This is a new episode of the data manipulation affair around Weizmann Institute of Science in Rehovot, Israel (and another guest post by “Smut Clyde“), with the hope that Israeli researchers and their state officials finally step in and investigate what goes on in this institute, supported by external experts from the academic community. There are many good and honest researchers working at Weizmann, the dishonest deeds which used to happen (and maybe still happen at Weizmann) should never throw a shadow upon their work. This can only be achieved by an open debate in the scientific community worldwide and a large, independent investigation inside Weizmann.
My earlier article about Weizmann’s “stars” of research integrity, and the follow-up guest post by Smut Clyde, prompted a wider scrutiny by internet sleuths on PubPeer. One of them was the well-known pseudonymous Claire Francis. In this case presented below, while Ofer Lider, associate professor of Immunology at Weizmann, was dying from leukaemia, his dedicated colleagues were apparently secretly stuffing manipulated data into his publications. They continued to do so even after his death in 2004, and now Lider papers are being plucked apart on PubPeer. There are many co-authors, and the scientific narrative mostly circles around a “visionary” diabetes cure, the substance DiaPep277, a peptide fragment of the ubiquitous heatshock protein Hsp60.
What was done to the scientific and human legacy of the immunologist Ofer Lider, is the basically academic equivalent of urinating of someone’s literal grave. If this won’t make Weizmann leadership feel shame, I do not know what would. It was a travesty of two retracted papers and a meeting abstract of a DiaPep277 clinical trial by the trainwreck company Andromeda Biotech, and it made world news in 2014. What came out only now, thanks to the sleuths of PubPeer, is that this scandal of clinical trial statistics was supported by a preclinical cornucopia of rigged western blots made at Weizmann, which all served the purpose of delivering a promise of a diabetes “vaccine”.
Those are the main protagonists:
- Irun Cohen, Ofer Lider’s former PhD advisor, now Weizmann’s professor emeritus. He is a visionary who postulated that the immune system is actually a computer. Cohen’s great mind also birthed the idea for DiaPep277, after he first appointed Hsp60 to be the immunologic cause of diabetes, based apparently on his educated guess, and then set up to prove this experimentally, and in a clinical trial in Israel. Cohen used to be listed as clinical advisory board member at Andromeda Biotech.
- Dana Elias, former PhD student of Cohen’s. Until 2014, where she was found guilty of “serious misconduct”, programme director at Andromeda and now “consultant,self-employed”.
- Itamar Raz, clinical diabetologist, former Head of the Diabetes Unit at Hadassah University Hospital, presently head of the Israeli National Council of Diabetes. According to the three retraction notices (here, here and here), Raz was a hapless victim of misconduct by his coauthors from Andromeda, together with Cohen he declared: “we were not involved in the assembly of the raw data”. Yet Raz used to chair the Steering Committee of Andromeda for this very study, in fact he was listed as the official Principal Investigator for the DiaPep277 clinical trial.
- Alexandra Zanin-Zhorov, PhD student in Ofer Lider’s lab, first author on all Lider’s papers flagged on PubPeer, now according to her LinkedIn profile Vice-President at the pharma company Kadmon In New York, though the company’s website doesn’t list her anywhere. Cohen is presently Kadmon’s scientific advisor.

Pictures from an Inhibition, by Smut Clyde
Here is a foretaste of what is to come, to whet your appetite during a lengthy exposition.
This post returns to the theme of illustrations in the biomedical literature. Specifically, to the theme of electrophoresis gels – which are both the raw material of research, as well as its final conclusive data.
A long series of science-fiction thrillers teaches us that the name ‘Andromeda’ has bad associations; things always end badly. But the lesson was lost on the people who created Andromeda Biotech Ltd. to commercialise a synthetic polypeptide, DiaPep277, on behalf of the Weizmann Institute.
I am loath to spoil the suspense, but I should say here at the start that DiaPep277 proved in the end to have no clinical value in slowing the progression of Type-1 Diabetes (T1D). And there is no shame in that, for most promising new drugs and interventions leave their efficacy behind them in the laboratory when they advance out into clinical trials. This ensures that tabloid journalists have a constant supply of ‘New Wonder Cure for X’ to write about, and provides pharmaceutical companies with an excuse to charge 10-fold when one of their new drugs is (initially) sellable.
So T1D is an autoimmune disease, caused when a hyper-sensitive immune system targets the insulin-producing ß-cells of the pancreas for detruction. The idea was that the specific antigen they over-react to is HSP-60 — one of the family of Heat Shock Proteins (which is totally the name of my new Cramps tribute band). HSP-60 is ubiquitous in human tissues, so it was not clear why an irritable immune system should take special umbrage to its presence in the pancreas; nor was it obvious why stimulating the immune response, with injections of the synthetic analog DiaPep, should reduce the damage it wreaks.
This leads us to Irun Cohen of the Department of Immunology of the Weizmann Institute, and his radical maverick Immunological Homunculus paradigm. The details of this escape me, but other laboratories have been slow to take it up, rather like Cap’n Redbeard Rum and his radical views on maritime crew levels. Readers may be familiar with the Weizmann Institute.
Now Professor Cohen is the first to admit that his ‘autoimmune vaccination’ concept is rather out there, but he is all about the Conceptual Paradigm Shift, and the Big-Picture Perspective, and the Interdisciplinary Out-of-the-Box Thinking. Also the Bold Metaphors.
“How did we arrive at the concept of vaccination therapy and how did we suspect that peptide p277 might turn out to be an effective vaccine. How does one dare go from mice to people?”
“The idea of vaccination for autoimmune disease was quite foreign to the thinking current at the time among autoimmunologists; the vaccination concept reflected my initial training in infectious disease”.
So far he has not been driven to confront the scientific establishment from the turret of a castle laboratory, brandishing a fist at the storm-clouds and shouting “You hidebound fools! You mocked my theories and robbed me of my Nobel! But I’ll show you all!!” Let us hope that it does not progress to that.
In company with his PhD student and protegée Dana Elias, Cohen developed his insight into the “HSP-autoimmunity-causes-T1D” theory, and from there to this 277 polypeptide — tailored to contain the antigenic flags of HSP-60, but boiled down to only 24 amino acids for easy production from vats of recombinant E. coli. There was a 1991 mouse-model paper in PNAS and a 1994 letter to Lancet. Dr Elias was one of six scientists employed by Andromeda Biotech who declined to comment on or cooperate with the retraction of DiaPep-clinical-trial papers in late 2014.
But I have got ahead of myself there. The company Peptor had been spun off from the Weizmann in 1993, vested with the intellectual-property rights to DiaPep277 and tasked with bringing it to market. It sold the rights to Avantis in 2002 but re-acquired them in 2003. In 2004 Peptor begat DeveloGen, which in 2007 begat Andromeda Biotech.
Initial-stage trials from several groups (2007-2009) agreed that DiaPep277 had no serious side-effects on recipients, and did provoke responses of some kind from immune systems, although any remission in the progress of T1D was difficult to discern (prompting efforts to broaden the scope of potential patients, and simultaneously to narrow it in search of some subgroup who would benefit). There was enough evidence to begin large-scale multi-centre trials, with distinguished diabetologist Itamar Raz recruited by now to oversee the project. And there was gloating from the Weizmann over the vindication and the discomfiture of the skeptics and nay-sayers.

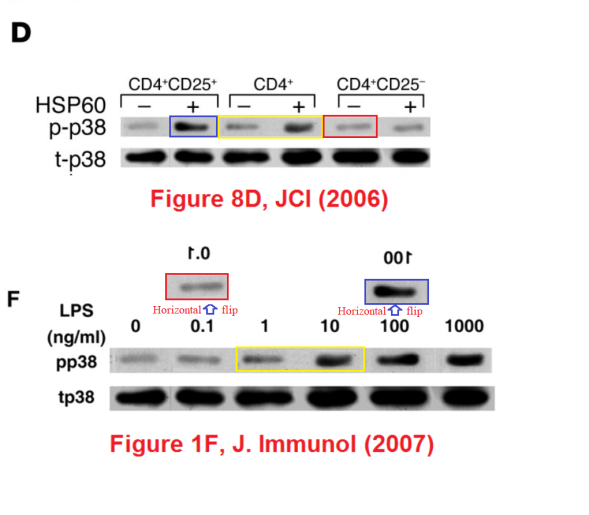
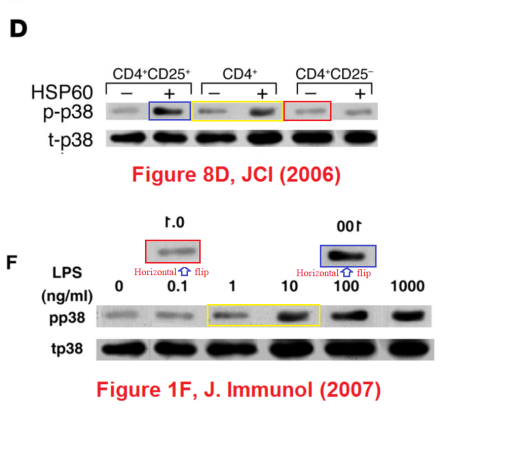
Meanwhile a group at the Department of Immunology had been tasked with finding (a) evidence to support the rickety rationale for vaccinating against diabetes, and (b) specific cellular mechanisms through which DiaPep277 might work the immunomodulatory magic expected from it, and a series of papers emerged. The artistic quality of the illustrations for these papers brought them to the attention of the anonymous whistle-blowers, data-integrity extremists and blackboard monitors who frequent PubPeer. Now, with the backstory out of the way, I will plagiarise their contributions.Perhaps we should begin with “Heat shock protein 60 enhances CD4+ CD25+ regulatory T cell function via innate TLR2 signaling” (Zanin-Zhorov et al., J. Clinical Investigation, 2006), for it became the subject of press releases from the Weizmann to showcase the team (admitting in passing that the development of DiaPep had hitherto been driven by intuition and aspirational metaphors). Contrast adjustments to Figures 8C and 8D expose changes in the background, delineating the second and first lanes of the p-Pyk2 and t-p38 bands respectively: suggesting that these lanes were not originally part of the same experimental session as their neighbours, but were spliced in digitally.
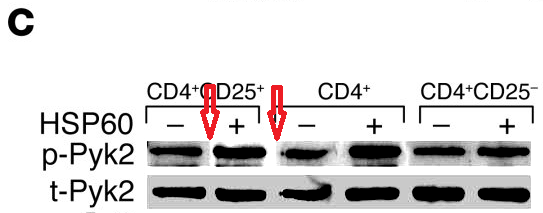
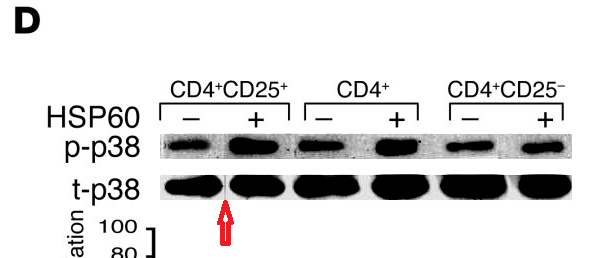
These bands show the amounts of a particular protein in its phosphorylated form, and also unphosphorylated, so that the difference in signal of the former can be compared with the equal amounts of the latter present inside the cells. You learn how much of the protein is phosphorylated, and in this case active, under different conditions. Lanes distinguish conditions: three types of T cells, with and without added HSP-60. The splice lines also delineate the 5th lane of the p-ERK lane in Figure 8E, purportedly from CD4+CD25- cells. Except it is a copy of the first lane (CD4+CD25+ cells), flipped horizontally to reduce its self-similarity. The point of the exercise had been to demonstrate the equivalence of these two cell types in the absence of HSP-60, and one has to conclude that the actual evidence was not strong enough.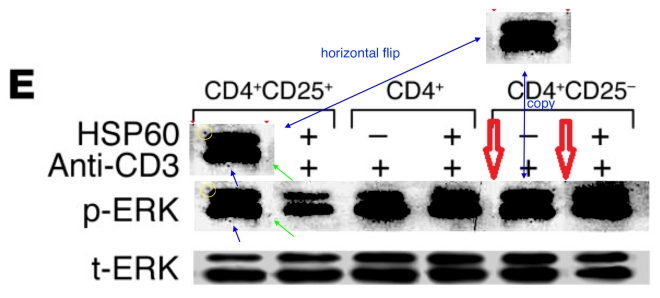
Or perhaps we should begin in media res with “Heat Shock Protein 60 activates cytokine-associated…” (Zanin-Zhorov et al., J. Immunology, 2007), for it is a tour-de-force, on which the entire panoply of cosmetological enhancements has been deployed. The construction of the illustrative bands involved the duplication of numerous proteins, variously transformed. With their duplicate lanes only horizontally flipped, Figures 5F and 5H seem anodyne.
Figure 6A reveals another horizontally-flipped copy in its SOCS3 band, while in 6D, the overall darkness of the 6th lane of the SOCS3 band has been increased by a selective over-exposure, as is revealed by adjusting the contrast for that panel.
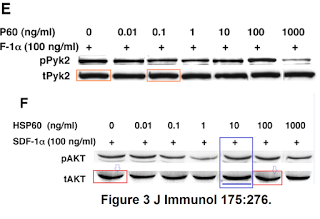
In Figures 3E and 3F, as well as the replication of lanes within their respective reference bands tPyk2 and tAKT, the sixth lane of tAKT appears to be a copy of the corresponding pAKT lane… over-exposed, and with a crude attempt to erase the supernumerary streak just beneath it (characteristic of pAKT).
A whole three-lane neighbourhood was rotated 180° to construct the tMLC reference band in Figure 4B, while 4A can boast a duplicated lane in the pMLC band and a slightly-rotated duplicate tMLC lane.

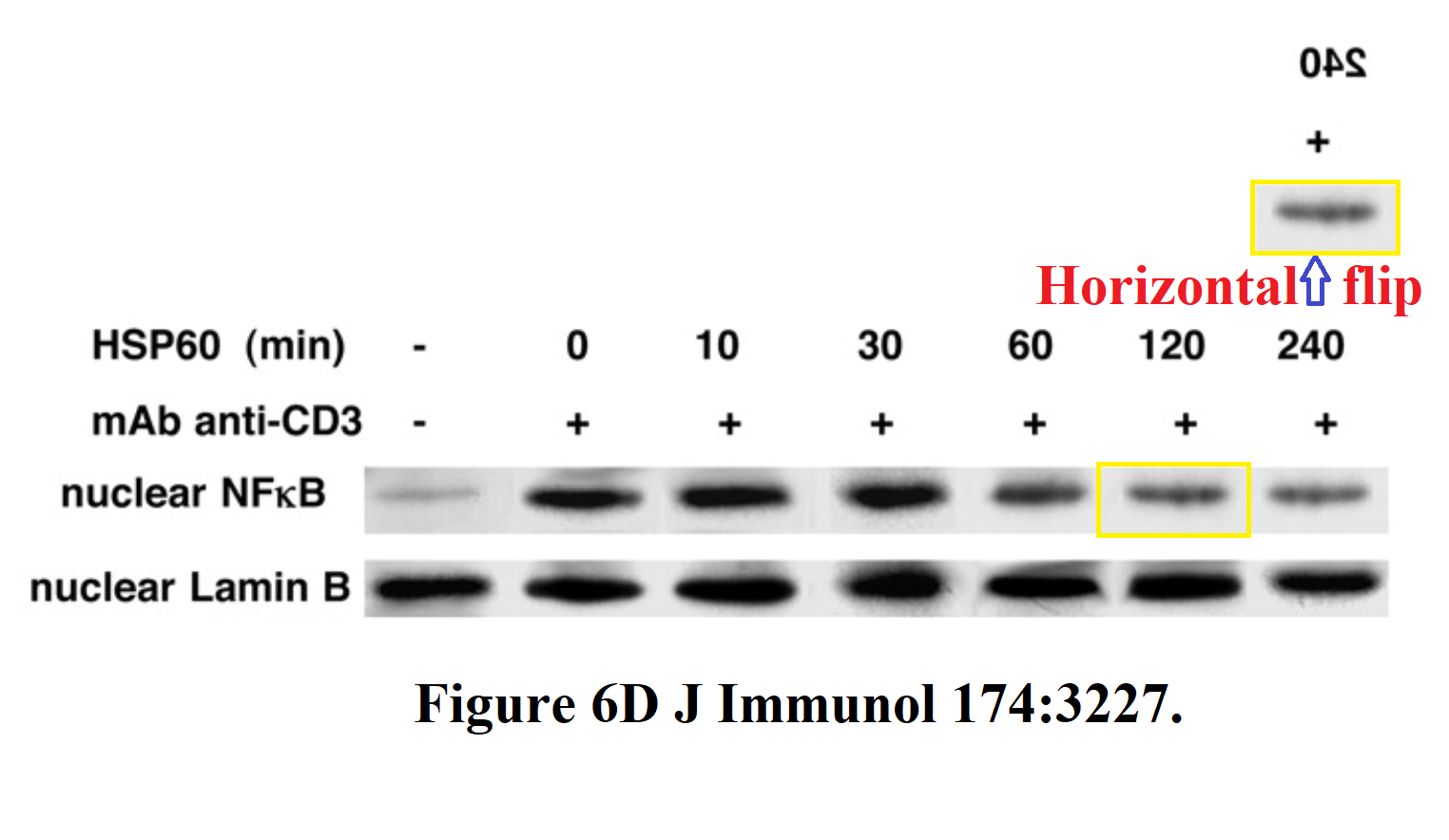
The same panoply is on show in “Heat shock protein 60 inhibits Th1-mediated hepatitis
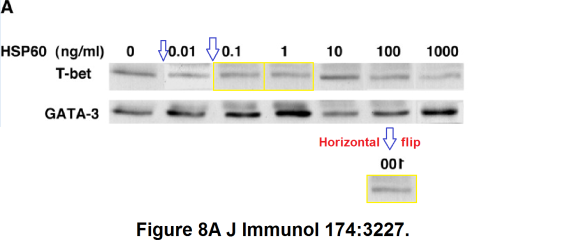
model…” (Zanin-Shorov et al., J of Immunology, 2005). Attention first turned to splices around the second lane in the T-bet band of Figure 8A, which appears “looks slotted into panel” in the manner of intarsia; then on the fact that three of the other lanes are identical (though mirrored in one appearance).
Further showcases of extensive lane re-use are Figures 6A and 6E:
Figures 6B and 6D display only a single flipped lane each to make up their full complement:
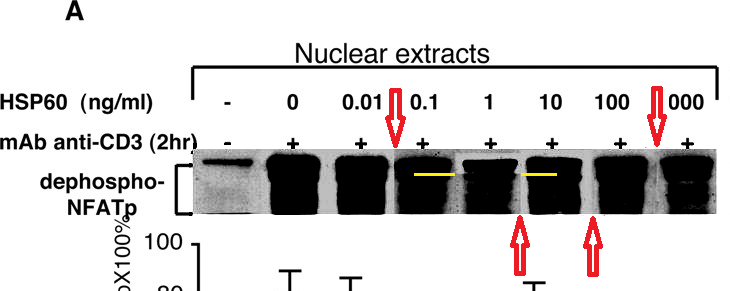
Figures 7A, 7B, 7C provide some visual variety. Here the point of each lane is the amount of phosphorylated protein expressed in the top streak, relative to the dephosphorylated protein in all the other streaks, so the streaks are not widely resolved and the lanes resemble a collection of flowerpots. On close inspection, some of these flowerpots appear twice or even thrice within and between the separate panels (reflected and sometimes at longer exposures), representing different conditions — sometimes purported as extracts of cell nuclei and sometimes of cytoplasm.
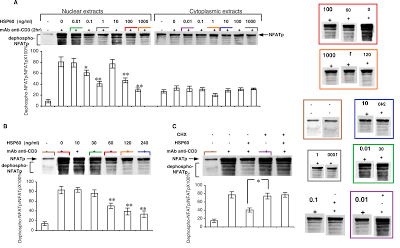 So Figure 7 was necessarily assembled like a stamp collection, in Photoshop rather than in the laboratory, and it was no surprise that adjusting the contrast would reveal vertical splices around separate flowerpots.
So Figure 7 was necessarily assembled like a stamp collection, in Photoshop rather than in the laboratory, and it was no surprise that adjusting the contrast would reveal vertical splices around separate flowerpots.
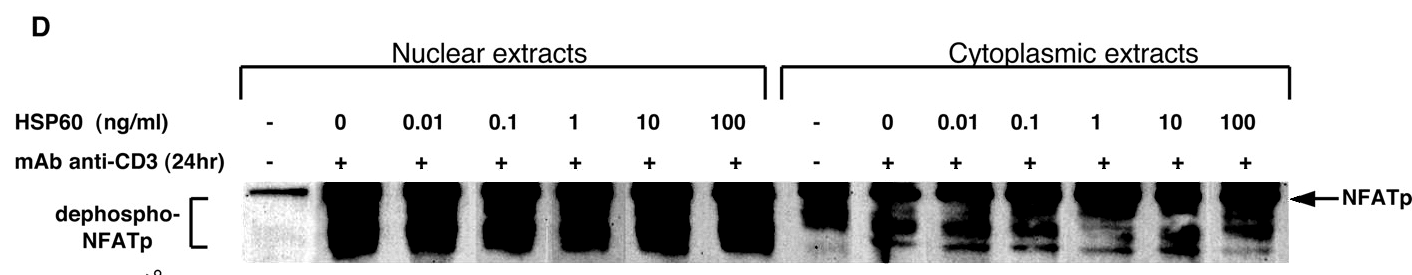
It was a surprise that one lane would have horizontal splice lines, indicative of a top streak that had been underexposed relative to the rest of the lane (lowering the ratio of protein content).
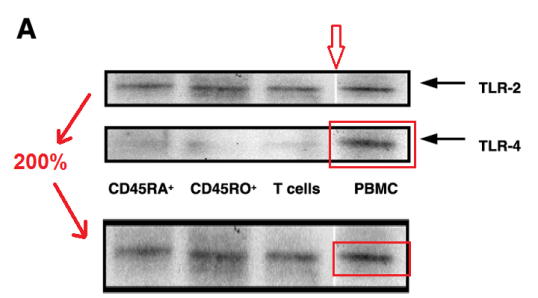
“T cells respond to heat shock protein 60 via TLR2” (Zanin-Shorov et al., FASEB, 2003) is less of a rich confection. In Figure 2A, the PBMC lane in the TLR-4 band was compressed vertically to 50% to produce the corresponding horizontal streak in the TLR-2 band, although its insertion there left a wide splice.
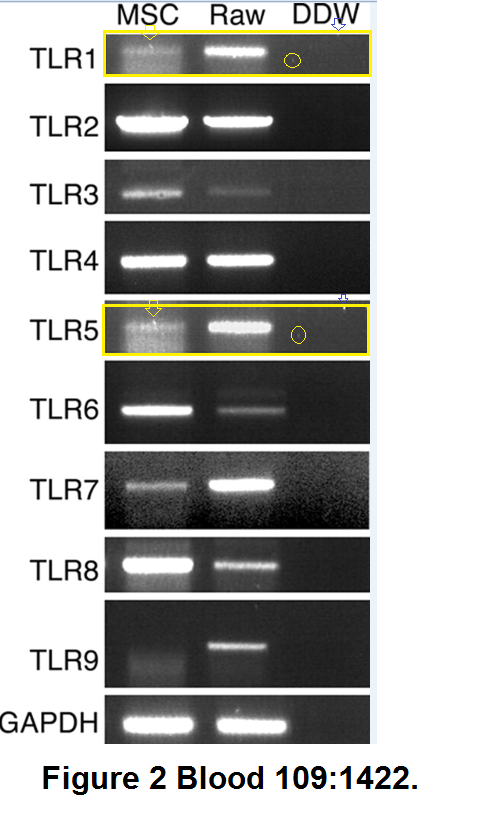 “Toll-like receptors and their ligands control mesenchymal stem cell functions” (Pevsner-Fischer et al., Blood, 2007), “Fibronectin-associated Fas ligands… ” (Zanin-Zhorov et al, J. Immunology, 2003), and “Cutting edge: T cells respond to lipopolysaccharide innately via TLR4 signaling” (Zanin-Zhorov et al., J Immunology, 2007) were not directly related to the vindication of the DiaPep277 vaccination, but we include them anyway for the sake of the largely-overlapping co-authorship and methods.The first is illustrated with RNA-PCA reactions rather than electrophoresis results for proteins, but the beautification tenchique is familiar: the vertical expansion of a copied gel to reduce its self-similarity.In the second, the pPyk2 (phosphorylated Pyk2) and tPyk2 (not phosphorylated, total Pyk2) band are reused between Figures 4B and 4C, while two of the pPyK2 lanes within 4C are mirror-image copies.
“Toll-like receptors and their ligands control mesenchymal stem cell functions” (Pevsner-Fischer et al., Blood, 2007), “Fibronectin-associated Fas ligands… ” (Zanin-Zhorov et al, J. Immunology, 2003), and “Cutting edge: T cells respond to lipopolysaccharide innately via TLR4 signaling” (Zanin-Zhorov et al., J Immunology, 2007) were not directly related to the vindication of the DiaPep277 vaccination, but we include them anyway for the sake of the largely-overlapping co-authorship and methods.The first is illustrated with RNA-PCA reactions rather than electrophoresis results for proteins, but the beautification tenchique is familiar: the vertical expansion of a copied gel to reduce its self-similarity.In the second, the pPyk2 (phosphorylated Pyk2) and tPyk2 (not phosphorylated, total Pyk2) band are reused between Figures 4B and 4C, while two of the pPyK2 lanes within 4C are mirror-image copies.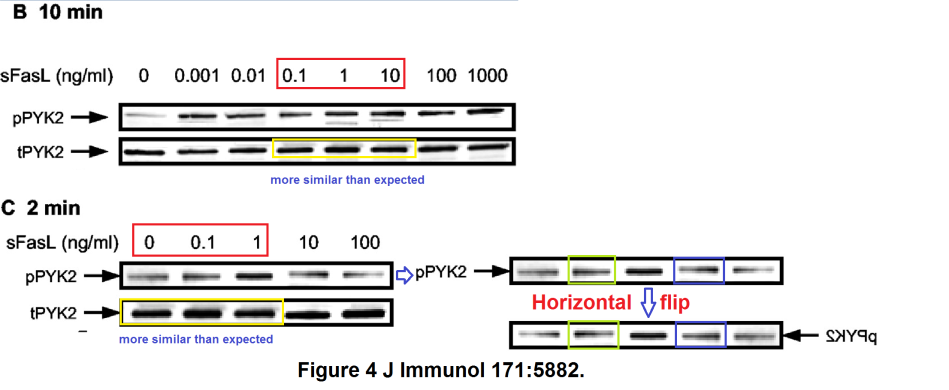
Bear in mind that lanes in the tERK band of Figure 5D are loading controls, to normalise the corresponding lanes in the pERK band. So it is cause for concern when we adjust the contrast on that panel and find that the second pERK lane is simply a copy of its neighbour (horizontally stretched), while the corresponding tERK lane has a surrounding moat of splices, as if the band had been assembled from multiple sources.
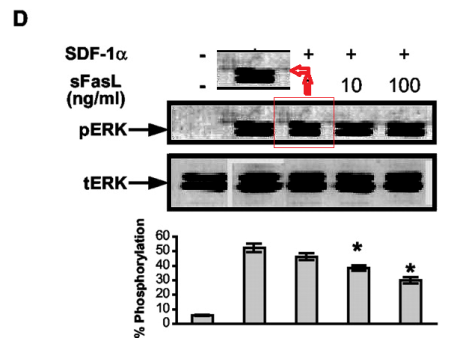
In the third of these out-takes (“Cutting edge: T cells respond to lipopolysaccharide innately via TLR4 signaling“), the 3rd and 6th lanes of the pPyk2 band in Figure 1E are the same, apart from a horizontal flip.

In Figure 1F, the p-p38 band shows the phosphorylation of a protein in response to six levels of lipopolysaccharide, relative to the background levels of protein in the t-p38 band. Rather alarmingly, the same t-p38 control band had previously appeared in 2006 (where we started), showing the background in three varieties of T-cell, with or without HSP-60. Indeed, lanes 1, 3 and 4 of the p-p38 band had also appeared in the same context, but with a different lane 6, while lanes 2 and 5 swapped places and flipped horizontally.
That doesn’t happen by accident.
Two or three other papers from the team targeted the question of DiaPep277’s mechanism, but they were not illustrated with gel images and have escaped the cold unsympathetic scrutiny of the PubPeer contributors.
Finally, Zanin-Zhorov and Cohen reviewed this work in Frontiers in Immunology (2013), providing an entry into their oeuvre by way of the References list. Of interest here is the authors’ declaration that they had no commercial relationship that could be construed as a Conflict of Interest (despite Cohen’s position as Andromeda’s “Clinical Advisory Board Member”). It may be that the term “COI” is understood differently in the Frontiers universe, but in 2014, in the Clinical-trial report, Cohen did feel that board membership was worth citing.
 But again I am getting ahead of myself for mentioning the 2014 Diabetes Care paper, which was retracted the same year.
But again I am getting ahead of myself for mentioning the 2014 Diabetes Care paper, which was retracted the same year.
Briefly: As the DIA-AID trial drew to a close, preliminary results were positive for one clinical end-point, but this posed the puzzle that a second clinical criterion showed no difference between DiaPep277 and placebo… it was as if someone had gone through the data, improving results for the first criterion by finding excuses to exclude unresponsive patients. And this was indeed what had happened. And there were retractions, and gnashing of teeth, and the new owners of Andromeda Biotech were not well-pleased, for on the basis of those preliminary results they had paid well for the company, thinking that they were acquiring a pharmaceutical goldmine.
Of course those clinical-trial shenanigans are not directly linked to the image cosmetology in the Weizmann Department of Immunology. But both could be symptomatic of a wider systemic problem — a shared attitude that if results of an experiment are not sufficiently convincing, but you have a strong enough feeling about what the results should have looked like, then it is fine to improve them with the appropriate erasure of defects.
Let us hope the whole story doesn’t inspire a sequel.

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism will be most appreciated!
€5.00












I am surprised that after the two retracted papers in Diabetes Care in 2014 that the Weizmann Institute did not review others papers by Irun Cohen?
LikeLike
Pingback: Ottavo comandamento... - Ocasapiens - Blog - Repubblica.it
I received today this email from Michal Neeman, Vice President of the Weizmann Institute,
The Weizmann Vice-president then explained in her next email:
LikeLike
Excellent response!
LikeLike
Are they getting the lawyer out?
LikeLike
Why do you write that? You do not think the response of Cohen and the vice president is good?
LikeLike
It is not obvious what it means. Would Irun Cohen bring the possible scientific misconduct to the notice of the Weizmann Vice president if he were happy?
LikeLike
The country has fewer inhabitants than New Jersey. It may be difficult to find scientists, or others, who are not conflicted.
LikeLiked by 1 person
Not sure how the connection to the number of inhabitants in New Jersey is relevant. You think that form 8 million people they cannot find suitable people? And who told you they restrict themselves to Israelies? My point is that you decided already the process is corrupted and this raises doubts from my side that you are indeed objective. You seem to have already decided before the Weizmann makes it investigation and this is really pity. I think that Irun asking to be investigated is the only right thing to do and I am happy he did.
LikeLike
Two papers about clinical work were retracted in 2014, and a meeting abstract was retracted in 2014. How come that did not lead to a review (looking at) of other papers at that time? All they had to do was look at the figures in the papers. Agreed that you need a modicum of memory to spot image reuse to represent different things in other papers, but even without that there is a large amount of problematic data which can be seen on the page. The failure to review other papers by the same authors as the retractions suggests that a willingness to root out the problem was lacking. Call that corruption if you like.
It is possible that a review was carried out, but if it is not made public that is secretiveness, lack of transparency in management-speak.
An old stoat trying to blame young weasels?
LikeLike
New Jersey has more inhabitants than Israel (8.9 vs 8.5 million). To put in perspective for the U.S. audience.
LikeLike
Despite the fact I’m not an immunologist, I do not think that the idea of a vaccine to an autoimmune disease is far-fetched. My argument: Allergy is also an over-reactive immune reaction to an antigen. For example, in Israel, where Weizmann Institute is located, the incidence of peanut allergy in children is much lower than the US. A study showed that babies are given a peanut-based snack called bamba early on, and as a result of this early exposure, children don’t develop peanut allergy. My point is that exposure to an antigen in a form of vaccine can lead to immunological tolerance towards that antigen. Hence, the same notion may apply to HSP60 over-expressed in pancreatic beta cells of T1D patients, although I’m not convinced HSP60 is involved in the disease pathogenesis.
This post provides a large amount of evidence on a huge scientific misconduct, but attacking the hypothesis is unnecessary. For example – “it was not clear why … was it obvious why stimulating the immune response, with injections of the synthetic analog DiaPep, should reduce the damage it wreaks.”
LikeLike
Kfir, You make a good point that attacking the hypothesis is unnecessary.
LikeLike
You are right, it was not fair to criticise Cohen’s original inuition of immunising against autoimmunity just because it was not worked out thoroughly. How scientists generate their hypotheses is irrelevant as long as they are testable. In another universe, his team did find an antigen that prevented T1D, and his inuition would have been entirely sensible.
But he did try it, and not just on diabetes… there was another Weizmann research project to find a vaccine against autoimmune arthritis.
LikeLike
The example with peanuts cannot be compared with what the HSP60 study. Peanuts represent a foreign substans and vaccine against peanut proteins is therefore possible and used to induce tolerance. To widen this concept to also include widely expressed proteins can be problematic. This is the reason why some immunetherapy studies ended with tragedy since the CARS attached proteins located differently than the primary target. In the HSP60 study I have problems understanding the logic behind the concept.
LikeLike
You are right, it’s difficult to understand the concept. However, this blogs deals with research integrity, and is not a debate on scientific ideas.
LikeLike
Was it a case of BSBB?
LikeLike
“according to her LinkedIn profile Vice-President at the pharma company Kadmon In New York, though the company’s website doesn’t list her anywhere.”
Based on the evidence that Alexandra Zanin-Zhorov can be linked to Kadmon research papers, a company email, and news articles related to their research, I believe she is a Kadmon employee and that the title of VP is referring to her role in the immunology department, not necessarily the entire company.
The idea of a vaccine for autoimmune diseases has been long fetishshized by researchers. In fact, a type 1 diabetes (you only mention “diabetes” in your post, but I like to clearly differentiate between them) vaccine was supposed to be in a phase II trial in the US, but little to no data has been released after the phase 1 trial. To the poster above, this also isn’t my field of expertise, but I have difficulty equating a basic allergy with an autoimmune disease. Unfortunately, autoimmune diseases tend to have only theories surrounding their root cause and, without this information, you’re likely only targeting their symptoms using a vaccine, which strikes me as pointless. How much long term efficacy can a vaccine have unless a scientist becomes lucky and targets the triggering mechanism? I’m not holding my breath for anything useful to show up any time soon.
LikeLike
Pingback: How Emilie Marcus and Cell covered up misconduct at Weizmann – For Better Science
Pingback: Collages by Paul Workman, from the Golden Age of Biological Imaging – For Better Science
http://www.fasebj.org/doi/pdf/10.1096/fj.02-1139fjeRET
Retraction
The article “T cells respond to heat shock protein 60 via TLR2: activation of adhesion and inhibition of chemokine receptors,” by Alexandra Zanin-Zhorov, Gabriel Nussbaum, Susanne Franitza, Irun R. Cohen, and Ofer Lider, publishedinprintasanFJExpresssummaryinFASEBJ.August200317:1567–1569(https://doi.org/10.1096/fj.02-1139fje), andasafull-lengtharticleonlineathttp://www.fasebj.org/doi/pdf/10.1096/fj.02-1139fje,hasbeenretractedduetoa finding of misconduct by the Weizmann Institute of Science. According to the institution, “the listed manuscript included clear manipulation of a figure amounting to scientific misconduct.”
DOI:10.1096/fj.02-1139fjeRET
LikeLike
That was perhaps the least problematical paper of the series, so I imagine that the Institute has sent similar requests to the other journals, and FASEBJ is just faster to act.
LikeLike
2nd 2018 retraction.
Pubpeer history
https://pubpeer.com/publications/08B08B9681EE15361E3BBC71938605
2018 retraction notice.
https://www.jci.org/articles/view/121856
Heat shock protein 60 enhances CD4+CD25+ regulatory T cell function via innate TLR2 signaling
Alexandra Zanin-Zhorov, Liora Cahalon, Guy Tal, Raanan Margalit, Ofer Lider, and Irun R. Cohen
First published May 7, 2018 – More info
See the related article at Heat shock protein 60 enhances CD4+CD25+ regulatory T cell function via innate TLR2 signaling.
Original citation: J Clin Invest. 2006;116(7):2022–2032. https://doi.org/10.1172/JCI28423
Citation for this retraction: J Clin Invest. https://doi.org/10.1172/JCI121856
At the request of the corresponding author, the JCI is retracting this article. The authors were recently made aware of duplicated bands in Figure 8E as well as portions of Figure 8D that were reused in another publication (Zanin-Zhorov A., et al. Cutting edge: T cells respond to lipopolysaccharide innately via TLR4 signaling. J Immunol. 2007;179(1):41–44.). After an extensive internal review, it became apparent that these duplications were introduced during figure assembly. The authors have stated that, after being made aware of the duplicated bands, they repeated the gel experiments and the results supported the conclusions of the original study. Moreover, other studies have subsequently confirmed and extended the primary conclusions of the manuscript. Nevertheless, the authors wish to retract this article because of the importance of accuracy and transparency in the published scientific literature.
The authors apologize for these errors.
LikeLike
Pingback: Another dead scientist framed with manipulated data? – For Better Science
J Biol Chem. 2016 May 20;291(21):11072-82. doi: 10.1074/jbc.M115.699041. Epub 2016 Mar 22.
FXYD5 Protein Has a Pro-inflammatory Role in Epithelial Cells.
Lubarski-Gotliv I1, Asher C2, Dada LA3, Garty H2.
Author information
1 From the Department of Biological Chemistry, Weizmann Institute of Science, Rehovot 7610001, Israel and irina.lubarski@weizmann.ac.il.
2 From the Department of Biological Chemistry, Weizmann Institute of Science, Rehovot 7610001, Israel and.
3 Division of Pulmonary and Critical Care Medicine, Northwestern University, Chicago, Illinois 60611.
Figure 7D.
See: https://imgur.com/nrKkhRc
https://www.weizmann.ac.il/WeizmannCompass/sections/people-behind-the-science/prof-haim-garty
Prof. Haim Garty
A life intertwined with the Weizmann Institute
PEOPLE BEHIND THE SCIENCE
DATE: DECEMBER 29, 2014
SOURCE:
WEIZMANN DIRECT VOL. 2 ISSUE 1
Prof. Haim Garty, the Vice-President of the Weizmann Institute of Science, passed away on November 29 [2014].
LikeLike
Pingback: Hyperbaric Oxygen Therapy extends life, the telomeres, and everything – For Better Science
Pingback: The Ballad of Claudio Hetz – For Better Science
Pingback: Who killed Margarita Lorenzo’s reputation? – For Better Science