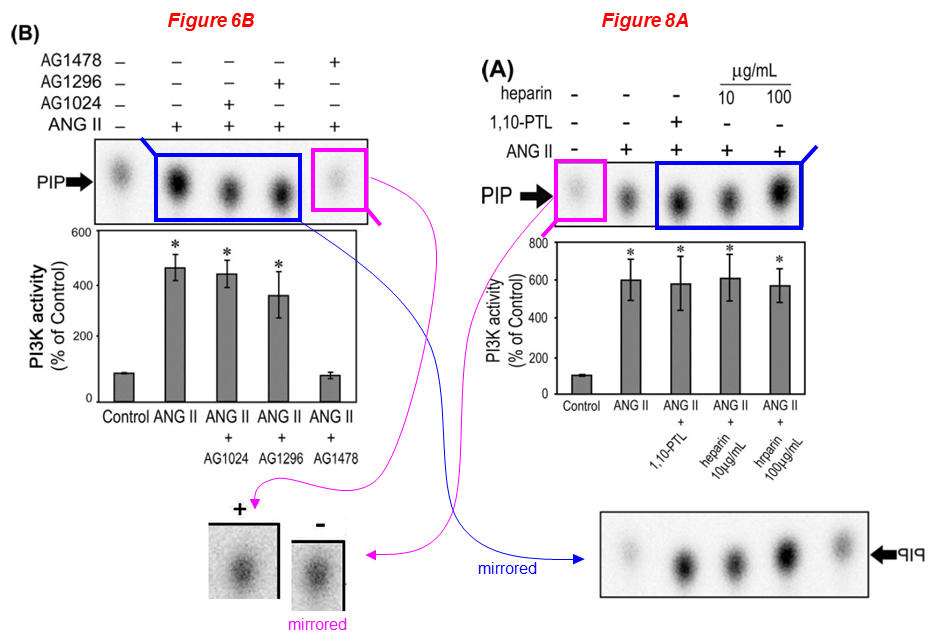
Lei Wei
Please meet Lei Wei, associate professor of orthopaedics at WAMS, whose research aims understanding the mechanism of cartilage development to cartilage degeneration. Dr Wei received about $2 million in NIH grants over the last ten years, and sports 20 papers on PubPeer. Look at this gem:
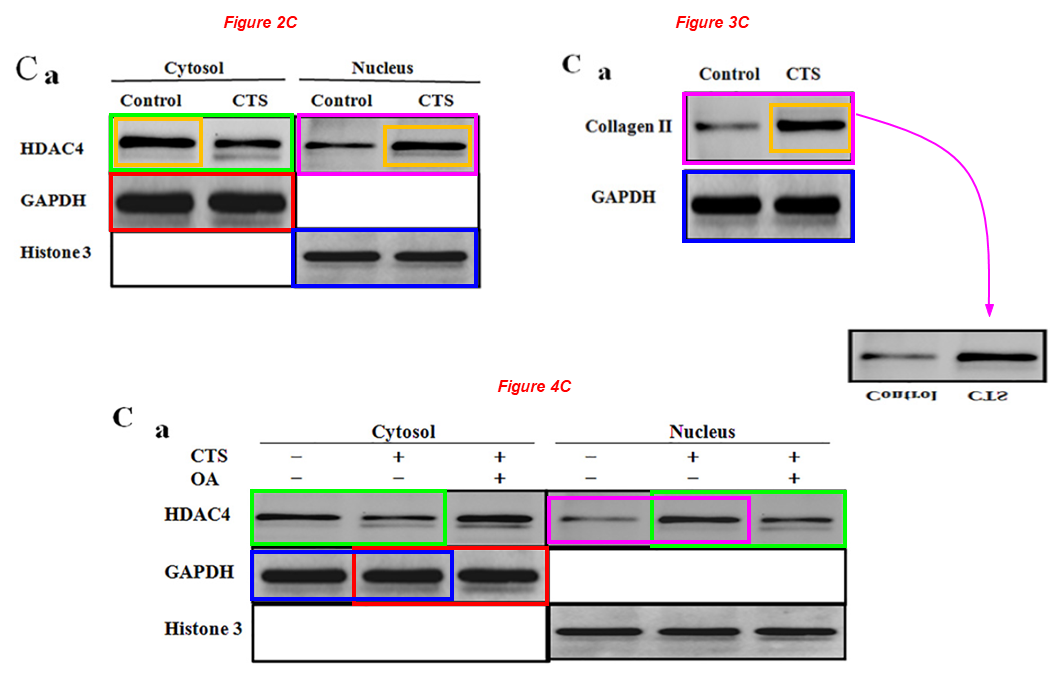
Chongwei Chen, Xiaochun Wei, Shaowei Wang, Qiang Jiao, Yang Zhang, Guoqing Du, Xiaohu Wang, Fangyuan Wei, Jianzhong Zhang, Lei Wei* Compression regulates gene expression of chondrocytes through HDAC4 nuclear relocation via PP2A-dependent HDAC4 dephosphorylation Biochimica et Biophysica Acta (2016) doi: 10.1016/j.bbamcr.2016.04.018
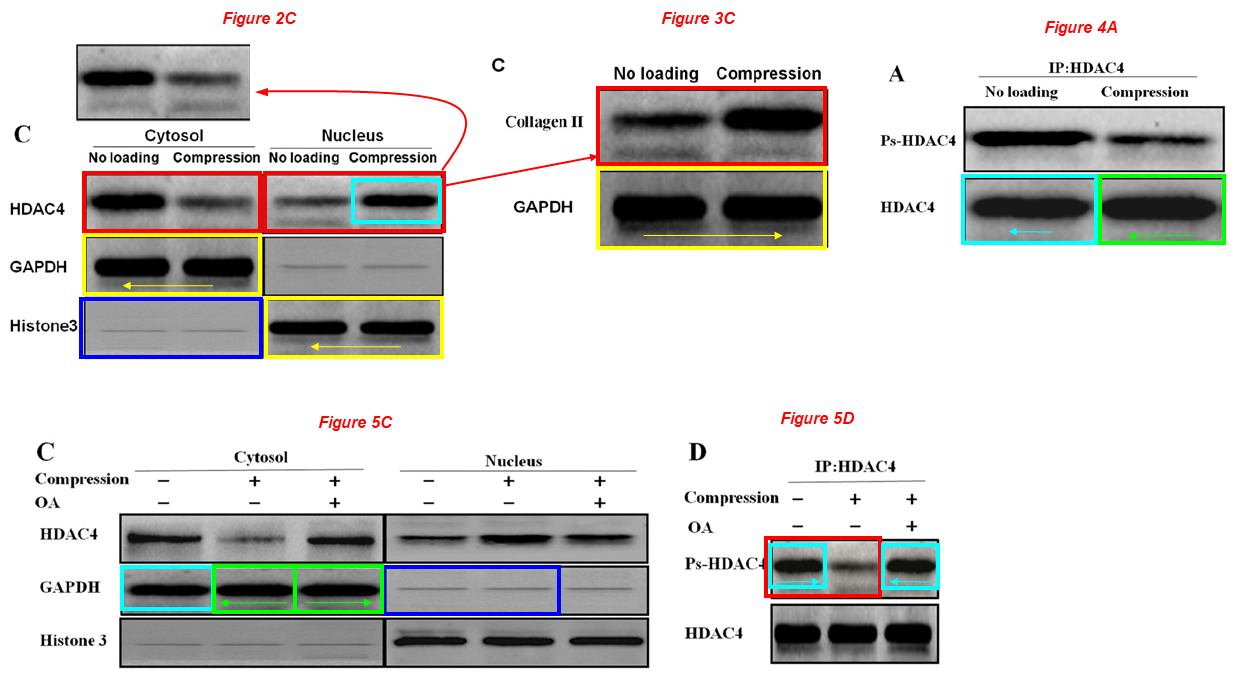
The other HDAC4 band of Figure 4A also apears twice (and mirrored) in Figure 5C as GAPDH (green boxes).
Two GAPDH bands of Figure 2C appear in the same figure as Histone 3, but have been compressed vertically (yellow boxes). The same two bands reappear mirrored in figure 3C.
Two Histone 3 bands of Figure 2C reappear as GAPDH bands in Figure 5C (blue boxes).
Unfortunately for the authors, the next study was plublished at PLOS One, a journal that usually shows little tolerance for blatant fraud. One lab, one style, let’s hope PLOS One will retract this:
Chongwei Chen, Xiaochun Wei, Zhi Lv , Xiaojuan Sun, Shaowei Wang, Yang Zhang, Qiang Jiao, Xiaohu Wang, Yongping Li, Lei Wei* Cyclic Equibiaxial Tensile Strain Alters Gene Expression of Chondrocytes via Histone Deacetylase 4 Shuttling PLOS One (2016) doi: 10.1371/journal.pone.0154951
Regarding the next study, are we really sure that ipriflavone attenuates cartilage degeneration? I wouldn’t necessarily suggest to set up any clinical trial on that:
Li Guo, Xiaochun Wei, Zhiwei Zhang, Xiaojian Wang, Chunli Wang, Pengcui Li, Chunfang Wang, Lei Wei* Ipriflavone attenuates the degeneration of cartilage by blocking the Indian hedgehog pathway Arthritis Research & Therapy (2019) doi: 10.1186/s13075-019-1895-x
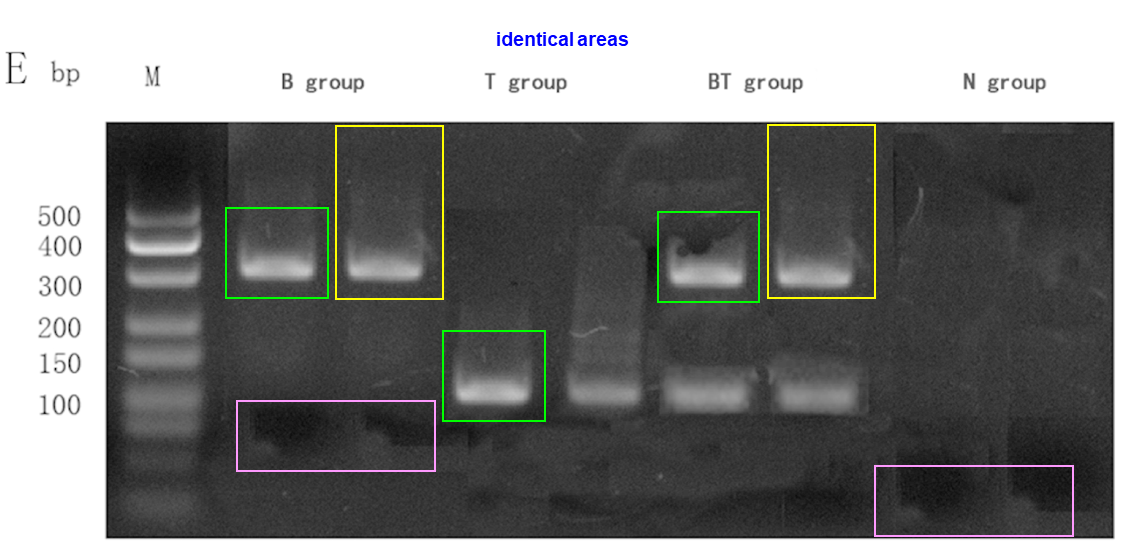
Here a wonderful collaboration between Lei Wei and some Chinese friends at Kunming Medical University, this time it’s about a fake DNA gel in Figure 6:
Xin Wang, Yanlin Li*, Rui Han, Chuan He, Guoliang Wang, Jianwei Wang, Jiali Zheng, Ming Pei, Mei Pei, Lei Wei Demineralized bone matrix combined bone marrow mesenchymal stem cells, bone morphogenetic protein-2 and transforming growth factor-β3 gene promoted pig cartilage defect repair PLOS One (2014) doi: 10.1371/journal.pone.0116061
Qian Chen
Please meet Qian Chen, Michael G. Ehrlich Endowed Chair in Orthopaedic Research, professor of Medical Science, Vice Chair for Research in the Department of Orthopaedics at the Warren Alpert Medical School, and director of Center of Biomedical Research Excellence in Skeletal Health and Repair in Rhode Island Hospital. Dr Chen received $12 million in NIH grants over the past ten years, and sports 10 papers on PubPeer.
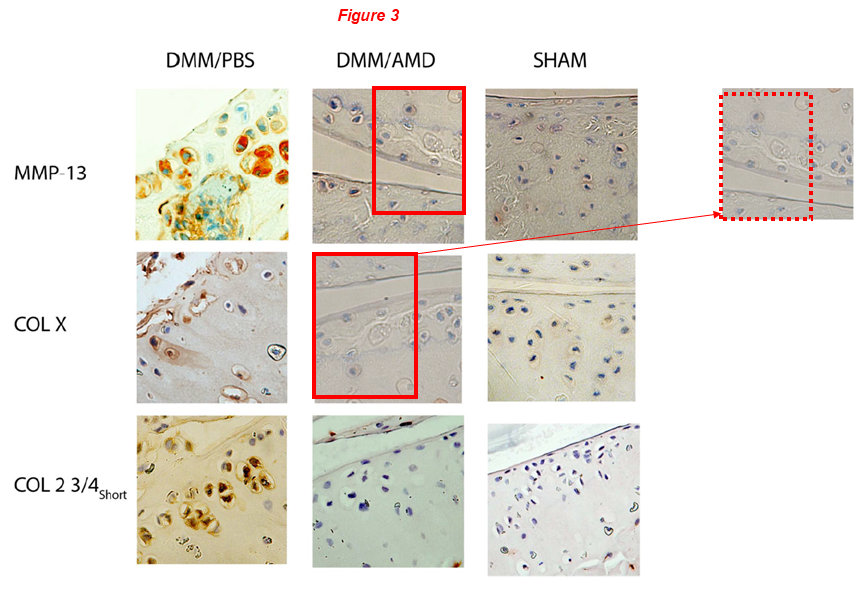
Let’s start with this one, the lead author is Lei Wei (discussed above), while Chen is a middle author. Two micrographs in Figure 3 overlap, but represent different immunostainings for MMP-13 and COL-X (red boxes):
Nathan P. Thomas, Pengcui Li, Braden C. Fleming, Qian Chen, Xiaochun Wei, Pan Xiao-Hua, Gang Li , Lei Wei* Attenuation of cartilage pathogenesis in post-traumatic osteoarthritis (PTOA) in mice by blocking the stromal derived factor 1 receptor (CXCR4) with the specific inhibitor, AMD3100 Journal of Orthopaedic Research (2015) doi: 10.1002/jor.22862
Additionally, two micrographs were taken from prior Lei’s work which sounds like a title of a novel (“Disrupting the Indian hedgehog“):
Jingming Zhou , Qian Chen, Beate Lanske , Braden C Fleming , Richard Terek , Xiaochun Wei , Ge Zhang , Shaowei Wang , Kai Li , Lei Wei Disrupting the Indian hedgehog signaling pathway in vivo attenuates surgically induced osteoarthritis progression in Col2a1-CreER T2 ; Ihhfl/fl mice Arthritis Research & Therapy (2014) doi: 10.1186/ar4437
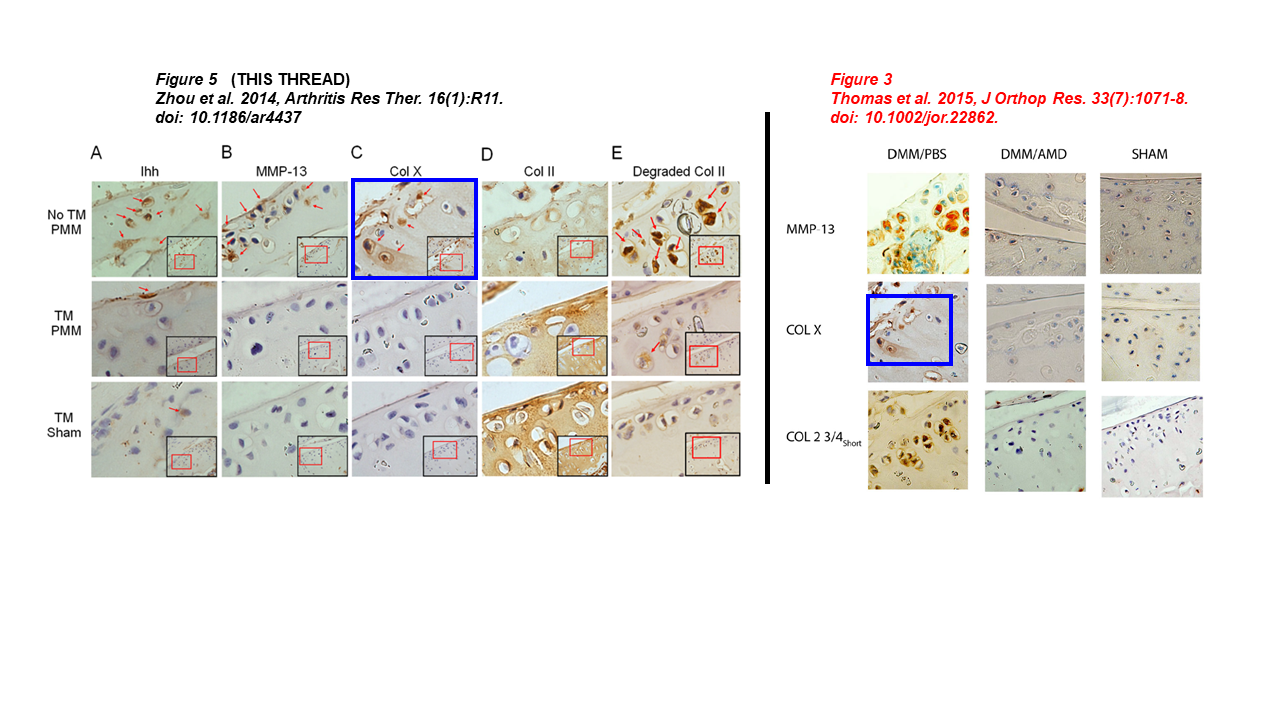
Dr Chen commented at PubPeer about the first image above:
“I just want to point out that the two images under question in #1 are different immunohistochemistry images, one reacted with an antibody against MMP-13 and the other reacted with an antibody against COLX. The antibody staining patterns and intensities are clearly different, although they may be from adjacent sections in the same tissue block. […]“
This is wrong, the overlapping area is pixel-by-pixel-identical, therefore those pictures can’t represent consecutive sections. Either Dr Chen is trolling, or he is incompetent, and I don’t know what is worse. Dr Chen also added:
“I cannot comment on the specifics of the data in inquiry beyond what I described above, since the experiments were done in the laboratory of Dr. Lei Wei, the corresponding author of both papers. Dr. Lei Wei retired several years ago. Any comments by him may be delayed since Dr. Wei is undergoing cancer treatment.“
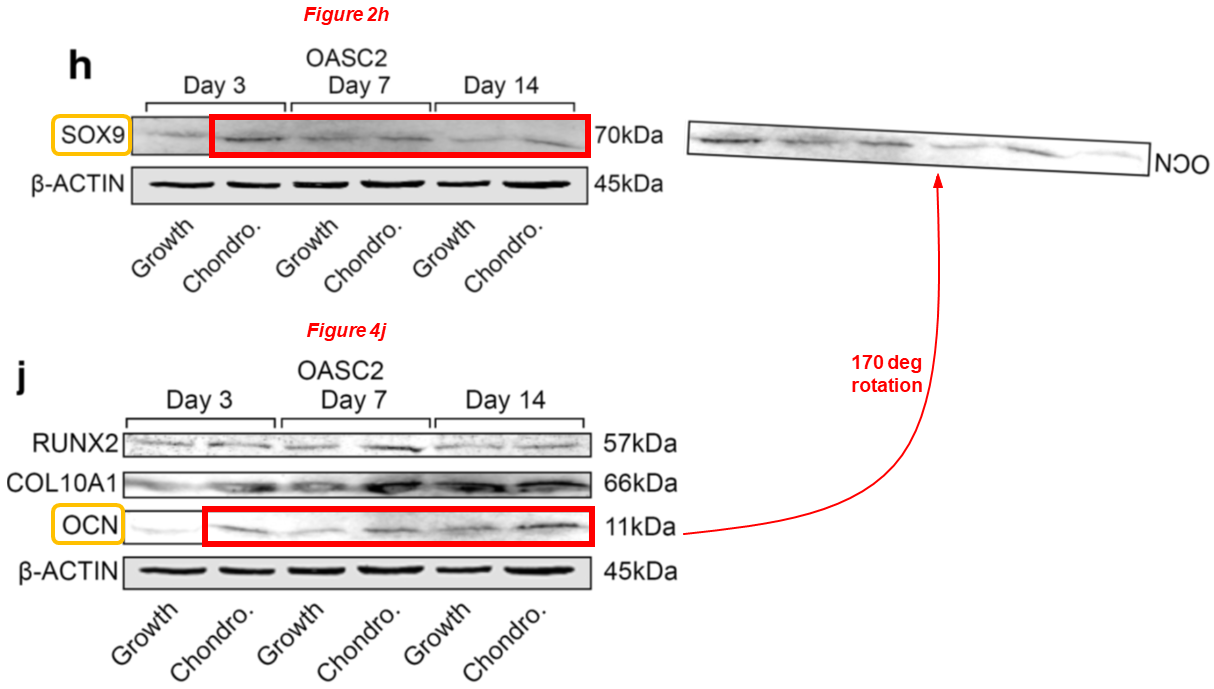
We wish Dr Lei a speedy recovery. Now let’s see some papers where Chen is the lead author. This one for example, where a duplicated and rotated blot describes SOX9 and OCN:
Nan Hu, Yun Gao, Chathuraka T. Jayasuriya, Wenguang Liu, Heng Du, Jing Ding, Meng Feng, Qian Chen* Chondrogenic induction of human osteoarthritic cartilage-derived mesenchymal stem cells activates mineralization and hypertrophic and osteogenic gene expression through a mechanomiR Arthritis Research & Therapy (2019) doi: 10.1186/s13075-019-1949-0
A major forgery in the next one, where a band of Figure 4 reappears rescaled in Figure 5, they supposedly describe different recombinant MATN3 versions probed with either anti-Flag or anti-V5.
Yue Zhang, Zheng-ke Wang, Jun-ming Luo, Katsuaki Kanbe, Qian Chen* Multiple functions of the von Willebrand Factor A domain in matrilins: secretion, assembly, and proteolysis Journal of Orthopaedic Surgery and Research (2008) doi: 10.1186/1749-799x-3-21
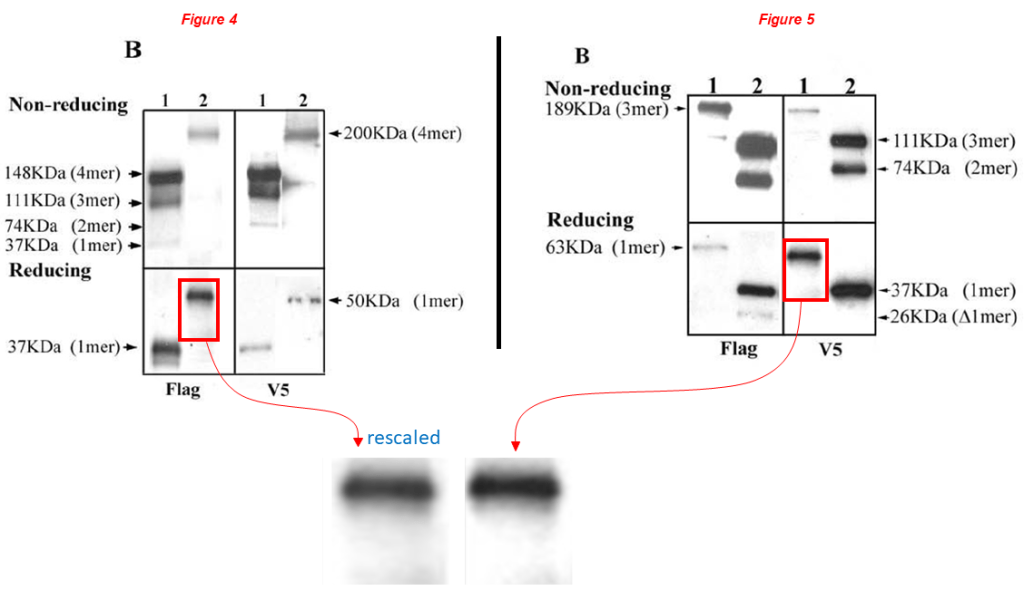
Dr Chen commented bitterly at PubPeer on the issues above (highlights mine):
“The images in the paper were the original data without digital manipulation. They include not only the main band but also other light bands and artifacts in the lane during exposure, which may serve as a signature for the lane. First, the two main bands are clearly different even after digital rescaling and manipulation by Aneurus inconstans. Second, other artifacts are different between the two lanes. For example, there is a small dot in the lower left of the indicated “Flag” lane in Figure 4. There is no such a dot at the corresponding position in the V5 lane in Figure 5. Unfortunately, such differences were cut off from the enlarged and manipulated image areas by Aneurus inconstans even though they should be included in the red box areas as indicated.“
Chen’s comment is quite blatantly deceptive. The absence of the dot beneath the band in Figure 5 isn’t an argument to refute the concern. The dot was just excluded by the authors with a convenient cropping. As a consequence, the dot wasn’t included in the enlarged comparison, as it is irrelevant to the duplicated area.
Chen with Lei Wei again:
Lei Wei*, Xiao-juan Sun, Zhengke Wang, Qian Chen CD95-induced osteoarthritic chondrocyte apoptosis and necrosis: dependency on p38 mitogen-activated protein kinase Arthritis Research & Therapy (2006) doi: 10.1186/ar1891
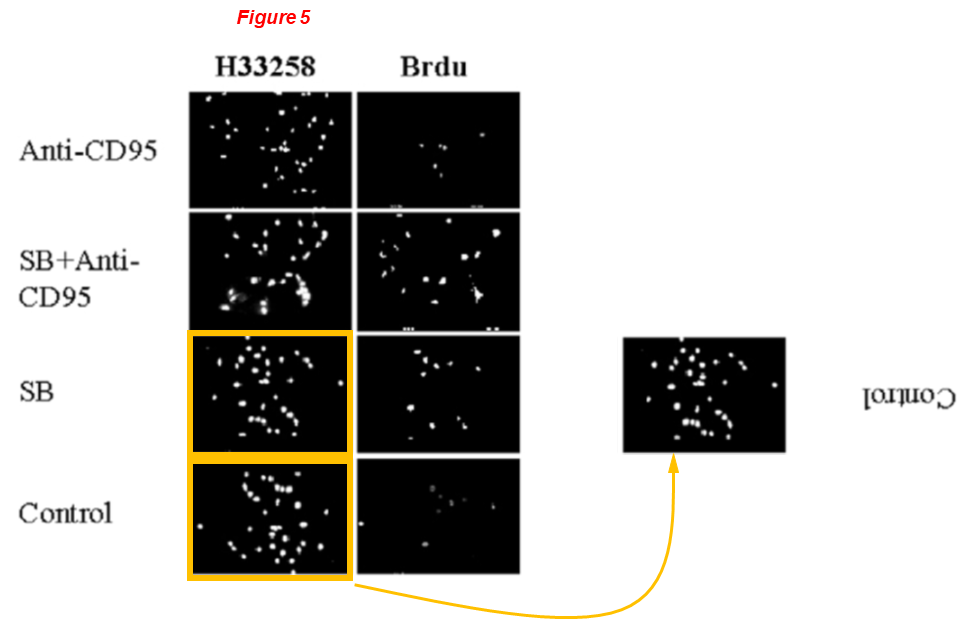
Figure 5: micrographs of H33258 staining for control cells and SB-treated cells are identical (yellow boxes), just rotated by 180 deg.
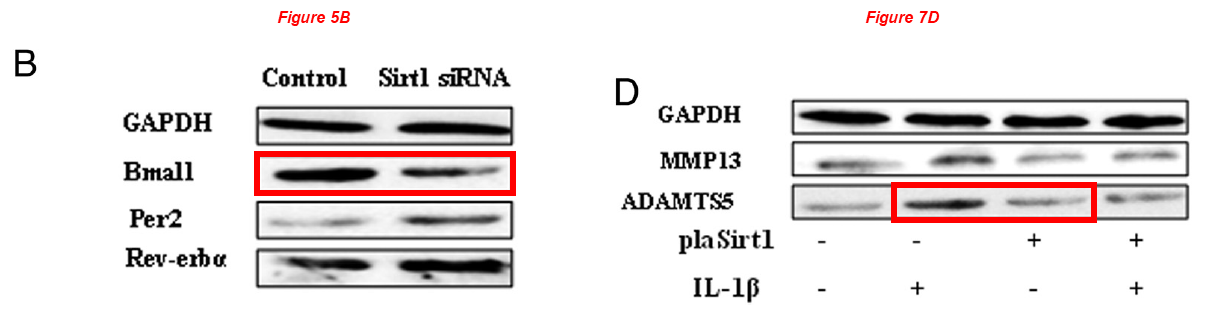
In the next one Chen and Chinese friends investigate the role of Bma11 in cartilage gene expression, but the Bma11 of Figure 5B describes ADAMTS5 in Figure 7D:
Wei Yang, Xiaomin Kang, Jiali Liu, Huixia Li, Zhengmin Ma, Xinxin Jin, Zhuang Qian, Tianping Xie, Na Qin, Dongxu Feng, Wenjie Pan, Qian Chen, Hongzhi Sun*, Shufang Wu* Clock Gene Bmal1 Modulates Human Cartilage Gene Expression by Crosstalk With Sirt1 Endocrinology (2016) doi: 10.1210/en.2015-2042
In the next paper Chen and his Korean friends investigate the beneficial effects of ginseng to prevent and treat muscle atrophy, same blots describe different things across papers:
Ga-Yeon Go, Ayoung Jo, Dong-Wan Seo, Woo-Young Kim, Yong Kee Kim, Eui-Young So, Qian Chen, Jong-Sun Kang, Gyu-Un Bae*, Sang-Jin Lee* Ginsenoside Rb1 and Rb2 upregulate Akt/mTOR signaling–mediated muscular hypertrophy and myoblast differentiation Journal of Ginseng Research (2020) doi: 10.1016/j.jgr.2019.01.007
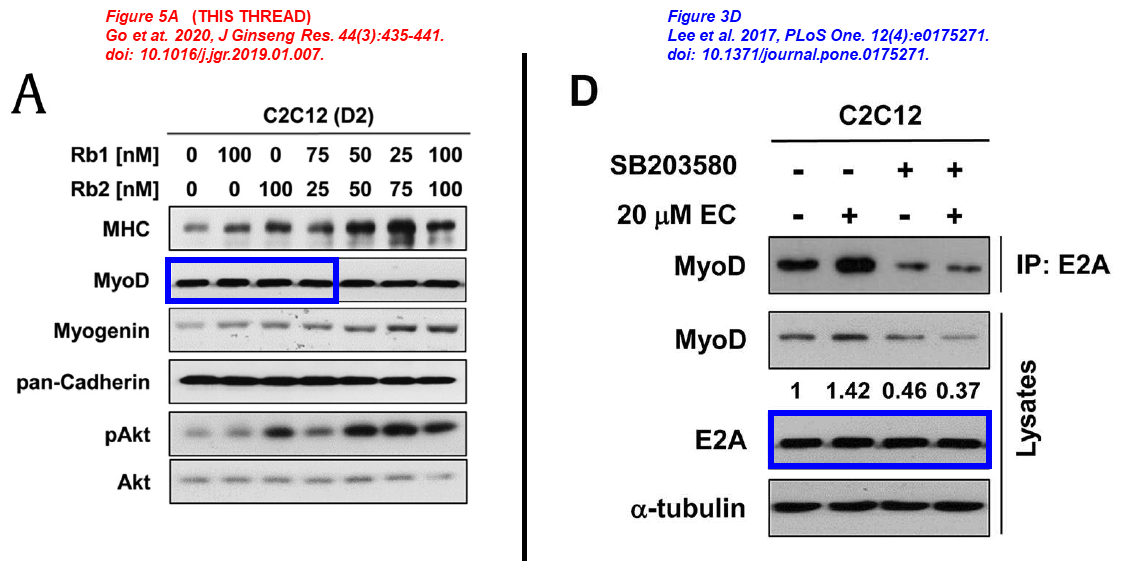
Sang-Jin Lee, Young-Eun Leem , Ga-Yeon Go , Younhee Choi , Yoo Jin Song , Insol Kim , Do Yoon Kim , Yong Kee Kim , Dong-Wan Seo , Jong-Sun Kang , Gyu-Un Bae Epicatechin elicits MyoD-dependent myoblast differentiation and myogenic conversion of fibroblasts PLOS One (2017) doi: 10.1371/journal.pone.0175271
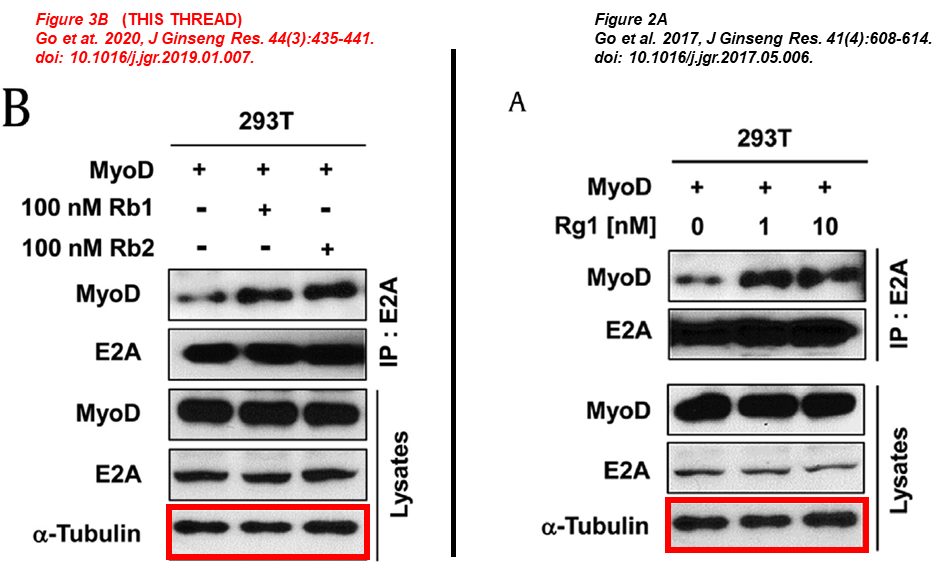
Ga-Yeon Go , Sang-Jin Lee, Ayoung Jo , Jaecheol Lee , Dong-Wan Seo , Jong-Sun Kang , Si-Kwan Kim , Su-Nam Kim , Yong Kee Kim, Gyu-Un Bae Ginsenoside Rg1 from Panax ginseng enhances myoblast differentiation and myotube growth Journal of Ginseng Research (2017) doi: 10.1016/j.jgr.2017.05.006
Rujun Gong
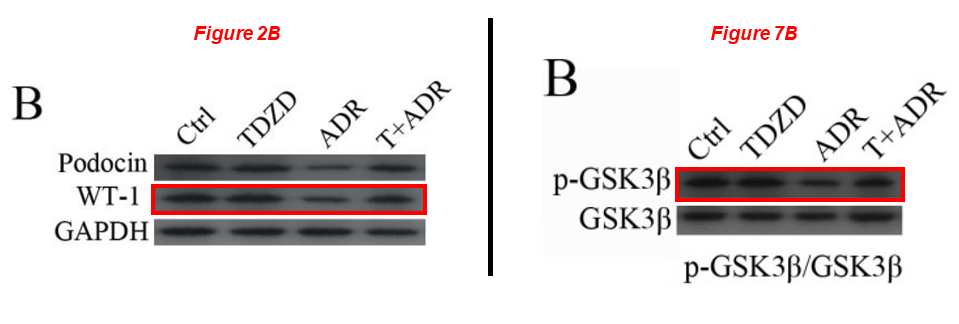
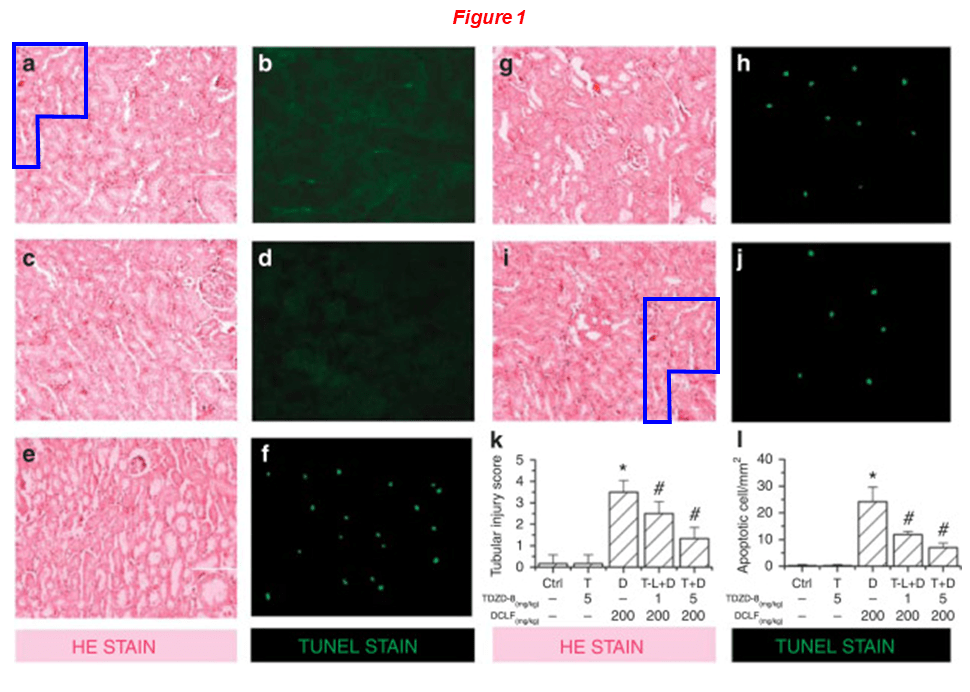
Please meet Rujun Gong, since 2018 professor of medicine and director of kidney research at the University of Toledo in Ohio, yet from 2004 to 2018 at Brown. Dr Gong’s research was funded with NIH grants worth $5 million over the last decade, he sports 10 papers on PubPeer. Here is one from Gong’s time at WAMS, showing that pharmacological targenting of GSK3β protects against podocytopathy and proteinuria. Of course, anything is possible when you fake a GSK3β blot:
Zhen Wang, Hui Bao, Yan Ge, Shougang Zhuang, Ai Peng, Rujun Gong* Pharmacological targeting of GSK3β confers protection against podocytopathy and proteinuria by desensitizing mitochondrial permeability transition British Journal of Pharmacology (2015) doi: 10.1111/bph.12952
Below, on the left a cluster of TKPT cells appears in two micrographs describing different samples (yellow boxes), but the surrounding cells are different. Moreover, micrographs 1a and 1i overlap (blue shapes).
Hao Bao, Yan Ge, Shougang Zhuang, Lance D. Dworkin, Zhihong Liu, Rujun Gong* Inhibition of glycogen synthase kinase-3β prevents NSAID-induced acute kidney injury Kidney International (2012) doi: 10.1038/ki.2011.443
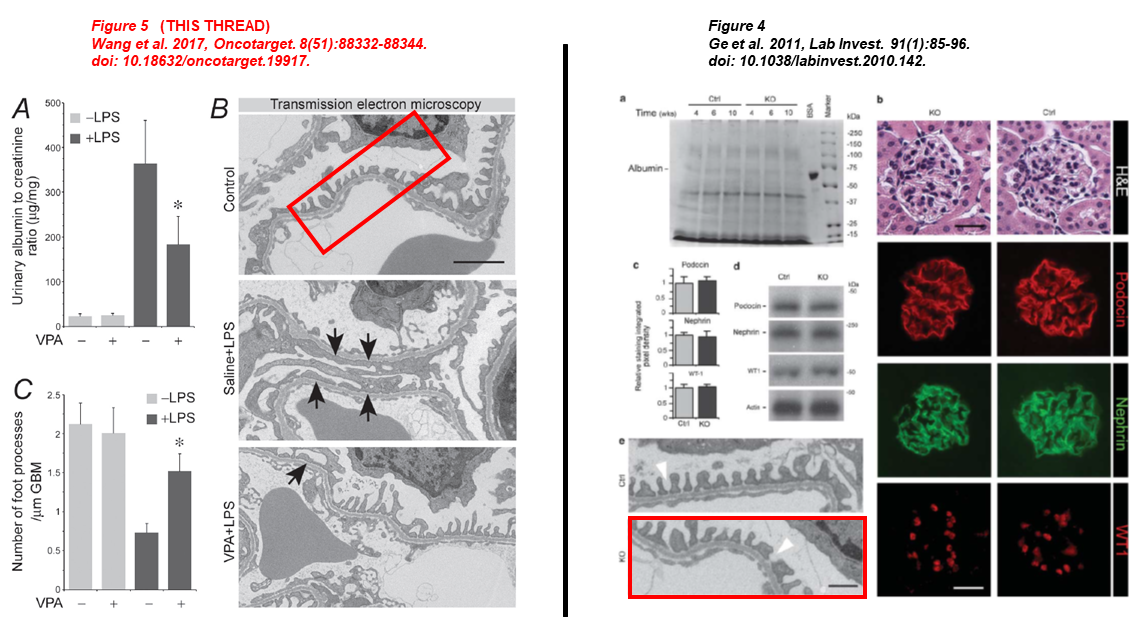
In the next one, the control TEM micrograph of Figure 5 was representing a GSK3β KO mouse six years before in Ge et al. 2011 (red boxes):
- Pei Wang, Sijie Zhou, Yan Ge, Minglei Lu, Zhangsuo Liu, Rujun Gong* Valproate hampers podocyte acquisition of immune phenotypes via intercepting the GSK3β facilitated NFkB activation Oncotarget (2017) doi: 10.18632/oncotarget.19917
- Yan Ge , Jin Si , Li Tian , Shougang Zhuang, Lance D Dworkin , Rujun Gong Conditional ablation of glycogen synthase kinase 3β in postnatal mouse kidney Laboratory Investigation (2011) doi: 10.1038/labinvest.2010.142
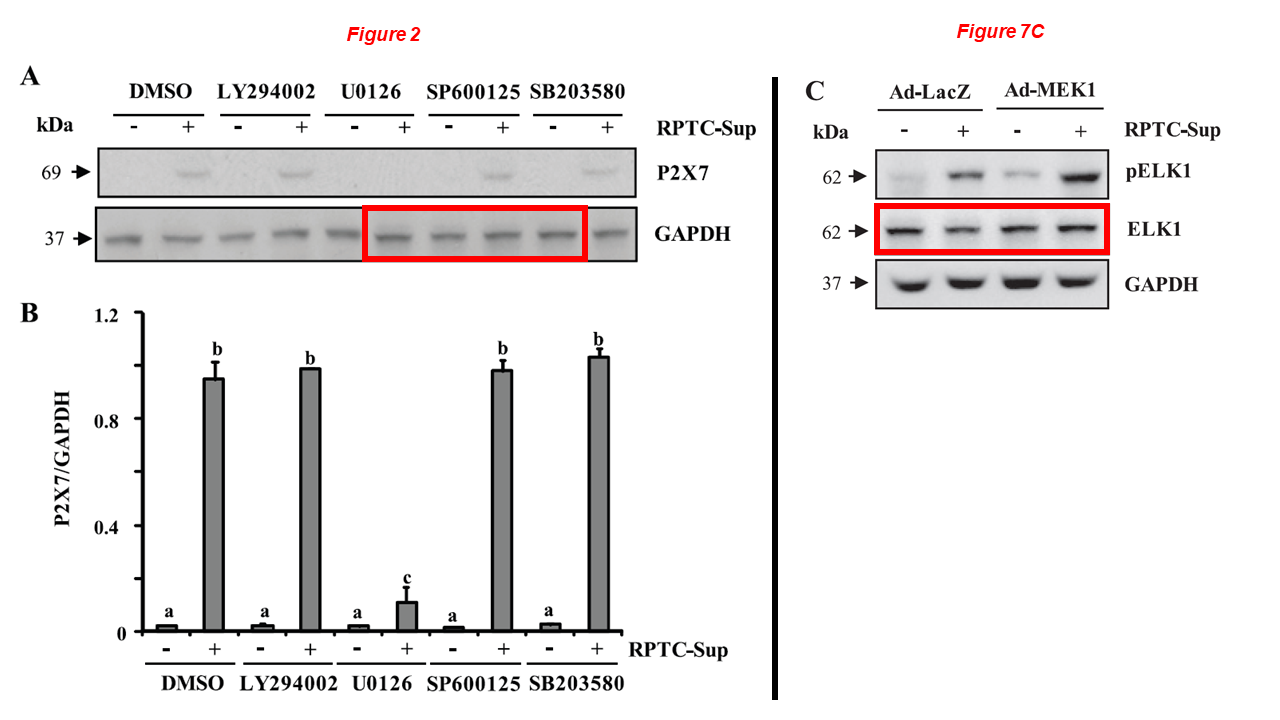
When he was still at Brown, Gong had fruitful collaborations with the legendary Shougang Zhuang (encountered above). Look at this beauty:
Murugavel Ponnusamy, Na Liu, Rujun Gong, Haidong Yan, Shougang Zhuang* ERK pathway mediates P2X7 expression and cell death in renal interstitial fibroblasts exposed to necrotic renal epithelial cells AJP Renal Physiology (2011) doi: 10.1152/ajprenal.00215.2011
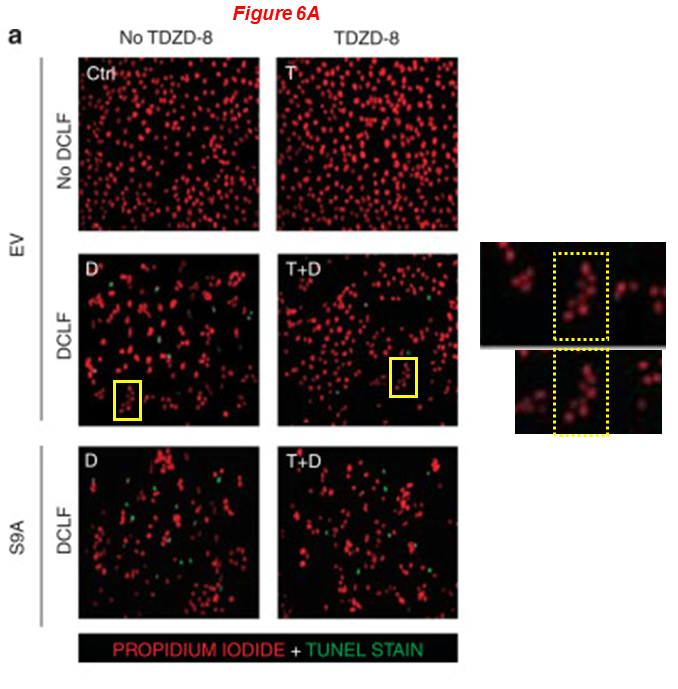
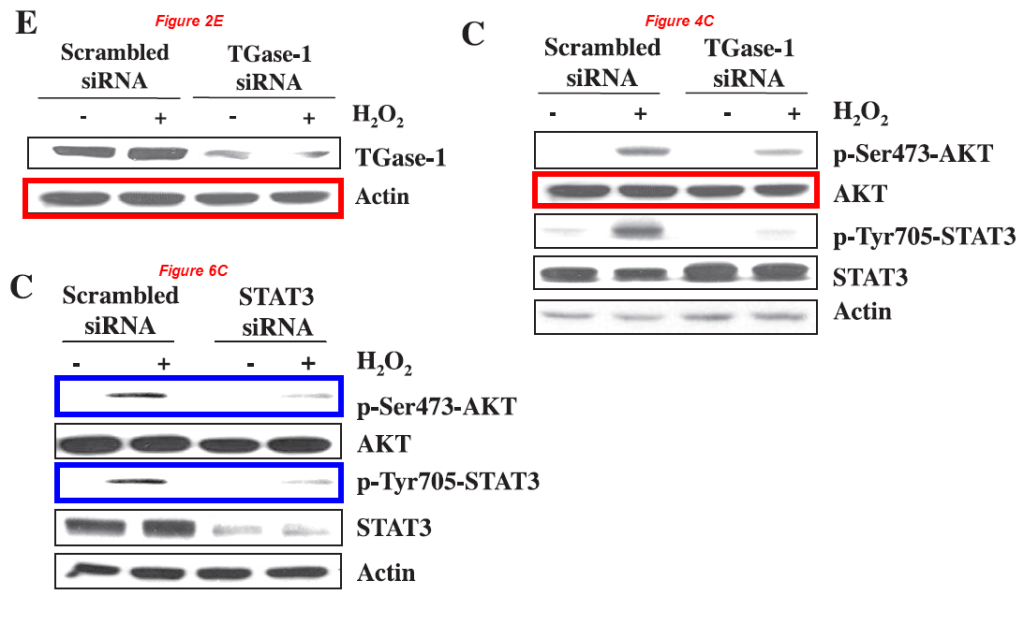
Yet another one with Gong and Zhuang, and Eugene Chin is also on board!
Murugavel Ponnusamy, Maoyin Pang, Pavan Kumar Annamaraju, Zhu Zhang, Rujun Gong, Y. Eugene Chin, Shougang Zhuang* Transglutaminase-1 protects renal epithelial cells from hydrogen peroxide-induced apoptosis through activation of STAT3 and AKT signaling pathways AJP Renal Physiology (2009) doi: 10.1152/ajprenal.00251.2009
Grant money
Here we have the math: $46 million of taxpayers money were awarded in the last ten years to the people discussed above, for carrying out research rife with questionable data. The steady erosion of trust in science is accompanied by even more tangible economic damage. No matter which research institution we look into, sadly we always manage to find dozens of researchers with a disturbing history of troubling data, even at top universities.
The depth of the rabbit hole
Several other Brown scholars were found having troubling records, I’ll briefly mention some of them below, they are mostly dead:
- The former chief of the Division of Gastroenterology at WAMS, the late Jack Wands (passed away in 2023), and his protégé Xiaoqun Dong have already had their fifteen minutes of fame in April 2025 Shorts, respective PubPeer records are here and here.
- There are also the emeritus professor José Behar and the late Piero Biancani (passed away in 2021), both used to collaborate with the still active Brown professor Weibiao Cao. These three guys have a dozen of very problematic articles (see here and here).
- Worth mentioning is also the late Devasis Chatterjee (passed away in 2021), former collaborator of Eugene Chin, employed at Brown from 2001 to 2018, here is his PubPeer record. Elisabeth Bik wrote about him in her guest post on For Better Science in 2018:
When gel bands go marching in, by Elisabeth Bik
This guest post by Elisabeth Bik will conclude the Fraud Triptych started by Smut Clyde. We shall meet a former mentee of Paul B Fisher, Sujit Bhutia, who is now busy fabricating data at his own lab at National Institute of Technology in India. Our next encounter will be with Fisher’s and Benjamin Bonavida’s past…
And last but not least, let us not forget Jack A. Elias, chief physician at Yale, Dean of Medicine at WAMS, and senior vice president for health affairs at Brown. Dr Elias sports several disconcerting papers on PubPeer, one got retracted in 2024 (not yet listed in Retraction Watch Database, but in August 2024 Shorts). Elias represents another example of how the fish stinks from the head down. On his Brown webpage, it is stated with emphasis and pride that “Dr. Elias is leading an ambitious $300 million effort to increase translational research, create new opportunities for students at all levels of training, and to train the next generation of physician-scientists. External research funding in the Division of Biology and Medicine has increased by greater than 118 percent during his tenure“.
Many data irregularities described in this article were identified with the help of ImageTwin.
- Adapted from Good Will Hunting, Miramax Films, 1997. ↩︎
- Csaba Szabo provided us with a statement about his colleaboration with Sellke, exceprts:
“By 2007, my group had been working on a hydrogen sulfide–releasing compound with
potential organ-protective effects. We had already demonstrated efficacy in a mouse model of
myocardial infarction through a separate collaboration. The next logical step was to test
whether these effects would scale to large animal models. Frank had exactly such a model. I
approached him to explore a collaboration. He agreed. The compound itself had been
developed by a biotech company, which also supported the project through a sponsored
research agreement. This was not simply an academic exercise; the stakes were higher. The
data generated in Frank’s lab were intended for, and ultimately included in regulatory
submissions, when the compound was scheduled to enter clinical trials.
The division of labor was clear. We provided the compound and provided the overall
experimental design. The actual experiments were conducted entirely in his laboratory. I was
not involved in the design or execution of certain ancillary assays, such as Western blot
analyses. My role included conceptual input and participation in manuscript preparation.
Before submission, I reviewed the data, including the figures with the blots. I saw nothing
that raised concern. Neither did the reviewers, the editors, or, presumably, the readers. Until
17 years later. Then, recently, a series of concerns emerged. Corrado identified repeated
image duplications and other irregularities across a large number of papers from his group.
My reaction was immediate: shock. I had no reason to suspect problems in Frank’s work. He
has long been regarded as a serious, committed investigator. Even now, I find it difficult to
reconcile these findings with the person I know.[…]
I have immediately contacted Frank and gave him some suggestions: Don’t try to explain
away what is unexplainable. Go back and track and check all the data generated around the
same time. Not only the Western blots, but everything else. Start a serious internal
investigation, or assist in every way in a University-initiated investigation, if it comes to that.
Find those who were responsible, and make sure the consequences are serious. And, be
proactive and initiate yourself the retractions of the papers.” ↩︎

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00






0 comments on “Mad School of Brown University”