On 14 December 2021, Veronika Skvortsova, Russian health minister between 2012 and 2020, and now head of Russian government’s Federal Medico-Biological Agency (FMBA), announced a new high-tech cure for COVID-19. It is a nebuliser spray for inhalation, containing an siRNA (small interfering RNA), which in Russian is called “Малая интерферирующая РНК”, hence the drug’s acronym “Мир-19” (Mir-19). The Russian word “Мир” means peace (or world), and it was also the name of the first ever space station. A likely continuation in naming Russian COVID-19 drugs after Soviet space achievements, since the vaccine Sputnik V was named after the first ever satellite. And because Putin’s Russia is the most peace-loving nation on earth.
Propaganda successes
Russia keeps conquering the coronavirus with home-made breakthrough medicines, but so far Kremlin’s only successful tool to fight the deadly pandemic was to fake the COVID-19 statistics. But even there, it doesn’t work convincingly, because the Russian officials haven’t (yet) learned to fake the national excess death numbers, and those tell of a ongoing catastrophe with almost a million dead.
Prior to the pandemic, Putin’s regime and its Health Minister Skvortsova tried to win the war on cancer by faking cancer statistics. It didn’t really work either.
In early 2020, when the COVID-19 pandemic began and the world started to place its hopes into remdesivir and chloroquine, Russia proudly deployed Arbidol, an old Soviet antiviral drug from 1970ies. Economically, arbidol was a huge success and made some pharma-business-owning oligarchs in Russia even richer than they already were. Clinically, arbidol’s effect against COVID-19 was zero (although we can’t exclude it made things worse). In September 2021, the Russian Health Ministry half-heartedly admitted that arbidol was kind of useless, but allowed its indication for moderate COVID-19 in non-risk group. Also the hydroxychloroquine and azithromycin combo became standard therapy for COVID-19 in Russia early in 2020.
Later in 2020, Russia established the old US drug favipiravir as the COVID-19 cure, and marketed it under Russian brand names. Just in November 2021, Russian news announced a medical breakthrough, a new domestic drug approved which was again favipiravir. This antiviral drug was proven in various clinical studies abroad to be useless against coronavirus infection, and it is also known to be highly toxic, especially in pregnancy. Never mind, because Russian health authorities anyway advises pregnant women to get an abortion if they catch COVID-19.
On top of everything, due to constant onslaught of state propaganda against the “evil” western vaccines, Russians have been steadily refusing all kinds of COVID-19 vaccines, the vaccination rates are disastrously low, and of the circulating immunisation certificates a large number is most likely faked. It may not be too convincing when Putin’s speaker Peskov now calls vaccine-refusers “dangerous fools”, while he himself until very recently refused to get vaccinated and was seen wearing bleach packets to ward off the coronavirus miasma.
Plus, even the Russian authorities seem to be worried their Sputnik V and other national vaccines may not be efficient against the new virus variants, regardless of public announcements constantly reassuring otherwise. If these patriotic vaccines were ever efficient against SARS-CoV2 at all, there are good reasons to doubt it. They sure were never tested properly before official approval by Russian Health Ministry.
New domestic and patriotic drugs are urgently needed, since, as the state-controlled TV keeps warning, the western stuff is toxic and turns you into monkeys (sic!).

Peace-19
So on 14 December 2021, the FMBA director and former Minister of Health Professor Skvortsova gave a seminar. You can download the slides, read the transcript and even watch the video here.
There is a new breakthrough medicine, it’s called Peace-19, and its already approved by the Health Ministry!
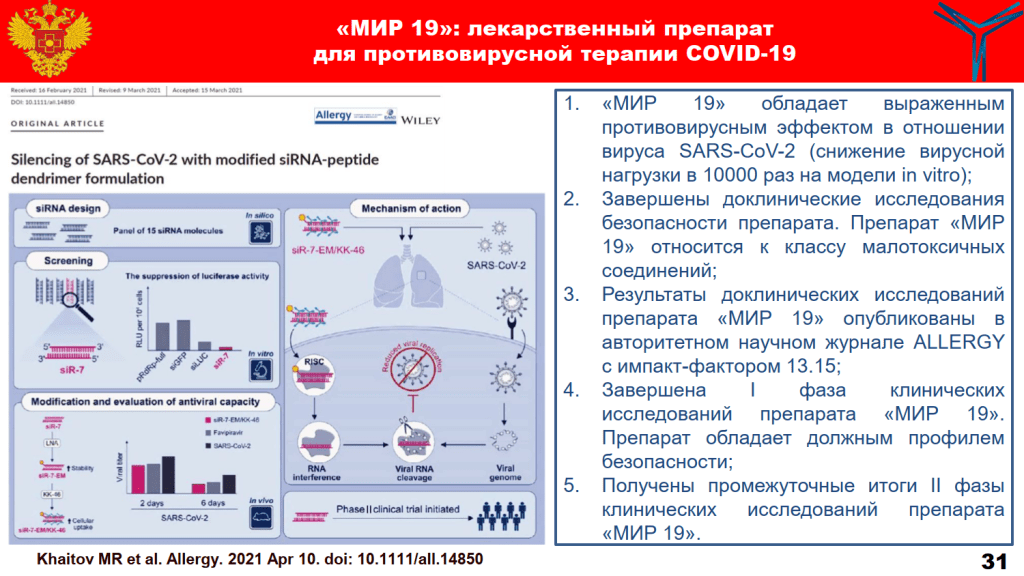
Mir-19 is an siRNA against the RdRP (RNA-dependent RNA polymerase) enzyme coded in the RNA genome of the SARS-CoV2 virus. Another ingredient is a specially designed cationic dendrimeric peptide as carrier. The drug, which we are told will works against all SARS-CoV2 variants, including omicron, has been allegedly tested in phase 1 and phase 2 clinical trials. Yet there is no trial registration, the clinical test results are not published, and no details of these trials have been released except that they of course were resounding successes. Important is that Mir-19 has received national approval on 22 December 2021 and is now being deployed in Russian hospitals.

FMBA informed on that occasion (translated from Russian):
“Preclinical studies have revealed a pronounced antiviral effect of the drug against SARS-CoV-2 (reducing the viral load by 10 thousand times) and its low toxicity. The results are published in the world’s leading highly rated journal on immunology “Allergy”.
Phase I clinical trials were approved on December 30, 2020. During the first phase of the study, the safety and good tolerance of the drug was shown in healthy volunteers.
On March 11, 2021, the State Research Center “Institute of Immunology” of the Federal Medical and Biological Agency received a patent No. 2746362 for a drug for the treatment of coronavirus infection of the SARS-CoV-2 type.
On April 14, 2021, official permission was obtained to conduct Phase II clinical trials, which began on April 26, 2021 at the clinical centers of the FMBA of Russia in Moscow and the Moscow Region, as well as in St. Petersburg and the Leningrad Region.
In the course of clinical trials, the safety and effectiveness of the drug “MIR 19” has been proven. As for safety – the drug has no effect on the human body, the target is the virus, therefore, the development of adverse reactions in patients has not been established. When studying the effectiveness, the drug was tested in a hospital in the “red zone”, in patients with moderate severity of COVID-19, where the effectiveness of the drug was proved in comparison with standard therapy. Based on the results of the completion of the second phase of clinical trials, which showed highly reliable advantages of the MIR 19 drug over standard therapy, a report was generated and sent for the drug registration procedure.”
That’s all the public is allowed to know about the phase 1 and 2 clinical tests. Expect the results to be published in The Lancet, to accompany the several Sputnik V papers there. Expect the Kremlin will be decrying “russophobia” when other countries will refuse to buy and approve Mir-19 because it has been never properly tested for safety or efficacy. Expect some Russia-friendly countries will buy and approve it anyway because it will be published in Lancet.
Until then, the only thing we have on Mir-19 is a paper with hamsters, from April 2021, in an obscure Wiley journal named Allergy. Skvortsova is last author, and she explained in her FMBA talk that this was actually a “world’s leading highly rated journal in immunology“. If she says so.
Musa Khaitov , Alexandra Nikonova , Igor Shilovskiy , Ksenia Kozhikhova , Ilya Kofiadi , Lyudmila Vishnyakova , Alexander Nikolskii , Pia Gattinger , Valeria Kovchina , Ekaterina Barvinskaia , Kirill Yumashev , Valeriy Smirnov , Artem Maerle , Ivan Kozlov , Artem Shatilov , Anastasiia Timofeeva , Sergey Andreev , Olesya Koloskova , Nadezhda Kuznetsova , Daria Vasina , Maria Nikiforova, Sergei Rybalkin, Ilya Sergeev, Dmitriy Trofimov, Alexander Martynov, Igor Berzin, Vladimir Gushchin, Aleksey Kovalchuk, Sergei Borisevich, Rudolf Valenta, Rakhim Khaitov, Veronica Skvortsova Silencing of SARS‐CoV‐2 with modified siRNA‐peptide dendrimer formulation Allergy (2021) doi: 10.1111/all.14850

In fact, even Skvortsova’s own FMBA presentation from 14 December 2021 admitted that the phase 2 clinical trial wasn’t completed, all they had was “intermediate conclusions”. In Russia, this suffices for a national approval and immediate deployment in hospitals.
The phase 3 clinical trial, as with Sputnik V, if it ever happens, will be “real-world” studies on the Russian population after the drug’s national approval and deployment. You can imagine the data forgery opportunities there.
Even Putin’s eternal speaker Dmitry Peskov recently admitted that Russia has its very own concept of which data is needed for drug approval, and is now fabricating assembling the clinical and technical documentation for WHO to eventually approve Sputnik V.
The degenerate West tests its drugs in phase 3 and publishes the results before submitting it to authorities, what losers. In Russia, the final efficacy and safety of Mir-19 was unambiguously proven by Skvortsova’s announcement to have taken it herself:
“In an interview with Rossiya-24, the head of the Federal Biomedical Agency Veronika Skvortsova said that she had tried inhalations of a new drug for the treatment of coronavirus infection Mir-19 after she had contact with a sick patient.
“The drug has a sweetish taste, it must be inhaled within 10 minutes. If you breathe the drug after contact with a sick person, the viral load on the body drops sharply, ” said Skvortsova on air.
The head of the FMBA explained that in the second phase of clinical trials Mir-19 demonstrates an advantage over existing drugs for COVID-19, and also noted that the new drug will be effective even against a new strain – omicron.”
I don’t know, how does this Mir-19 compare to the standard Soviet therapy for everything, from flu to acute radiation exposure, which is drinking vodka?
Because the new COVID-19 drug is approved and clinically deployed in Russian hospitals now, there is at least a safety sheet. Which states Mir-19 should not be used at all on patients with severe COVID-19, children, those over 65, or those with cancer, and avoided for those with diabetes, autoimmune diseases, any cardiovascular, liver, kidney or neurological problems. Basically, not on any risk groups. The idea is apparently to use Mir-19 on healthy young people who generally recover from COVID-19 by themselves and then to declare yet another victory for Russian science.

Fighting the Cytokine Storm
In the unlikely case Mir-19 fails to stop the pandemic, Skvortsova announced another miracle drug in her 14 December 2021 lecture at FMBA. It’s an opioid receptor agonist peptide, named “Лейтрагин” (Leytragin or Leutragin) and that one is already available in Russian pharmacies. The authors say it helps against the Cytokine Storm and somehow a sirtuin (SIRT1), popular target of resveratrol and NAD+ quackery, is involved!
Also here, no clinical studies were published, the drug was approved based on FMBA’s request and this preclinical paper in Hindawi from September 2021:
Vladislav N. Karkischenko , Veronika I. Skvortsova , Melik T. Gasanov , Yuriy V. Fokin , Maxim S. Nesterov , Nataliya V. Petrova , Oxana V. Alimkina , Igor A. Pomytkin Inhaled [D-Ala]-Dynorphin 1-6 Prevents Hyperacetylation and Release of High Mobility Group Box 1 in a Mouse Model of Acute Lung Injury Journal of Immunology Research (2021) doi: 10.1155/2021/4414544
Its abstract declared:
“In this study, we demonstrate that inhaled [D-Ala2]-dynorphin 1-6 (leytragin), a peptide agonist of δ-opioid receptors, significantly inhibits HMGB1 secretion in mice with lipopolysaccharide- (LPS-) induced acute lung injury. The mechanism of action involves preventing HMGB1’s hyperacetylation at critical lysine residues within nuclear localization sites, as well as promoting the expression of sirtuin 1 (SIRT1), an enzyme known to deacetylate HMGB1. Leytragin’s effects are mediated by opioid receptors, since naloxone, an antagonist of opioid receptors, abrogates the leytragin effect on SIRT1 expression.”

Thing is, Leytragin was approved by Russian Health Ministry already in May 2021, weeks before the above paper was even submitted to that Hindawi journal. The official announcement goes:
““From March to June 2020, the Research Center for Biomedical Technologies of the FMBA of Russia worked intensively, almost around the clock. The head of the Agency, Veronika Skvortsova, was tasked with developing new effective approaches to treating patients with COVID-19. The Institute was able to quickly develop a non-infectious model of acute respiratory distress syndrome in mice. This syndrome is one of the main causes of death in coronavirus infection. A variety of candidate molecules were screened for its prevention and relief, and the most effective and safest agents were selected. On April 15, 2020, clinical studies of Leitragin began, which were carried out in several scientific and clinical centers of the FMBA of Russia. Studies have confirmed the high efficacy and safety of this drug in patients with COVID-19 pneumonia compared to standard therapy”, said Vladislav Karkishchenko, director of the Scientific Center for Biomedical Technologies of the FMBA of Russia.
In the course of the studies, in which 320 patients with a moderate course of the new coronavirus infection took part, it was found that in the group of patients receiving Leytragin, there were not only not a single death, but also there were no cases of the transition of the disease to a more severe form. At the same time, the recovery time of patients receiving Leutragin was reduced to 8 days, compared with 14 days in the control group receiving standard therapy.”
That data is of course secret. Skvortsova also mentioned in her talk that leytragin reduces the duration of hospitalisation by 1.4 times, and is therefore indicated for severe COVID-19. Needless to say, none of that was ever published. Just the mouse tests in Hindawi, submitted after the drug’s national approval.
The new vaccine
For the even more unlikely case that the coronavirus can’t be stopped by Mir-19 or leytragin, Skvortsova surprises Putin with yet another scientific breakthrough. It is yet another COVID-19 vaccine, to complement Sputnik V and two or three other approved Russian vaccines, all of which are certified to be at least 100% efficient, including against delta and omicron variants.
FMBA’s new vaccine is called “Конвасэл” (Konvasel) and is made of the recombinant N-Protein of the SARS-CoV2, the idea being that since the coronavirus is mutating its spike protein, another target is needed. The new Russian vaccine has been duly tested on mice, hamsters and even rabbits, and this is obviously enough.
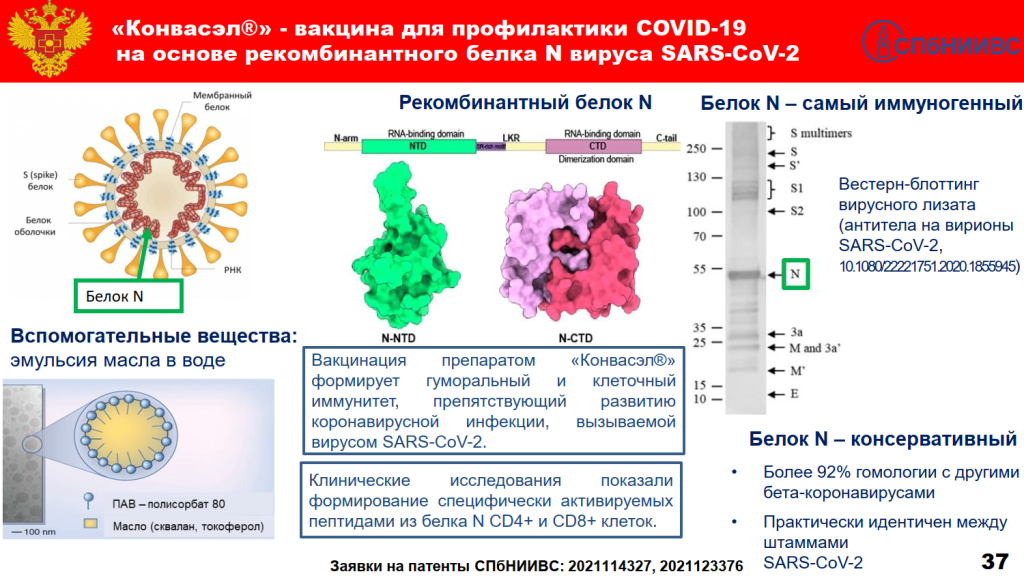
Two days after Skvortsova’s presentation from 14.12.2021, FMBA applied to its colleagues at the Health Ministry for the emergency approval of Konvasel. The official announcement by Interfax mentions:
“In 2020, the vaccine passed preclinical trials… The Ministry of Health allowed FMBA to begin clinical trials on July 19, 2021. The agency reported that the study is being carried out among 200 volunteers between the ages of 18 and 60.
In early September 2012 [sic!], Skvortsova told reporters that the second phase of clinical trials of the vaccine should be completed by the end of 2021, and the registration of the drug expected in 2022.”
In her talk from 14 December 2021, Skvortsova merely spoke of intermediate data from the phase 1-2 clinical trial which has been submitted to the authorities. In Russia, this is more than enough to inject the entire population.

I’m not sure what Skvortsova and her FMBA are really doing. They may think they are inventing COVD-19 miracle drugs on a conveyor belt, but in reality it all looks like Potemkin’s villages to impress Putin. The joke is of course on the Russian people who will have to literally swallow all this virtually untested half-baked stuff.
Unless of course, Mir-19 is really meant to become a political tool of global “diplomacy” like Sputnik V. And a money maker.
I thank all my donors for supporting my reporting. You can be one of them!
Make a one-time donation:
I thank all my donors for supporting my reporting. You can be one of them!
Make a monthly donation:
Choose an amount
Or enter a custom amount
Your contribution is appreciated.
Your contribution is appreciated.


In other Russian news: the supreme court just dissolved Memorial and declared it a Nazi spy organisation.
Memorial was the first Soviet NGO founded in 1980ies. It began by exposing mass murders, deportations and other crimes of Stalinism, under Putin Memorial Memorial has been recording human right abuses and political imprisonments.
The court decided now that Memorial are Nazis and foreign spies because:
Memorial criticises Stalin which means its defames the Great Patriotic War (WW2), which means Memorial endorses Nazism, which means Memorial must be paid by the russophobic USA and Germany hence Memorial are foreign spies.
https://t.me/theinsider/9630
LikeLike
Did I miss something or there is a typo for the year here
“In the unlikely case Mir-19 fails to stop the pandemic, Skvortsova announced another miracle drug in her December 2014”
2014?
LikeLike
Sorry, 14 December 2021. Like Interfax , I mistype the year!
LikeLike
Pingback: Teoria e pratica del campo elettrostatico – ocasapiens
I was actually wondering if it was a typo or a meta joke ^^
LikeLike
Regarding Mir-19, unaware of the Russian work we reported that the genomic region corresponding to RNA-dependent RNA polymerase (nsp12) has an open structure. We recommended it as a possible target site for antisense oligonucleotides (Computational Biology & Chemistry 94 (2021) 107570).
LikeLike
This is another big problem of the so-called ” Covid-19 pandemic”. The development of therapies against the “supposed Covid-19 disease’
LikeLike