America is about to lose the science race to China. The US National Science Foundation (NSF) warned in Nature News that “China is closing gap with United States on research spending”, adding that “Preliminary data from 2019 suggest that China has already surpassed the United States in R&D spending”. In 2018, NSF announced in Nature News that “For the first time, China has overtaken the United States in terms of the total number of science publications”. These are serious developments, apparently the Chinese fraudsters are much more ruthless and efficient than ours.
We cannot afford a Photoshop gap.
It makes the matters only worse that the Communist Party of China resorted to not only legalising, but actively promoting quackery in the form of Traditional Chinese Medicine (TCM), which serves to further boost the country’s research output. There, China found an ally with Indian nationalists who deploy masses of Ayurveda papers to establish India’s dominance. Data forgers of both nations seem to agree on the magic of curcumin, which this post by Smut Clyde will celebrate.
No help can be expected from scholarly publishers, quite the opposite. Elsevier operates outlets like Journal of Traditional Chinese Medicine and Journal of Traditional Chinese Medical Sciences, Springer Nature offers journals like Chinese Medicine and BMC Complementary and Alternative Medicine, while even society publishers like the Royal Society of Chemistry are eager to accept TCM quackery in allegedly serious journals.
If we are to beat China in the research publication race we must make Photoshop training mandatory for every PhD student. We must also open our science for alchemy, homeopathy, snake oils and other quackery. Ideally combined with stem cells and nanotechnology.
Luckily there is Smut Clyde who will now show you the ropes of Chinese artisan fraud. Read, observe and learn how to create peer-reviewed papers from generously applied TCM and skilful Photoshop.
The alchemical life-force herbalism of Traditional Chinese Medicine does work!
By Smut Clyde
The present-day perversely-incentivised ecology of scholarly publication inspires many emotions, ranging from elation to despair depending on the attitude of Reviewer #3. Among these emotions is “Marvel”, when the editors and publisher allow an author to insert new not-obviously-faked results into an already-published paper to replace the deprecated original figures (“These corrections do not affect any of the major conclusions of the paper”… they never do). Here is an analogy by Elisabeth Bik:
“If a sports professional had won a race but their blood/urine sample taken just before the race had doping in it, would you accept a clean sample from two weeks later? Would that make it ok?“

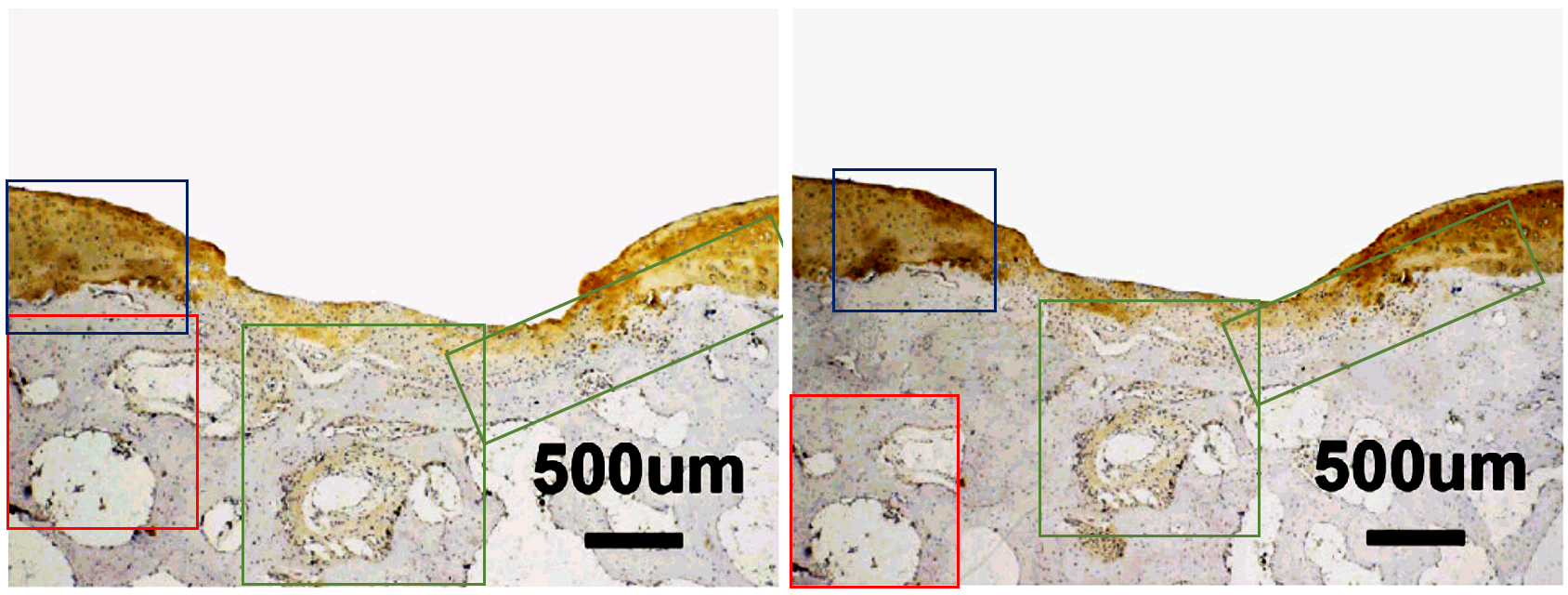
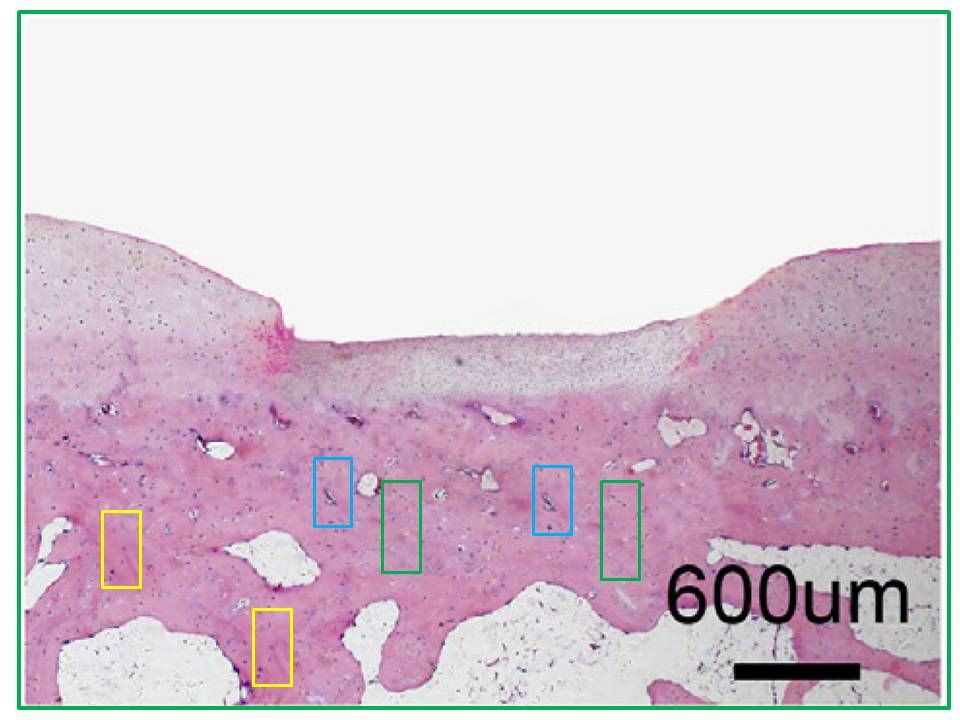
Another case in point is this close-up slice of nanotech-enhanced stem-cell bone regeneration after injury. They are 4-week QD-SMCC and PEI-si treatments from Fig. 5D of Wu et al. (2016) [H]. The components of the two adjacent panels in the Figure are the same but horizontally flipped and inserted in a different context… that is, one or other was captured within the special microscope sold as “Photoshop”, and the best explanation for the lack of effort spent disguising its origins is that it was Friday afternoon and everyone in the lab was in a hurry to knock off work and head down the road to the Karaoke bar.
The QD-SMCC (Quantum Dot) panel was replaced in a Corrigendum of October 1 2019. Two years earlier, an October 19 2017 Corrigendum of the same paper had replaced the 2-week QD-SMCC slice of Fig. 6A, after “incorrect uploading” had caused it to consist of the same elements as another panel but in different assemblage.
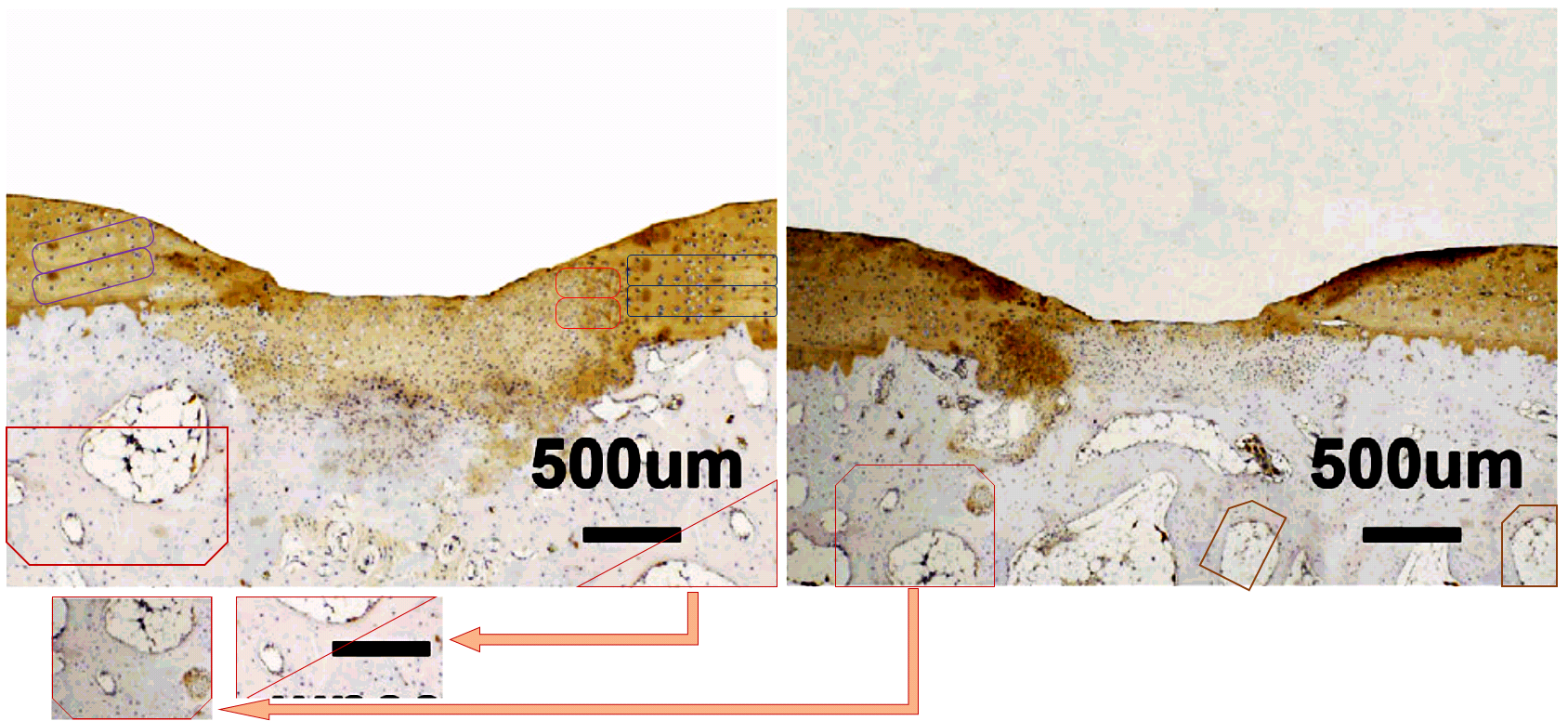
Now other frames from Figs 5A and 6A are not so blatant, but like the microscopic serial number stamped on the snake scale in the Animoid-Row scene from Bladerunner, telltale glitches reveal a synthetic nature. Two from 5A had suffered some kind of accident in the matter replicator…
…while the recycling between the first two panels of the 4-week row of 6A is too complex for a single diagram to express.
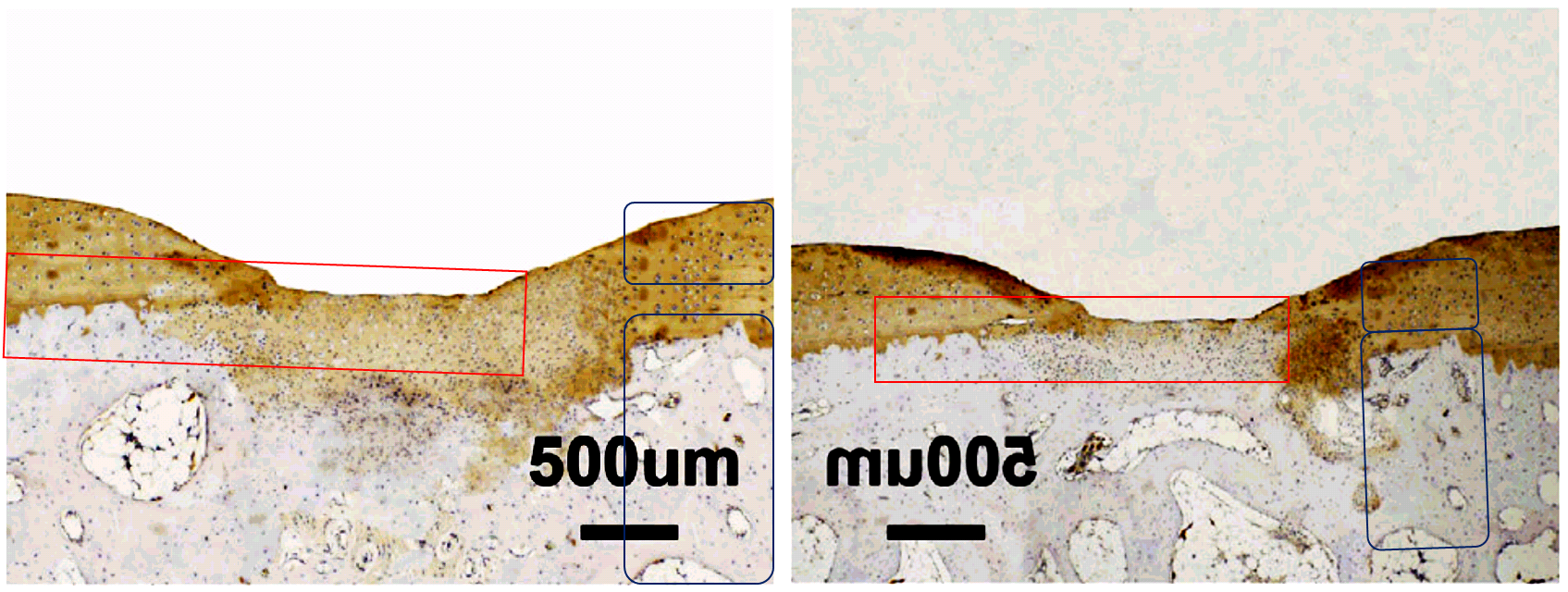
I assume from the opportunities for correction not taken that the authors stand by these images, which survived both rounds of amendation. Authors here are Jinmin Zhao and colleagues from the orthopedic surgery group at Guangxi Medical University, Nanning. I will come back to them after a brief digression. Can anyone confirm, by the way, that Guangxi is a real bricks-and-concrete campus and not a collage painstakingly assembled in Photoshop?
First I want to touch on the oeuvre of Pengwen Wang and Jinzhou Tian, from the Key Laboratory of Chinese Internal Medicine, Dongzhimen Hospital, Beijing University of Chinese Medicine (BUCM), Beijing. As well as the institutions, the topics are different: rather than improving bone regeneration, Wang et al. are curing Alzheimer’s Disease with curcumin. But both groups of papers house figures that are better judged by the standards of aesthetics than by those of laboratory technique, and surely an adventurous art historian (looking at you, James Elkins!) can be persuaded to write a lengthy critique, with erudite comparisons to Tobey’s all-over tachisme innovations.
“It’s got to be prepared.” “Treated,” said Benji. “Diced.”
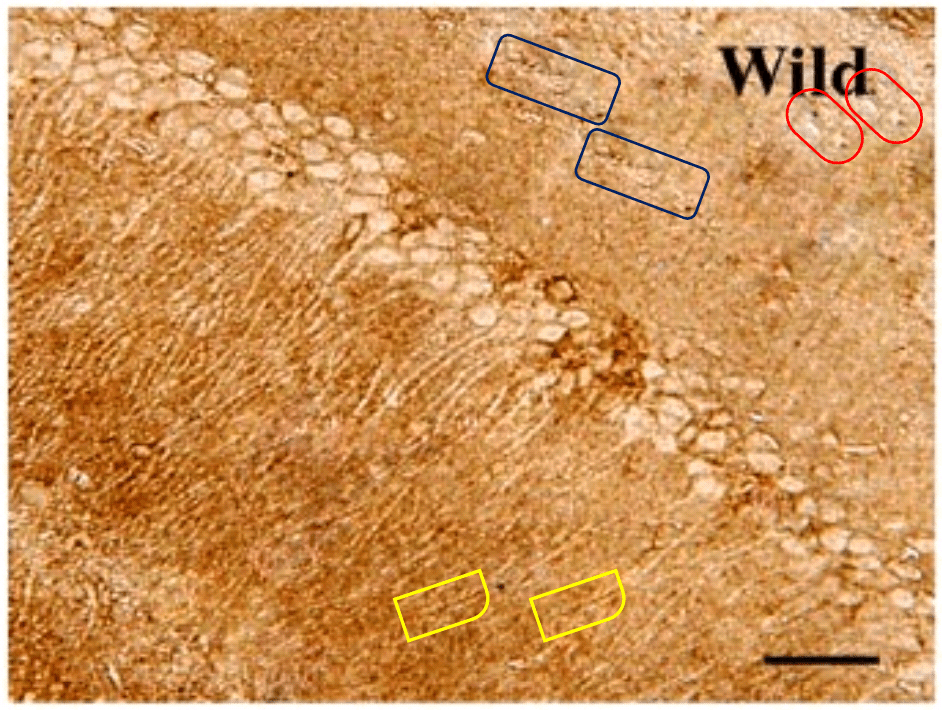
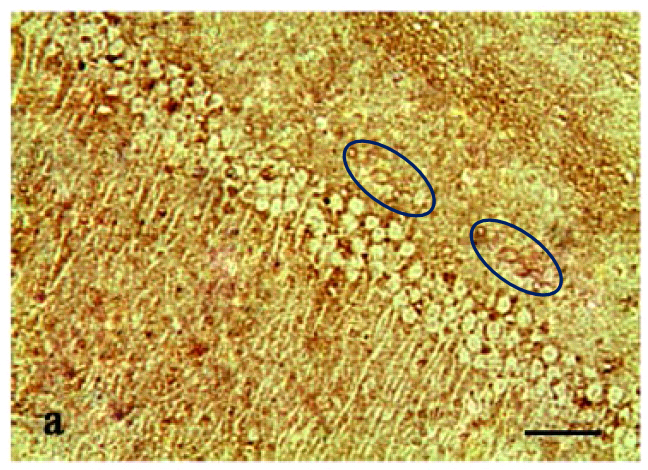
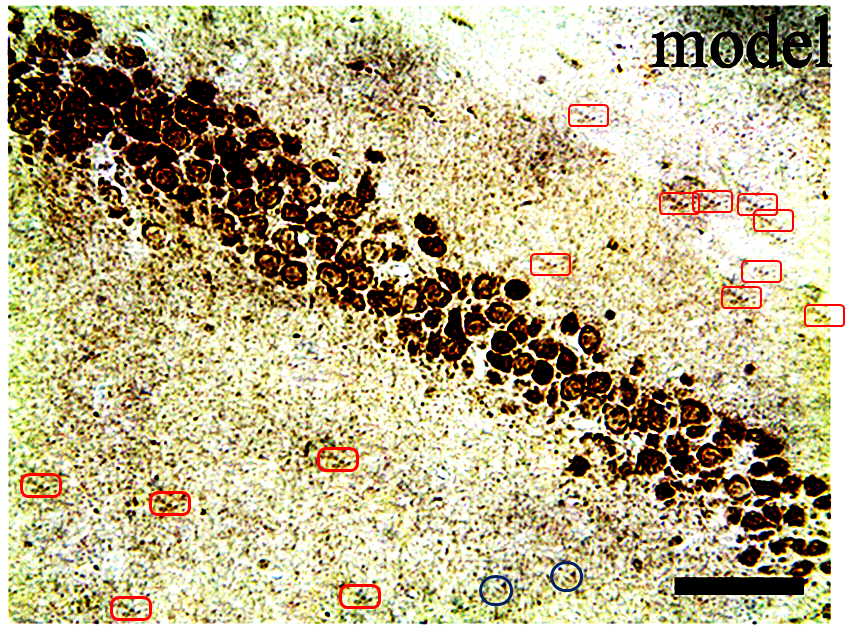
Are these the sucker sides of octopoidal tentacles? A deposit of ichthyosaur vertebrae? ¹ Lost souls at the mercy of a perpetual hurricane in the Second Circle of Hell? They also remind me of Turner’s ‘Heidelberg Schloss‘. But the strips of circles are neither suckers, vertebrae nor a torrent of spectators’ faces, but the soma of neurons. For these scenes purport to be slices of rodent hippocampus, illustrating how different medication regimes slow the ravages of Alzheimer’s (AD) and the coagulation of amyloid protein into plaques.


In Figs 5 and 6 of Wang et al. (2017) [7], two pairs of panels overlap at the corners, making it unlikely that they do represent different conditions. To be fair to the authors, this could easily be an error, as the obviousness of the overlap is reduced by rotation, and a mirror-flip for one pair, and minor airbrush removal of some fallen-from-favour cells from one or other image of each pair.
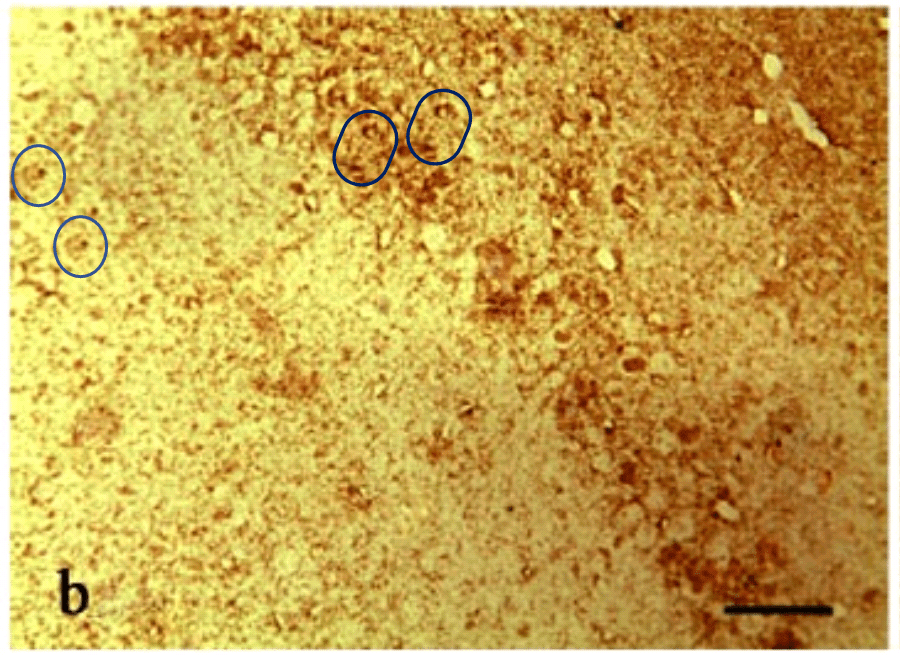
The airbrushing continues into the panels of Figs 2, 3, 4, 7, 8, 10, 11 and in particular Fig. 9, with minor glitches in the matrix. Perhaps these feature duplications were intended as a form of steganographic signature, like the deliferate misteaks that cartographers used to insert in their maps so as to provide conclusive proof of plagiarism if anyone pirated their creations.
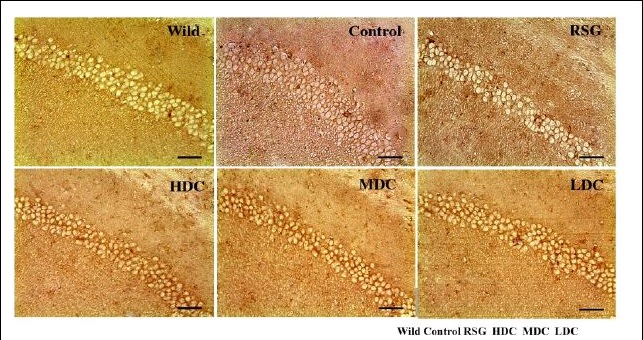
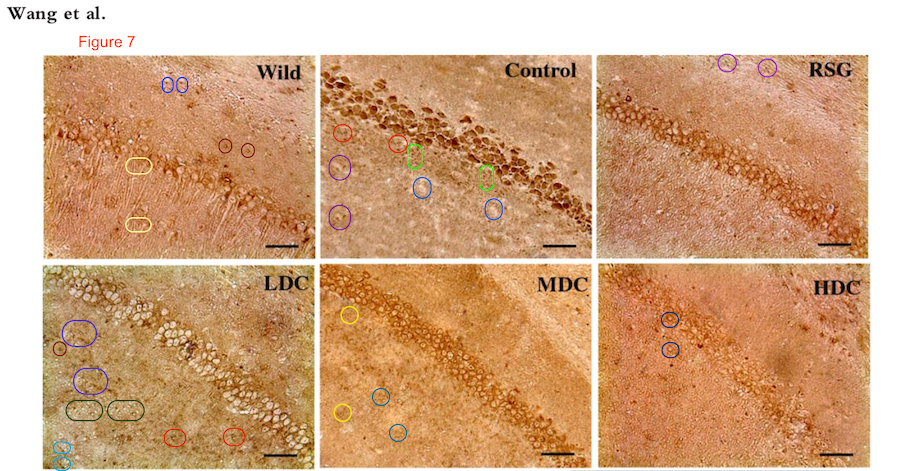
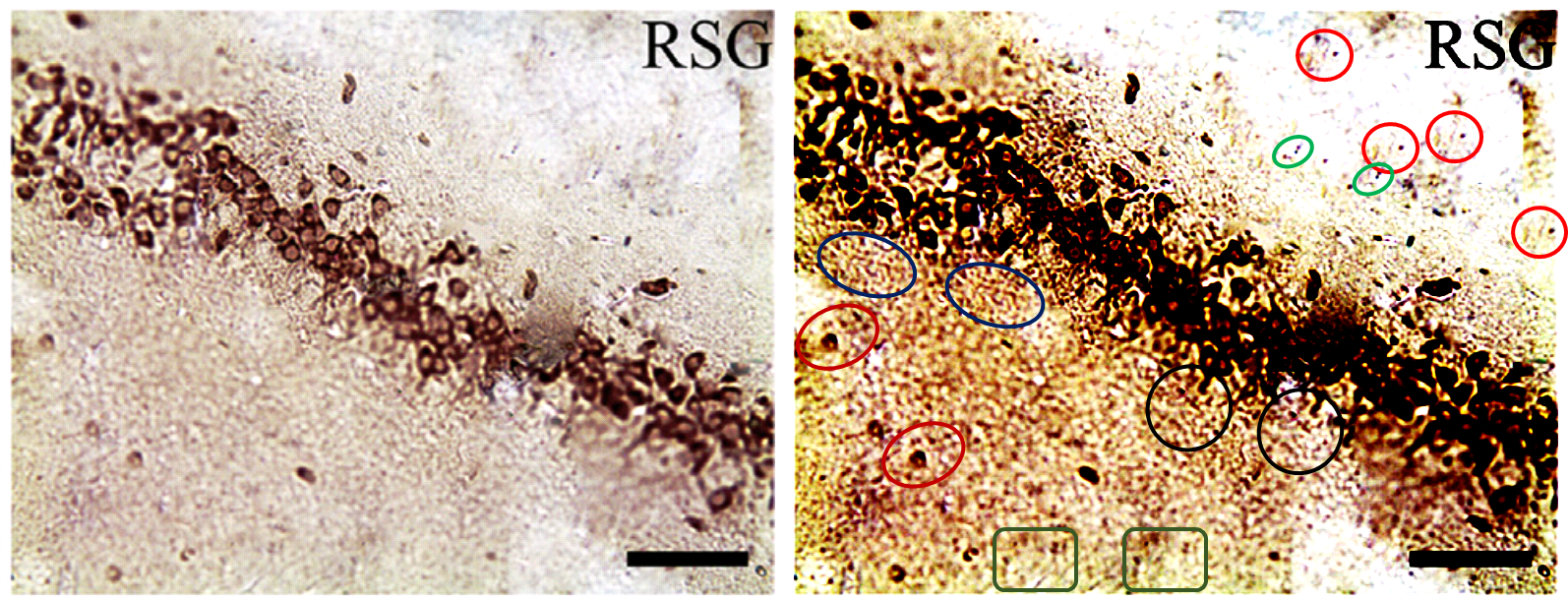
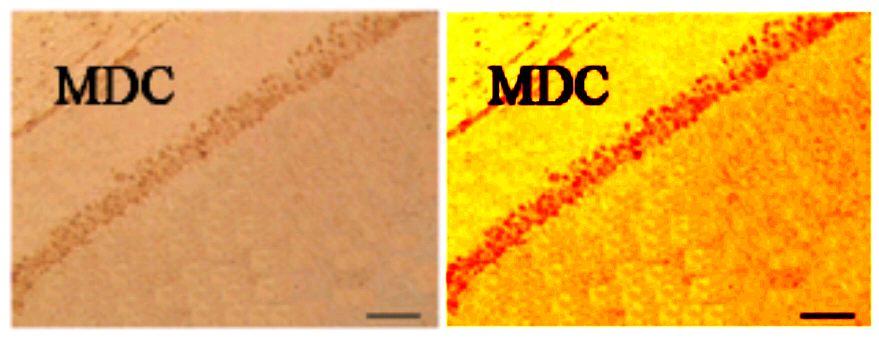
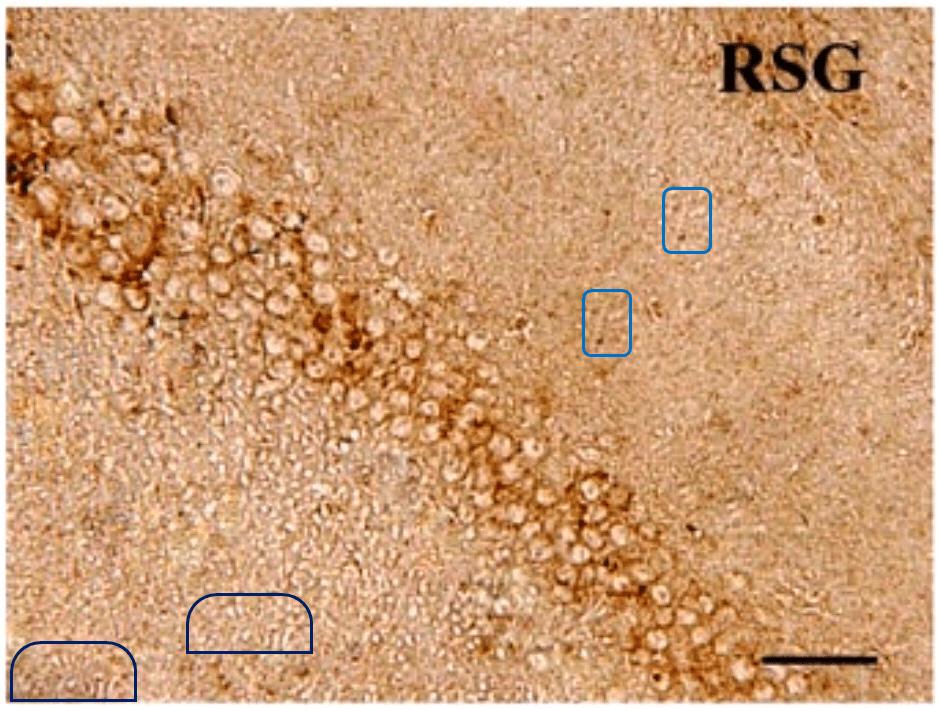
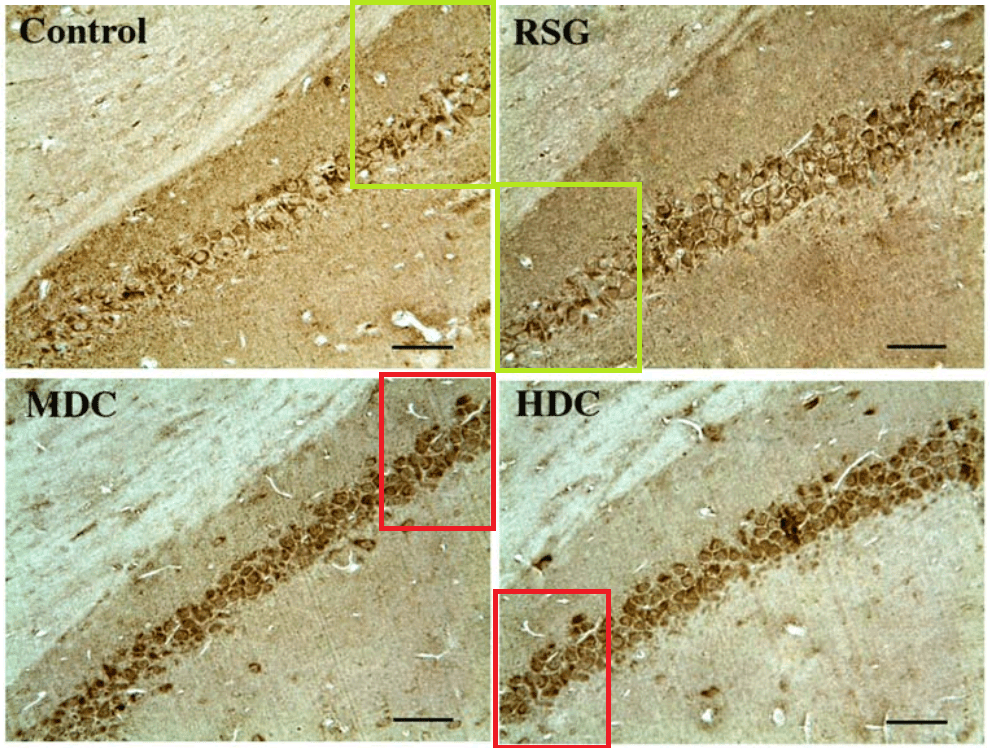
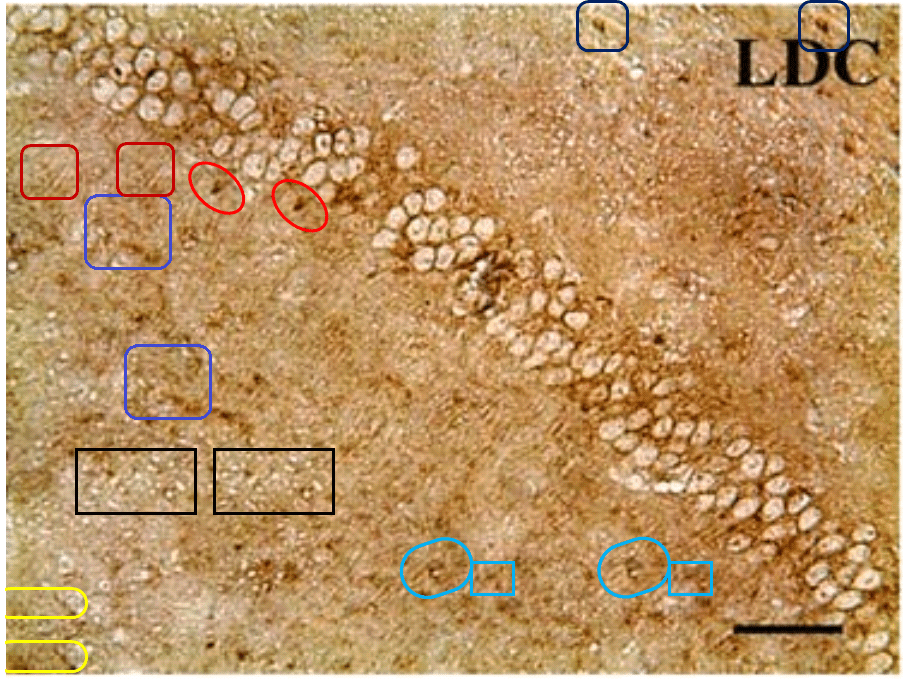
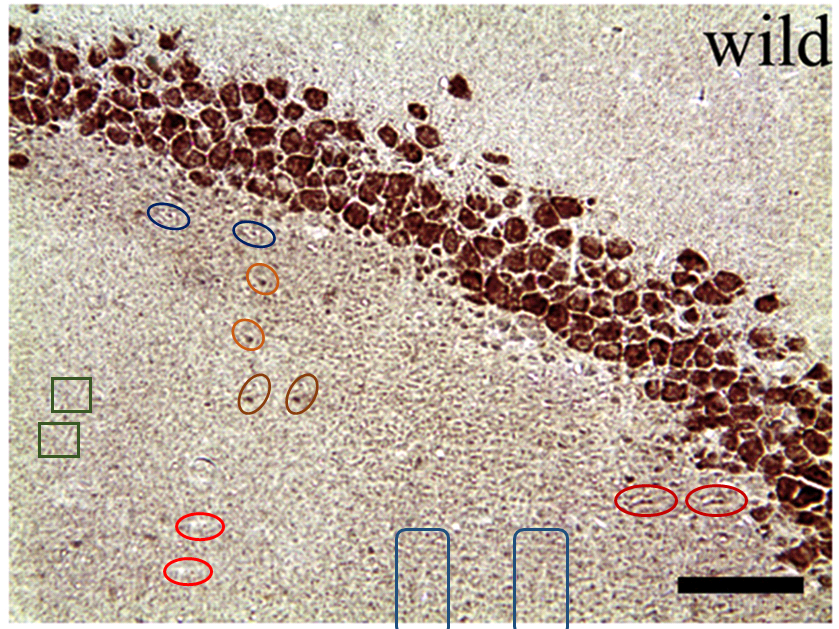
The duplicates hint at the fruitfulness of closer inspection, and of comparisons between panels. Indeed, the LDC and MDC frames of Fig. 9 share the same diagonal band of quizzical faces and axonal filaments like lines scraped into a wet canvas by a painter using the pointy end of the paintbrush. Yet the off-diagonal wedges of the two panels are quite different… which is to say that at least one of them is a Photoshop confection.
Wang and Tian’s presence in PubPeer is limited, with only nine relevant papers. The earliest is from 2012 (Shi et al. [1]), back when curcumin was not neuroprotective in isolation and only worked in the form of turmeric root as part of a herbal melange. [1] comes to our notice because four of the six hippocampic slices in Fig 1 resurfaced after four years as Fig. 1 of Shi et al. (2016) [6] — illustrating the beneficial effects of the same herbalist treatment for AD, but rearranged and relabelled for different conditions.
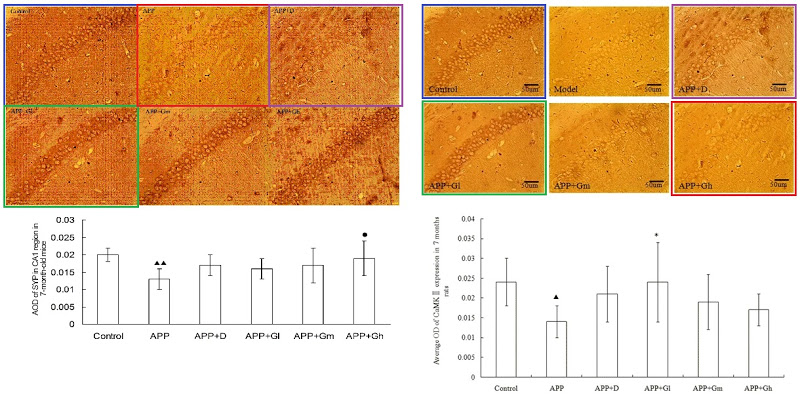
Elsewhere in [6], another three slices ring the changes of permutations across Figs 2, 4 and 5, representing different age groups and the expression of different proteins. All this reuse sows a suspicion that elegance of appearance was more important than any connection between an illustration and the therapeutic regime it putatively illustrated. The suspicion is bolstered by Fig, 8 of Mana et al. (2019) [9] — most recent in this nonet of papers — where two pairs of panels depicting the different conditions are also overlapping at the corners (albeit with one rotation).

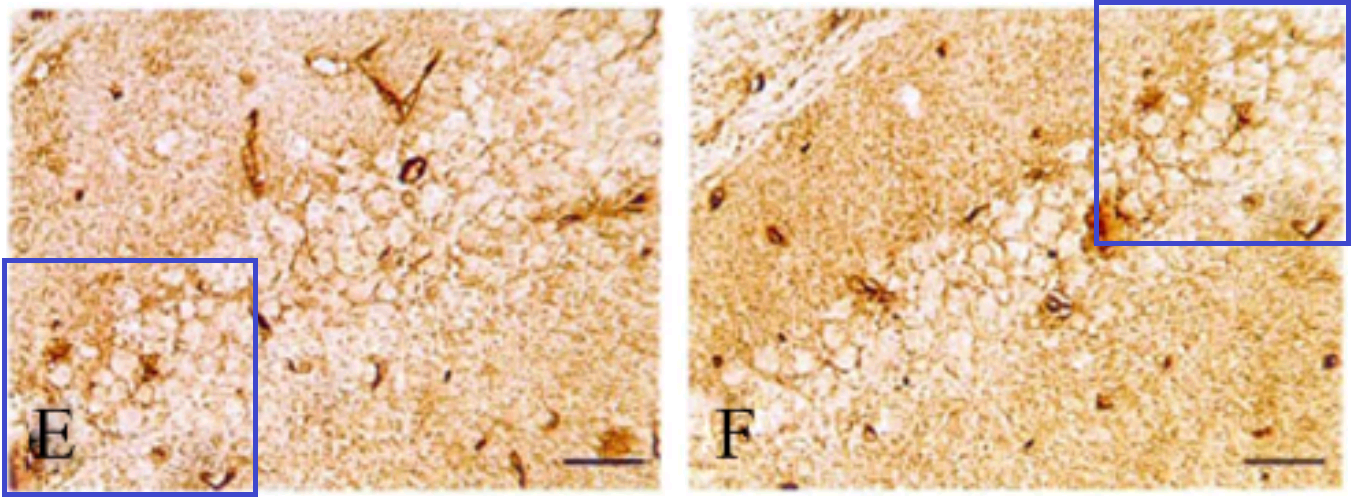
… and the slice of cortex shared between a 7-month-old mouse and an 11-month-old mouse receiving different treatments in Figs 1 and 3 of Shi et al. (2013) [2].
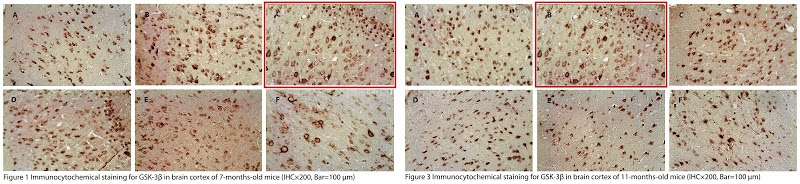
Image duplication is all very well but it is not as challenging and provides less satisfaction than spotting the replication of features within an image. Wang et al. (2014) [3] offers Fig. 6, with two corner overlaps, but more to my tastes are the glitches scampering through Figs 2, 4, 5 and 7 like Pokemons with no-one pursuing them.²
No doubt there are many more, but the downside of training yourself to spot them is the sleepless nights and the haunted dreams, or so I hear from a friend.
Figs 2 and 3 of He et al. (2016) [5] are gorgeously limned in gold-leaf like a Byzantine icon so it seems sacrilegious to desecrate them with annotations around the clone-stamps.
 The pinnacle of this artistic tradition was Chen et al. (2018) [8], in which the curative power of curcumin was assisted by images which not only overlap, but are woven from clone-stamps like the threads of a tapestry.
The pinnacle of this artistic tradition was Chen et al. (2018) [8], in which the curative power of curcumin was assisted by images which not only overlap, but are woven from clone-stamps like the threads of a tapestry.

The recurring trails of tiny footprints across the backgrounds of some images suggest that the textures are purely fictive: composed in Photoshop with painstaking lapidary layers of clone-stamping.
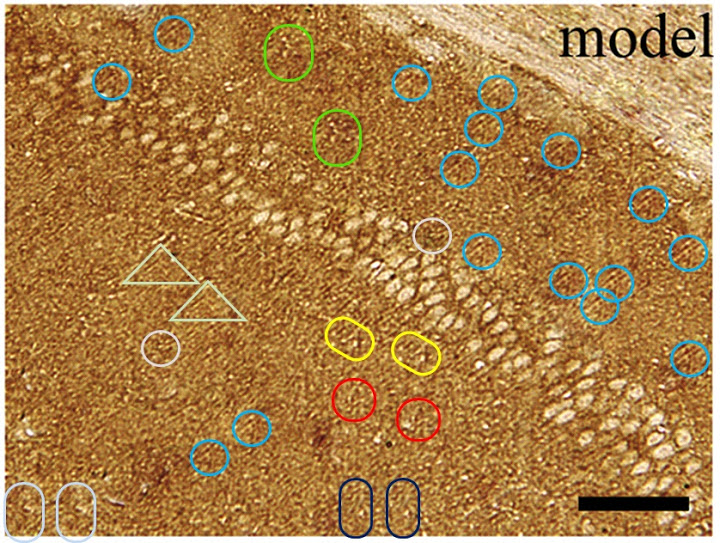
The problem when some images were clearly built up from a blank canvas is that the glitches and hiccups in other more-plausible images could merely be superficial enhancements to otherwise veridical microphotographs, or they could be remaining traces (elsewhere concealed) of the process of construction.. There is no way of telling. I can only be sure that some of these, like the Model panel of Fig 2a, took a great deal of dedication.
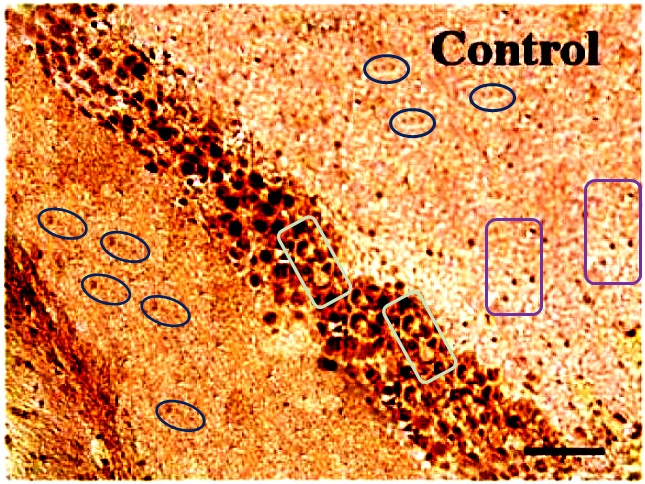
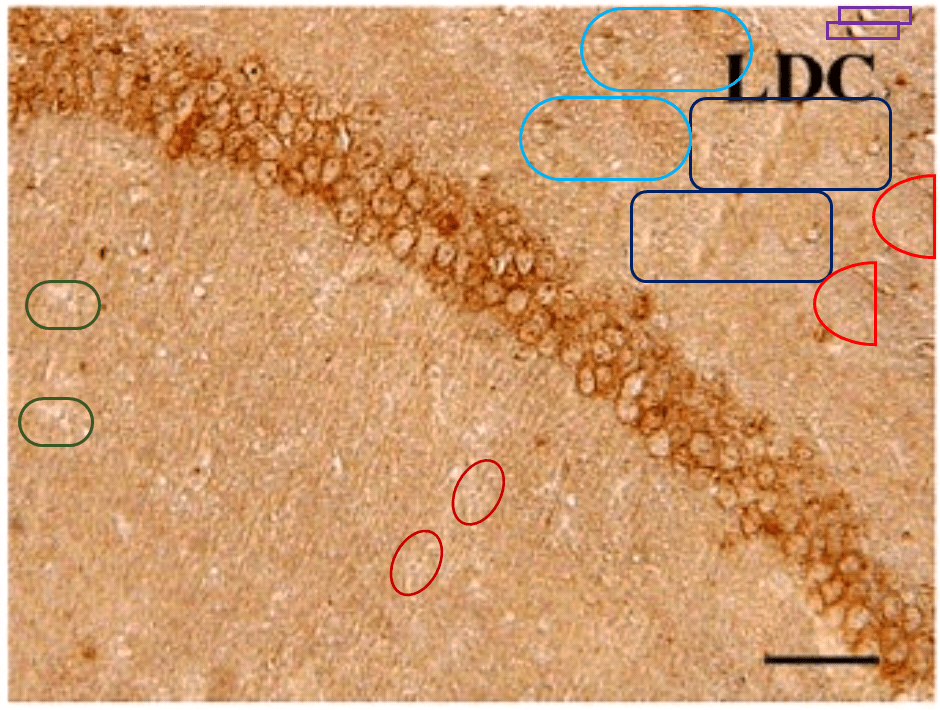
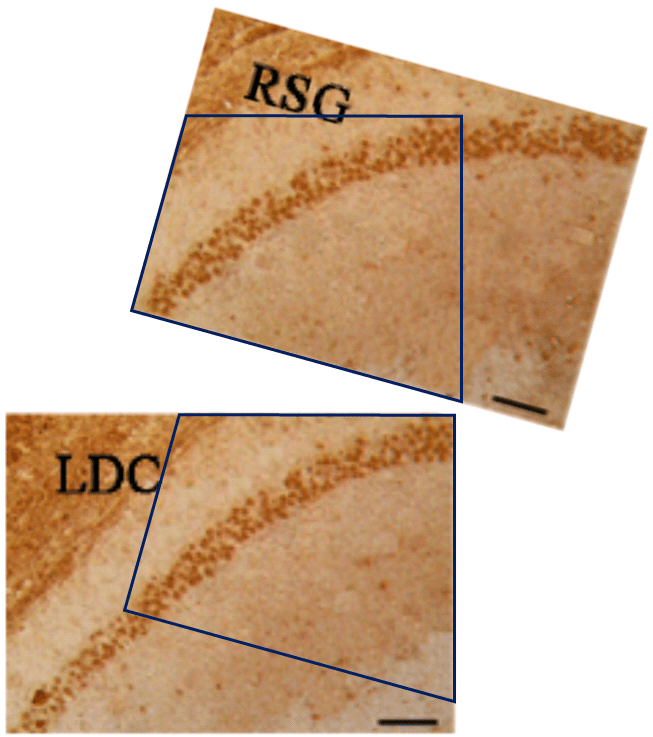
For another example where quilt-weaving took over from histochemistry we can go back to Fig. 4 (Control panel) of [7] — below at left, contrast-heightened to highlight the top layer of clone-stamping. Below at right, Fig.10 (LDC panel). It would be wrong to devote disproportionate space to [7] so I will refer readers to the PubPeer thread. With 10 x 6 panels for different treatments, stained to highlight the expression of various proteins, there is no end of material for people who want to practice their skills, hardly any of it analysed so far.
Compare those careful creations with the insouciant non-naturality of these hellish scenes: Fig. 1(d) from Feng et al. (2016) [4]. Their garish palette is a side-effect of contrast enhancement. The resolution is less than ideal, but it doesn’t obscure the brick-work regularity of the copy-pasted blocks stacked up to compose each scene.

Figs 1(a), 1(b), 1(c), 1(e) and 1(f) are equally fraught with the sharp edges and regularity of crude fabrications. The insouciant non-naturality here is a far cry from the delicate multi-layered digital glazes that went into [8] and [7]. Four of the six panels in Fig. 1(c) overlap, but that is the least of their problems.
All this should be viewed in the context of the recent Lysenkoist decree from Chinese leadership that the alchemical life-force herbalism of Traditional Chinese Medicine does work. The edict might partly have been motivated to uphold Chinese cultural pride, and partly because the entrepreneurs who marked TCM-branded products within China have become wondrously wealthy, thereby amplifying the value of their opinions. Medical researchers are therefore obliged, if they want to advance in their academic careers, to interlard their publications with reports (obtained one way or another) of treating diseases with herbal-mixture decoctions. Especially researchers who are affiliated to the “Key Laboratory of Chinese Internal Medicine” at “Beijing University of Chinese Medicine”. The BUCM even has its own Elsevier-published open-access journal — the J. of Traditional Chinese Medical Sciences.
Consequences of this incentive include the advertisements produced by Wen Jiang (and funded by Welsh charities) for fungus-and-cockroach extracts from a cancer-cure tycoon; and nano-encapsulated dried-leech soup for treating strokes in diabetic rats.
Now back in 2011 (and again in 2017), Tian reported promising results in a Phase-III human trial (conducted 2007-2009) of Shenwu capsules: a different six-herb treatment for AD. While in 2013, a human trial of Fufangdanshen herb tablets for vascular dementia delivered “significant improvements”, and another trial is underway. From the perspective of this prior success it seems like a step backwards to exploratory work on AD in mice with turmeric (as an extract or a synergistic blend with fungi and endangered plant species). To be scrupulously fair to Wang and Tian, though, the Alzheimers-research establishment in Europe and US have been reporting the same kind of dramatic-improvement breakthroughs (which go nowhere) for at least as long.
Cartilago Delenda Est
Now we can return at last to Guangxi Medical University, where Jinmin Zhao’s circle of researchers have experienced equally strong incentives to find positive results to report for TCM. This will be an overview of their oeuvre rather than a detailed dive into each of the 28 relevant PubPeer threads. I draw heavily on the contributions to those threads from Indigofera Tanganykensis.
Guangxi is not a wealthy province, leaving the GMU faculty no room to be fastidious about what they do to win research funding. The results are (a) a creatively eclectic program of research into natural and semi-synthetic compounds that might potentially accelerate bone / cartilage regeneration; and (b) an explosion of acronyms in the titles of papers: EGCG, LDQN-C, ZXHA-TC, though also Andrographolide, Baicalin, Daidzin and cobra-venom NGF.
In keeping with the opening theme, here is a correction (issued 29 Aug 2019) to Wei et al. (2015) [G], involving three panels in Fig 7a and two panels in 7b. As overlapping details blown up from a single larger panorama, they were not really suitable to illustrate scenes of chondrocytes growing in vitro with different amounts of anti-oxidant growth stimulation. A correction seems appropriate here in the absence of manipulation, but even better would have been to avoid the mistake and match images to conditions in the first place.
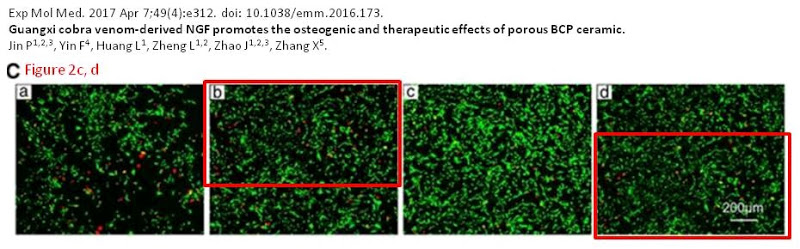
Mid-to-late 2019 was a busy time for amendations. On 5 July 2019 one was issued for Jin et al. (2017) [L], Fig. 2A. These corrections were made pro-actively without public nagging and the authors deserve kudos for noticing the error, which might never have come to attention otherwise.
Then on 26 September 2019, this correction to Jiang et al. (2016) [K], affecting Fig. 5C. Perhaps journal editors could invite other scholars to preview manuscripts and check for minor administrative stumbles like this before the need to amend them arises… we could call them, just thinking aloud here, “peer reviewers”.
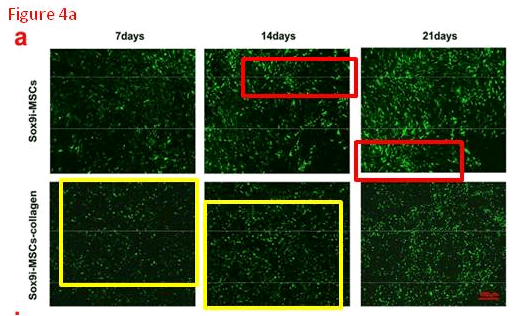
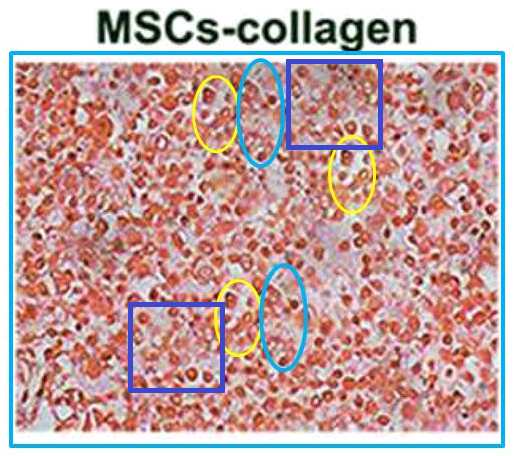
For the sake of narrative expedience I skipped a correction on 1 September 2019, for Jiang et al. (2018) [M]. Yes, the six panels of Fig. 4(a) overlap and could only have come from at most four original images. Of more interest, though, and as yet uncorrected, is the ‘MSCs-Collagen’ panel of Fig. 5(a). It could be a cross-section of some kind of German sausage, except sausage contents do not have replicated areas.
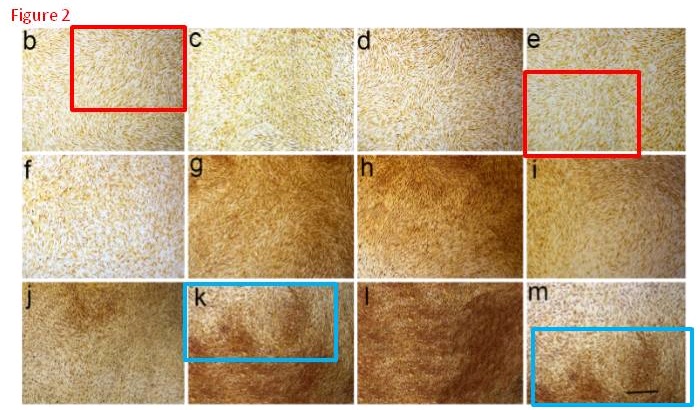
For some variety, how about these images?
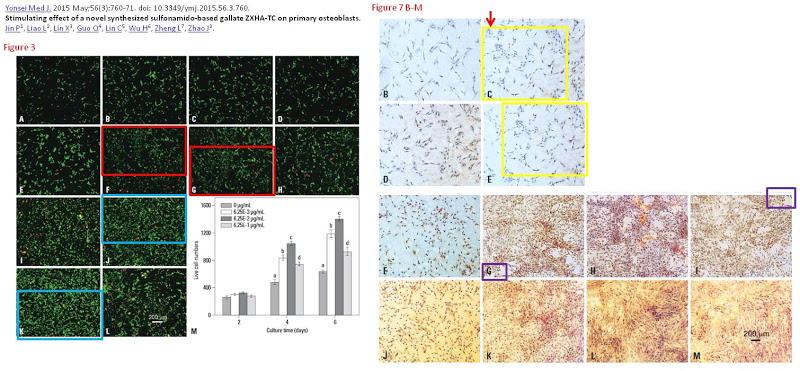
They are panels from Fig. 7 of Jin et al. (2015) [D], annotated with boxes to highlight recurring cell clusters. Nominally they depict “ALP staining of primary osteoblasts at different concentrations (0 μg/mL, 6.25×10-3 μg/mL, 6.25×10-2 μg/mL, and 6.25×10-1 μg/mL) of ZXHA-TC”. They call out, though, to hang in the Tate Britain, alongside Turner’s impressionistic storms.
They are silent tornadoes of agitation, like the cavernous windswept spaces of Dorothea Tanning, but osteoblasts swirling instead of drapery. Whichever author assembled these images should consider illustrating the Divine Comedy, with Fig. 7M to go with Canto V. Personally I would have been tempted to save trouble by conducting experiments, even at the risk of a less aesthetically-pleasing outcome.³
An Erratum was published June 24 2019, but it only addressed three pairs of overlaps while these panels went unmentioned.
” We regret to state that incorrect images were uploaded in Fig. 3F, Fig. 3J, and Fig. 7C. The authors apologize for these errors.”
I am loath to bore readers by continuing this litany of corrections for protein-labelled tissue-culture overlaps. Thus we can skip the 25 June 2019 alternation for Fig 5. of Xu et al. (2016) [I]. Also the alteration for Figs. 4B, F and H, Fig. 7F, and Fig. S3 of Jiang et al. (2019) [O], although it was recent, and included some Western Blots. We turn instead to protein-labelled tissue-culture overlaps where the Errata are not yet on-line.
Fig. 3 from Lu et al. (2014) [B] and Fig. 3A from Lu et al. (2015) [E] deserve our attention because the colours are so restful to the eye (also Fig. 5 from [B]).
From Jiang et al. (2015) [C], Figs 4 and 5:
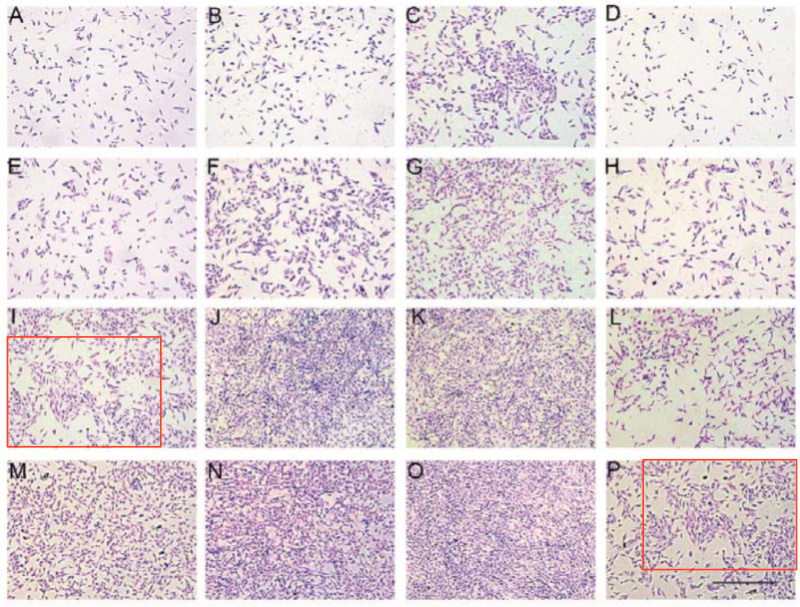

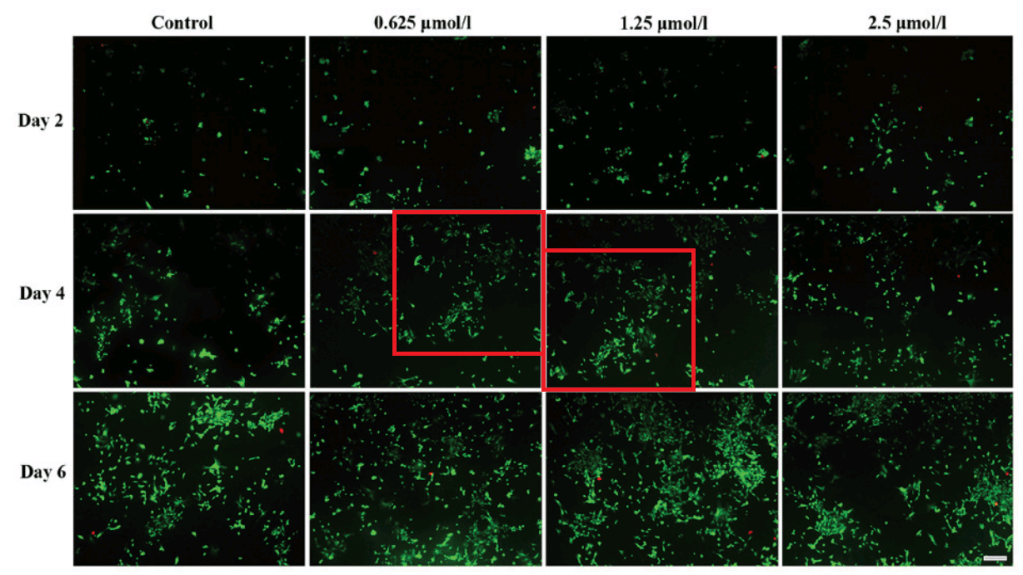
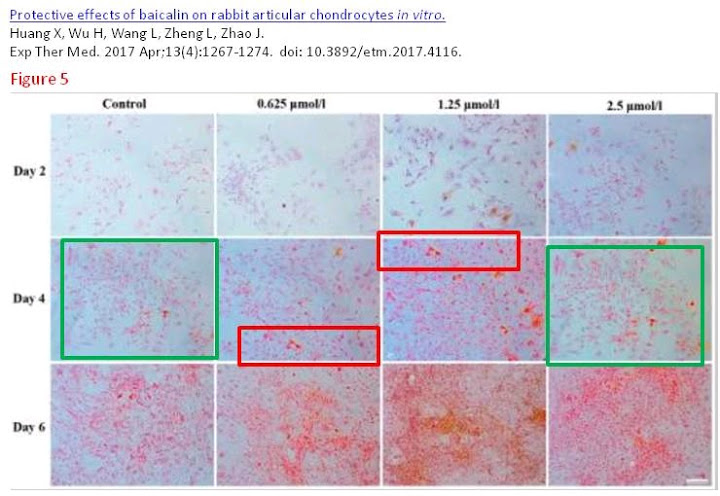
From Huang et al. (2017) [J], Figs 3 and 5:
Figs 4B of Lu et al. (2016) [F] and 4D of Qin et al. (2019) [P] are a little more problematical. In the former, a single stained slice of cartilage was rotated between its duplicate appearances.
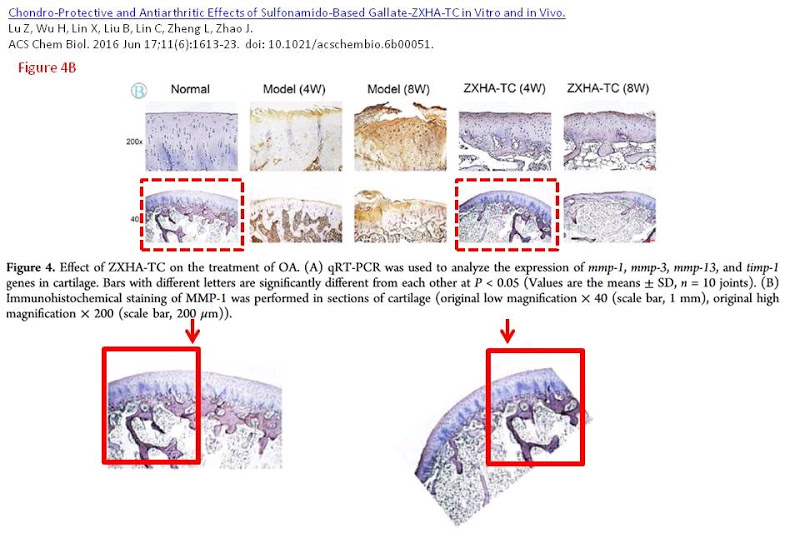
The latter involved a 90° rotation of the duplicated panel (Fig 3 of [P] included another doppelganger).
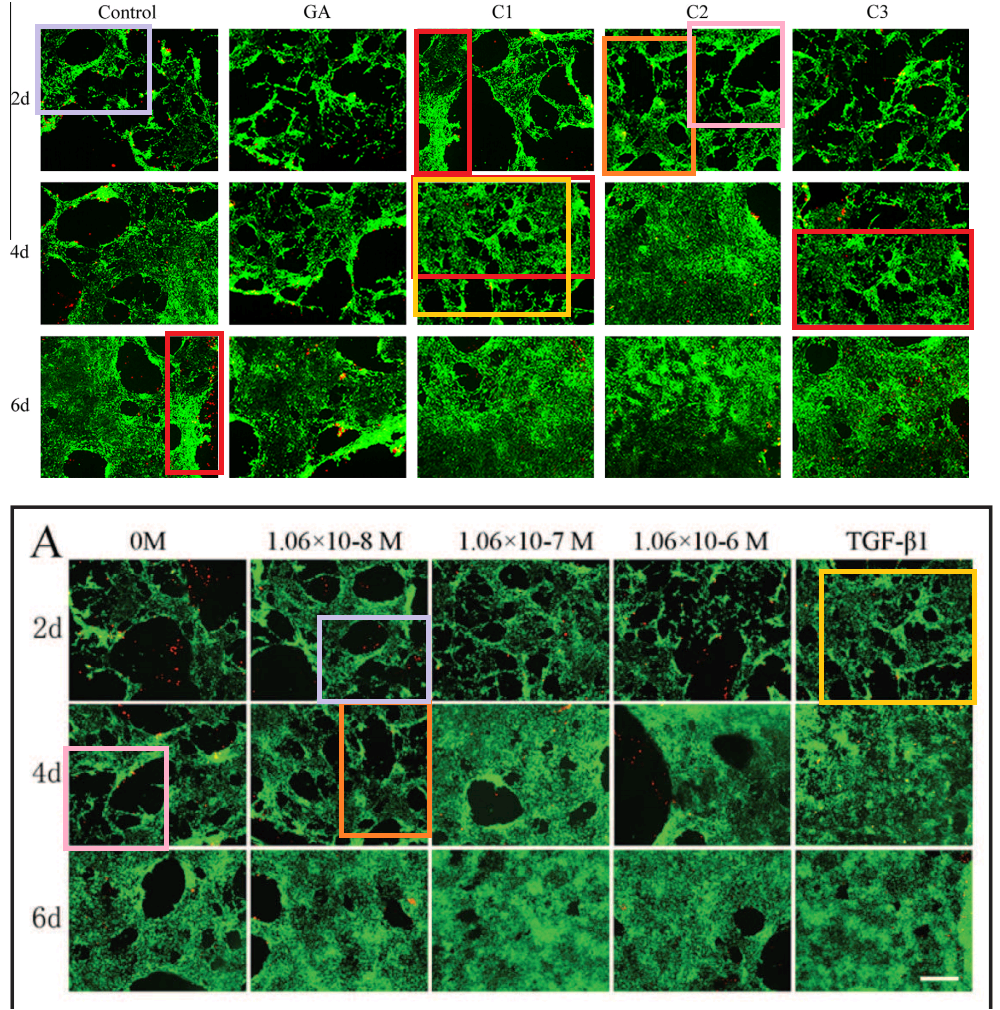
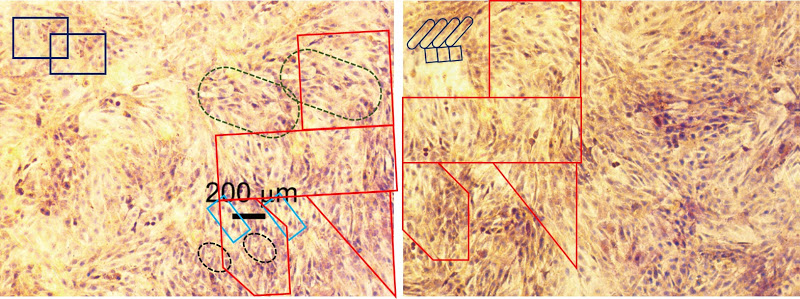
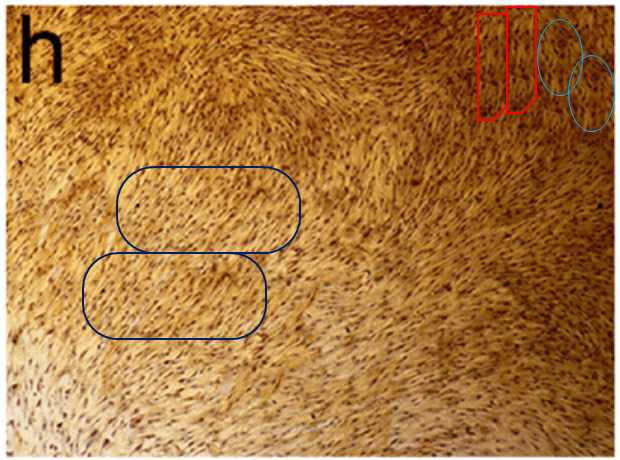
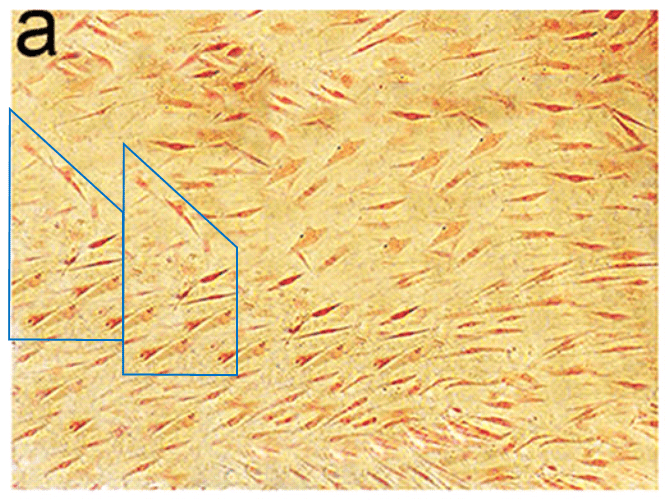
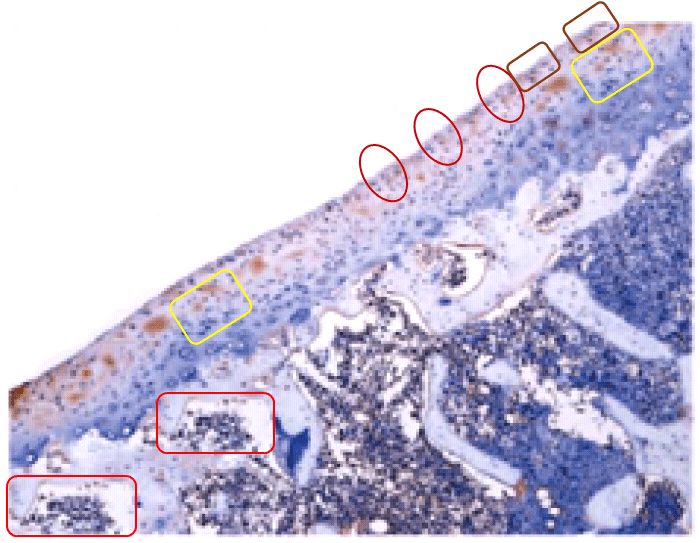
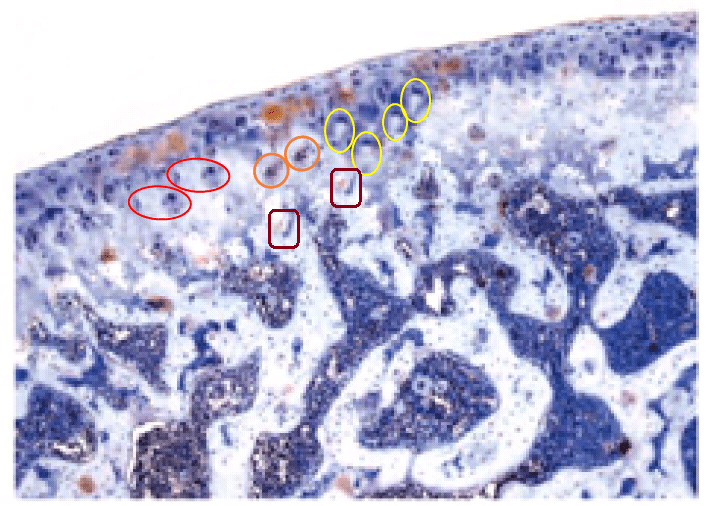
All this is leading up to Jin et al. (2014) [A], and in my impatience to get there I shall leave other examples for interested readers to find for themselves. For [A] brings us back into Turner territory. Indigofera parsed the panels of Fig. 5 into their digital brushstrokes:
Fig. 6 is more of the same but we do not want to attract criticism from Koestler’s Society for the Prevention of Cruelty to Dead Horses by flogging it. One panel each from Figs 2 and 3 suffice to make the point. Any resemblance to Günther Uecker’s nail/shadow painting/sculptures is purely coincidental.
So much effort must have gone into the construction of these frames that I can forgive the authors for using them more than once in Fig. 2.
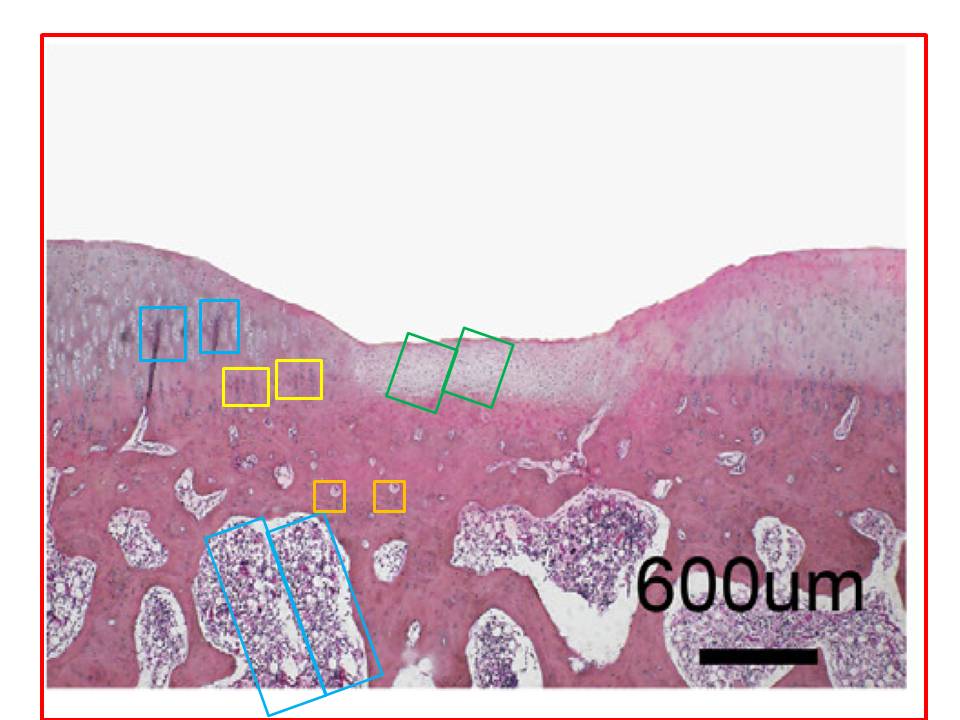
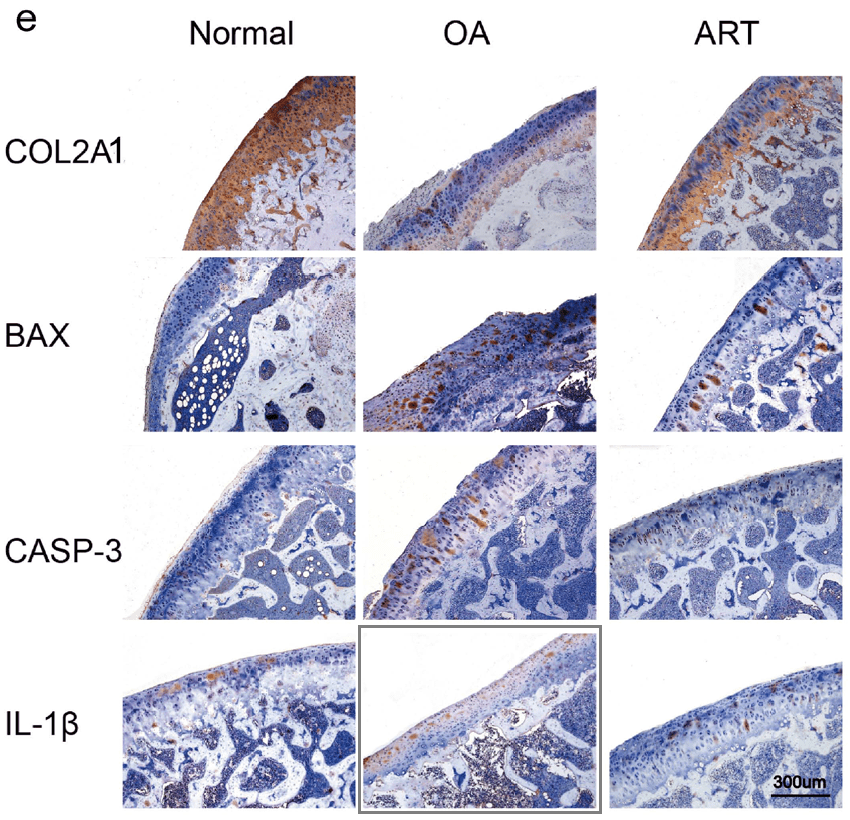
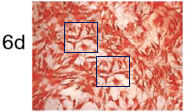
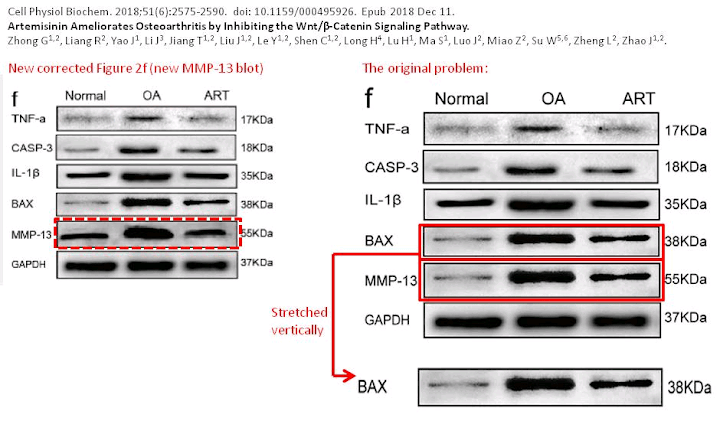
To round off this whole discussion, Zhong et al. (2018) [N] brings us more-or-less back to where we began with some protein-specific stained cartilage slices. The panels of Fig 3(e) are probably not entirely manufactured, but they are not entirely natural either.
This is in addition to the unusual appearance of the Normal / 6-day panel of Fig. 2(d) (below, left).
An Erratum was issued in September 2019, but it only addressed the borked Western Blot of Fig. 2(f) (above, right).
The overall effect is to leave me concerned for the general research culture at Guangxi Medical University. Prof. Jinmin Zhao is President of GMU but I am guessing that he is primarily involved in writing the grant applications, with little time left for oversight responsibility for the projects themselves, or what comes out of them. Frequent co-author Li Zheng may be closer to the lab-bench and seems to be leading the project of amending the scientific record, with comments at PubPeer about a correction in progress. Transforming an institutional culture may be even harder than persuading a journal to issue an Erratum, so I wish Dr Zheng the best of luck.
- Berlin Ichthyosaur Death Assemblage” is in fact the name of my German Death Metal band.
- At press time, [3] is marked at the journal website as “Retracted”. There has not been time for a Retraction Notice explaining the reasons, or for the change of status to percolate through to the PubMed database.
- Photoshop manipulations do not completely disguise the overlap between Panels 7L and 7M of [D].
Sources
- “A combination extract of ginseng, epimedium, polygala, and tuber curcumae increases synaptophysin expression in APPV717I transgenic mice”, Jing Shi, Jinzhou Tian, Xuekai Zhang, Mingqing Wei, Long Yin, Pengwen Wang, Yongyan Wang (2012). Chinese Medicine doi: 10.1186/1749-8546-7-13 [PubPeer].
- “A combination extract of Renshen (Panax Ginseng), Yinyanghuo (Herba Epimedii Brevicornus), Yuanzhi (Radix Palygalae) and Jianghuang (Rhizoma Curcumae Longae) decreases glycogen synthase kinase 3β expression in brain cortex of APPV717I transgenic mice”, Jing Shi, Jinzhou Tian, Xuekai Zhang, Chuiyou Zeng, Mingqing Wei, Pengwen Wang, Yongyan Wang (2013). Journal of Traditional Chinese Medicine doi: 10.1016/s0254-6272(13)60127-2 [PubPeer].
- “Mechanisms and effects of curcumin on spatial learning and memory improvement in APPswe/PS1dE9 mice”, Pengwen Wang, Caixin Su, Ruisheng Li, Hong Wang, Ying Ren, Haiyun Sun, Jinduo Yang, Jianning Sun, Jing Shi, Jinzhou Tian, Shucui Jiang (2014). Journal of Neuroscience Research doi: 10.1002/jnr.23322 [PubPeer].
- “Curcumin ameliorates insulin signalling pathway in brain of Alzheimer’s disease transgenic mice”, Hui-li Feng, Hui-zi Dang, Hui Fan, Xiao-pei Chen, Ying-xue Rao, Ying Ren, Jin-duo Yang, Jing Shi, Peng-wen Wang, Jin-zhou Tian (2016). International Journal of Immunopathology & Pharmacology doi: 10.1177/0394632016659494 [Pubpeer].
- “Effects of curcumin on synapses in APPswe/PS1dE9 mice”, Yingkun He, Pengwen Wang, Peng Wei, Huili Feng, Ying Ren, Jinduo Yang, Yingxue Rao, Jing Shi, Jinzhou Tian (2016). International Journal of Immunopathology & Pharmacology doi: 10.1177/0394632016638099 [PubPeer].
- “Herbal formula GAPT prevents beta amyloid deposition induced Ca(2+)/Calmodulin-dependent protein kinase II and Ca(2+)/Calmodulin-dependent protein phosphatase 2B imbalance in APPV717I mice”, Jing Shi, Xuekai Zhang, Long Yin, Mingqing Wei, Jingnian Ni, Ting Li, Pengwen Wang, Jinzhou Tian, Yongyan Wang (2016). BMC Complementary & Alternative Medicine doi: 10.1186/s12906-016-1144-7 [PubPeer].
- “Curcumin regulates insulin pathways and glucose metabolism in the brains of APPswe/PS1dE9 mice”, Pengwen Wang, Caixin Su, Huili Feng, Xiaopei Chen, Yunfang Dong, Yingxue Rao, Ying Ren, Jinduo Yang, Jing Shi, Jinzhou Tian, Shucui Jiang (2017). International Journal of Immunopathology & Pharmacology doi: 10.1177/0394632016688025 [PubPeer].
- “Curcumin can influence synaptic dysfunction in APPswe/PS1dE9 mice”, Fang Chen, Yingkun He, Pengwen Wang, Peng Wei, Huili Feng, Yingxue Rao, Jing Shi, Jinzhou Tian (2018). Journal of Traditional Chinese Medical Sciences doi: 10.1016/j.jtcms.2018.03.005 [PubPeer].
- “Effect of Chinese herbal compound GAPT on the early brain glucose metabolism of APP/PS1 transgenic mice”, Lulu Mana, Huili Feng, Yunfang Dong, Yahan Wang, Jing Shi, Jinzhou Tian, Pengwen Wang (2019). International Journal of Immunopathology & Pharmacology doi: 10.1177/2058738419841482 [PubPeer].
- [A]. “Epigallocatechin-3-gallate (EGCG) as a pro-osteogenic agent to enhance osteogenic differentiation of mesenchymal stem cells from human bone marrow: an in vitro study”, Pan Jin, Huayu Wu, Guojie Xu, Li Zheng, Jinmin Zhao (2014). Cell & Tissue Research doi: 10.1007/s00441-014-1797-9 [PubPeer].
- [B]. “A novel synthesized sulfonamido-based gallic acid – LDQN-C: Effects on chondrocytes growth and phenotype maintenance”, Zhenhui Lu, Shixiu Wei, Huayu Wu, Xiao Lin, Cuiwu Lin, Buming Liu, Li Zheng, Jinmin Zhao (2014). Bioorganic Chemistry doi: 10.1016/j.bioorg.2014.09.005 [PubPeer].
- [C]. “Andrographolide Exerts Pro-Osteogenic Effect by Activation of Wnt/β-Catenin Signaling Pathway in Vitro”, Tongmeng Jiang, Bo Zhou, Li Huang, Huayu Wu, Jiancheng Huang, Tihong Liang, Hui Liu, Li Zheng, Jinmin Zhao (2015). Cellular Physiology & Biochemistry doi: 10.1159/000430196 [PubPeer].
- [D]. “Stimulating effect of a novel synthesized sulfonamido-based gallate ZXHA-TC on primary osteoblasts”, Pan Jin, Liang Liao, Xiao Lin, Qinggong Guo, Cuiwu Lin, Huayu Wu, Li Zheng, Jinmin Zhao (2015). Yonsei Medical Journal doi: 10.3349/ymj.2015.56.3.760 [PubPeer].
- [E]. “Stimulating Effect of a Newly Synthesized Sulfonamido-Basedgallate on Articular Chondrocytes in Vitro”, Zhenhui Lu, Liqin Wang, Hongmei Pan, Xiao Lin, Cuiwu Lin, Buming Liu, Li Zheng, Jinmin Zhao (2015). Cellular Physiology & Biochemistry doi: 10.1159/000430243 [PubPeer].
- [F]. “Chondro-Protective and Antiarthritic Effects of Sulfonamido-Based Gallate–ZXHA-TC in Vitro and in Vivo”, Zhenhui Lu, Huayu Wu, Xiao Lin, Buming Liu, Cuiwu Lin, Li Zheng, Jinmin Zhao (2016). ACS Chemical Biology doi: 10.1021/acschembio.6b00051 [PubPeer].
- [G] “A Novel Synthesized Sulfonamido-Based Gallate—JEZ-C as Potential Therapeutic Agents for Osteoarthritis”, Shixiu Wei, Zhenhui Lu, Yunfeng Zou, Xiao Lin, Cuiwu Lin, Buming Liu, Li Zheng, Jinmin Zhao (2015). PLoS ONE doi: 10.1371/journal.pone.0125930 [PubPeer].
- [H]. “Functional quantum dot-siRNA nanoplexes to regulate chondrogenic differentiation of mesenchymal stem cells”, Yang Wu, Bo Zhou, Fuben Xu, Xiaoyong Wang, Gang Liu, Li Zheng, Jinmin Zhao, Xingdong Zhang (2016). Acta Biomaterialia doi: 10.1016/j.actbio.2016.09.008 [PubPeer].
- [I] “Pro-neurogenic effect of β-asarone on RSC96 Schwann cells in vitro”, Fuben Xu, Huayu Wu, Kun Zhang, Peizhen Lv, Li Zheng, Jinmin Zhao (2016). In Vitro Cellular & Developmental Biology – Animal doi: 10.1007/s11626-015-9980-1 [PubPeer].
- [J]. “Protective effects of baicalin on rabbit articular chondrocytes in vitro”, Xianyuan Huang, Huayu Wu, Liqin Wang, Li Zheng, Jinmin Zhao (2017). Experimental & Therapeutic Medicine doi: 10.3892/etm.2017.4116 [PubPeer].
- [K]. “In vitro expansion impaired the stemness of early passage mesenchymal stem cells for treatment of cartilage defects”, Tongmeng Jiang, Guojie Xu, Qiuyan Wang, Lihui Yang, Li Zheng, Jinmin Zhao, Xingdong Zhang (2017). Cell Death & Disease doi: 10.1038/cddis.2017.215 [PubPeer].
- [L]. “Guangxi cobra venom-derived NGF promotes the osteogenic and therapeutic effects of porous BCP ceramic”, Pan Jin, Fuqiang Yin, Li Huang, Li Zheng, Jinmin Zhao, Xingdong Zhang (2017). Experimental & Molecular Medicine doi: 10.1038/emm.2016.173 [PubPeer].
- [M]. “The role of Sox9 in collagen hydrogel-mediated chondrogenic differentiation of adult mesenchymal stem cells (MSCs)”, Xianfang Jiang, Xianyuan Huang, Tongmeng Jiang, Li Zheng, Jinmin Zhao, Xingdong Zhang (2018). Biomaterials Science doi: 10.1039/c8bm00317c [PubPeer].
- [N]. “Artemisinin Ameliorates Osteoarthritis by Inhibiting the Wnt/β-Catenin Signaling Pathway”, Gang Zhong, Ruiming Liang, Jun Yao, Jia Li, Tongmeng Jiang, Jianwei Liu, Yiguan Le, Chong Shen, Huiping Long, Huiping Lu, Shiting Ma, Jun Luo, Zhikang Miao, Wei Su, Li Zheng, Jinmin Zhao (2018). Cellular Physiology & Biochemistry doi: 10.1159/000495926 [PubPeer].
- [O]. “Untangling the response of bone tumor cells and bone forming cells to matrix stiffness and adhesion ligand density by means of hydrogels”, Tongmeng Jiang, Jinmin Zhao, Shan Yu, Zhengwei Mao, Changyou Gao, Ye Zhu, Chuanbin Mao, Li Zheng (2019). Biomaterials doi: 10.1016/j.biomaterials.2018.10.015 [PubPeer].
- [P]. “miR-17-5p Regulates Heterotopic Ossification by Targeting ANKH in Ankylosing Spondylitis”, Xiong Qin, Bo Zhu, Tongmeng Jiang, Jiachang Tan, Zhenjie Wu, Zhenchao Yuan, Li Zheng, Jinmin Zhao (2019). Molecular Therapy – Nucleic Acids doi: 10.1016/j.omtn.2019.10.003 [PubPeer].

Donate!
If you are interested to support my work, you can leave here a small tip of $5. Or several of small tips, just increase the amount as you like (2x=€10; 5x=€25). Your generous patronage of my journalism, however small it appears to you, will greatly help me with my legal costs.
€5.00





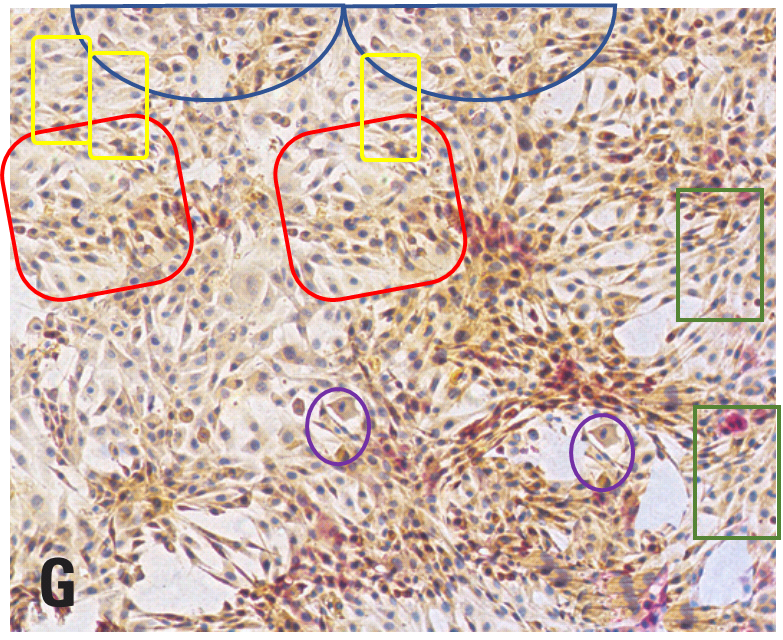
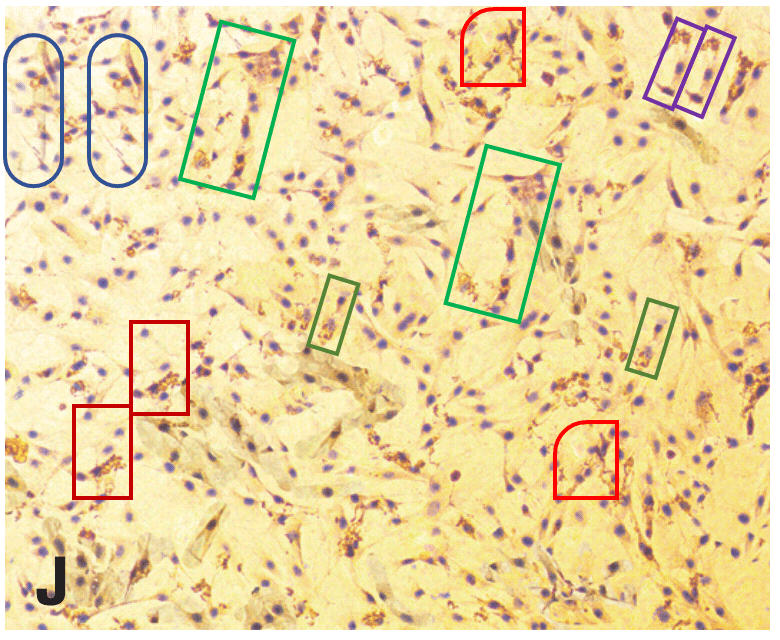

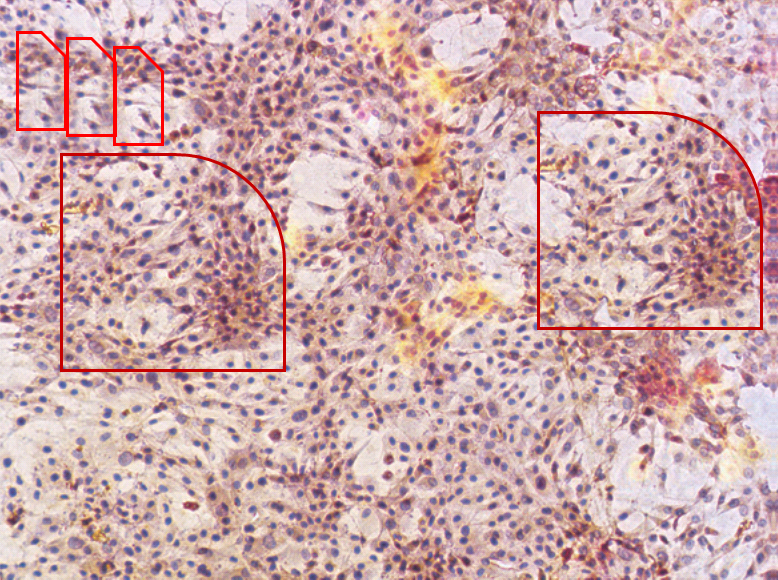
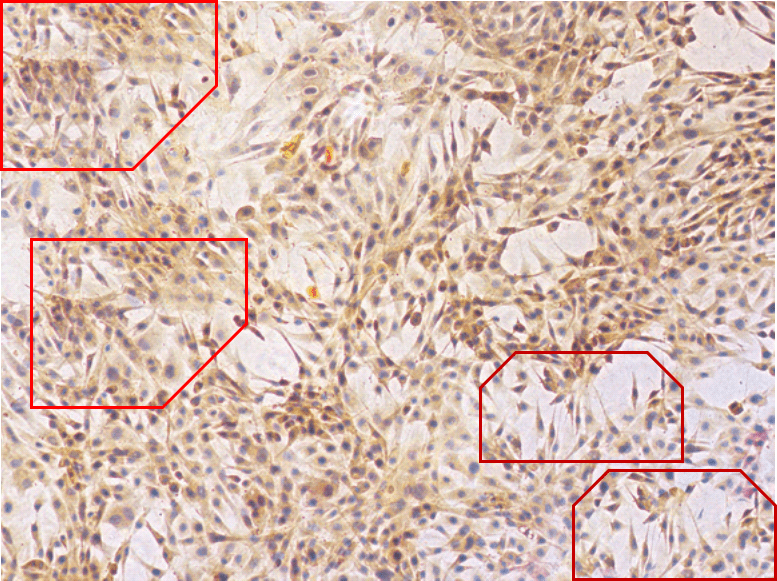

Great summary Smut. For the papers analyzed here, these 2 groups of PS artists received at least 87 millions RMB of research grant from Chinese gov to support their endeavor. What a waste of money for a developing country!
LikeLike
Great work! But this is only what is visible. Think about all the problems we don’t see!
LikeLike
And guess what? They probably want to get out of the polluted stink-hole they are in, and come to the US (or somewhere else with less pollution) and do “good” science. Given enough publications in prestigious journals, they may be able to pull it off, like “Mike”. And no, this is not a racist comment. Ever look at the air quality in major Chinese cities?
LikeLike
You would think that the publishers have learned something after all the Pubpeer disclosures?
They must care about the science!
Think again. I just took a quick look at some random new issues of Journal of Cellular Biochemistry (which
should be renamed to Chinese Journal of Cellular Biochemistry). Duplications everywhere!
It is a complete tragedy! Resources are spent on publishing and indexing misleading research data. Public funds spent on scientific misinformation!
LikeLike
As posted on PubPeer:
2020 retraction notice. https://onlinelibrary.wiley.com/doi/abs/10.1002/jnr.24580?af=R
The above article, published online on 23 November 2013 in Wiley Online Library (wileyonlinelibrary.com), has been retracted by agreement between the journal’s Editor‐in‐Chief, Dr Eric M. Prager, and Wiley Periodicals, Inc. The retraction has been agreed due to the unreliable nature of the figures presented. Concerns were originally raised via PubPeer (https://pubpeer.com/publications/AFF7C528364ED2B17D56AFF1C9BAFD). After an investigation conducted by the journal’s Editor‐in‐Chief, in accordance with the guidelines from the Committee on Publication Ethics (COPE), it was determined that the figures presented in the article could not be validated. Consequently, the main findings of the article, and the conclusions drawn, can no longer be relied upon. As recommended by COPE’s guidelines, we have therefore decided to retract the article. We have attempted to contact the authors for comment but have been unsuccessful in our efforts.
REFERENCE
Wang, P., Su, C., Li, R., Wang, H., Ren, Y., Sun, H., … Jiang, S. (2014). Mechanisms and effects of curcumin on spatial learning and memory improvement in APPswe/PS1dE9 mice. Journal of Neuroscience Research, 92, 218–231. https://doi.org/10.1002/jnr.23322
LikeLike
We are about to submit a paper on the (faint) antibacterial effects of turmeric (curcumin) and neem, that we used to produce composite materials. Now I’m quite worried that people will think that (A) our materials are incredibly less effective than other ones in literature based on the same molecules or (B) our results are part of the Indian/Chinese quackery.
LikeLike
Pingback: The full-service paper mill and its Chinese customers – For Better Science
Pingback: How to quickly write and submit manuscripts to SCI-indexed Journals – For Better Science
“Retracted : GABARAP promotes bone marrow mesenchymal stem cells‐based the osteoarthritis cartilage regeneration through the inhibition of PI3K/AKT/mTOR signaling pathway” (retracted 30 November 2020).
https://pubpeer.com/publications/ECDC4ED8FEB42F8599428D0DEC695F
“RETRACTED ARTICLE: Chondro-protective effects of polydatin in osteoarthritis through its effect on restoring dysregulated autophagy via modulating MAPK, and PI3K/Akt signaling pathways” (retracted 25 September 2019).
https://pubpeer.com/publications/69BB7E582BD129EBC7FEA908C22042
LikeLike
TigerBB8 reports that Zhenhui Lu “has successfully secured her promotion to an associate professor just recently.”
Not a huge surprise when serial-last-author Jinmin Zhao remains President of Guangxi Medical University.
LikeLike