Arthur Caplan, internationally respected professor of bioethics and director of the Division of Medical Ethics at the New York University, requested publicly retractions of the bioethics reviews on regenerative medicine he co-authored with the scandal surgeon Paolo Macchiarini. Caplan notified the journals with his retraction requests, but said to have received no response from them yet. Not only this: Caplan calls for all works of Macchiarini to be retracted.
Macchiarini is now an international pariah, he recently again lost his job, this time with the Kazan Federal University in Russia (see my report). His publications are under investigation at his former workplace the Karolinska Institutet in Stockholm (where he was previously sacked), the Swedish state announced to soon charge him with manslaughter, due to 3 lethal plastic trachea transplants he performed at Karolinska.
This is the statement Caplan provided me with over email:
“The level of fraud, deception and misconduct in the work of Paolo Macchiarini, most recently affiliated with Kazan Federal University, is such that all of his published work ought to be withdrawn or retracted, especially papers he co-authored on ethics (some with me).
I hope action can promptly be taken to remove his writings from all peer-reviewed journals”.
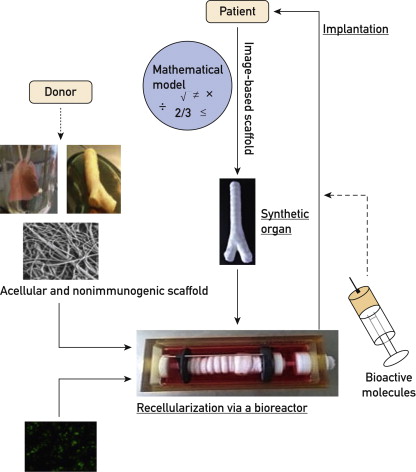
The New York bioethicist co-authored three reviews with Macchiarini, all of which deal with whole organ bioengineering, a technology where a dead decellurised donor organ or an artificial scaffold is allegedly brought to live by the use of patient’s own cells, like those from the bone marrow. There is little actual science to support this concept, which did not stop Macchiarini to put in into practice. Not on animals, but on humans. The result was: 8 patients died horribly from plastic trachea grafts implanted by Macchiarini, the cadaveric scaffold-based tracheas had several lethal outcomes and left survivors mutilated and in permanent emergency care (see my reports e.g. here, here and here).
These are the bioethics reviews Caplan co-authored with Macchiarini, which he now says to have asked journals to retract:
Ethics of bioengineering organs and tissues.
Taylor DA, Caplan AL, Macchiarini P.
Expert Opin Biol Ther. 2014 Jul;14(7):879-82. doi: 10.1517/14712598.2014.915308.
Abstract:
“Tissue-engineered medical products are now entering the clinical testing phase of development. Therefore, an open discussion is warranted regarding ethical issues that may arise as these novel ‘combination’ products move forward, such as when to conduct clinical trials, how to regulate such trials, when and how to responsibly introduce these strategies into clinical practice and how to maintain a positive public perception of the tissue-engineering field as a whole. These issues are discussed, and recommendations are provided for conducting first-in-human clinical studies”.
The first author of this bioethics opinion piece is Doris Taylor, who works at the Texas Heart Institute in Houston on heart regeneration. Just like Macchiarini, Taylor uses the magic of bone marrow cells seeded on dead decellurised donor tissue. Luckily, no one allowed Taylor to test this in patients. Nevertheless, Nature reported in 2013 very admiringly about her achievements on this field. Nothing resembling actual hearts has been reported from that Texan lab since, as it is often the case with bombastic regenerative medicine announcements which lack basic science. Taylor’s and Macchiarini’s common work on bioengineered oesophagus (Sjöqvist et al 2014) was recently retracted by the journal Nature Communications, after Swedish authorities found misconduct and data manipulation (see my report here). As I found out, the Texan heart maker previously tried to help Macchiarini get a professorship at her institute and the University of Texas, but did not succeed despite her best efforts (see my report).
Engineered whole organs and complex tissues.
Badylak SF, Weiss DJ, Caplan A, Macchiarini P.
Lancet. 2012 Mar 10;379(9819):943-52. doi: 10.1016/S0140-6736(12)60073-7.
Abstract:
“End-stage organ failure is a key challenge for the medical community because of the ageing population and the severe shortage of suitable donor organs available. Equally, injuries to or congenital absence of complex tissues such as the trachea, oesophagus, or skeletal muscle have few therapeutic options. A new approach to treatment involves the use of three-dimensional biological scaffolds made of allogeneic or xenogeneic extracellular matrix derived from non-autologous sources. These scaffolds can act as an inductive template for functional tissue and organ reconstruction after recellularisation with autologous stem cells or differentiated cells. Such an approach has been used successfully for the repair and reconstruction of several complex tissues such as trachea, oesophagus, and skeletal muscle in animal models and human beings, and, guided by appropriate scientific and ethical oversight, could serve as a platform for the engineering of whole organs and other tissues”.
The first author of this review paper, Stephen Badylak, is deputy director of the McGowan Institute for Regenerative Medicine at the University of Pittsburgh. In April 2017, Badylak became Co-Chairman of the scientific advisory board of the Boston-based company Biostage. Never heard of them? It’s because the company changed its name, after its previous one, Harvard Apparatus, became infamous. This company manufactured the plastic tracheas which Macchiarini implanted into 3 patients in Russia. 2 of them are dead, one survived because the graft could be removed in time. The former Harvard Apparatus CEO, David Green, was filmed by a TV crew as he was talking one of the victims, Yulia Tuulik, into agreeing to that lethal transplant (details mentioned here). The new Biostage still has Green in its team, and now Badylak. They currently work to develop bioengineered plastic oesophagus which they hope to be able to test soon on humans.
There is also a minor literature review which Caplan co-authored with Macchiarini and his international team:
Whole organ and tissue reconstruction in thoracic regenerative surgery.
Lim ML, Jungebluth P, Ajalloueian F, Friedrich LH, Gilevich I, Grinnemo KH, Gubareva E, Haag JC, Lemon G, Sjöqvist S, Caplan AL, Macchiarini P.
Mayo Clin Proc. 2013 Oct;88(10):1151-66. doi: 10.1016/j.mayocp.2013.03.011.
I will now contact the journals and will bring an update should I learn anything. Note: The Lancet previously ceased all communications with me.

“Paolo Macchiarini Is Not Guilty of Scientific Misconduct.” – The Lancet
LikeLiked by 1 person
Pingback: Paolo Macchiarini, ennesima puntata - Ocasapiens - Blog - Repubblica.it
I hope this will help you in the cases where you are unfairly sued.
LikeLike
Green’s linkedin says he left Biostage but is a director at Harvard Bioscence. … are those two companies linked? Thanks
LikeLike
they are the same, as I explained above. Harvard Apparatus renamed itself to Biostage after Macchiarini scandal.
LikeLike
Pingback: Expert review: Macchiarini papers on plastic trachea transplants are misconduct – For Better Science
Pingback: Whom to sacrifice for Human Challenge Trials? – For Better Science