England. A world superpower. Not just in everything else, but also in science, which is world-beating. English spitfire pluckiness destroyed the Huns in two world wars and in Brexit, but also in scientific performance, both impact-factor and money-wise. Brexit Britain will be free of all diseases, because whatever an English scientist does, conclusions remain not affected.
The secret to English science supremacy is not just the limitless capacity of certain English researchers to fabricate exciting results and phony cures, anyone can do that. It is the unique arrogance and corruption of English universities and of British health authorities who regularly decree all data fakeries to be quality science, outside statute of limitations, and verified independently by many citations, while telling whistleblowers to get lost.
It is a game only the crooks can win. And tax payers, charity donors and patients pay the price.
Not matter what you hear from Brexit, it’s not like England is not tolerant and welcoming. Clean of foreign detritus, free Britain can now freely choose what kind of foreigner it will let it. The foreigner must bring the right skill, like Amato Giaccia did, when he arrived to Oxford from Stanford in USA, trailed by a string of questionable research. Brexit and the COVID-19 pandemic showcased what the English ruling elites value most: defrauding and bullshitting the public. Science is just another coffer to plunder in this scam.
I previously celebrated the English science genius, with superior English minds like regenerative medicine enthusiast Martin Birchall, who took over Paolo Macchiarini‘s deadly technology to run some trachea transplants of his own. Birchall was acquitted in full by his own UCL, including for data irregularities, foreign scapegoats were assigned. Also Master of Birkbeck Sir David Latchman, CBE, was saved by UCL from unruly investigators whose reports were overruled by officials to determine no misconduct. Dame Kay Davies, DBE, was defended by her University of Oxford thanks to statute of limitations of 3 years for research misconduct charges, and by the publisher Oxford University Press where she as Editor-in-Chief retaliated against a fraud whistleblower. Paul Workman and Alan Ashworth, current and former director of ICR London, were both absolved in full, nothing at all worth retracting was found, while minor characters (not their protegees though) got the blame. Richard Marais of Cancer Research UK in Manchester weathered a research fraud scandal in his lab and a mountain of bullying accusations, he is still director of the institute.
Now, I shall celebrate more such talents, based on the sleuthing of Clare Francis. Cancer is cured, brain diseases vanquished, all thanks to the English scholars I want you to admire. Please raise, tuck in your shirt and comb your hair. The honour is all yours:

Nick Lemoine, FRCPath FMedSci
We shall start with Nicholas Lemoine. This cancer researcher is professor at the Queen Mary University of London and director of the Cancer Research UK Barts Centre. He is also an extremely high-ranking expert at the MRC (Medical Research Council), which means Dr Nick decides which biomedical researchers in UK gets funded and which don’t:
“He has served as Chair of the Clinical Training and Career Development Panel at the Medical Research Council, and as Vice-Chair of the MRC Stratified Medicine Expert Panel. He has previously served as Chair of the MRC Stem Cell Strategic Grant and Fellowship Panels, and has been a member of the MRC’s Molecular & Cellular Medicine Board.“
As medical director of NIHR Clinical Research Network, Dr Nick is now in charge of UK’s COVID-19 response:
So what kind of science are you, a humble British scientist seeking to eke out a meagre grant from MRC or NIHR, expected to deliver in order to impress Professor Lemoine? Well, you could study from his own papers, here is one:
Iman El-Hariry, Massimo Pignatelli, Nicholas R. Lemoine FGF-1 and FGF-2 regulate the expression of E-cadherin and catenins in pancreatic adenocarcinoma International Journal of Cancer (2001) DOI: 10.1002/ijc.1515
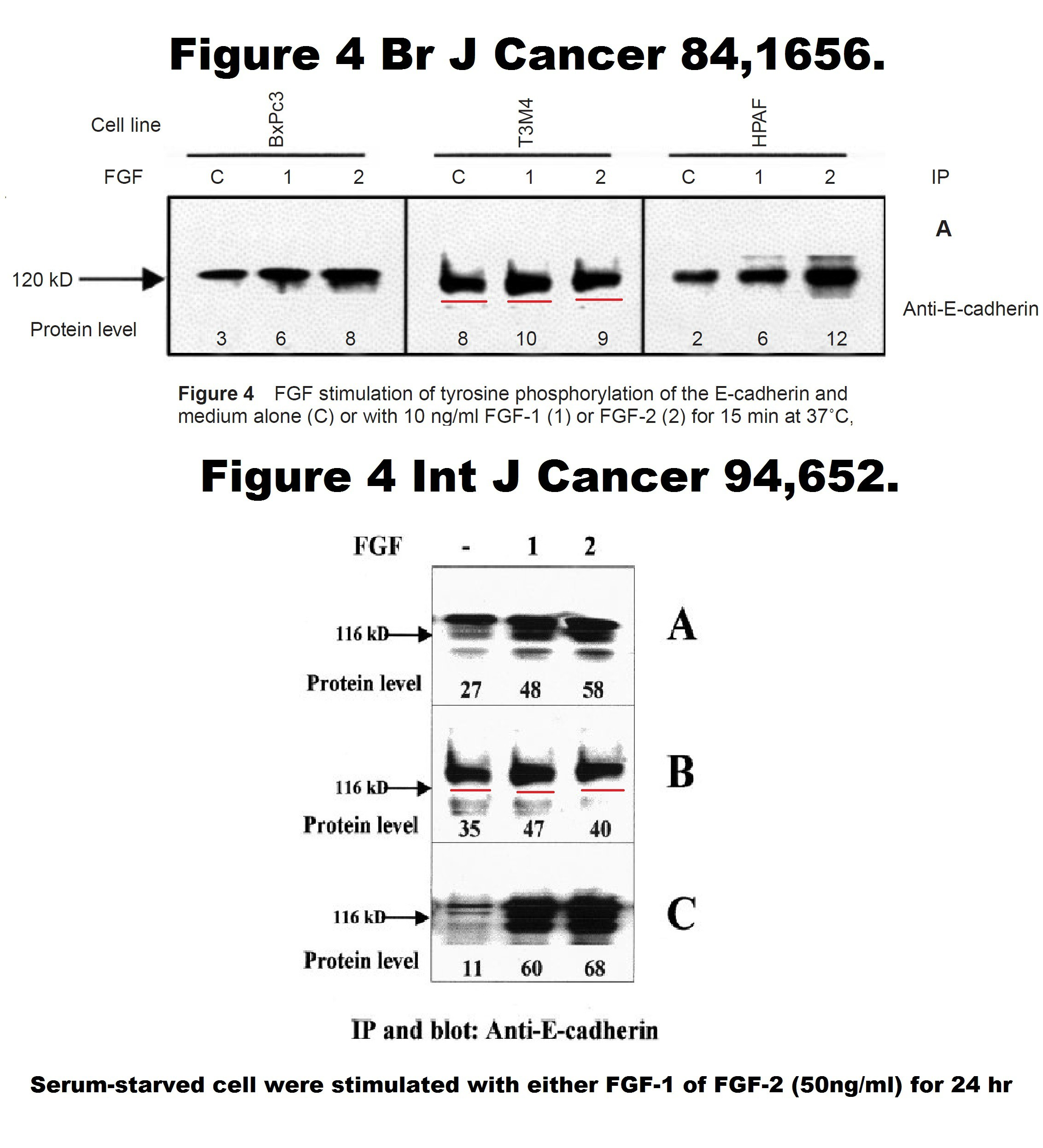
As you see, the Figure 4 is fake, it has 3 cloned bands. Which means, the conclusions are not affected. A correction was issued by the publisher Wiley in 2019:
“There is an error in the experimental conditions reported for the data presented in Figure 4 panel B. We apologize for these circumstances, which required clarification of the data.”
As it happens, Ashworth’s mentee and first author on a bunch of hilariously fake papers, Sarah Martin, is presently faculty member and Deputy Centre Lead at the Lemoine-ruled Bart’s and, this is really cruel: director of the graduate school. Now you see which qualifications you need to get employed at Barts, plus they probably sack PhD students for refusing to fake data there.
Maybe they use this paper by Limoine and his Barts colleagues to teach the students how to do cancer research properly:
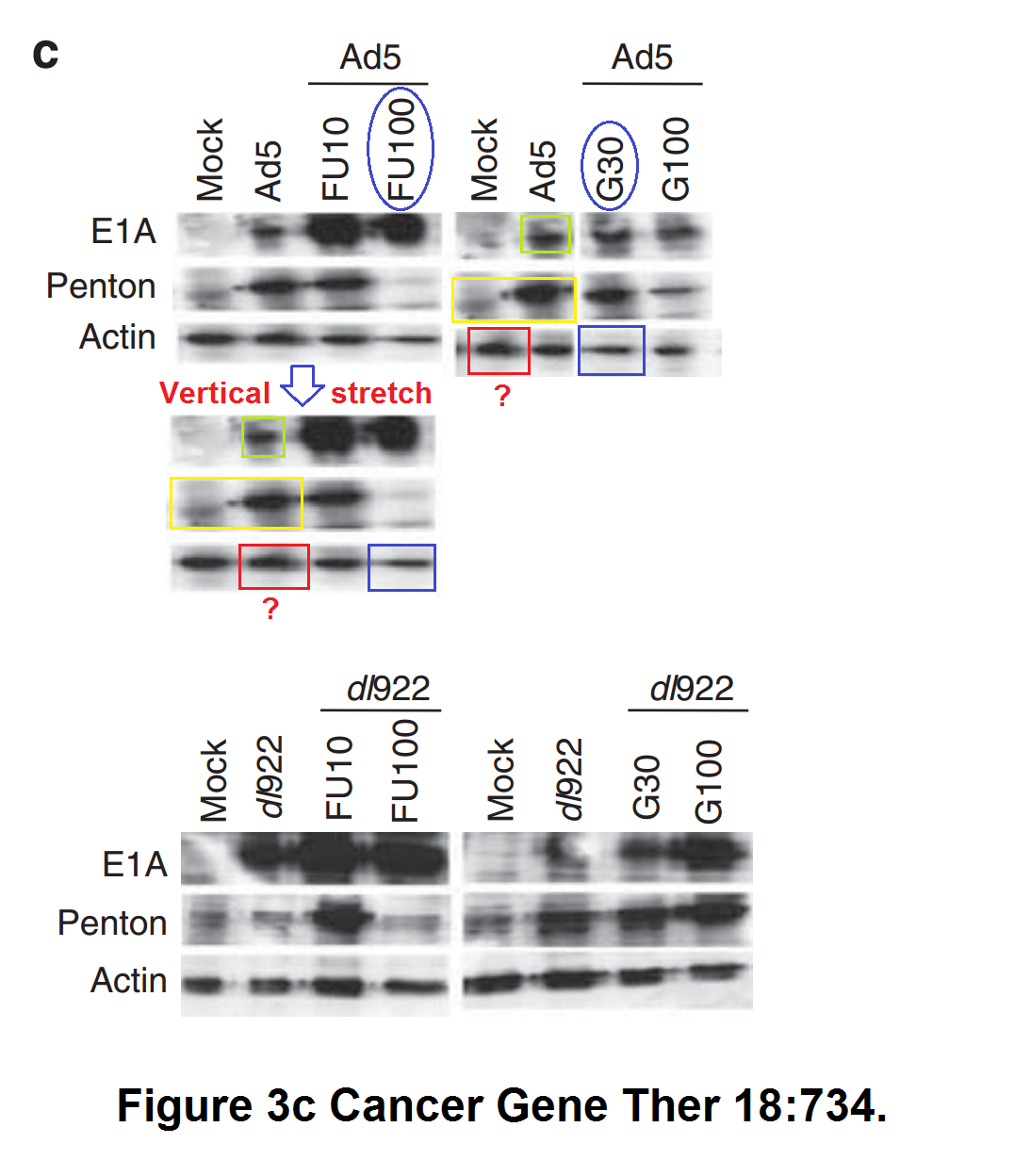
M Bhattacharyya, J Francis, A Eddouadi, N R Lemoine, G Halldén An oncolytic adenovirus defective in pRb-binding (dl922–947) can efficiently eliminate pancreatic cancer cells and tumors in vivo in combination with 5-FU or gemcitabine Cancer Gene Therapy (2011) doi: 10.1038/cgt.2011.45
Nothing was done. The Editor-in-Chief of this Nature Research Group journal is another professor in England, Georgios Giamas, owner of his own PubPeer record, and protege of the controversial, yet perfectly safe in his job, Imperial College professor Justin Stebbing. Giamas also holds a visiting professorship at Imperial, and he will definitely never do anything about the following paper by Limoine in Cancer Gene Therapy:
I A McNeish, T Tenev, S Bell, M Marani, G Vassaux, N Lemoine Herpes simplex virus thymidine kinase/ganciclovir–induced cell death is enhanced by co-expression of caspase-3 in ovarian carcinoma cells Cancer Gene Therapy (2001) doi: 10.1038/sj.cgt.7700305
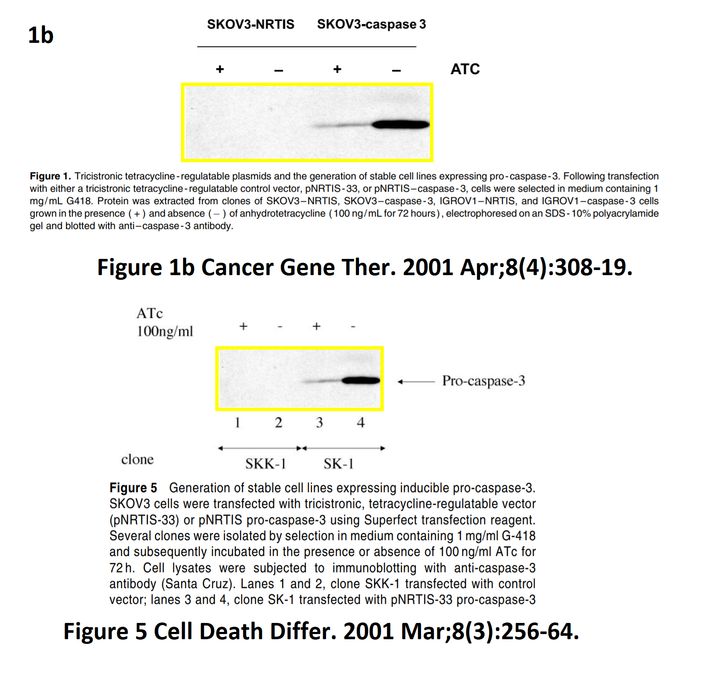
In November 2018, the first author and another Imperial College professor Ian McNeish posted on PubPeer the full investigative report by Limoine’s employer, Barts College, which was also approved by Imperial. McNeish, Limoine and the unnamed investigator agree that the raw data doesn’t exist (anymore, they say), that the two papers were submitted and reviewed simultaneously and that the gel bands are indeed duplicated. The investigator’s conclusions, as quoted by McNeish:
“None of these issues in my opinion change the conclusions made in the papers. I see no merit in retracting these important publications or publishing an erratum at this late stage, although agree that there have been errors made in producing the figures. […] The investigation stated that the conclusions made from the published studies were valid and important, even accounting for these reporting errors.“
This is the standard attitude of all English and in fact British universities. The wording is typical English passive-aggressiveness towards whistleblowers warning them never to waste the university’s time ever again or meet their due fate.
Below some more of Limoine papers, the rest is on PubPeer. The following is 7 years old, when it was posted the raw data was rather fresh, sometimes even piping hot. A key coauthor on all these papers is Yaohe Wang, another professor at Barts.
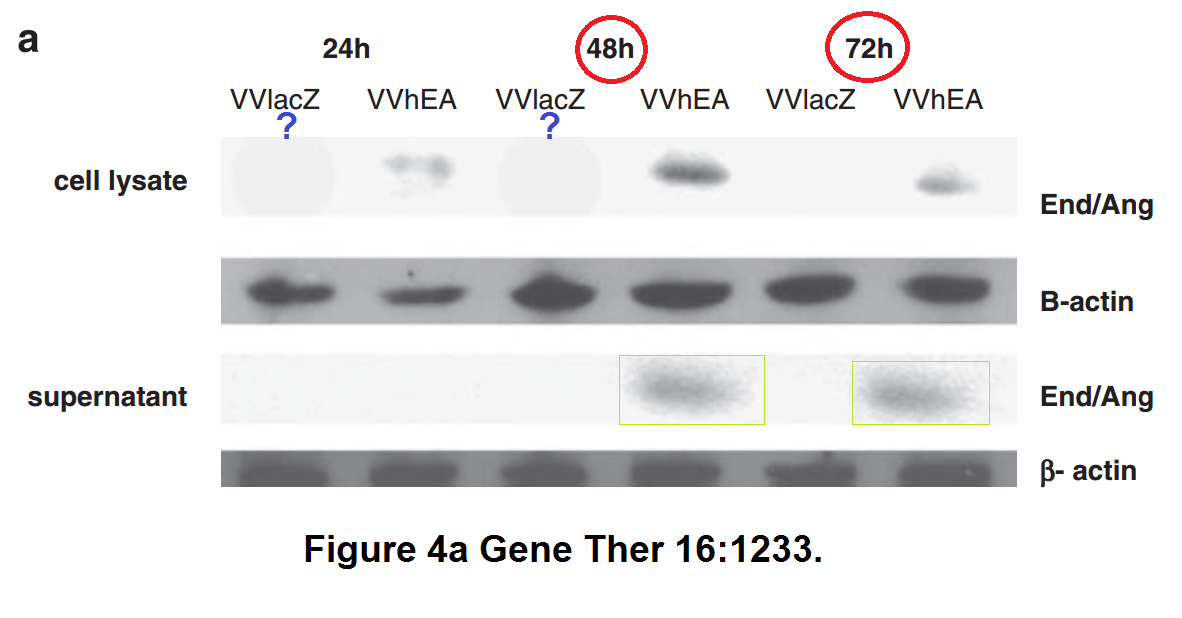
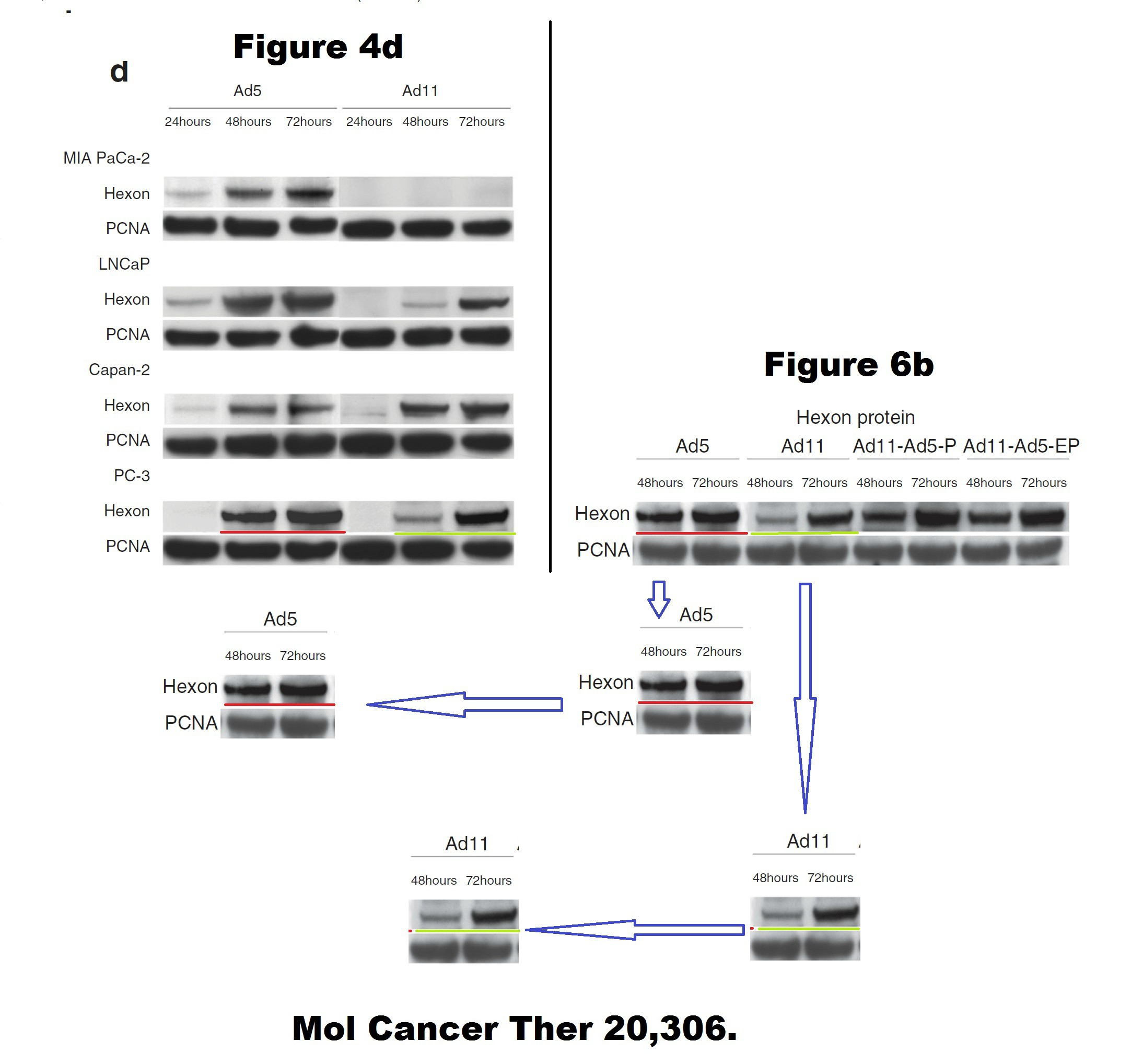
Barts College apparently decided instead to wait until this Photoshop travesty passes the 10 year statute of limitations. Expect another passive-aggressive investigative report in 2025, declaring Limoine and Wang innocent and all conclusions valid and not affected.
Peter St George-Hyslop, OC, FRS, FRSC, FRCPC
I think this name is so impressive, you can’t be more of English upper class, both in science and in society with a name like this. Of course Professor Peter St George-Hyslop is located in Cambridge, where he works on clinical neuroscience, previously he was in Canada where he was proclaimed “one of the most cited authors in the field of Alzheimer’s disease research”.
There are a number of papers coauthored by St George-Hyslop on PubPeer, here is one from his old lab at the University of Toronto:
Fusheng Chen, Gang Yu, Shigeki Arawaka, Masaki Nishimura, Toshitaka Kawarai, Haung Yu, Anurag Tandon, Agnes Supala, You Qiang Song, Ekaterina Rogaeva, Paul Milman, Christine Sato, Cong Yu, Christopher Janus, Julie Lee, Lixin Song, Lili Zhang, Paul E. Fraser, P. H. St George-Hyslop Nicastrin binds to membrane-tethered Notch Nature Cell Biology (2001) doi: 10.1038/35087069
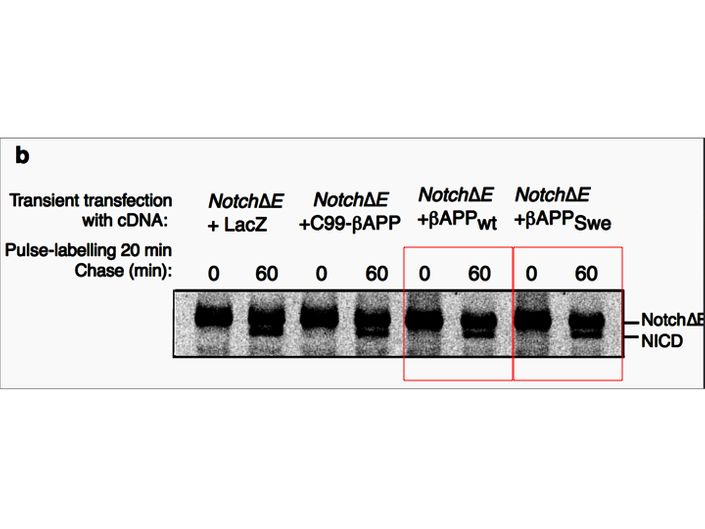
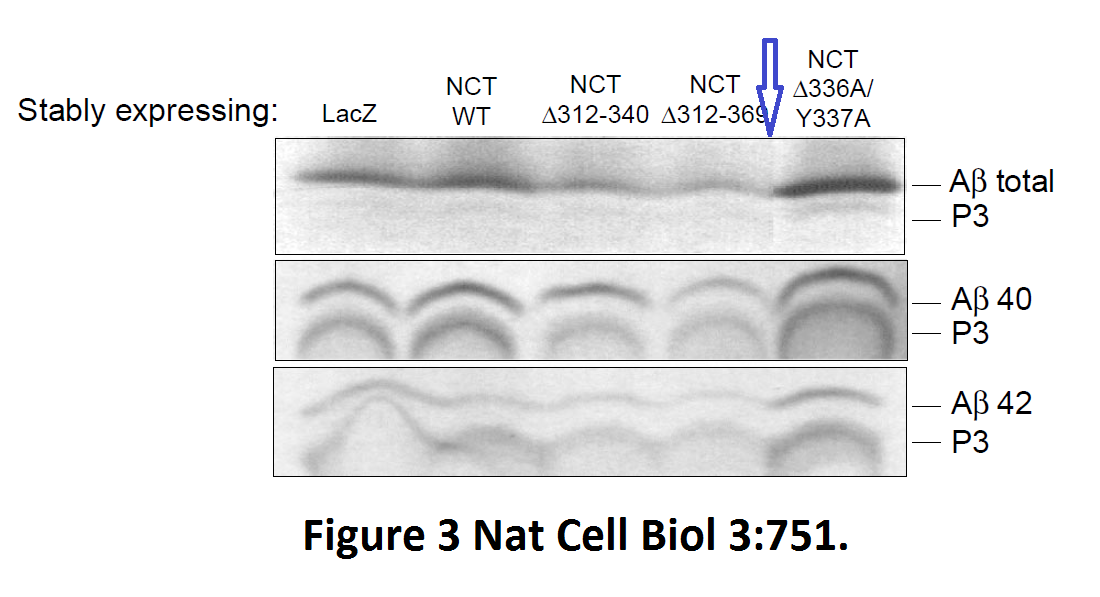
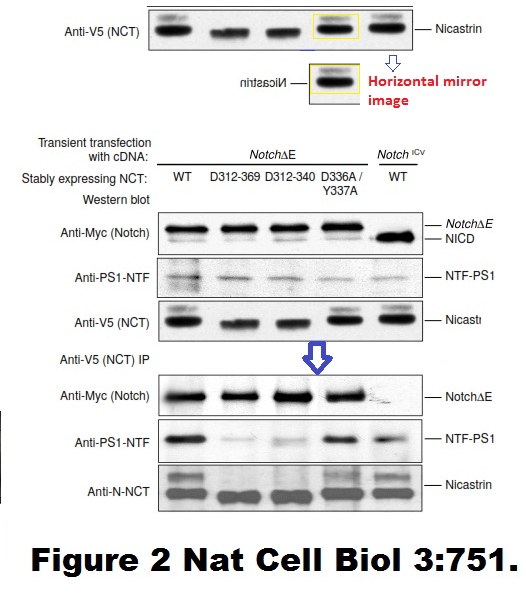
Cloned gel bands, spotted by Clare Francis. No action at all form that elite Nature-themed journal. Another St George-Hyslop fabrication, also done with Toronto colleagues:
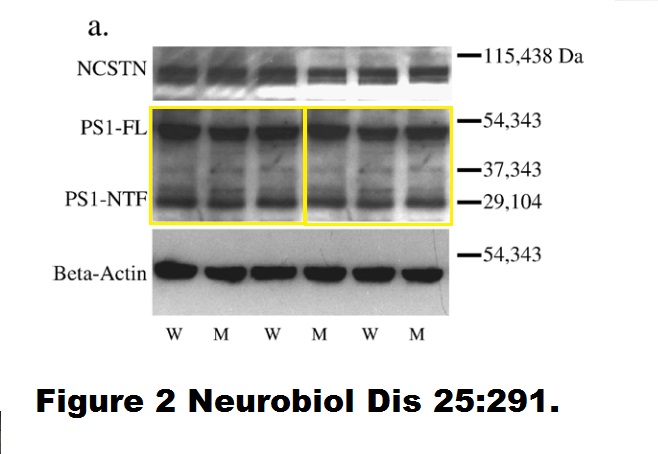
Sonya Brijbassi, Zareen Amtul, Susan Newbigging, David Westaway, Peter St George-Hyslop, Richard F. Rozmahel Excess of nicastrin in brain results in heterozygosity having no effect on endogenous APP processing and amyloid peptide levels in vivo Neurobiology of Disease (2007) doi: 10.1016/j.nbd.2006.09.013
But the worst ones were done in collaboration with Frederic Checler, professor of neuroscience at CNRS in France. Like this, where the authors shamelessly double-published to increase their publication record:
Julie Dunys, Toshitaka Kawarai, Emilie Giaime, Sherwin Wilk, M. Herrant, P. Auberger, Peter St George-Hyslop, Cristine Alves Da Costa, Frédéric Checler Study on the putative contribution of caspases and the proteasome to the degradation of Aph-1a and Pen-2 Neuro-degenerative diseases (2007) doi: 10.1159/000101840
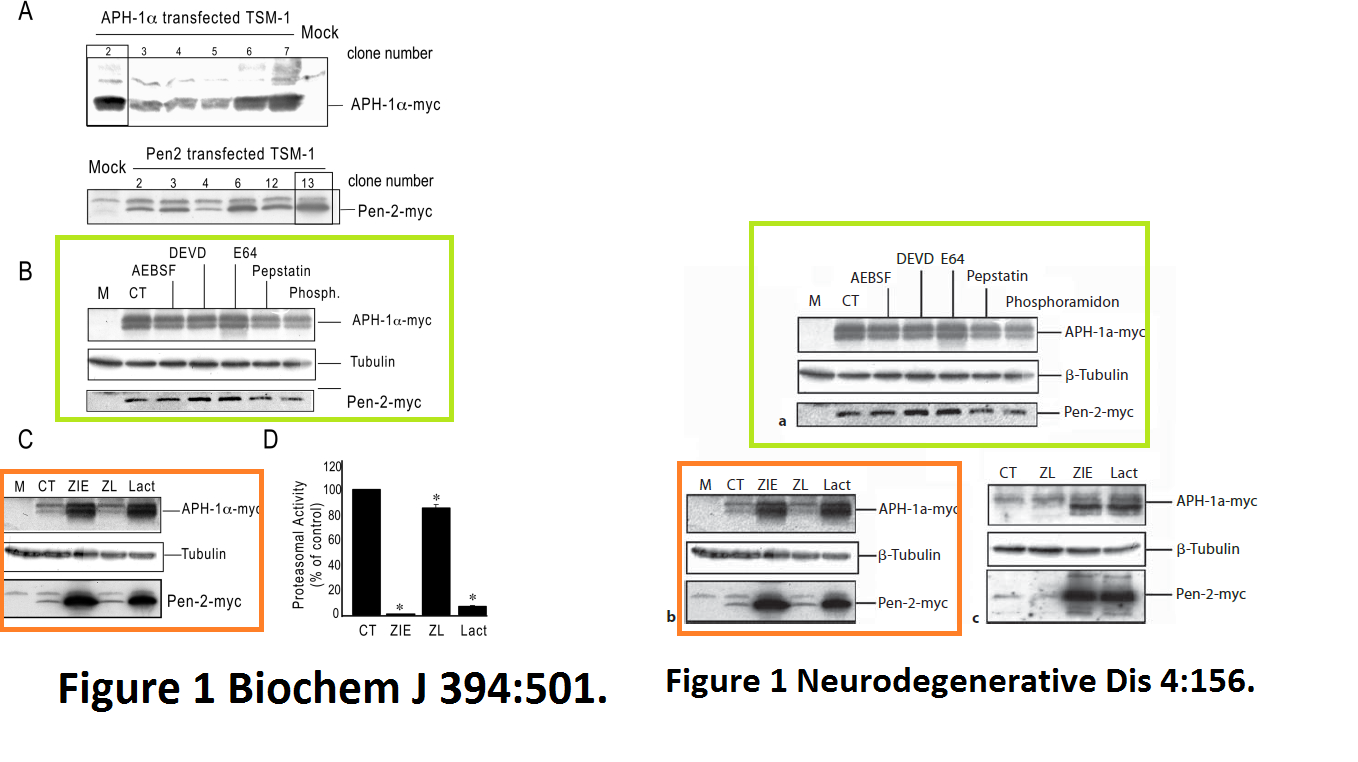
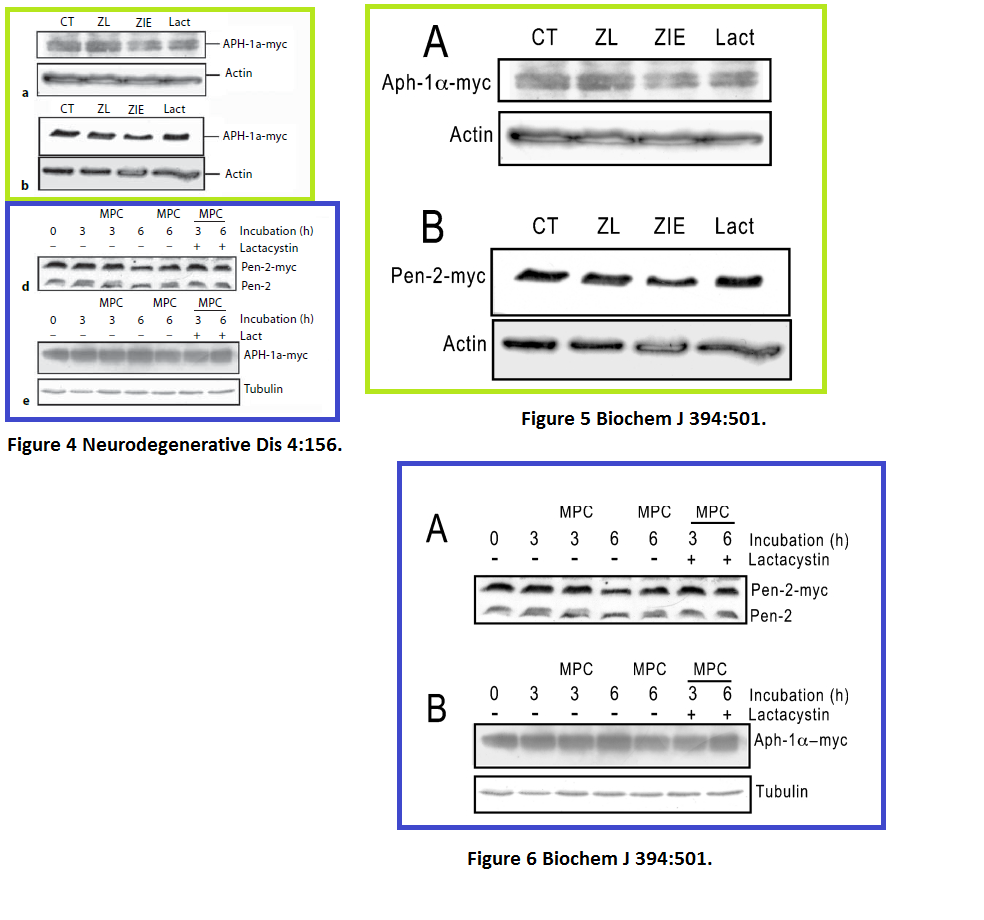
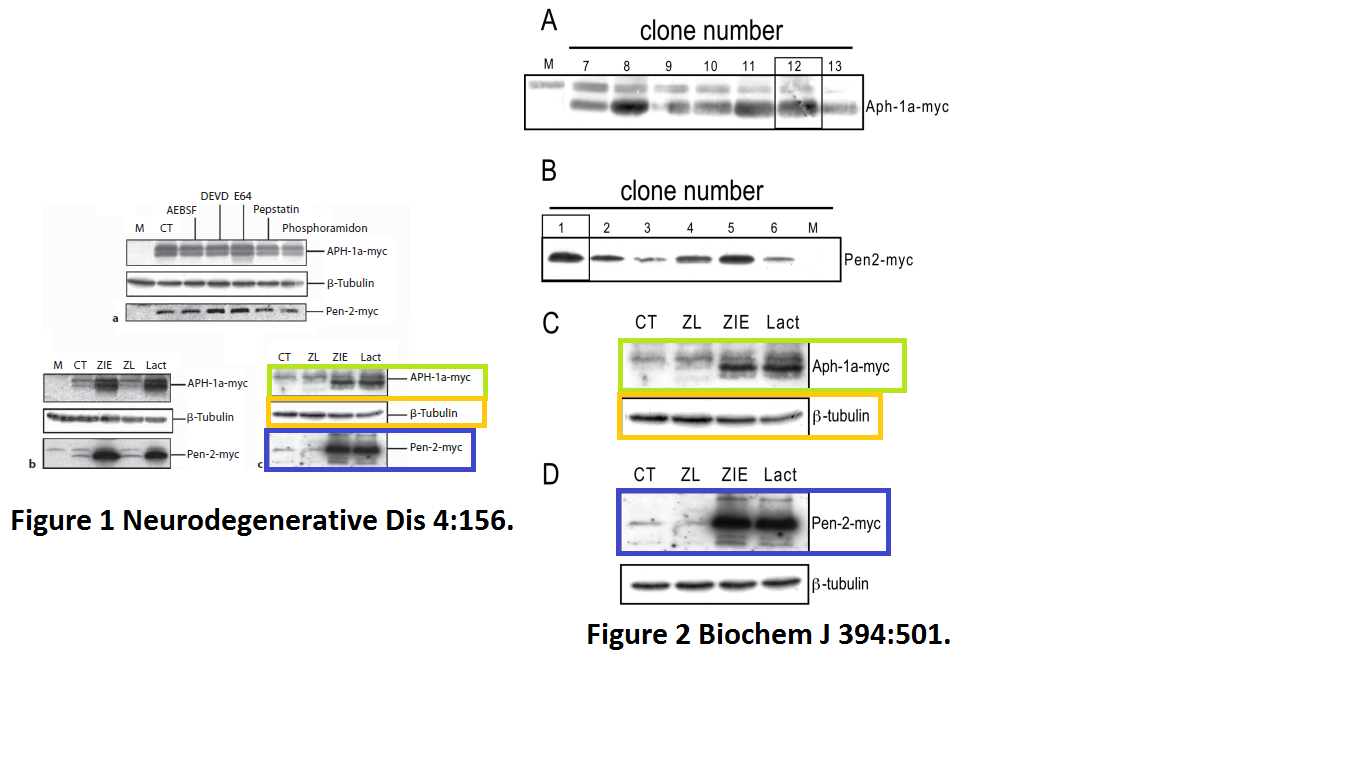
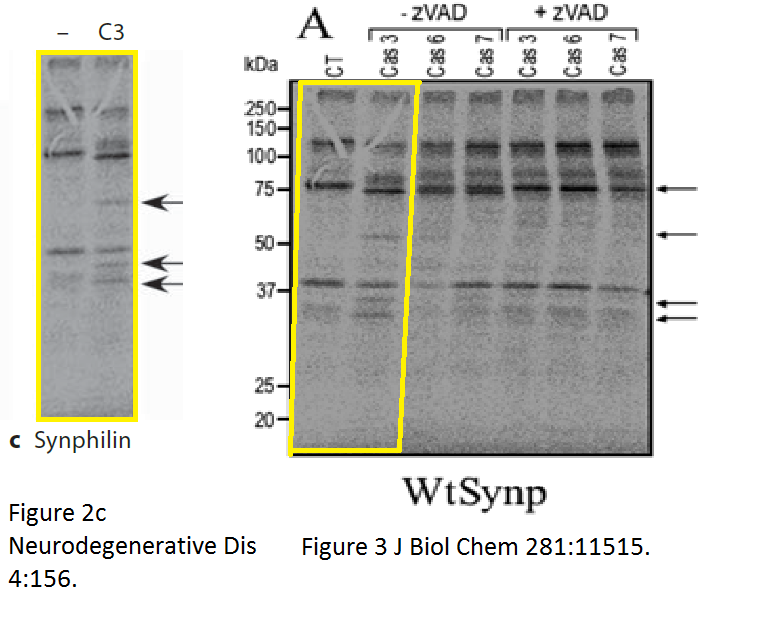
There was more data recycling, basically that paper in Neurodeg Dis is a copy of the previously published paper Dunys et al Biochemical J 2006, with all other authors the same. Neither journal, one by Portland Press, one by Karger, cares, never mind for research integrity, not even for their own infringed copyright.
Here more by St George-Hylop and his French ally, some gel band copy-pasting:
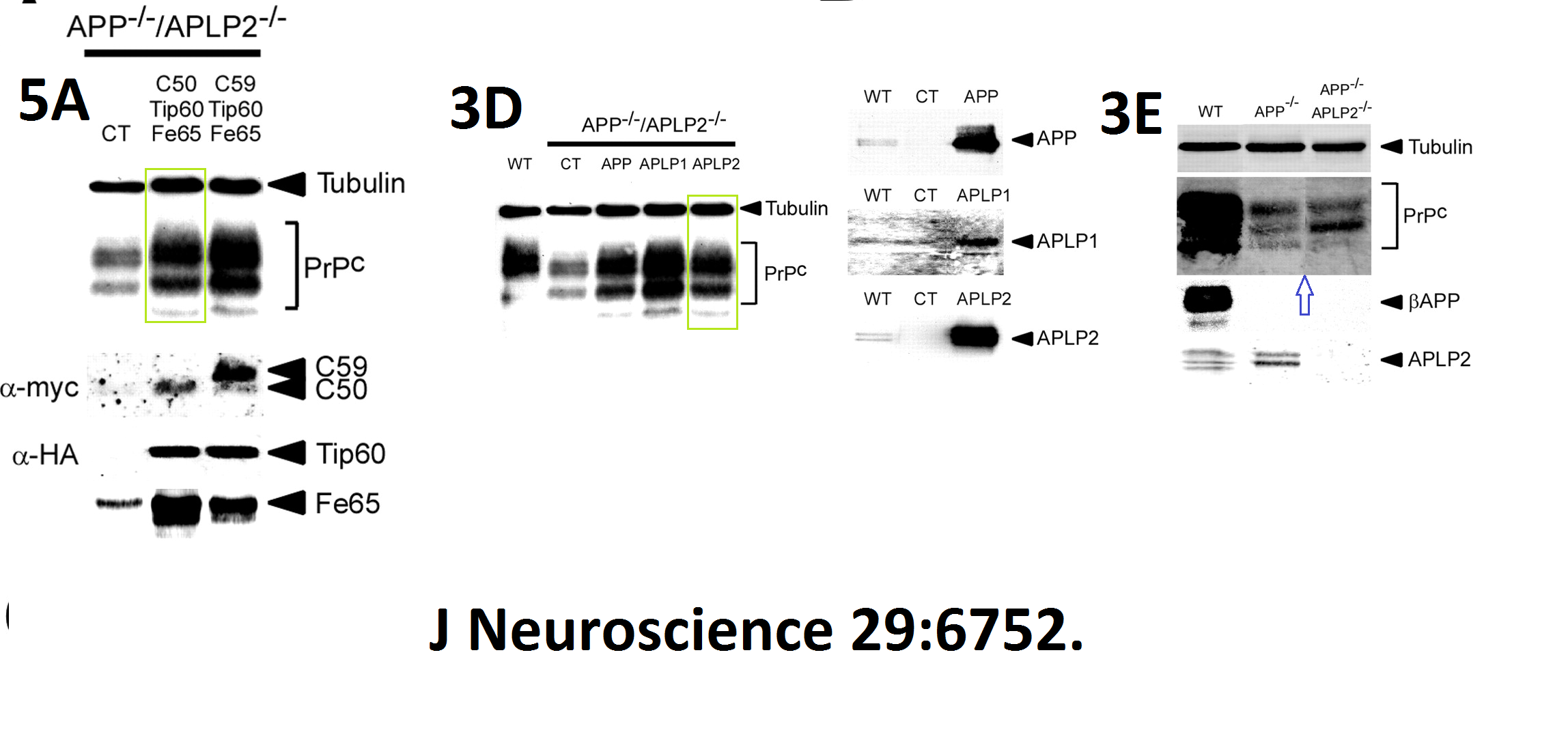
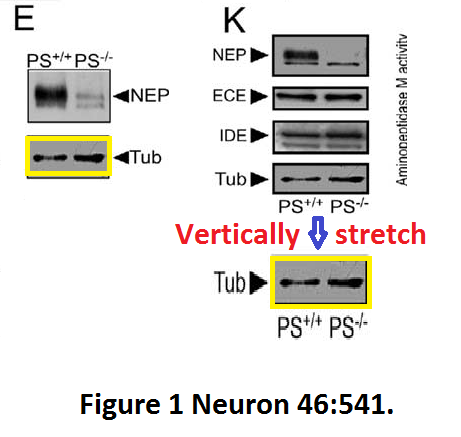
Here another interesting collaboration of our esteemed English upper class scientist, this time with Masaya Tohyama in Japan:
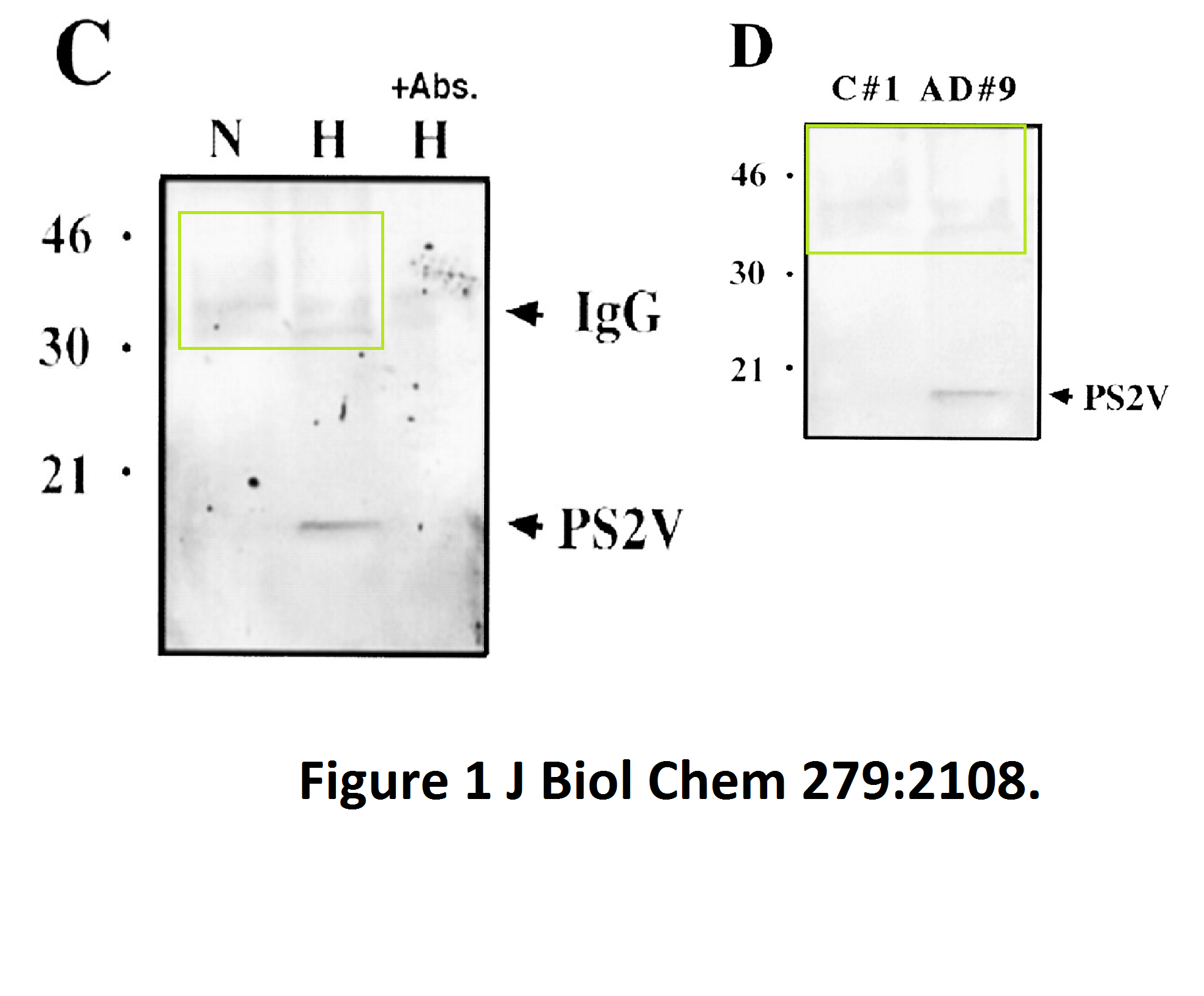
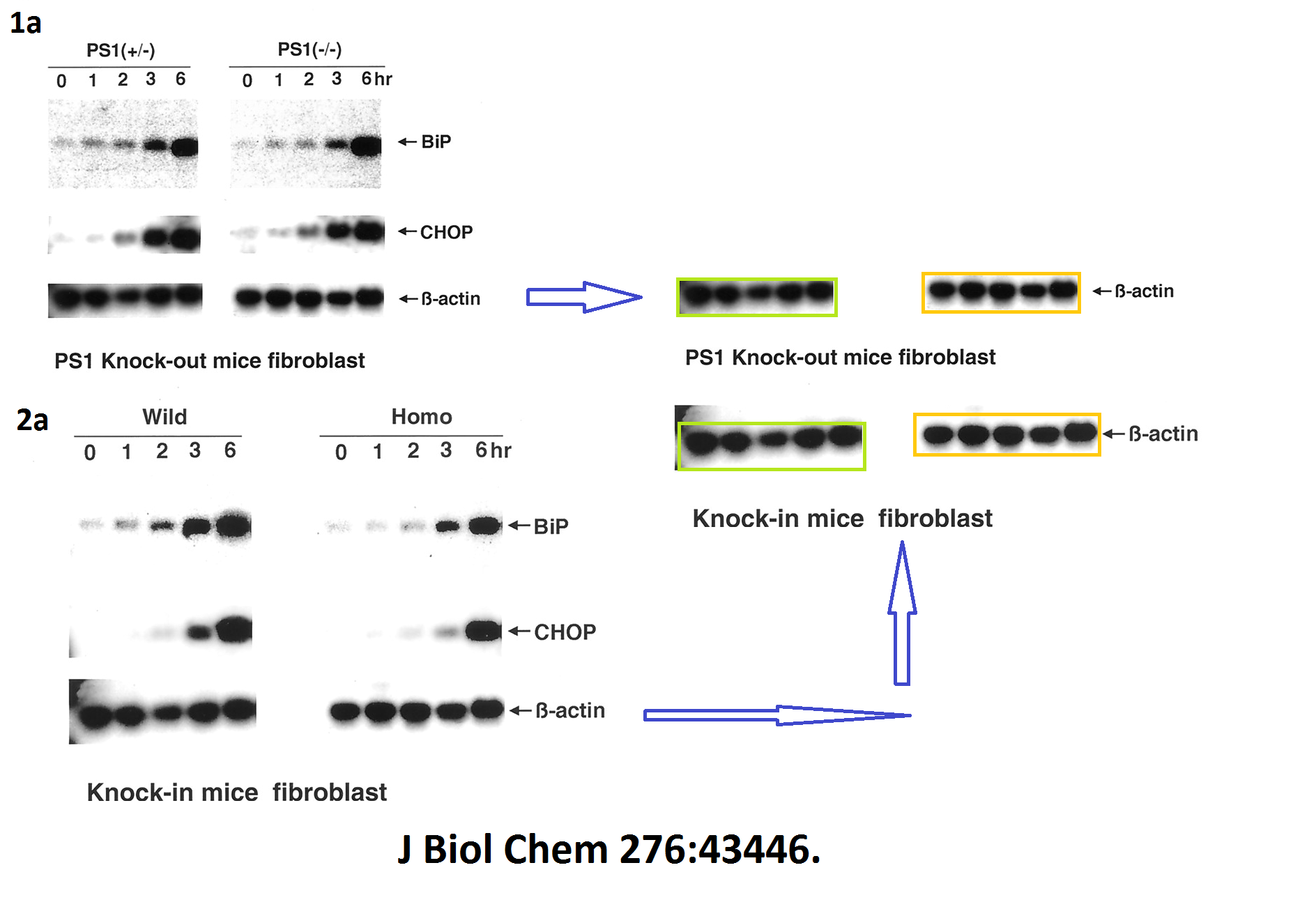
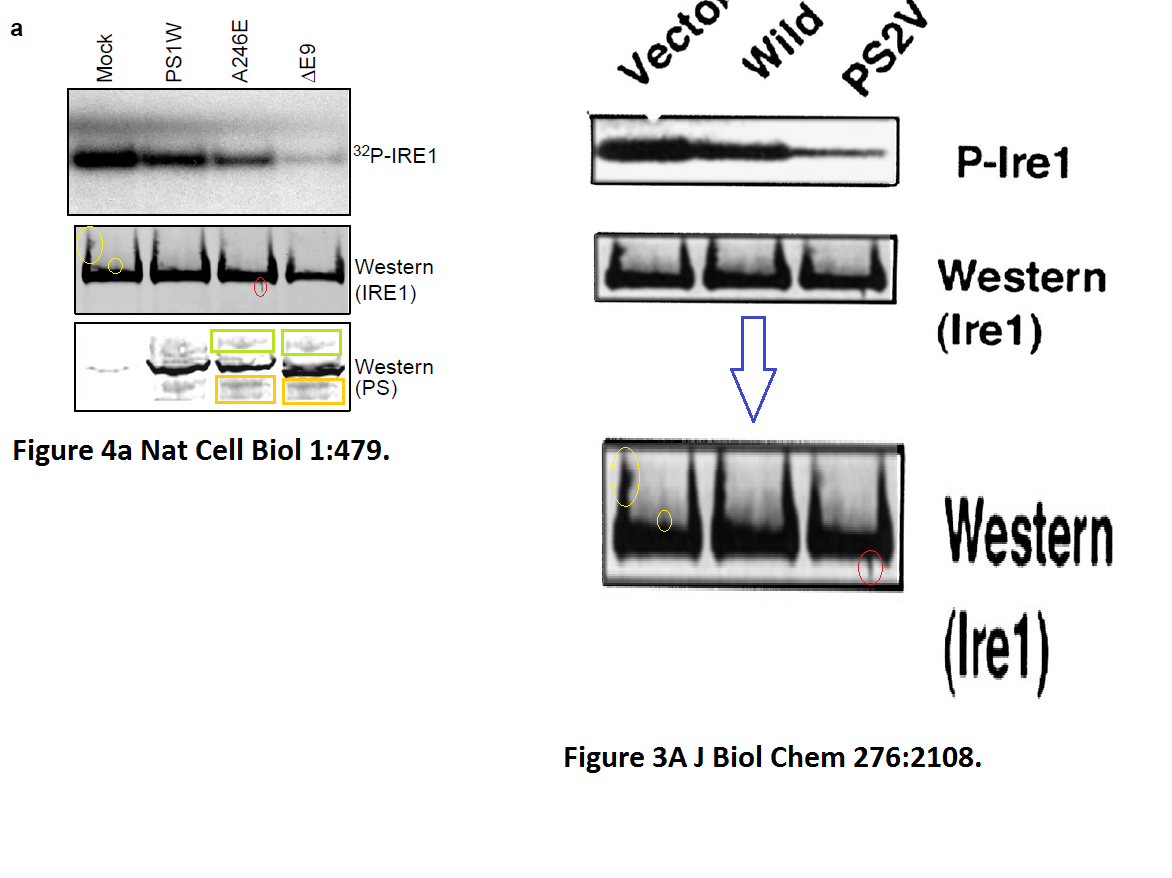
The evidence is years old, but nothing has happened so far. Yet one St George-Hyslop coauthored publication was retracted in 2020. Its first author Sabine Wislet is professor in Belgium and has a number of other papers questioned on PubPeer, including another retraction.
Sabine Wislet-Gendebien, Cheryl D’Souza, Toshitaka Kawarai, Peter St George-Hyslop, David Westaway, Paul Fraser, Anurag Tandon Cytosolic proteins regulate alpha-synuclein dissociation from presynaptic membranes The Journal of biological chemistry (2006) doi: 10.1074/jbc.m605965200
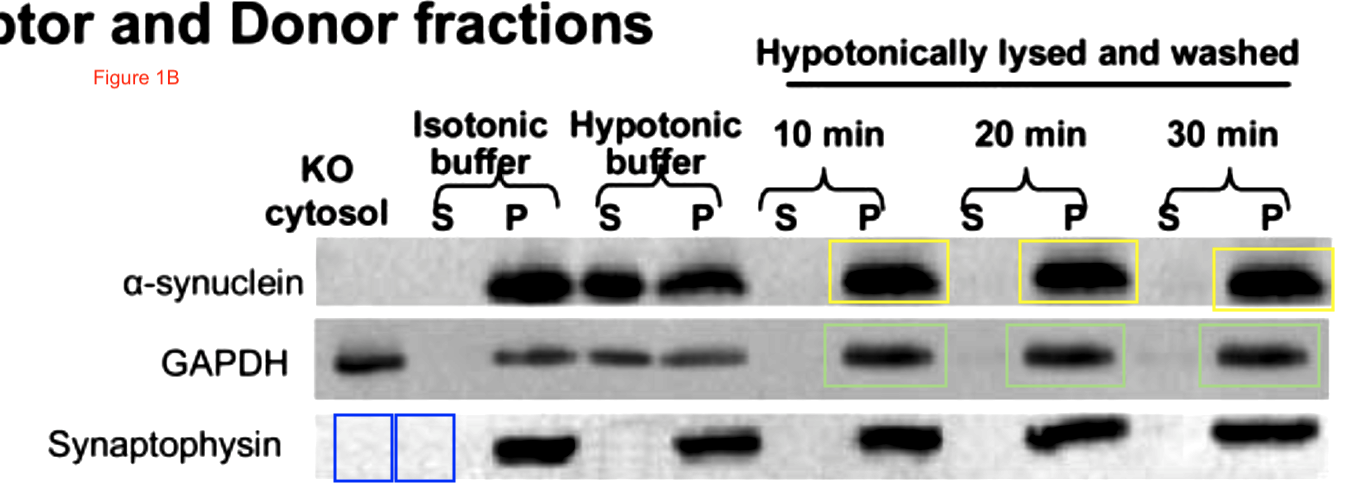
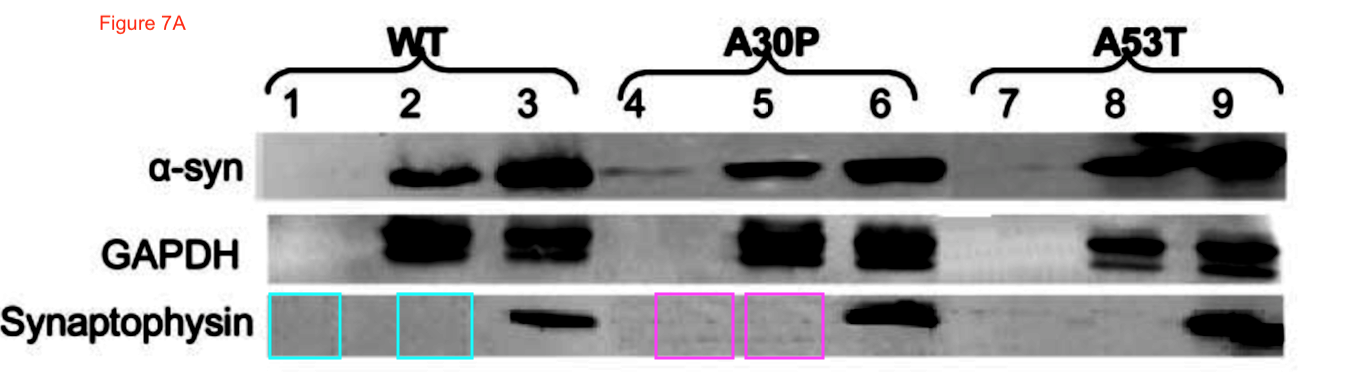
Now the fun starts. Clare Francis of course notified the University of Cambridge and the Wellcome Trust (which funds St Seorge-Hyslop’s research). This was their decision, as penned to Clare Francis by the Policies & Governance Officer of Wellcome Trust, Lucy Douch, in February 2017:
“Further to your email of 17 August 2016, we raised your concerns about Professor Peter St George-Hyslop with the University of Cambridge, the employing institution.
The University conducted an investigation under its Misconduct in Research policy. The investigation concluded that the allegations made are entirely unfounded.”
This is not even passive aggressive, it sounds almost like a legal threat from Wellcome Trust. In fact, this is a proud English Fuck You to every single whistleblower who dares to impugn the greatness of English science genius.
Although, to be fair, I suspect the Cambridge University probably has same statute of limitations on research fraud as Oxford, of 3 years. Because there was nothing for them to investigate in those older paper, they declared the allegations as “entirely unfounded”.
If you enjoyed St George-Hyslop’s aristocratic antics, you should also have a look at the PubPeer record of another Cambridge professor, Dame Carol Mary Black, DBE, FRCP, BSD, Principal of the Newnham College. Here a sample, with the Dame Commander of the Order of the British Empire as last author, Denton et al JBC 2005:

Xin Lu, FRS
Britain is open to foreigners, as we know. Not the lowly kind who don’t know how to get rich quick. If you know how to play the game, you can become part of the English science elite. Xin Lu studied in her home land China, and in UK she eventually arrived to the Ludwig Cancer Centre, first at its London branch and later to take over the directorship of the central seat in Oxford.
Yes, Oxford is the university with 3 year statute of limitation for research misconduct. Nothing to see here at all:
Daniele Bergamaschi, Yardena Samuels, Alexandra Sullivan, Marketa Zvelebil, Hilde Breyssens, Andrea Bisso, Giannino Del Sal, Nelofer Syed, Paul Smith, Milena Gasco, Tim Crook, Xin Lu iASPP preferentially binds p53 proline-rich region and modulates apoptotic function of codon 72–polymorphic p53 Nature Genetics (2006) doi: 10.1038/ng1879
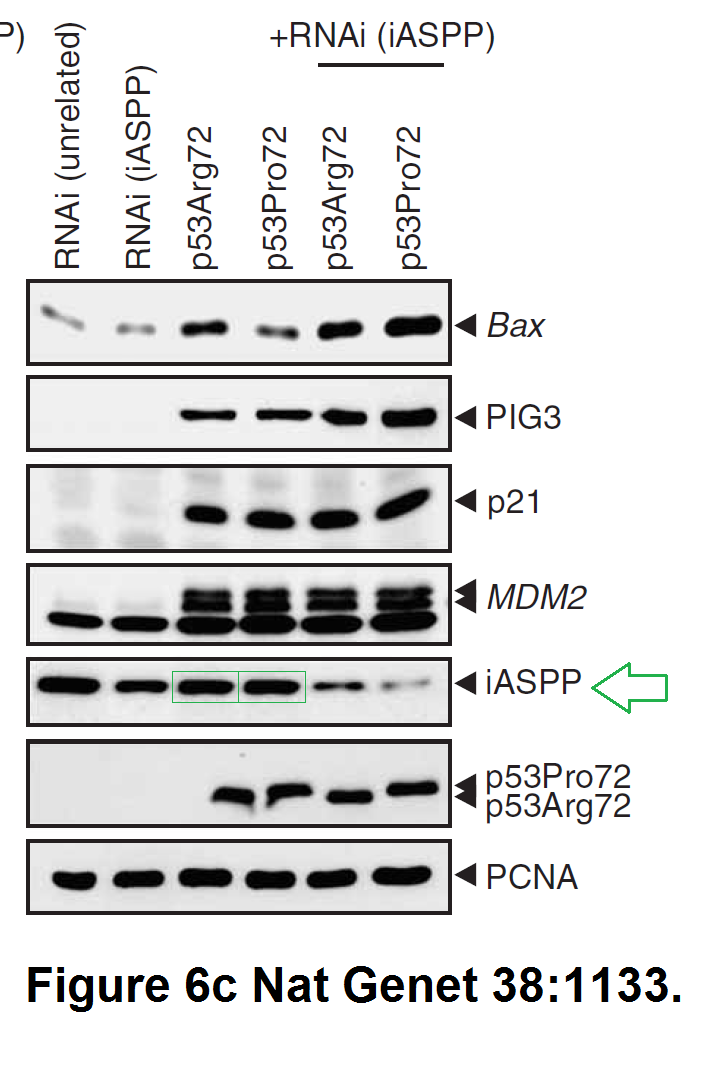
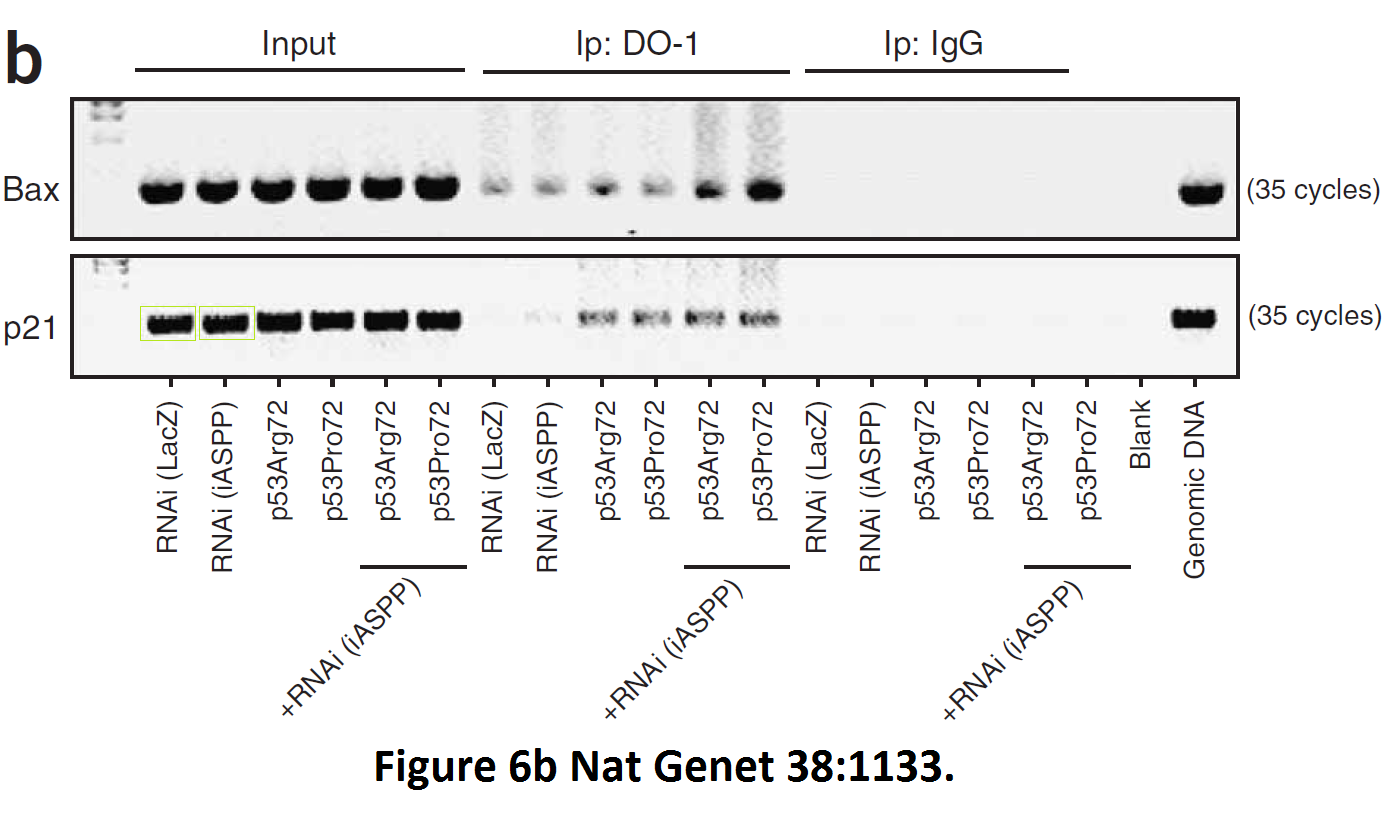
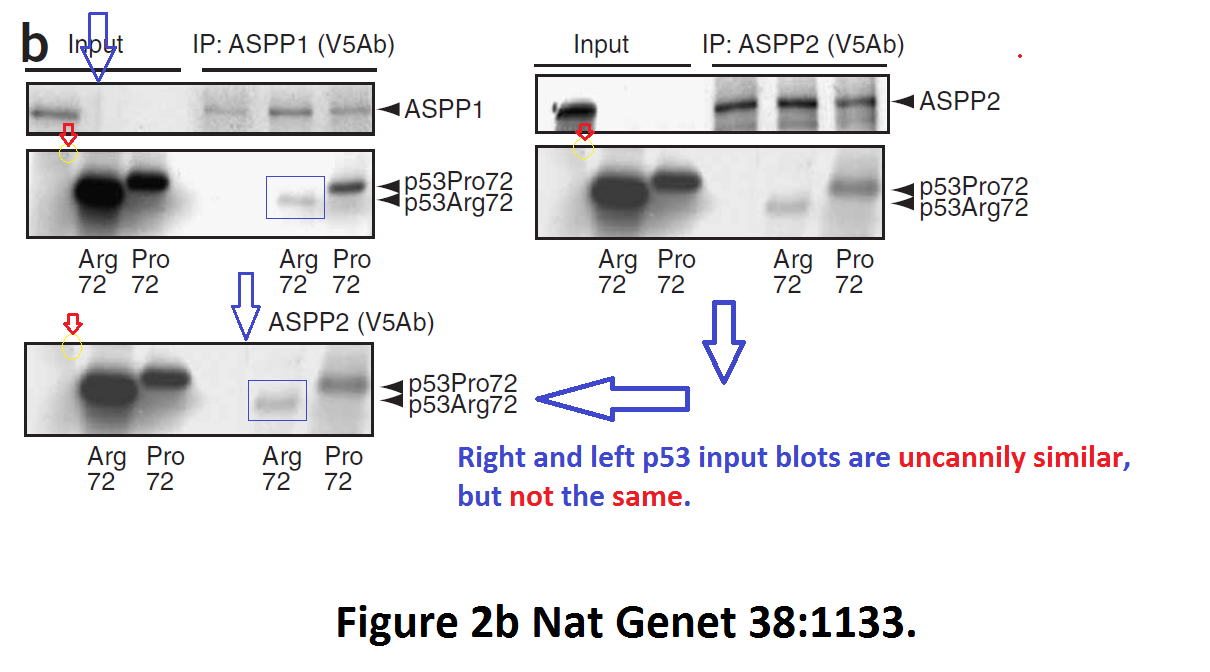
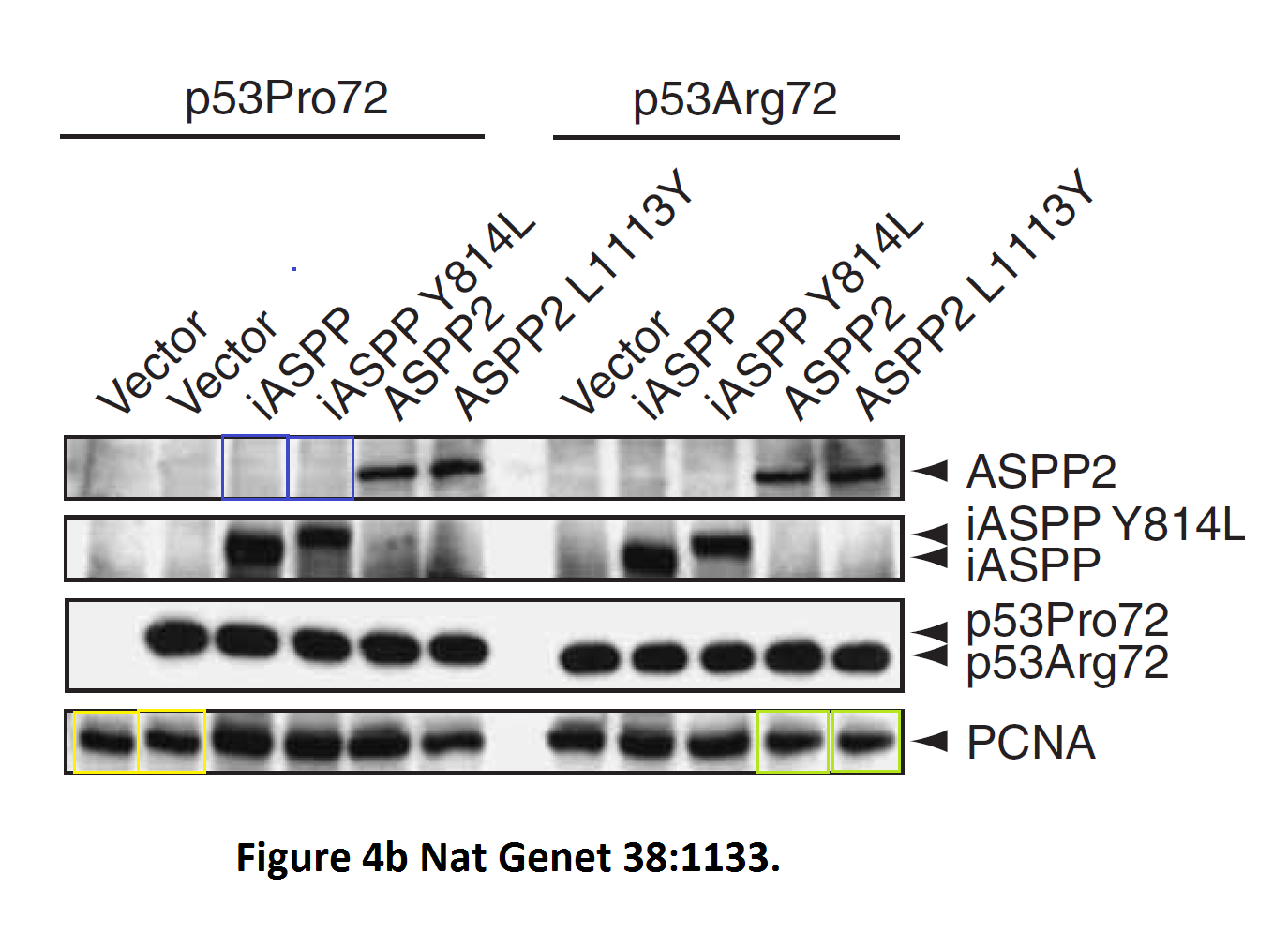
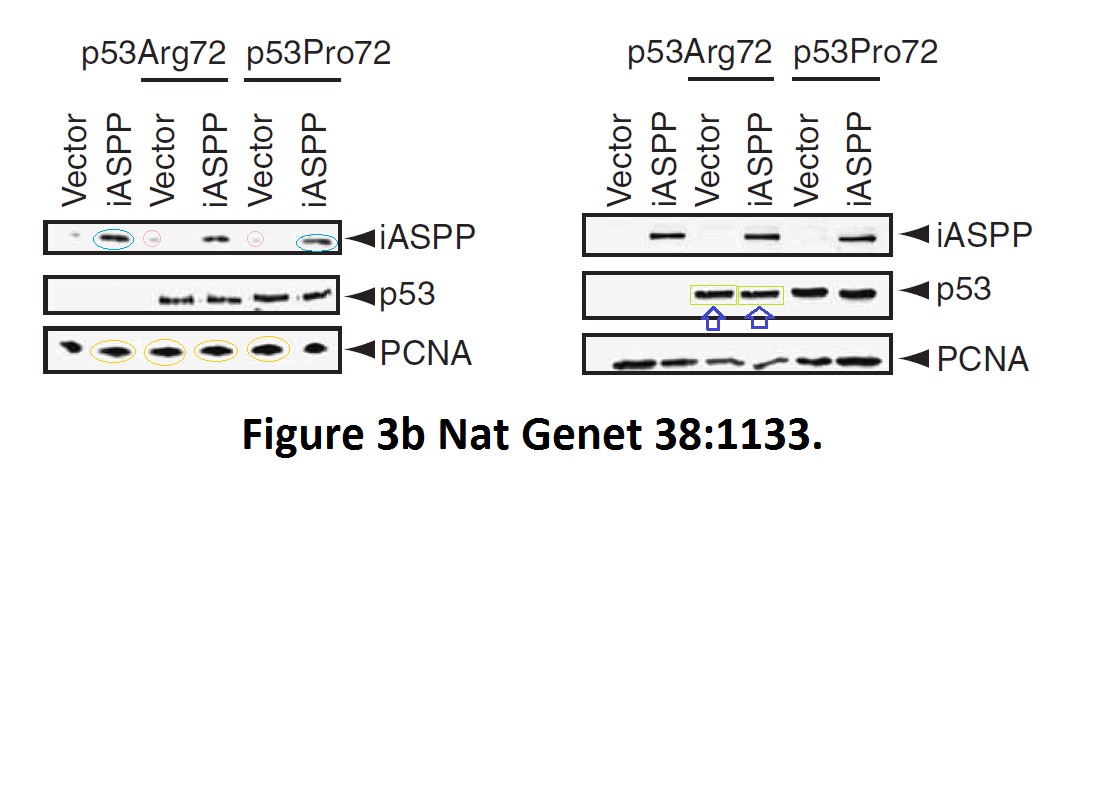
So many cloned gel bands, and yes, Clare Francis informed the University of Oxford of this and other masterpieces. The first author on that paper, Daniele Bergamaschi is now lecturer at the Queen Mary University of London, his lab is at Barts. You see again which kind of talents Barts boss Lemoine expects from his principal investigators. Bergamaschi also authored this work of art with Lu:
Daniele Bergamaschi, Yardena Samuels, Boquan Jin, Sai Duraisingham, Tim Crook, Xin Lu ASPP1 and ASPP2: common activators of p53 family members Molecular and Cellular Biology (2004) doi: 10.1128/mcb.24.3.1341-1350.2004
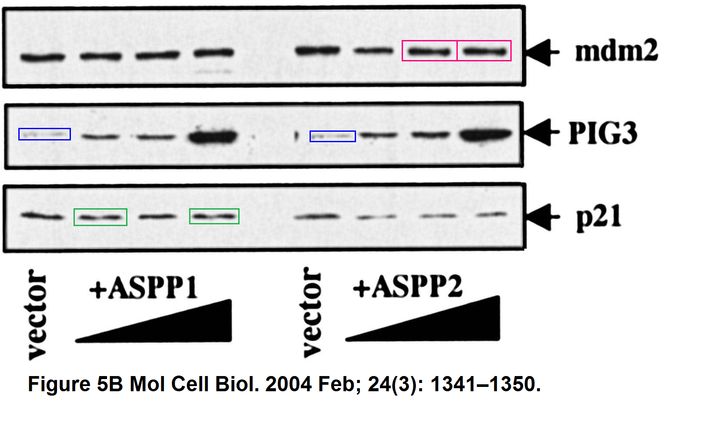
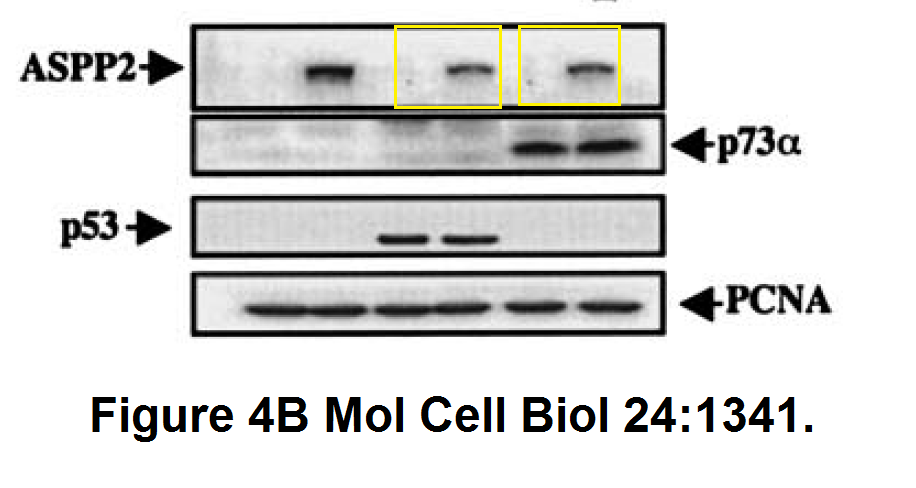
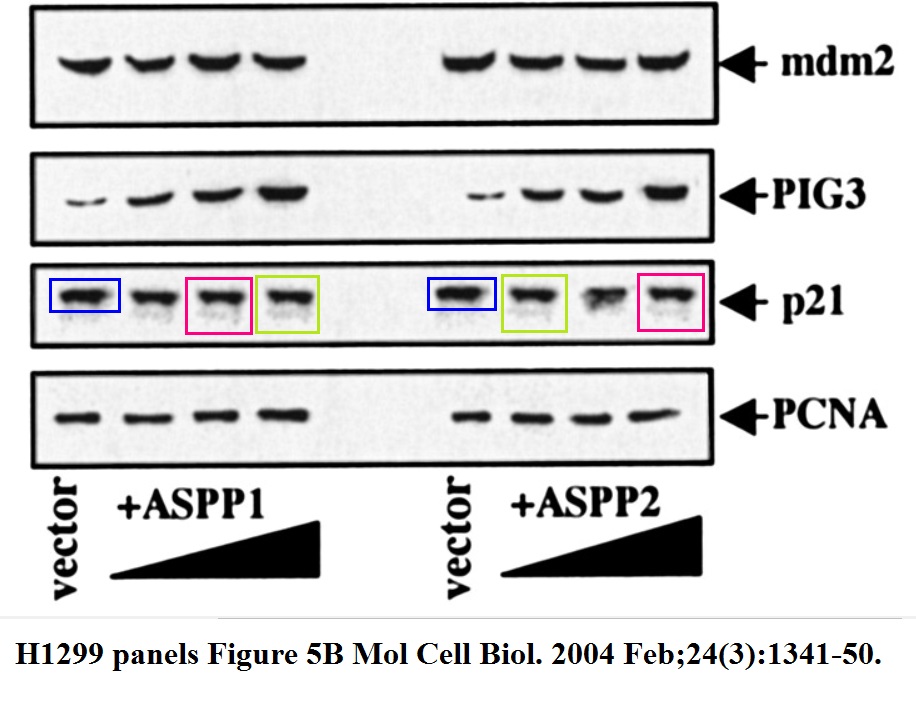
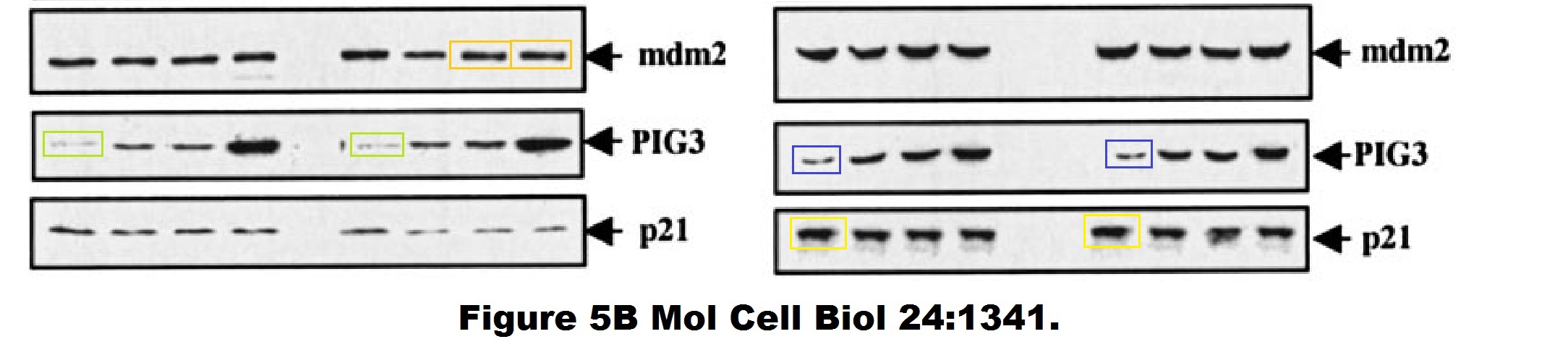
And this one by Bergamaschi and Lu, in Oncogene (remember the Editor-in-Chief Justin Stebbing?):
Daniele Bergamaschi, Yardena Samuels, Shan Zhong, Xin Lu Mdm2 and mdmX prevent ASPP1 and ASPP2 from stimulating p53 without targeting p53 for degradation Oncogene (2005) doi: 10.1038/sj.onc.1208535
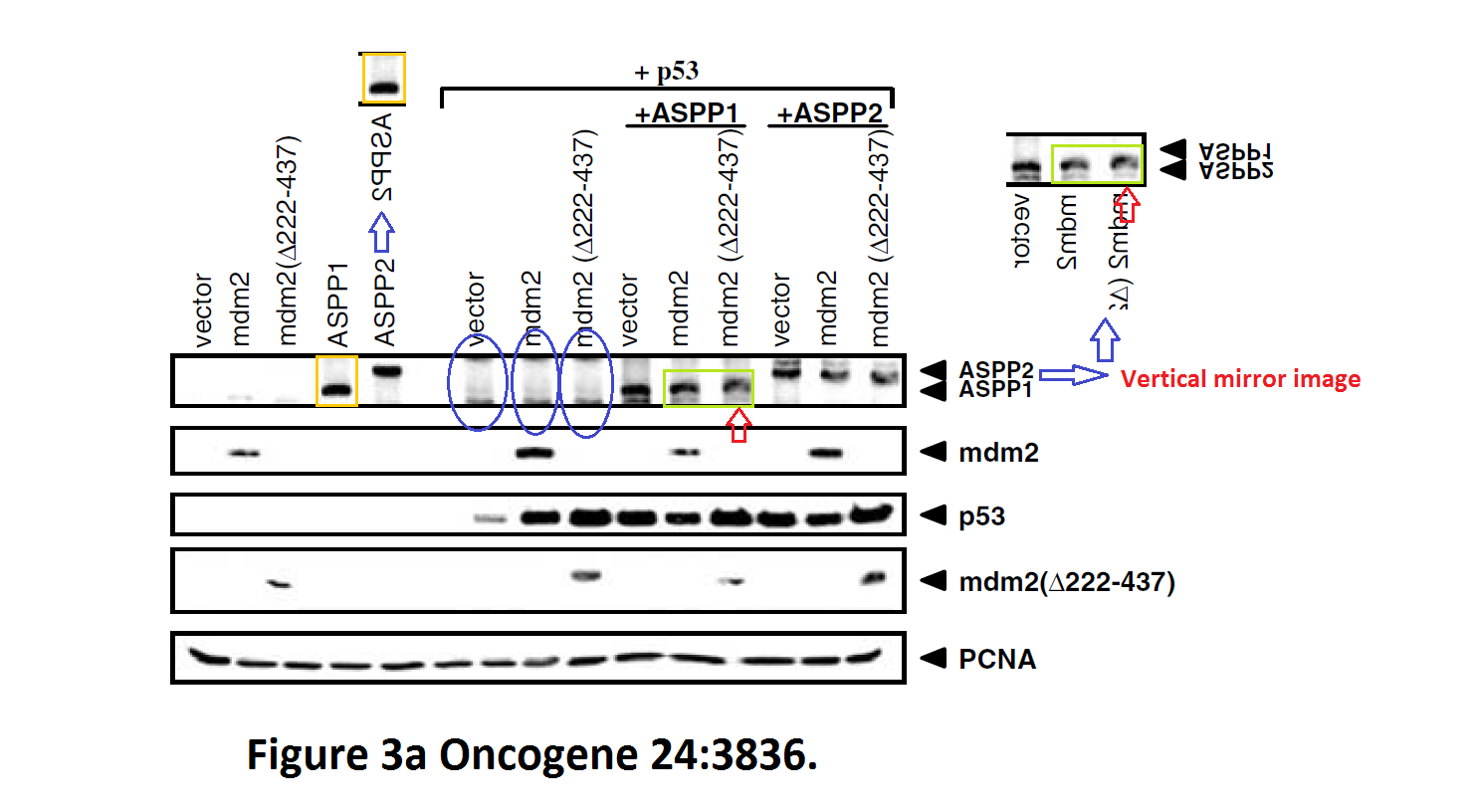
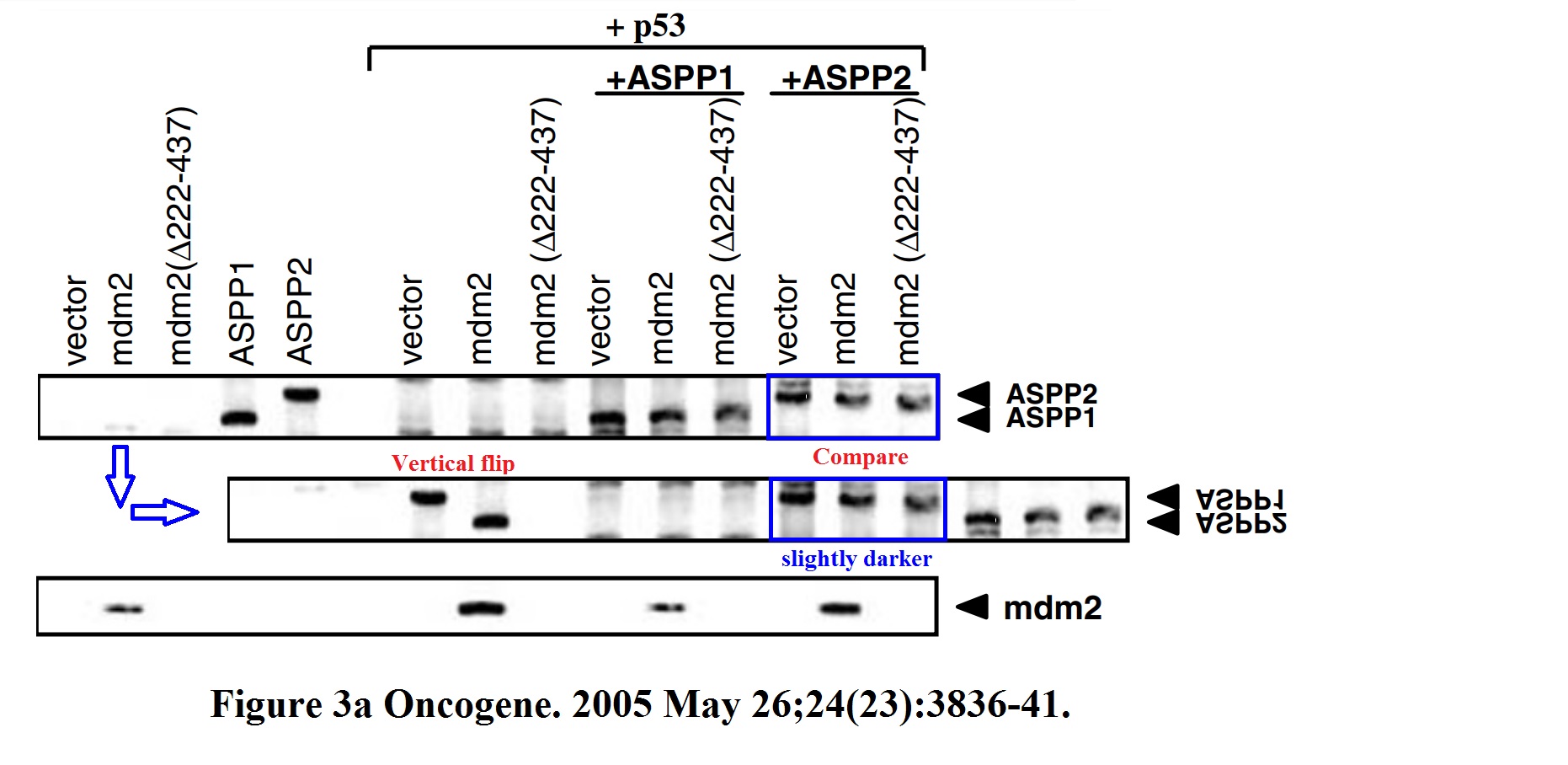
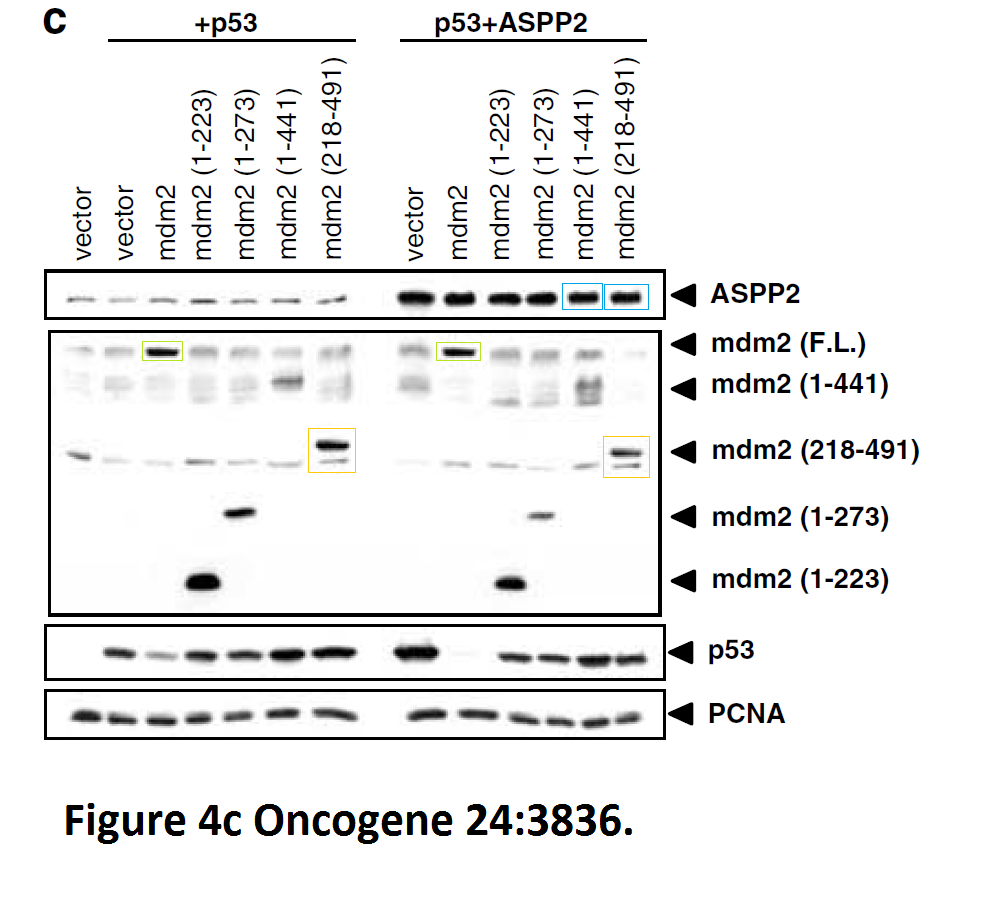
I hope you are learning from Bergamaschi and Lu how to make career in English science. It’s not that difficult, all you need is the will to succeed. Another common author on all these papers is Yardena Samuels. She went back to Israel where she is now professor at the Weizmann Institute. Here more by Bergamaschi (and Samuels):
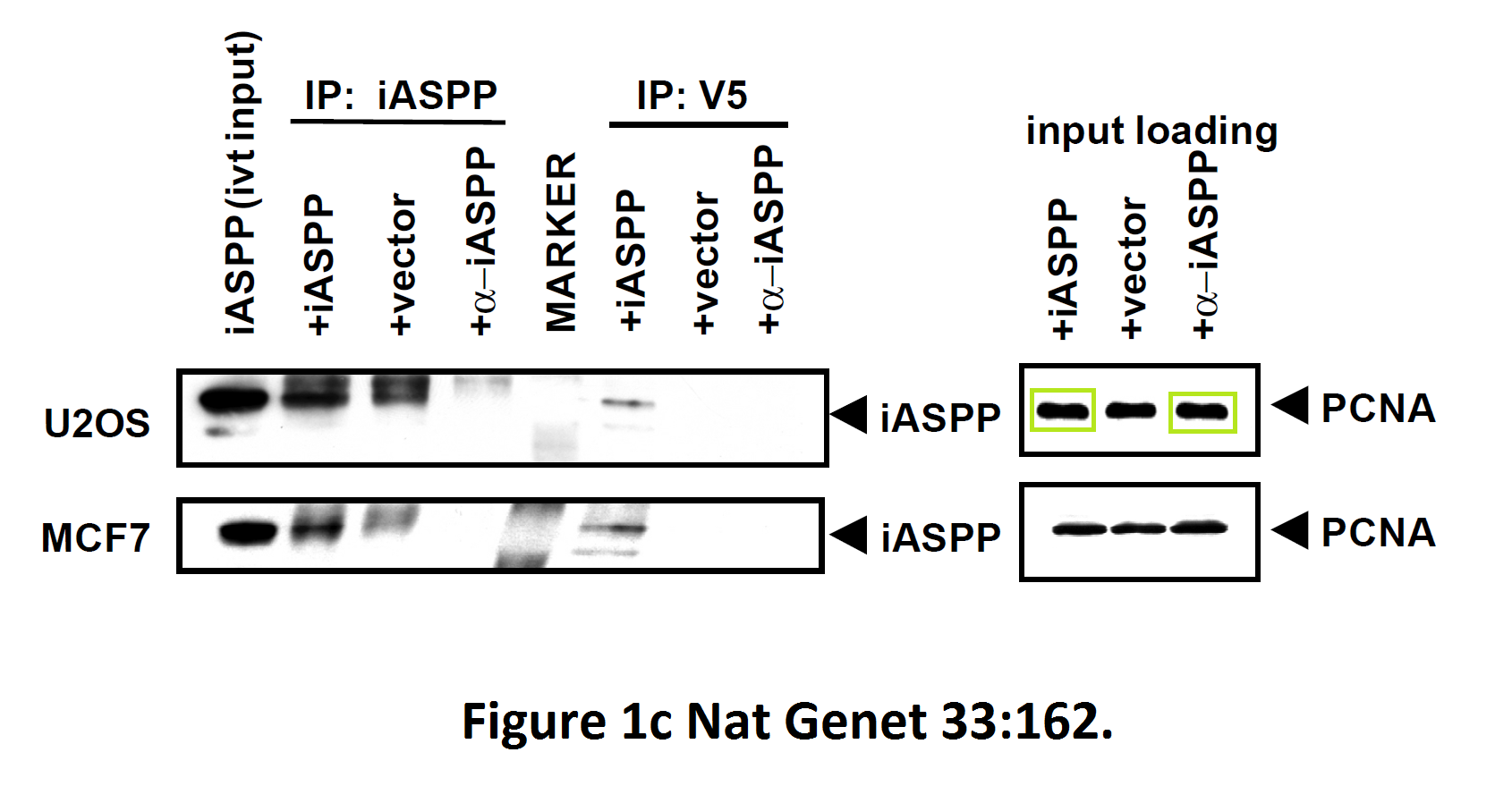
The last author on one of those papers is Geoffrey Smith, head of Pathology Department at University of Cambridge and member of all possible learned societies including the German Leopoldina. People of this calibre can’t be associated with bad science, so don’t expect even a correction.
But the next two papers from Lu lab doesn’t have Bergamaschi among its authors. Here one:

Damian B. S. Yap, Jung-Kuang Hsieh, Shan Zhong, Vicky Heath, Barry Gusterson, Tim Crook, Xin Lu Ser392 phosphorylation regulates the oncogenic function of mutant p53 Cancer Research (2004) doi: 10.1158/0008-5472.can-1305-2
These are two very fraudulent figures, bands were erased in one version by a rascal. But this paper, like others, is too old for anyone to give a toss about. Here something more recent:
Min Lu, Hilde Breyssens, Victoria Salter, Shan Zhong, Ying Hu, Caroline Baer, Indrika Ratnayaka, Alex Sullivan, Nicholas R. Brown, Jane Endicott, Stefan Knapp, Benedikt M. Kessler, Mark R. Middleton, Christian Siebold, E. Yvonne Jones, Elena V. Sviderskaya, Jonathan Cebon, Thomas John, Otavia L. Caballero, Colin R. Goding, Xin Lu Restoring p53 function in human melanoma cells by inhibiting MDM2 and cyclin B1/CDK1-phosphorylated nuclear iASPP Cancer Cell (2013) doi: 10.1016/j.ccr.2013.03.013
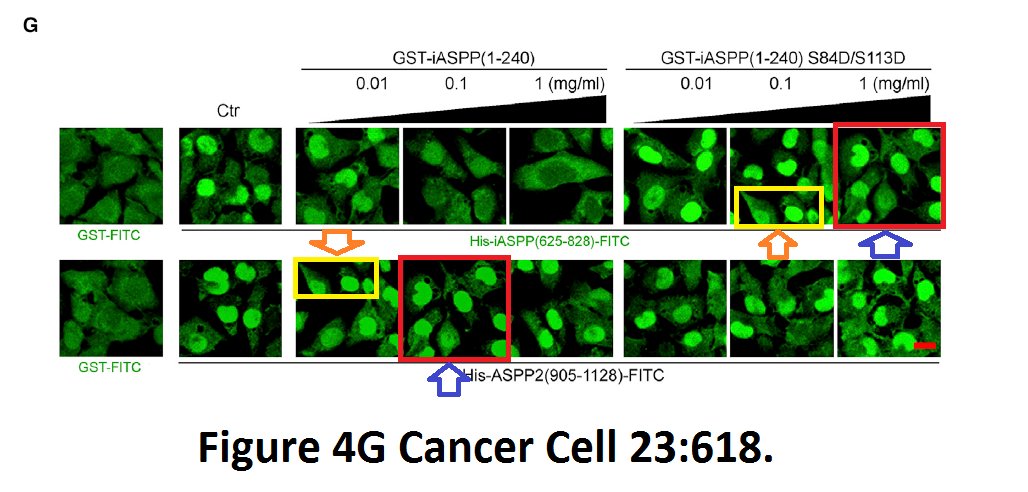
An Erratum was issued by Cell Press in 2016 and declared:
“This error does not affect any of the findings reported in the paper.”
With English research fabrications it is just like with Brexit: get over it, they won. Science is an English colony and the rotten upper classes are busy plundering it.

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Or you can donate to Barts or Ludwig centres for cancer research!
€5.00


J Biol Chem. 2003 May 2;278(18):16059-72. doi: 10.1074/jbc.M212935200. Epub 2003 Feb 17.
The histone deacetylase 9 gene encodes multiple protein isoforms
Kevin Petrie 1, Fabien Guidez, Louise Howell, Lyn Healy, Samuel Waxman, Mel Greaves, Arthur Zelent
Affiliation
1Leukemia Research Fund Centre, Institute of Cancer Research, 237 Fulham Road, London SW3 6JB, UK.
PMID: 12590135 DOI: 10.1074/jbc.M212935200
Such high quality (images that is)!
6B much more similar than expected.
https://pubpeer.com/publications/C806968D6FB27828C665A97E5CD571#1
Figure 9. Much more similar than expected.
https://pubpeer.com/publications/C806968D6FB27828C665A97E5CD571#3
LikeLike
Mol Cancer Res. 2010 Jan;8(1):24-34. doi: 10.1158/1541-7786.MCR-09-0432. Epub 2010 Jan 12.
FOXM1 confers acquired cisplatin resistance in breast cancer cells
Jimmy M-M Kwok 1, Barrie Peck, Lara J Monteiro, Helma D C Schwenen, Julie Millour, R Charles Coombes, Stephen S Myatt, Eric W-F Lam
Affiliation
1Cancer Research-UK Laboratories, Department of Oncology, Medical Research Council Cyclotron Building, Imperial College London, Hammersmith Hospital Campus, Du Cane Road, London W12 0NN, United Kingdom.
PMID: 20068070 PMCID: PMC2809047 DOI: 10.1158/1541-7786.MCR-09-0432
https://pubpeer.com/publications/0C5F99E1AC3E1483B880AC5D247B46
Figure 6C. Much more similar than expected after vertical stretch (also appears in Ph.D. thesis).
https://pubpeer.com/publications/0C5F99E1AC3E1483B880AC5D247B46#5
LikeLike
J Cell Sci. 2012 Jul 1;125(Pt 13):3153-63. doi: 10.1242/jcs.100511. Epub 2012 Mar 27.
A novel regulatory mechanism links PLCγ1 to PDK1
Claudio Raimondi 1, Anissa Chikh, Ann P Wheeler, Tania Maffucci, Marco Falasca
Affiliation
1Centre for Diabetes, Blizard Institute, Queen Mary University of London, Barts and The London School of Medicine and Dentistry, London, UK.
PMID: 22454520 PMCID: PMC3434861 DOI: 10.1242/jcs.100511
https://pubpeer.com/publications/2C8112926FA0AE1307E2F42FE004E3
2017 correction.
https://jcs.biologists.org/content/130/5/1016
There was an error published in J. Cell Sci. 125, 3153–3163.
In Fig. 7A, the MDA-MB-231 shPLCγ1+GSK2334470 sample was mislabelled as shPLCγ1+GSK2337740. Additionally, that image was inadvertently duplicated as the A375M shPLCγ1 image in panel B. The correct Fig. 7 is shown below. There are no changes to the figure legend, which is accurate. This error does not affect the conclusions of the study.
N.B. “This error does not affect the conclusions of the study.”
LikeLike
“If you enjoyed St George-Hyslop’s aristocratic antics, you should also have a look at the PubPeer record of another Cambridge professor, Dame Carol Mary Black, DBE, FRCP, BSD, Principal of the Newnham College.”
Fibrogenesis Tissue Repair. 2011 Mar 31;4(1):9. doi: 10.1186/1755-1536-4-9.
Thrombospondin 1 is a key mediator of transforming growth factor β-mediated cell contractility in systemic sclerosis via a mitogen-activated protein kinase kinase (MEK)/extracellular signal-regulated kinase (ERK)-dependent mechanism
Yunliang Chen # 1, Andrew Leask # 2, David J Abraham # 3, Laura Kennedy 2, Xu Shi-Wen 3, Christopher P Denton 3, Carol M Black 3, Liaquat S Verjee 4, Mark Eastwood 1
Affiliations collapse
Affiliations
1School of Life Sciences, University of Westminster, London, UK.
2Canadian Institute of Health Research Group in Skeletal Development and Remodelling, Division of Oral Biology and Department of Physiology and Pharmacology, Schulich School of Dentistry, University of Western Ontario, London, Ontario, Canada.
3Department of Inflammation, Centre for Rheumatology, University College London, London, UK.
4Kennedy Institute of Rheumatology, Imperial College London, London, UK.
#Contributed equally.
PMID: 21453480 PMCID: PMC3077328 DOI: 10.1186/1755-1536-4-9
https://pubpeer.com/publications/FCF8AC52C33CCEB2770BAA4C0AF1E7
Figure 2A. Much more similar than expected.
https://pubpeer.com/publications/FCF8AC52C33CCEB2770BAA4C0AF1E7#2
Figure 5B. Much more similar than expected.
https://pubpeer.com/publications/FCF8AC52C33CCEB2770BAA4C0AF1E7#1
Figure 6A. Much more similar than expected.
https://pubpeer.com/publications/FCF8AC52C33CCEB2770BAA4C0AF1E7#3
Figure 6A Fibrogenesis Tissue Repair. 2011 Mar 31;4(1):9 much more similar to figure 1 Am J Pathol. 2005 Dec;167(6):1699-711 after horizontal mirroring.
https://pubpeer.com/publications/FCF8AC52C33CCEB2770BAA4C0AF1E7#4
Figure 6A Fibrogenesis Tissue Repair. 2011 Mar 31;4(1):9 much more similar to figure 3A Am J Pathol. 2005 Dec;167(6):1699-711
https://pubpeer.com/publications/FCF8AC52C33CCEB2770BAA4C0AF1E7#5
2015 erratum.
http://fibrogenesis.biomedcentral.com/articles/10.1186/s13069-015-0021-1
Erratum
“After publication of this work [1], the authors became aware of some errors in the figures with respect to the loading controls for the western blots in Figure Two panel A (Figure 1 here), Figure Five panel B (Figure 2 here) and Figure Six panel A (Figure 3 here). These errors were due to genuine mistakes in generating the figures from templates and incorrect cropping of the western blot X-ray film images. These errors had no impact on the scientific conclusions of the article. The experiments reported in these figures have been repeated and new images produced.”
This does not deal with the overlap between Fibrogenesis Tissue Repair 2011 Mar 31;4(1):9. and Am J Pathol 167,1699
N.B. “These errors had no impact on the scientific conclusions of the article.”
LikeLike
J Vasc Surg. 2010 Mar;51(3):689-99. doi: 10.1016/j.jvs.2009.10.044.
Angiogenic effects of stromal cell-derived factor-1 (SDF-1/CXCL12) variants in vitro and the in vivo expressions of CXCL12 variants and CXCR4 in human critical leg ischemia
Teik K Ho 1, Janice Tsui, Shiwen Xu, Patricia Leoni, David J Abraham, Daryll M Baker
Affiliations collapse
Affiliation
1Vascular Unit, University Department of Surgery, The Royal Free and University College Medical School, University College London (Hampstead Campus), London, United Kingdom. t.ho@medsch.ucl.ac.uk
PMID: 20206813 DOI: 10.1016/j.jvs.2009.10.044
Figue 4A. Much more similar than expected.
https://pubpeer.com/publications/01640AE748C7E24D1489B7E1F71A78#6
A November 2015 correction for figure 4A has been published. http://www.jvascsurg.org/article/S0741-5214(15)00286-4/fulltext
“In the March 2010 issue of the Journal of Vascular Surgery, the article by Ho et al (Ho TK, Tsui J, Xu S, Leoni P, Abraham DJ, Baker DM. Angiogenic effects of stromal cell-derived factor-1 (SDF-1/CXCL12) variants in vitro and the in vivo expressions of CXCL12 variants and CXCR4 in human critical leg ischemia. J Vasc Surg 2010;51:689-99) contained an error in the blots of panel A in Figure 4. The corrected version of Figure 4, A is as it appears below.”
The correction does not mention the nature of the error.
The correction seems to be incomplete. It does not mention the repeated elements in the right Phos p38 panel, or in both Phos Akt panels.
LikeLike
“If you enjoyed St George-Hyslop’s aristocratic antics, you should also have a look at the PubPeer record of another Cambridge professor, Dame Carol Mary Black, DBE, FRCP, BSD, Principal of the Newnham College.”
Mol Biol Cell. 2004 Jun;15(6):2707-19. doi: 10.1091/mbc.e03-12-0902. Epub 2004 Mar 26.
Endothelin-1 promotes myofibroblast induction through the ETA receptor via a rac/phosphoinositide 3-kinase/Akt-dependent pathway and is essential for the enhanced contractile phenotype of fibrotic fibroblasts
Xu Shi-Wen 1, Yunliang Chen, Christopher P Denton, Mark Eastwood, Elisabetta A Renzoni, George Bou-Gharios, Jeremy D Pearson, Michael Dashwood, Roland M du Bois, Carol M Black, Andrew Leask, David J Abraham
Affiliation
1Centre for Rheumatology, Royal Free and University College Medical School, London NW3 2PF, United Kingdom.
PMID: 15047866 PMCID: PMC420095 DOI: 10.1091/mbc.e03-12-0902
Data figure 3 Mol Biol Cell. 2004 Jun;15(6):2707-19 very similar to data in 3 earlier papers even though the times of treatment are not the same..
https://pubpeer.com/publications/E654830F2CBAB5A490494764F4B34E#3
Internal problematic data Mol Biol Cell. 2004 Jun;15(6):2707-19
https://pubpeer.com/publications/E654830F2CBAB5A490494764F4B34E#2
LikeLike
Continuation
Zebedee
February 14, 2021
” I suspect the Cambridge University probably has same statute of limitations on research fraud as Oxford, of 3 years.”
https://medgen.medschl.cam.ac.uk/professor-eamonn-maher/
“Prior to taking up this post in 2013 he was Professor of Medical Genetics and Academic Lead for the Centre for Rare Diseases and Personalised Medicine at the University of Birmingham.”
Cancer Res. 2005 Apr 1;65(7):2690-7. doi: 10.1158/0008-5472.CAN-04-3593.
Transcriptional regulation of cyclin A2 by RASSF1A through the enhanced binding of p120E4F to the cyclin A2 promoter
Jalal Ahmed-Choudhury 1, Angelo Agathanggelou, Sarah L Fenton, Christopher Ricketts, Geoffrey J Clark,
Eamonn R Maher, Farida Latif
Affiliation
1Section of Medical and Molecular Genetics, Division of Reproductive and Child Health, Institute of Biomedical Research, University of Birmingham, Birmingham, United Kingdom.
PMID: 15805267
DOI: 10.1158/0008-5472.CAN-04-3593
Figure 1D Cancer Res. 2005 Apr 1;65(7):2690-7 very similar to figure 5B Br J Cancer 91,2071(2004).
https://pubpeer.com/publications/18B7316A533BDBB01AA9DA4E397B25#6
For Br J Cancer 91,2071(2004).See:https://pubpeer.com/publications/E307A428E33EE9D077A51C01E40180
Figure 5A. Much more similar than expected.
https://pubpeer.com/publications/18B7316A533BDBB01AA9DA4E397B25#9
LikeLike
“Science is an English colony and the rotten upper classes are busy plundering it.”
That’s quite a good analogy. Colonies can be inside country, they do not have to be abroad.
Directors of institutes are the equivalent of governors of colonies and given free rein to do what they want, even though their science may not be up to much (unreal data does not science make).
Nothing like the worker/management councils you’d find at a German research institute.
CEO and President.
https://www.icr.ac.uk/our-research/researchers-and-teams/professor-paul-workman
https://forbetterscience.com/2018/01/30/collages-by-paul-workman-from-the-golden-age-of-biological-imaging/
https://pubpeer.com/search?q=paul+workman
Previous director of same institute.
https://cancer.ucsf.edu/people/profiles/ashworth.alan
“Prior to joining UCSF in January 2015, he served as Chief Executive of the Institute of Cancer Research (ICR) and the Director of the Breakthrough Breast Cancer Research Centre in London, United Kingdom.”
https://pubpeer.com/search?q=alan+ashworth
Director of institute.
https://en.wikipedia.org/wiki/Nick_Lemoine
Hawking “gene therapy” for decades, but never achieved it.
https://forbetterscience.com/2021/02/10/the-english-science-supremacy/
https://pubpeer.com/search?q=nicholas+lemoine
To be fair to England it does occur north of the border.
https://www.ed.ac.uk/profile/prof-david-argyle
Dean of Veterinary Medicine and Head of School and is also the Deputy Head of The College of Medicine and Veterinary Medicne
https://forbetterscience.com/2021/01/04/david-argyle-can-bullying-lead-to-bad-science/
https://pubpeer.com/search?q=david+argyle
https://pubpeer.com/publications/2F2DA8CF087E5F2BB08FFF1B3DEE41
R Soc Open Sci. 2016 Sep 21;3(9):160384. doi: 10.1098/rsos.160384. eCollection 2016 Sep.
The natural selection of bad science
Paul E Smaldino 1, Richard McElreath 2
Affiliations collapse
Affiliations
1Cognitive and Information Sciences , University of California , Merced, CA 95343, USA.
2Department of Human Behavior , Ecology, and Culture, Max Planck Institute for Evolutionary Anthropology , Leipzig, Germany.
PMID: 27703703 PMCID: PMC5043322 DOI: 10.1098/rsos.160384
“Poor research design and data analysis encourage false-positive findings. Such poor methods persist despite perennial calls for improvement, suggesting that they result from something more than just misunderstanding. The persistence of poor methods results partly from incentives that favour them, leading to the natural selection of bad science. This dynamic requires no conscious strategizing-no deliberate cheating nor loafing-by scientists, only that publication is a principal factor for career advancement. Some normative methods of analysis have almost certainly been selected to further publication instead of discovery. In order to improve the culture of science, a shift must be made away from correcting misunderstandings and towards rewarding understanding. We support this argument with empirical evidence and computational modelling. We first present a 60-year meta-analysis of statistical power in the behavioural sciences and show that power has not improved despite repeated demonstrations of the necessity of increasing power. To demonstrate the logical consequences of structural incentives, we then present a dynamic model of scientific communities in which competing laboratories investigate novel or previously published hypotheses using culturally transmitted research methods. As in the real world, successful labs produce more ‘progeny,’ such that their methods are more often copied and their students are more likely to start labs of their own. Selection for high output leads to poorer methods and increasingly high false discovery rates. We additionally show that replication slows but does not stop the process of methodological deterioration. Improving the quality of research requires change at the institutional level.”
“Improving the quality of research requires change at the institutional level.”
How is that ever going to happen with practitioners of poor research methods in charge?
The heads of the U.K. research councils know what is going on. Perhaps they should go too.
LikeLike
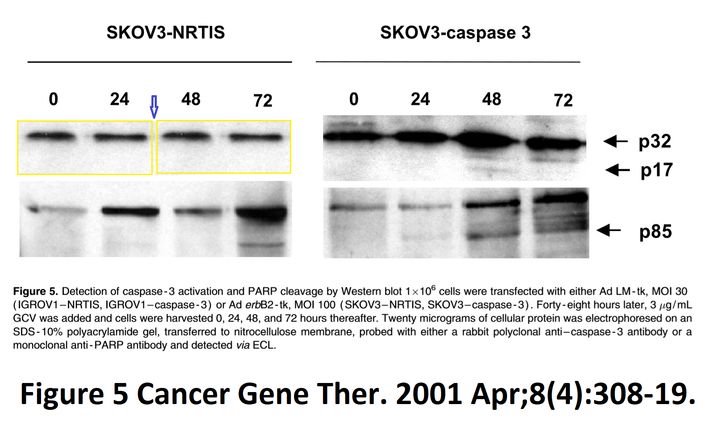
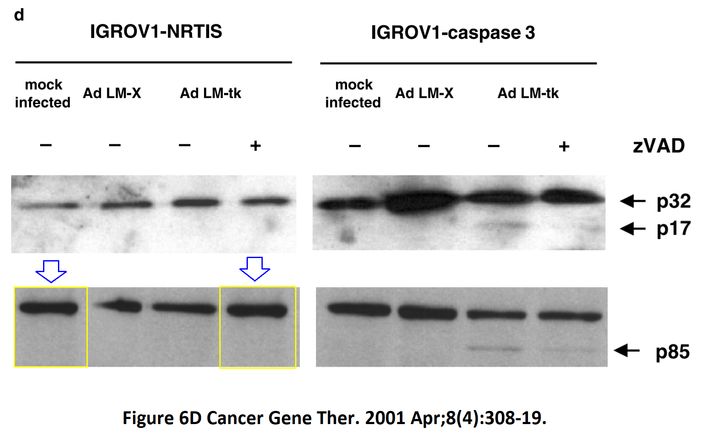
“I A McNeish, T Tenev, S Bell, M Marani, G Vassaux, N Lemoine Herpes simplex virus thymidine kinase/ganciclovir–induced cell death is enhanced by co-expression of caspase-3 in ovarian carcinoma cells Cancer Gene Therapy (2001) doi: 10.1038/sj.cgt.7700305”
Pubpeer does not really do justice to figures 5 and 6d.
Figure 5.
https://pubpeer.com/publications/E76D4CD96F41333C88C84A8EFBC57C#1
Figure 6d.
https://pubpeer.com/publications/E76D4CD96F41333C88C84A8EFBC57C#2
LikeLike
PLoS One. 2018 May 22;13(5):e0196528. doi: 10.1371/journal.pone.0196528. eCollection 2018.
TDP-43 causes neurotoxicity and cytoskeletal dysfunction in primary cortical neurons
Pranetha Baskaran 1, Christopher Shaw 2, Sarah Guthrie 1
Affiliations collapse
Affiliations
1Department of Developmental Neurobiology, King’s College, Guy’s Campus, London, United Kingdom.
2Maurice Wohl Clinical Neuroscience Institute, King’s College, London, United Kingdom.
PMID: 29787572 PMCID: PMC5963761 DOI: 10.1371/journal.pone.0196528
Is there an explanation for figure 4?
Figure 4A.https://pubpeer.com/publications/043CD581FC7EE5FCB7B67E52F8E3A9#6
Figure 4B.https://pubpeer.com/publications/043CD581FC7EE5FCB7B67E52F8E3A9#5
Figure 4C.https://pubpeer.com/publications/043CD581FC7EE5FCB7B67E52F8E3A9#3
Figure 4D.https://pubpeer.com/publications/043CD581FC7EE5FCB7B67E52F8E3A9#4
LikeLike
J Inflamm (Lond). 2010 Jul 16;7:33. doi: 10.1186/1476-9255-7-33.
Cigarette Smoke Exposure Alters mSin3a and Mi-2alpha/beta Expression; implications in the control of pro-inflammatory gene transcription and glucocorticoid function
John A Marwick 1, Christopher S Stevenson, Kian Fan Chung, Ian M Adcock, Paul A Kirkham
Affiliation
1Section of Airways Disease, National Heart & Lung Institute, Imperial College London, UK. john.Marwick@ed.ac.uk.
PMID: 20637110
PMCID: PMC2912298
DOI: 10.1186/1476-9255-7-33
https://pubpeer.com/publications/E3DBF1694E7948A3D524B494FF7203
Figures 1 and 2. Much more similar than expected.
Figures 2 and 4. Much more similar than expected.
LikeLike
Am J Physiol Lung Cell Mol Physiol. 2005 Jan;288(1):L68-76. doi: 10.1152/ajplung.00156.2004. Epub 2004 Sep 17.
Regulation of TGF-beta 1-induced connective tissue growth factor expression in airway smooth muscle cells
Shaoping Xie 1, Maria B Sukkar, Razao Issa, Ute Oltmanns, Andrew G Nicholson, Kian Fan Chung
Affiliations collapse
Affiliation
1Thoracic Medicine, National Heart and Lung Institute, Imperial College, Dovehouse St., London SW3 6LY, UK.
PMID: 15377500 DOI: 10.1152/ajplung.00156.2004
https://pubpeer.com/publications/4CD6A828FA0A0ACC1AFAD4BC80BEC6
Figures 3A and 4D. Much more similar than expected.
Figure 8B. Much more similar than expected.
Figures 3A, 4D, 7A and 9B.
Much more similar than expected.
Figures 3A and 4A. Much more similar than expected.
LikeLike
J Immunol. 2007 Jun 1;178(11):7366-75. doi: 10.4049/jimmunol.178.11.7366.
Corticosteroid inhibition of growth-related oncogene protein-alpha via mitogen-activated kinase phosphatase-1 in airway smooth muscle cells
Razao Issa 1, Shaoping Xie, Nadia Khorasani, Maria Sukkar, Ian M Adcock, Kang-Yun Lee, Kian Fan Chung
Affiliations collapse
Affiliation
1Experimental Medicine, Airway Studies Section, National Heart and Lung Institute, Imperial College, London, United Kingdom.
PMID: 17513787
https://pubpeer.com/publications/6F0D881352809757414BC333BF69F1
Figures 4C and 7B. Much more similar than expected.
LikeLike
Am J Respir Cell Mol Biol. 2006 Oct;35(4):407-14. doi: 10.1165/rcmb.2006-0012OC. Epub 2006 May 11.
Neutrophil-Derived Elastase Induces TGF-β1 Secretion in Human Airway Smooth Muscle via NF-κB Pathway
Kang-Yun Lee , Shu-Chuan Ho , Horng-Chyuan Lin , Shu-Min Lin , Chien-Ying Liu , Chien-Da Huang , Chun-Hua Wang , Kian Fan Chung , and Han-Pin Kuo
-Author Affiliations
Department of Thoracic Medicine, Chang Gung Memorial Hospital, Chang Gung University College of Medicine, Taipei, Taiwan; and Department of Thoracic Medicine, National Heart and Lung Institute, Imperial College, London, United Kingdom
https://doi.org/10.1165/rcmb.2006-0012OC
https://pubpeer.com/publications/9DEEB4ABCBC1A564871A94934DFB50
Figure 6. Much more similar than expected.
LikeLike
Am J Respir Cell Mol Biol. 2011 Oct;45(4):746-52. doi: 10.1165/rcmb.2010-0304OC. Epub 2011 Feb 4.
Hydrogen sulfide inhibits proliferation and release of IL-8 from human airway smooth muscle cells
Mark M Perry 1, Christopher K Hui, Matthew Whiteman, Mark E Wood, Ian Adcock, Paul Kirkham, Charalambos Michaeloudes, Kian Fan Chung
Affiliation
Experimental Studies, National Heart and Lung Institute, Imperial College London, London, United Kingdom.
PMID: 21297080 PMCID: PMC3577139 DOI: 10.1165/rcmb.2010-0304OC
Problematic data figures 4B and 6A. Much more similar after horizontal flip and slight tilting than expected.
https://pubpeer.com/publications/00164861E2812B06BF811763961528
LikeLike
J Inflamm (Lond). 2010 Feb 13;7(1):11. doi: 10.1186/1476-9255-7-11.
Cigarette smoke regulates VEGFR2-mediated survival signaling in rat lungs
John A Marwick 1, Indika Edirisinghe, Gnanapragasam Arunachalam, Christopher S Stevenson, William Macnee, Paul A Kirkham, Irfan Rahman
Affiliation
1National Heart and Lung Institute, Imperial College London, UK. p.kirkham@imperial.ac.uk.
PMID: 20205917 PMCID: PMC2831890 DOI: 10.1186/1476-9255-7-11
https://pubpeer.com/publications/01BAE656F97EED0947C378F78C5539#1
Figure 7. Much more similar than expected.
https://imgur.com/4W3D5X8
Disclaimer: These potential image duplications have been detected automatically using ImageTwin http://dx.doi.org/10.1145/3019612.3019875 Although results have been curated, the automatic algorithm may have generated false positive results and cannot take into account all scientific explanations for apparent duplications like stripped and reprobed blots, time series, serial sections and reused controls.
Comment #2.
https://pubpeer.com/publications/01BAE656F97EED0947C378F78C5539#2
LikeLike
Senior author J Inflamm (Lond). 2010 Feb 13;7(1):11. doi: 10.1186/1476-9255-7-11, Irfan Rahman was first noticed in this retraction watch post.
https://retractionwatch.com/2012/12/28/scientists-retract-paper-because-theyre-not-satisfied-with-the-quality-of-some-of-the-data/
https://www.liebertpub.com/doi/abs/10.1089/ars.2012.5170?prevSearch=Retraction&searchHistoryKey=
Antioxidants & Redox SignalingVol. 18, No. 12Retraction
RETRACTION: Cigarette Smoke–Induced Oxidative/Nitrosative Stress Impairs VEGF- and Fluid Shear Stress–Mediated Signaling in Endothelial Cells. Edirisinghe I, Arunachalam G, Wong C, Yao H, Rahman A, Phipps RP, Jin ZG, and Rahman I. Antioxid Redox Signal 12: 1355–1369, 2010
Published Online:14 Mar 2013https://doi.org/10.1089/ars.2012.5170
“The corresponding author has sought retraction of this work from ARS. The author statement is copied below: We, the authors, wish to retract “Cigarette Smoke–Induced Oxidative/Nitrosative Stress Impairs VEGF- and Fluid Shear Stress–Mediated Signaling in Endothelial Cells” by Edirisinghe et al. (Antioxid Redox Signal 12: 1355–1369; DOI:10.1089/ars.2009.2874) because we are not satisfied with the quality of some of the data presented in the article. Overall, however, the data are reproducible and the conclusions drawn were not affected. We apologize for any inconvenience this may have caused to the readers.”
Despite: ” We, the authors, wish to retract.. because we are not satisfied with the quality of some of the data presented in the article”,
comes, ” the conclusions drawn were not affected”.
https://www.urmc.rochester.edu/people/23899711-irfan-rahman:
Under publications.
6/15/2010
Edirisinghe I, Arunachalam G, Wong C, Yao H, Rahman A, Phipps RP, Jin ZG, Rahman I. “Cigarette-smoke-induced oxidative/nitrosative stress impairs VEGF- and fluid-shear-stress-mediated signaling in endothelial cells.” Antioxidants & redox signaling.. 2010 Jun 15; 12(12):1355-69.
LikeLike
According to the penultimate author, Ian Adcock, there is an “internal review” into Respir Res. 2018 May 9;19(1):85.
See: https://pubpeer.com/publications/31921A79606A411CA361871C7D1308#6
Full Pubpeer comments here: https://pubpeer.com/publications/31921A79606A411CA361871C7D1308
Respir Res. 2018 May 9;19(1):85. doi: 10.1186/s12931-018-0788-x.
The anti-proliferative and anti-inflammatory response of COPD airway smooth muscle cells to hydrogen sulfide
Mark M Perry 1, Bernadett Tildy 2, Alberto Papi 3, Paolo Casolari 3, Gaetano Caramori 4, Karen Limbert Rempel 5, Andrew J Halayko 5, Ian Adcock 2, Kian Fan Chung 2
Affiliations
1School of Pharmacy & Biomedical Sciences, University of Portsmouth, St. Michael’s Building, White Swan Road, Portsmouth, PO1 2DT, UK. mark.perry@port.ac.uk.
2Airways Disease, National Heart and Lung Institute, Imperial College, London & Royal Brompton NIHR Biomedical Research Unit, London, SW3 6LY, UK.
3Sezione di Medicina Interna e Cardiorespiratoria, Centro Interdipartimentale per lo Studio delle Malattie Infiammatorie delle Vie Aeree e Patologie Fumo-Correlate (CEMICEF, formerly termed Centro di Ricerca su Asma e BPCO), Università di Ferrara, Ferrara, Italy.
4Unità Operativa Complessa di Pneumologia, Dipartimento di Scienze Biomediche, Odontoiatriche e delle Immagini Morfologiche e Funzionali (BIOMORF), Università degli Studi di Messina, Messina, Italy.
5Departments of Internal Medicine & Physiology, Respiratory Hospital, Sherbrook Street, Winnipeg, MB, R3A 1R9, Canada.
PMID: 29743070 PMCID: PMC5944010 DOI: 10.1186/s12931-018-0788-x
Figure 3a. Much more similar than expected.
Data very similar to day in an earlier paper.
LikeLike
2020 correction for:
BMC Pulm Med. 2017 Apr 21;17(1):68. doi: 10.1186/s12890-017-0413-7.
Water-pipe smoke condensate increases the internalization of Mycobacterium Bovis of type II alveolar epithelial cells (A549)
Esmaeil Mortaz 1 2, Shamila D Alipoor 1 3 4, Masoud Movassaghi 5, Mohammad Varahram 6, Jahangir Ghorbani 1, Gert Folkerts 7, Johan Garssen 7 8, Ian M Adcock 9 10
Affiliations collapse
Affiliations
1Clinical Tuberculosis and Epidemiology Research Center, National Research Institute of Tuberculosis and Lung Diseases (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran.
2Department of Immunology, Faculty of Medicine, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
3Institute of Medical Biotechnology, Molecular Medicine Department, National Institute of Genetic Engineering and Biotechnology (NIGEB), Tehran, Iran.
4Department of Biotechnology, School of Advanced Technologies in Medicine, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
5Department of Pathology and Laboratory Medicine, University of California, Los Angeles (UCLA), Los Angeles, CA, USA. MMovassaghi@mednet.ucla.edu.
6Mycobacteriology Research Center (MRC) National Research Institute of Tuberculosis and lung Diseases (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran.
7Division of Pharmacology, Utrecht Institute for Pharmaceutical Sciences, Faculty of Science, Utrecht University, Utrecht, The Netherlands.
8Nutricia Research Centre for Specialized Nutrition, Utrecht, The Netherlands.
9Cell and Molecular Biology Group, Airways Disease Section, National Heart and Lung Institute, Imperial College London, Dovehouse Street, London, UK.
10Priority Research Centre for Healthy Lungs, Hunter Medical Research Institute, The University of Newcastle, Newcastle, New South Wales, Australia.
PMID: 28431548 PMCID: PMC5401461 DOI: 10.1186/s12890-017-0413-7
So many pairs of author eyes, yet Actinopolyspora Biskrensis spotted what turned out to be image duplication.
https://pubpeer.com/publications/73F6989B11162BC1A5CB1026F96F9A#1
Figures 4a and 4c duplicated.
2020 correction. https://bmcpulmmed.biomedcentral.com/articles/10.1186/s12890-020-01244-1
Published: 22 September 2020
Correction to: BMC Pulm Med 17, 68 (2017)
https://doi.org/10.1186/s12890-017-0413-7
Following publication of the original article [1], the authors that there is an error in the FACs plots detailed in Fig. 4.
The error is that the plot of panel ‘C’ has been duplicated as the plot of panel ‘A’.
Please see the corrected figure in this correction article.
The authors apologize for any inconvenience caused.
LikeLike
Kian Fan Chung rhymes with two-for-one.
Eur Respir J. 2004 Jun;23(6):805-12. doi: 10.1183/09031936.04.00104304.
Effect of smoking on MAP kinase-induced modulation of IL-8 in human alveolar macrophages
AUTHOR INFORMATION https://erj.ersjournals.com/content/23/6/805.article-info
A. Koch 1 , M. Giembycz 2 , R.G. Stirling 2 , S. Lim 2 , I. Adcock 2 , K. Waßermann 1 , E. Erdmann 1 and K.F. Chung 2
1Dept of Pneumology, Clinic III for Internal Medicine, University of Cologne, Cologne, Germany and 2Thoracic Medicine, Imperial College at National Heart & Lung Institute, London, UK
A. Koch, University of Cologne, Medizinische Klinik III, Dept of Pneumology, Joseph-Stelzmann-Str. 9, 50924 Köln, Germany. Fax: 49 221 4787038 E‐mail: andrea.koch@uni‐koeln.de
PMID: 15218990 DOI: 10.1183/09031936.04.00104304
Am J Respir Cell Mol Biol. 2004 Mar;30(3):342-9. doi: 10.1165/rcmb.2003-0122OC. Epub 2003 Jul 18.
Mitogen-activated protein kinase modulation of nuclear factor-kappaB-induced granulocyte macrophage-colony-stimulating factor release from human alveolar macrophages
Andrea Koch 1, Mark Giembycz, Kazuhiro Ito, Sam Lim, Elen Jazrawi, Peter J Barnes, Ian Adcock, Erland Erdmann, K Fan Chung
Affiliations collapse
Affiliation
1University of Cologne, Medizinische Klinik III, Department of Pneumology, Joseph-Stelzmann-Str. 9, 50924 Köln (Cologne), Germany. andrea.koch@uni-koeln.de
Data figure 4a Eur Respir J. 2004 Jun;23(6):805-12 very similar to data figure 3B Am J Respir Cell Mol Biol. 2004 Mar;30(3):342-9.
LikeLike
Kian Fan Chung rhymes with two-for-one.
Am J Respir Crit Care Med. 2001 Oct 15;164(8 Pt 1):1403-9. doi: 10.1164/ajrccm.164.8.2010002.
Interleukin-5 induces CD34(+) eosinophil progenitor mobilization and eosinophil CCR3 expression in asthma
R G Stirling 1, E L van Rensen, P J Barnes, K F Chung
Affiliation
1Department of Thoracic Medicine, National Heart and Lung Institute, Imperial College School of Medicine, London, United Kingdom.
PMID: 11704586 DOI: 10.1164/ajrccm.164.8.2010002
https://www.atsjournals.org/doi/full/10.1164/ajrccm.166.4.16641
Duplicate publication
K. Fan Chung , Elizabeth L. J. van Rensen , Robert G. Stirling , and Peter J. Barnes
-Author Affiliations
National Heart and Lung Institute London, United Kingdom
https://doi.org/10.1164/ajrccm.166.4.16641 PubMed: 12186832
To the Editor :
We have recently published two manuscripts on the effects of interleukin (IL)-5 administration to patients with mild asthma, one in the American Journal of Respiratory and Critical Care Medicine (1), and the other in Thorax (2). We are addressing this letter to the Editors of both journals. The data described in both articles have been obtained from one experiment performed in the same group of patients with mild asthma. Whereas the article in the AJRCCM (1) reports primarily on the phenotypic changes in circulating blood eosinophils and CD34+ cells, the Thorax article (2) describes the changes in the airways with measurements of bronchial responsiveness and of eosinophil counts in sputum, together with eotaxin levels in the serum. Both manuscripts, however, detail the changes in blood eosinophils counts and serum IL-5 changes derived from these nine patients, which constitutes a partial duplication of the data described in the two papers. At the time of submission of the manuscripts to the two journals, we did not inform the respective journals of the existence and submission of the other manuscript.
We would also like to take the opportunity of pointing out one mistake and two differences in the two manuscripts: (1) Table 1 in Thorax contains mistakes regarding the FEV1 values; Table 1 in the AJRCCM article shows the correct values. (2) The AJRCCM article quotes the median values in the text, whereas mean values are plotted in Figure 2, and the Thorax article shows the geometric mean. (3) The blood eosinophil counts were analyzed in the AJRCCM paper as paired t tests, whereas in the Thorax paper, we used repeated measures analysis of variance (as recommended by a reviewer), which does not provide significant differences in eosinophil counts.
We recognize that it has been an error of judgement on our part in having the experimental data from this study reported in two separate manuscripts, and also in not informing you (and the reviewers) of the existence of these two separate manuscripts at the time of submission. We have not intentionally set out to duplicate publication of our experimental data and regret that this has happened. We also would like to apologize for the concern that this may cause to the high reputation of both journals.
References
Stirling RG, van Rensen ELJ, Barnes PJ, Chung KF. Interleukin-5 induces CD34+ eosinophil progenitor mobilization and eosinophil CCR3 expression in asthma. Am J Respir Crit Care Med 2001;164:1403–1409.
van Rensen ELJ, Stirling RG, Scheerens J, Staples K, Sterk PJ, Barnes PJ, Chung KF. Evidence for systematic rather than pulmonary effects of interleukin-5 administration in asthma. Thorax 2001;56:935–940.
LikeLike
PLoS Med. 2009 May 12;6(5):e1000076. doi: 10.1371/journal.pmed.1000076. Epub 2009 May 19.
Suppression of GATA-3 nuclear import and phosphorylation: a novel mechanism of corticosteroid action in allergic disease
Kittipong Maneechotesuwan 1, Xin Yao, Kazuhiro Ito, Elen Jazrawi, Omar S Usmani, Ian M Adcock, Peter J Barnes
Affiliation
1Airway Disease Section, National Heart and Lung Institute, Imperial College, London, United Kingdom.
PMID: 19436703 PMCID: PMC2674207 DOI: 10.1371/journal.pmed.1000076
https://pubpeer.com/publications/77F84816A35F0EE53CD2713FAE4E35
Figure 1 PLoS Med. 2009 May 12;6(5):e1000076 very similar to
figure 1 J Immunol. 2007 Feb 15;178(4):2491-8.
2018 correction.
https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1002657
The PLOS Medicine Editors received comments from readers suggesting that the Western blot demonstrating nuclear translocation of GATA-3 in response to anti-CD3/CD28 stimulation in panel A of Fig 1 was labeled incorrectly. The authors have re-examined the original experimental images and would like to correct Fig 1. Please see the corrected version of panel A in Fig 1 below. The conclusions of the paper are unaffected by this correction.
Figure 2B PLoS Med. 2009 May 12;6(5):e1000076 very similar to
figure 6B J Immunol. 2007 Feb 15;178(4):2491-8.
LikeLike
J Immunol. 2007 Feb 15;178(4):2491-8. doi: 10.4049/jimmunol.178.4.2491.
Regulation of Th2 cytokine genes by p38 MAPK-mediated phosphorylation of GATA-3
Kittipong Maneechotesuwan 1, Yao Xin, Kazuhiro Ito, Elen Jazrawi, Kang-Yun Lee, Omar S Usmani, Peter J Barnes, Ian M Adcock
Affiliation
1Section of Airway Disease, National Heart and Lung Institute, Imperial College, London, United Kingdom.
PMID: 17277157 DOI: 10.4049/jimmunol.178.4.2491
https://pubpeer.com/publications/76B6FDEB0C24588364E9C8BBFEED55
Figures 4B and 4C much more similar than expected.
Figures 4A and 4B much more similar than expected.
LikeLike